Abstract
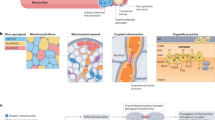
Perturbed diurnal rhythms are becoming increasingly evident as deleterious events in the pathology of metabolic diseases. Exercise is well characterized as a crucial intervention in the prevention and treatment of individuals with metabolic diseases. Little is known, however, regarding optimizing the timing of exercise bouts in order to maximize their health benefits. Furthermore, exercise is a potent modulator of skeletal muscle metabolism, and it is clear that skeletal muscle has a strong circadian profile. In humans, mitochondrial function peaks in the late afternoon, and the circadian clock might be inherently impaired in myotubes from patients with metabolic disease. Timing exercise bouts to coordinate with an individual’s circadian rhythms might be an efficacious strategy to optimize the health benefits of exercise. The role of exercise as a Zeitgeber can also be used as a tool in combating metabolic disease. Shift work is known to induce acute insulin resistance, and appropriately timed exercise might improve health markers in shift workers who are at risk of metabolic disease. In this Review, we discuss the literature regarding diurnal skeletal muscle metabolism and the interaction with exercise bouts at different times of the day to combat metabolic disease.
Key points
-
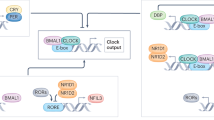
Skeletal muscle has an extensive network of clock-controlled genes, and dysregulation of its molecular clock can lead to deleterious metabolic consequences.
-
Physical strength and skeletal muscle mitochondrial function peak in the late afternoon, whereas low-energy sensitive signalling peaks in the morning.
-
Exercise is a robust Zeitgeber of skeletal muscle clocks, and exercise can reset the molecular circadian clock, thereby effectively ameliorating the negative effects of disrupted sleep patterns.
-
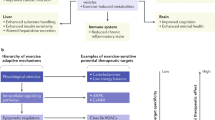
Optimizing the timing of exercise bouts could aid existing therapeutic interventions for the management of metabolic disease.
-
Divergent modalities of exercise can interact with the circadian rhythm, resulting in potent metabolic effects.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Gerhart-Hines, Z. & Lazar, M. A. Circadian metabolism in the light of evolution. Endocr. Rev. 36, 289–304 (2015).
Robinson, I. & Reddy, A. B. Molecular mechanisms of the circadian clockwork in mammals. FEBS Lett. 588, 2477–2483 (2014).
Panda, S. Circadian physiology of metabolism. Science 354, 1008–1015 (2016).
Dyar, K. A. et al. Muscle insulin sensitivity and glucose metabolism are controlled by the intrinsic muscle clock. Mol. Metab. 3, 29–41 (2014).
Schiaffino, S., Blaauw, B. & Dyar, K. A. The functional significance of the skeletal muscle clock: lessons from Bmal1 knockout models. Skelet. Muscle 6, 33 (2016).
Vetter, C. et al. Night shift work, genetic risk, and type 2 diabetes in the UK biobank. Diabetes Care 41, 762–769 (2018).
Bescos, R. et al. Four days of simulated shift work reduces insulin sensitivity in humans. Acta Physiol. (Oxf.) 223, e13039 (2018).
Peek, C. B. et al. Circadian clock interaction with HIF1α mediates oxygenic metabolism and anaerobic glycolysis in skeletal muscle. Cell Metab. 25, 86–92 (2017).
Saner, N. J., Bishop, D. J. & Bartlett, J. D. Is exercise a viable therapeutic intervention to mitigate mitochondrial dysfunction and insulin resistance induced by sleep loss? Sleep Med. Rev. 37, 60–68 (2018).
Wolff, G. & Esser, K. A. Scheduled exercise phase shifts the circadian clock in skeletal muscle. Med. Sci. Sports Exerc. 44, 1663–1670 (2012).
Gabriel, B. M. & Zierath, J. R. The limits of exercise physiology: from performance to health. Cell Metab. 25, 1000–1011 (2017).
