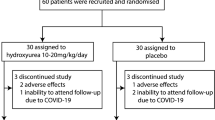
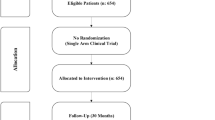
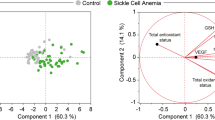
Abstract
For over 100 years, clinicians and scientists have been unravelling the consequences of the A to T substitution in the β-globin gene that produces haemoglobin S, which leads to the systemic manifestations of sickle cell disease (SCD), including vaso-occlusion, anaemia, haemolysis, organ injury and pain. However, despite growing understanding of the mechanisms of haemoglobin S polymerization and its effects on red blood cells, only two therapies for SCD — hydroxyurea and l-glutamine — are approved by the US Food and Drug Administration. Moreover, these treatment options do not fully address the manifestations of SCD, which arise from a complex network of interdependent pathophysiological processes. In this article, we review efforts to develop new drugs targeting these processes, including agents that reactivate fetal haemoglobin, anti-sickling agents, anti-adhesion agents, modulators of ischaemia–reperfusion and oxidative stress, agents that counteract free haemoglobin and haem, anti-inflammatory agents, anti-thrombotic agents and anti-platelet agents. We also discuss gene therapy, which holds promise of a cure, although its widespread application is currently limited by technical challenges and the expense of treatment. We thus propose that developing systems-oriented multi-agent strategies on the basis of SCD pathophysiology is needed to improve the quality of life and survival of people with SCD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bunn, H. F. Pathogenesis and treatment of sickle cell disease. N. Engl. J. Med. 337, 762–769 (1997).
Rees, D. C., Williams, T. N. & Gladwin, M. T. Sickle-cell disease. Lancet 376, 2018–2031 (2010).
Platt, O. S. et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. N. Engl. J. Med. 330, 1639–1644 (1994).
Kauf, T. L., Coates, T. D., Huazhi, L., Mody-Patel, N. & Hartzema, A. G. The cost of health care for children and adults with sickle cell disease. Am. J. Hematol. 84, 323–327 (2009).
Elmariah, H. et al. Factors associated with survival in a contemporary adult sickle cell disease cohort. Am. J. Hematol. 89, 530–535 (2014).
Arnold, S. D., Bhatia, M., Horan, J. & Krishnamurti, L. Haematopoietic stem cell transplantation for sickle cell disease — current practice and new approaches. Br. J. Haematol. 174, 515–525 (2016).
Fitzhugh, C. D., Abraham, A. A., Tisdale, J. F. & Hsieh, M. M. Hematopoietic stem cell transplantation for patients with sickle cell disease: progress and future directions. Hematol. Oncol. Clin. North Amer. 28, 1171–1185 (2014).
Hulbert, M. L. & Shenoy, S. Hematopoietic stem cell transplantation for sickle cell disease: progress and challenges. Pediatr. Blood Cancer 65, e27263 (2018).
Kassim, A. A. & Sharma, D. Hematopoietic stem cell transplantation for sickle cell disease: the changing landscape. Hematol. Oncol. Stem Cell Ther. 10, 259–266 (2017).
Oringanje, C., Nemecek, E. & Oniyangi, O. Hematopoietic stem cell transplantation for people with sickle cell disease. Cochrane Database Syst. Rev. 19, CD007001 (2016).
Walters, M. C. Update of hematopoietic cell transplantation for sickle cell disease. Curr. Opin. Hematol. 22, 227–233 (2015).
Eaton, W. A. & Bunn, H. F. Treating sickle cell disease by targeting HbS polymerization. Blood 129, 2719–2726 (2017).
Hebbel, R. P. Ischemia-reperfusion injury in sickle cell anemia: relationship to acute chest syndrome, endothelial dysfunction, arterial vasculopathy, and inflammatory pain. Hematol. Oncol. Clin. North Amer. 28, 181–198 (2014).
Hebbel, R. P., Vercellotti, G. & Nath, K. A. A systems biology consideration of the vasculopathy of sickle cell anemia: the need for multi-modality chemo-prophylaxsis. Cardiovascular Hematol. Disord. Drug Targets 9, 271–292 (2009).
Hebbel, R. P. Reconstructing sickle cell disease: a data-based analysis of the “hyperhemolysis paradigm” for pulmonary hypertension from the perspective of evidence-based medicine. Am. J. Hematol. 86, 123–154 (2011).
Hebbel, R. P., Osarogiagbon, R. & Kaul, D. The endothelial biology of sickle cell disease: inflammation and a chronic vasculopathy. Microcirculation 11, 129–151 (2004).
Zennadi, R. et al. Epinephrine acts through erythroid signaling pathways to activate sickle cell adhesion to endothelium via LW-αvβ3 interactions. Blood 104, 3774–3781 (2004).
Zennadi, R. et al. Erythrocyte plasma membrane-bound ERK1/2 activation promotes ICAM-4-mediated sickle red cell adhesion to endothelium. Blood 119, 1217–1227 (2012).
George, A. et al. Erythrocyte NADPH oxidase activity modulated by Rac GTPases, PKC, and plasma cytokines contributes to oxidative stress in sickle cell disease. Blood 121, 2099–2107 (2013).
Wun, T. et al. Platelet-erythrocyte adhesion in sickle cell disease. J. Investig. Med. 47, 121–127 (1999).
Wun, T. et al. Platelet activation and platelet-erythrocyte aggregates in patients with sickle cell anemia. J. Lab. Clin. Med. 129, 507–516 (1997).
Frenette, P. S. Sickle cell vaso-occlusion: multistep and multicellular paradigm. Curr. Opin. Hematol. 9, 101–106 (2002).