Vera, B. et al. Modifiable lifestyle behaviors, but not a genetic risk score, associate with metabolic syndrome in evening chronotypes. Sci. Rep. 8, 945 (2018).
Huang, W., Ramsey, K. M., Marcheva, B. & Bass, J. Circadian rhythms, sleep, and metabolism. J. Clin. Invest. 121, 2133–2141 (2011).
Atkinson, G., Fullick, S., Grindey, C. & Maclaren, D. Exercise, energy balance and the shift worker. Sports Med. 38, 671–685 (2008).
Patke, A. et al. Mutation of the human circadian clock gene CRY1 in familial delayed sleep phase disorder. Cell 169, 203–215 (2017).
Manoogian, E. N. C. & Panda, S. Circadian rhythms, time-restricted feeding, and healthy aging. Ageing Res. Rev. 39, 59–67 (2017).
Mure, L. S. et al. Diurnal transcriptome atlas of a primate across major neural and peripheral tissues. Science 359, eaao0318 (2018).
Chtourou, H. & Souissi, N. The effect of training at a specific time of day: a review. J. Strength Cond. Res. 26, 1984–2005 (2012).
Driver, H. S. & Taylor, S. R. Exercise and sleep. Sleep Med. Rev. 4, 387–402 (2000).
Perrin, L. et al. Transcriptomic analyses reveal rhythmic and CLOCK-driven pathways in human skeletal muscle. eLife 7, e34114 (2018).
Dashti, H. S. et al. CRY1 circadian gene variant interacts with carbohydrate intake for insulin resistance in two independent populations: Mediterranean and North American. Chronobiol. Int. 31, 660–667 (2014).
Raney, B. J. et al. ENCODE whole-genome data in the UCSC genome browser (2011 update). Nucleic Acids Res. 39, D871–D875 (2011).
Boyle, A. P. et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 22, 1790–1797 (2012).
Villard, J. et al. A functionally essential domain of RFX5 mediates activation of major histocompatibility complex class II promoters by promoting cooperative binding between RFX and NF-Y. Mol. Cell. Biol. 20, 3364–3376 (2000).
Dyar, K. A. et al. Transcriptional programming of lipid and amino acid metabolism by the skeletal muscle circadian clock. PLOS Biol. 16, e2005886 (2018).
Lamia, K. A. et al. AMPK regulates the circadian clock by cryptochrome phosphorylation and degradation. Science 326, 437–440 (2009).
Um, J. H. et al. Activation of 5′-AMP-activated kinase with diabetes drug metformin induces casein kinase Iepsilon (CKIepsilon)-dependent degradation of clock protein mPer2. J. Biol. Chem. 282, 20794–20798 (2007).
Jordan, S. D. et al. CRY1/2 selectively repress PPARdelta and limit exercise capacity. Cell Metab. 26, 243–255 (2017).
Lowe, M. et al. Cry2 is critical for circadian regulation of myogenic differentiation by Bclaf1-mediated mRNA stabilization of cyclin D1 and Tmem176b. Cell Rep. 22, 2118–2132 (2018).
Miller, B. H. et al. Circadian and CLOCK-controlled regulation of the mouse transcriptome and cell proliferation. Proc. Natl Acad. Sci. USA 104, 3342–3347 (2007).
Nakao, R. et al. Atypical expression of circadian clock genes in denervated mouse skeletal muscle. Chronobiol. Int. 32, 486–496 (2015).
Zambon, A. C. et al. Time- and exercise-dependent gene regulation in human skeletal muscle. Genome Biol. 4, R61 (2003).
Hansen, J. et al. Synchronized human skeletal myotubes of lean, obese and type 2 diabetic patients maintain circadian oscillation of clock genes. Sci. Rep. 6, 35047 (2016).
O’Connor, E., Kiely, C., O’Shea, D., Green, S. & Egana, M. Similar level of impairment in exercise performance and oxygen uptake kinetics in middle-aged men and women with type 2 diabetes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 303, R70–R76 (2012).
Phielix, E. & Mensink, M. Type 2 diabetes mellitus and skeletal muscle metabolic function. Physiol. Behav. 94, 252–258 (2008).
van Moorsel, D. et al. Demonstration of a day-night rhythm in human skeletal muscle oxidative capacity. Mol. Metab. 5, 635–645 (2016).
de Goede, P., Wefers, J., Brombacher, E. C., Schrauwen, P. & Kalsbeek, A. Circadian rhythms in mitochondrial respiration. J. Mol. Endocrinol. 60, R115–R130 (2018).
Liu, C., Li, S., Liu, T., Borjigin, J. & Lin, J. D. Transcriptional coactivator PGC-1α integrates the mammalian clock and energy metabolism. Nature 447, 477–481 (2007).
Yoon, Y., Galloway, C. A., Jhun, B. S. & Yu, T. Mitochondrial dynamics in diabetes. Antioxid. Redox Signal. 14, 439–457 (2011).
Oliva-Ramirez, J., Moreno-Altamirano, M. M., Pineda-Olvera, B., Cauich-Sanchez, P. & Sanchez-Garcia, F. J. Crosstalk between circadian rhythmicity, mitochondrial dynamics and macrophage bactericidal activity. Immunology 143, 490–497 (2014).
Schmitt, K. et al. Circadian control of DRP1 activity regulates mitochondrial dynamics and bioenergetics. Cell Metab. 27, 657–666 (2018).
Lassiter, D. G., Sjogren, R. J. O., Gabriel, B. M., Krook, A. & Zierath, J. R. AMPK activation negatively regulates GDAP1, which influences metabolic processes and circadian gene expression in skeletal muscle. Mol. Metab. 16, 12–23 (2018).
Xu, Y. et al. Modeling of a human circadian mutation yields insights into clock regulation by PER2. Cell 128, 59–70 (2007).
Zhao, Y. et al. Uncovering the mystery of opposite circadian rhythms between mouse and human leukocytes in humanized mice. Blood 130, 1995–2005 (2017).
Brehm, M. A., Shultz, L. D., Luban, J. & Greiner, D. L. Overcoming current limitations in humanized mouse research. J. Infect. Dis. 208 (Suppl. 2), S125–S130 (2013).
Atkinson, G. & Reilly, T. Circadian variation in sports performance. Sports Med. 21, 292–312 (1996).
Facer-Childs, E. & Brandstaetter, R. The impact of circadian phenotype and time since awakening on diurnal performance in athletes. Curr. Biol. 25, 518–522 (2015).
Fowler, P. M. et al. Greater effect of east versus west travel on jet lag, sleep, and team sport performance. Med. Sci. Sports Exerc. 49, 2548–2561 (2017).
Facer-Childs, E. & Brandstaetter, R. Circadian phenotype composition is a major predictor of diurnal physical performance in teams. Front. Neurol. 6, 208 (2015).
Egan, B. & Zierath, J. R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 17, 162–184 (2013).
Dyar, K. A. et al. The calcineurin-NFAT pathway controls activity-dependent circadian gene expression in slow skeletal muscle. Mol. Metab. 4, 823–833 (2015).
Atkinson, G., Coldwells, A., Reilly, T. & Waterhouse, J. A comparison of circadian rhythms in work performance between physically active and inactive subjects. Ergonomics 36, 273–281 (1993).
Coldwells, A., Atkinson, G. & Reilly, T. Sources of variation in back and leg dynamometry. Ergonomics 37, 79–86 (1994).
Wyse, J. P., Mercer, T. H. & Gleeson, N. P. Time-of-day dependence of isokinetic leg strength and associated interday variability. Br. J. Sports Med. 28, 167–170 (1994).
Gauthier, A., Davenne, D., Martin, A., Cometti, G. & Van Hoecke, J. Diurnal rhythm of the muscular performance of elbow flexors during isometric contractions. Chronobiol. Int. 13, 135–146 (1996).
Gauthier, A., Davenne, D., Gentil, C. & Van Hoecke, J. Circadian rhythm in the torque developed by elbow flexors during isometric contraction. Effect of sampling schedules. Chronobiol. Int. 14, 287–294 (1997).
Martin, A., Carpentier, A., Guissard, N., van Hoecke, J. & Duchateau, J. Effect of time of day on force variation in a human muscle. Muscle Nerve 22, 1380–1387 (1999).
Callard, D., Davenne, D., Gauthier, A., Lagarde, D. & Van Hoecke, J. Circadian rhythms in human muscular efficiency: continuous physical exercise versus continuous rest. A crossover study. Chronobiol. Int. 17, 693–704 (2000).
Souissi, N., Gauthier, A., Sesboue, B., Larue, J. & Davenne, D. Effects of regular training at the same time of day on diurnal fluctuations in muscular performance. J. Sports Sci. 20, 929–937 (2002).
Souissi, N., Sesboue, B., Gauthier, A., Larue, J. & Davenne, D. Effects of one night’s sleep deprivation on anaerobic performance the following day. Eur. J. Appl. Physiol. 89, 359–366 (2003).
Castaingts, V., Martin, A., Van Hoecke, J. & Perot, C. Neuromuscular efficiency of the triceps surae in induced and voluntary contractions: morning and evening evaluations. Chronobiol. Int. 21, 631–643 (2004).
Chtourou, H. et al. The effect of strength training at the same time of the day on the diurnal fluctuations of muscular anaerobic performances. J. Strength Cond. Res. 26, 217–225 (2012).
Souissi, N. et al. Effect of time of day and partial sleep deprivation on short-term, high-power output. Chronobiol. Int. 25, 1062–1076 (2008).
Taylor, K., Cronin, J. B., Gill, N., Chapman, D. W. & Sheppard, J. M. Warm-up affects diurnal variation in power output. Int. J. Sports Med. 32, 185–189 (2011).
Sedliak, M. et al. Morphological, molecular and hormonal adaptations to early morning versus afternoon resistance training. Chronobiol. Int. 35, 450–464 (2018).
Bernard, T., Giacomoni, M., Gavarry, O., Seymat, M. & Falgairette, G. Time-of-day effects in maximal anaerobic leg exercise. Eur. J. Appl. Physiol. Occup. Physiol. 77, 133–138 (1998).
Racinais, S., Perrey, S., Denis, R. & Bishop, D. Maximal power, but not fatigability, is greater during repeated sprints performed in the afternoon. Chronobiol. Int. 27, 855–864 (2010).
Lericollais, R., Gauthier, A., Bessot, N., Sesboue, B. & Davenne, D. Time-of-day effects on fatigue during a sustained anaerobic test in well-trained cyclists. Chronobiol. Int. 26, 1622–1635 (2009).
Souissi, N. et al. Diurnal variation in Wingate test performances: influence of active warm-up. Chronobiol. Int. 27, 640–652 (2010).
Fernandes, A. L. et al. Effect of time of day on performance, hormonal and metabolic response during a 1000-M cycling time trial. PLOS ONE 9, e109954 (2014).
Deschenes, M. R. et al. Chronobiological effects on exercise performance and selected physiological responses. Eur. J. Appl. Physiol. Occup. Physiol. 77, 249–256 (1998).
Waterhouse, J. et al. The circadian rhythm of core temperature: origin and some implications for exercise performance. Chronobiol. Int. 22, 207–225 (2005).
Boukelia, B., Fogarty, M. C., Davison, R. C. & Florida-James, G. D. Diurnal physiological and immunological responses to a 10-km run in highly trained athletes in an environmentally controlled condition of 6 degrees C. Eur. J. Appl. Physiol. 117, 1–6 (2017).
Dibner, C., Schibler, U. & Albrecht, U. The mammalian circadian timing system: organization and coordination of central and peripheral clocks. Annu. Rev. Physiol. 72, 517–549 (2010).
Edwards, B., Waterhouse, J., Reilly, T. & Atkinson, G. A comparison of the suitabilities of rectal, gut, and insulated axilla temperatures for measurement of the circadian rhythm of core temperature in field studies. Chronobiol. Int. 19, 579–597 (2002).
Gray, S. R., De Vito, G., Nimmo, M. A., Farina, D. & Ferguson, R. A. Skeletal muscle ATP turnover and muscle fiber conduction velocity are elevated at higher muscle temperatures during maximal power output development in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R376–R382 (2006).
Gray, S. R., Soderlund, K. & Ferguson, R. A. ATP and phosphocreatine utilization in single human muscle fibres during the development of maximal power output at elevated muscle temperatures. J. Sports Sci. 26, 701–707 (2008).
Gonzalez-Alonso, J. et al. Blood temperature and perfusion to exercising and non-exercising human limbs. Exp. Physiol. 100, 1118–1131 (2015).
Bass, J. & Lazar, M. A. Circadian time signatures of fitness and disease. Science 354, 994–999 (2016).
Buhr, E. D., Yoo, S. H. & Takahashi, J. S. Temperature as a universal resetting cue for mammalian circadian oscillators. Science 330, 379–385 (2010).
Racinais, S., Cocking, S. & Periard, J. D. Sports and environmental temperature: from warming-up to heating-up. Temperature (Austin) 4, 227–257 (2017).
Pullinger, S. A. et al. Diurnal variation in repeated sprint performance cannot be offset when rectal and muscle temperatures are at optimal levels (38.5 degrees C). Chronobiol. Int. 35, 1054–1065 (2018).
Deschenes, M. R. et al. Biorhythmic influences on functional capacity of human muscle and physiological responses. Med. Sci. Sports Exerc. 30, 1399–1407 (1998).
Morton, R. W. et al. Neither load nor systemic hormones determine resistance training-mediated hypertrophy or strength gains in resistance-trained young men. J. Appl. Physiol. 121, 129–138 (2016).
Atger, F., Mauvoisin, D., Weger, B., Gobet, C. & Gachon, F. Regulation of mammalian physiology by interconnected circadian and feeding rhythms. Front. Endocrinol. (Lausanne) 8, 42 (2017).
Mattson, M. P. et al. Meal frequency and timing in health and disease. Proc. Natl Acad. Sci. USA 111, 16647–16653 (2014).
Milton, K. Hunter-gatherer diets-a different perspective. Am. J. Clin. Nutr. 71, 665–667 (2000).
Crittenden, A. N. & Schnorr, S. L. Current views on hunter-gatherer nutrition and the evolution of the human diet. Am. J. Phys. Anthropol. 162 (Suppl. 63), 84–109 (2017).
Lamia, K. A., Storch, K. F. & Weitz, C. J. Physiological significance of a peripheral tissue circadian clock. Proc. Natl Acad. Sci. USA 105, 15172–15177 (2008).
Harfmann, B. D. et al. Muscle-specific loss of Bmal1 leads to disrupted tissue glucose metabolism and systemic glucose homeostasis. Skelet. Muscle 6, 12 (2016).
Moran-Ramos, S. et al. The suprachiasmatic nucleus drives day-night variations in postprandial triglyceride uptake into skeletal muscle and brown adipose tissue. Exp. Physiol. 102, 1584–1595 (2017).
Gabriel, B., Ratkevicius, A., Gray, P., Frenneaux, M. P. & Gray, S. R. High-intensity exercise attenuates postprandial lipaemia and markers of oxidative stress. Clin. Sci. 123, 313–321 (2012).
Gabriel, B. M. et al. The effect of high intensity interval exercise on postprandial triacylglycerol and leukocyte activation—monitored for 48h post exercise. PLOS ONE 8, e82669 (2013).
Sato, S., Parr, E. B., Devlin, B. L., Hawley, J. A. & Sassone-Corsi, P. Human metabolomics reveal daily variations under nutritional challenges specific to serum and skeletal muscle. Mol. Metab. 16, 1–11 (2018).
Jeukendrup, A. E. Periodized nutrition for athletes. Sports Med. 47, 51–63 (2017).
Marquet, L. A. et al. Enhanced endurance performance by periodization of carbohydrate intake: “Sleep Low” strategy. Med. Sci. Sports Exerc. 48, 663–672 (2016).
Lane, S. C. et al. Effects of sleeping with reduced carbohydrate availability on acute training responses. J. Appl. Physiol. 119, 643–655 (2015).
Hawley, J. A., Lundby, C., Cotter, J. D. & Burke, L. M. Maximizing cellular adaptation to endurance exercise in skeletal muscle. Cell Metab. 27, 962–976 (2018).
Kuusmaa, M. et al. Effects of morning versus evening combined strength and endurance training on physical performance, muscle hypertrophy, and serum hormone concentrations. Appl. Physiol. Nutr. Metab. 41, 1285–1294 (2016).
Matsumoto, C. S. et al. PI3K-PTEN dysregulation leads to mTOR-driven upregulation of the core clock gene BMAL1 in normal and malignant epithelial cells. Oncotarget 7, 42393–42407 (2016).
Ramanathan, C. et al. mTOR signaling regulates central and peripheral circadian clock function. PLOS Genet. 14, e1007369 (2018).
Liu, D. et al. mTOR signaling in VIP neurons regulates circadian clock synchrony and olfaction. Proc. Natl Acad. Sci. USA 115, E3296–E3304 (2018).
Reeds, P. J., Palmer, R. M., Hay, S. M. & McMillan, D. N. Protein synthesis in skeletal muscle measured at different times during a 24 hour period. Biosci. Rep. 6, 209–213 (1986).
Chang, S. W., Yoshihara, T., Machida, S. & Naito, H. Circadian rhythm of intracellular protein synthesis signaling in rat cardiac and skeletal muscles. Biochem. Biophys. Rep. 9, 153–158 (2017).
Gangwisch, J. E. et al. Sleep duration as a risk factor for diabetes incidence in a large US sample. Sleep 30, 1667–1673 (2007).
Ogilvie, R. P. & Patel, S. R. The epidemiology of sleep and obesity. Sleep Health 3, 383–388 (2017).
Zwighaft, Z. et al. Circadian clock control by polyamine levels through a mechanism that declines with age. Cell Metab. 22, 874–885 (2015).
Myllymaki, T. et al. Effects of vigorous late-night exercise on sleep quality and cardiac autonomic activity. J. Sleep Res. 20, 146–153 (2011).
Banno, M. et al. Exercise can improve sleep quality: a systematic review and meta-analysis. PeerJ 6, e5172 (2018).
Youngstedt, S. D., O’Connor, P. J. & Dishman, R. K. The effects of acute exercise on sleep: a quantitative synthesis. Sleep 20, 203–214 (1997).
Murray, K. et al. The relations between sleep, time of physical activity, and time outdoors among adult women. PLOS ONE 12, e0182013 (2017).
Figueiro, M. G. et al. The impact of daytime light exposures on sleep and mood in office workers. Sleep Health 3, 204–215 (2017).
Harrington, J. M. Health effects of shift work and extended hours of work. Occup. Environ. Med. 58, 68–72 (2001).
Buxton, O. M. et al. Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes 59, 2126–2133 (2010).
Eastman, C. I., Hoese, E. K., Youngstedt, S. D. & Liu, L. Phase-shifting human circadian rhythms with exercise during the night shift. Physiol. Behav. 58, 1287–1291 (1995).
Youngstedt, S. D. et al. Circadian phase-shifting effects of bright light, exercise, and bright light + exercise. J. Circadian Rhythms 14, 2 (2016).
Dempsey, P. C. et al. Interrupting prolonged sitting in type 2 diabetes: nocturnal persistence of improved glycaemic control. Diabetologia 60, 499–507 (2017).
Gabel, K. et al. Effects of 8-hour time restricted feeding on body weight and metabolic disease risk factors in obese adults: a pilot study. Nutr. Healthy. Aging 4, 345–353 (2018).
Friborg, O., Bjorvatn, B., Amponsah, B. & Pallesen, S. Associations between seasonal variations in day length (photoperiod), sleep timing, sleep quality and mood: a comparison between Ghana (5 degrees) and Norway (69 degrees). J. Sleep Res. 21, 176–184 (2012).
Renstrom, F. et al. Season-dependent associations of circadian rhythm-regulating loci (CRY1, CRY2 and MTNR1B) and glucose homeostasis: the GLACIER Study. Diabetologia 58, 997–1005 (2015).
Atkinson, G. & Drust, B. Seasonal rhythms and exercise. Clin. Sports Med. 24, e25–e34 (2005).
Honma, K., Honma, S., Kohsaka, M. & Fukuda, N. Seasonal variation in the human circadian rhythm: dissociation between sleep and temperature rhythm. Am. J. Physiol. 262, R885–R891 (1992).
Chen, L. & Yang, G. PPARs integrate the mammalian clock and energy metabolism. PPAR Res. 2014, 653017 (2014).
Acknowledgements
The authors are supported by grants from the Novo Nordisk Foundation (NNF14OC0011493, NNF14OC0009941 and NNF18CC0034900), the Wenner-Gren Foundation, the Swedish Research Council (2015–00165), the European Research Council (233285) and the Strategic Research Programme in Diabetes at Karolinska Institutet (2009–1068). The authors are grateful to B. Atkins for his contribution to figure design.
Author information
Authors and Affiliations
Contributions
B.M.G. and J.R.Z. contributed equally to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.`
Glossary
- Core clock
-
A set of protein-coding genes (active in nearly all mammalian cells) that oscillate in expression and activity in a circadian manner.
- Training
-
Repeated bouts of exercise resulting in physiological adaptations.
- Diurnal
-
A diurnal cycle is any pattern that recurs every 24 hours, not necessarily biological or intrinsic.
- Zeitgeber
-
A rhythmically occurring natural phenomenon that acts as a cue in the regulation of the body’s circadian rhythms.
- Oxygen consumption rate
-
The amount of oxygen consumed by metabolic processes in tissues, cells or organelles. When applied to measuring mitochondria, different metabolic states (states 1–5) of the mitochondria are used.
- Chronotype
-
The interindividual differences in the circadian phase of activity patterns and sleep–wake cycles.
- Maximal power output
-
Maximal intensity of exercise or skeletal muscle contraction measured by power output (Watts).
- Maximal sprint
-
A short burst of intense exercise after which the individual is momentarily unable to continue owing to fatigue.
- Cycle ergometer
-
A fixed cycling machine often used in fitness testing to estimate exercise intensity.
- Periodized nutrition
-
The strategic combined use of exercise training and nutrition, or nutrition alone, with the overall aim to improve the physiological response to exercise training.
- Voluntary muscle force
-
Skeletal muscle contraction force produced as a result of endogenous activation of motor neurons.
- Acute exercise
-
A single exercise bout, rather than exercise training.
- Sleep hygiene
-
Habits and practices that are conducive to sleeping well on a regular basis.
Rights and permissions
About this article
Cite this article
Gabriel, B.M., Zierath, J.R. Circadian rhythms and exercise — re-setting the clock in metabolic disease. Nat Rev Endocrinol 15, 197–206 (2019). https://doi.org/10.1038/s41574-018-0150-x
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41574-018-0150-x
This article is cited by
-
The relationship between nighttime exercise and problematic smartphone use before sleep and associated health issues: a cross-sectional study
BMC Public Health (2024)
-
From bedside to recovery: exercise therapy for prevention of post-intensive care syndrome
Journal of Intensive Care (2024)
-
Move the night way: how can physical activity facilitate adaptation to shift work?
Communications Biology (2024)
-
Best Time of Day for Strength and Endurance Training to Improve Health and Performance? A Systematic Review with Meta-analysis
Sports Medicine - Open (2023)
-
The relationship between the intake of branched-chain and aromatic amino acids and individuals' sleep quality based on body mass index, gender, and age
Journal of Health, Population and Nutrition (2023)