Frenette, P. S. Sickle cell vasoocclusion: heterotypic, multicellular aggregations driven by leukocyte adhesion. Microcirculation 11, 167–177 (2004).
Turhan, A., Weiss, L. A., Mohandas, N., Coller, B. S. & Frenette, P. S. Primary role for adherent leukocytes in sickle cell vascular occlusion: a new paradigm. Proc. Natl Acad. Sci. USA 99, 3047–3051 (2002).
Brittain, J. E., Knoll, C. M., Ataga, K. I., Orringer, E. P. & Parise, L. V. Fibronectin bridges monocytes and reticulocytes via integrin α4β1. Br. J. Haematol. 141, 872–881 (2008).
Hidalgo, A. et al. Heterotypic interactions enabled by polarized neutrophil microdomains mediate thromboinflammatory injury. Nat. Med. 15, 384–391 (2009).
Shiu, Y. T., Udden, M. M. & McIntire, L. V. Perfusion with sickle erythrocytes up-regulates ICAM-1 and VCAM-1 gene expression in cultured human endothelial cells. Blood 95, 3232–3241 (2000).
Li, H. & Lykotrafitis, G. Erythrocyte membrane model with explicit description of the lipid bilayer and the spectrin network. Biophys. J. 107, 642–653 (2014).
de Jong, K., Larkin, S. K., Styles, L. A., Bookchin, R. M. & Kuypers, F. A. Characterization of the phosphatidylserine-exposing subpopulation of sickle cells. Blood 98, (860–867 (2001).
Joiner, C. H., Jiang, M. & Franco, R. S. Deoxygenation-induced cation fluxes in sickle cells. IV. Modulation by external calcium. Am. J. Physiol. 269, C403–409 (1995).
Mankelow, T. J. et al. Autophagic vesicles on mature human reticulocytes explain phosphatidylserine-positive red cells in sickle cell disease. Blood 126, 1831–1834 (2015).
Kuypers, F. A. & de Jong, K. The role of phosphatidylserine in recognition and removal of erythrocytes. Cell. Mol. Biol. 50, 147–158 (2004).
Villagra, J. et al. Platelet activation in patients with sickle disease, hemolysis-associated pulmonary hypertension, and nitric oxide scavenging by cell-free hemoglobin. Blood 110, 2166–2172 (2007).
Brittain, H. A., Eckman, J. R., Swerlick, R. A., Howard, R. J. & Wick, T. M. Thrombospondin from activated platelets promotes sickle erythrocyte adherence to human microvascular endothelium under physiologic flow: a potential role for platelet activation in sickle cell vaso-occlusion. Blood 81, 2137–2143 (1993).
Setty, B. N., Kulkarni, S., Rao, A. K. & Stuart, M. J. Fetal hemoglobin in sickle cell disease: relationship to erythrocyte phosphatidylserine exposure and coagulation activation. Blood 96, 1119–1124 (2000).
Arumugam, P. I. et al. Genetic diminution of circulating prothrombin ameliorates multiorgan pathologies in sickle cell disease mice. Blood 126, 1844–1855 (2015).
Lee, S. P., Ataga, K. I., Orringer, E. P., Phillips, D. R. & Parise, L. V. Biologically active CD40 ligand is elevated in sickle cell anemia: potential role for platelet-mediated inflammation. Arterioscler. Thromb. Vasc. Biol. 26, 1626–1631 (2006).
Lee, S. P. et al. Phase I study of eptifibatide in patients with sickle cell anaemia. Br. J. Haematol. 139, 612–620 (2007).
Polanowska-Grabowska, R. et al. P-Selectin-mediated platelet-neutrophil aggregate formation activates neutrophils in mouse and human sickle cell disease. Arterioscler. Thromb. Vasc. Biol. 30, 2392–2399 (2010).
Bennewitz, M. F. et al. Lung vaso-occlusion in sickle cell disease mediated by arteriolar neutrophil-platelet microemboli. JCI Insight 2, e89761 (2017).
Dominical, V. M. et al. Prominent role of platelets in the formation of circulating neutrophil-red cell heterocellular aggregates in sickle cell anemia. Haematologica 99, e214–217 (2014).
Zennadi, R., Chien, A., Xu, K., Batchvarova, M. & Telen, M. J. Sickle red cells induce adhesion of lymphocytes and monocytes to endothelium. Blood 112, 3474–3483 (2008).
Belcher, J. D. et al. Heme oxygenase-1 is a modulator of inflammation and vaso-occlusion in transgenic sickle mice. J. Clin. Invest. 116, 808–816 (2006).
Ghosh, S. et al. Extracellular hemin crisis triggers acute chest syndrome in sickle mice. J. Clin. Invest. 123, 4809–4820 (2013).
Belcher, J. D. et al. Heme triggers TLR4 signaling leading to endothelial cell activation and vaso-occlusion in murine sickle cell disease. Blood 123, 377–390 (2014).
Bean, C. J. et al. Heme oxygenase-1 gene promoter polymorphism is associated with reduced incidence of acute chest syndrome among children with sickle cell disease. Blood 120, 3822–3828 (2012).
Belcher, J. D., Nath, K. A. & Vercellotti, G. M. Vasculotoxic and proinflammatory effects of plasma heme: cell signaling and cytoprotective responses. ISRN Oxidative Med. 2013, 831596 (2013).
Nagel, R. L. et al. Structural bases of the inhibitory effects of hemoglobin F and hemoglobin A2 on the polymerization of hemoglobin S. Proc. Natl Acad. Sci. USA 76, 670–672 (1979).
Akinsheye, I. et al. Fetal hemoglobin in sickle cell anemia. Blood 118, 19–27 (2011).
Estepp, J. H. et al. A clinically meaningful fetal hemoglobin threshold for children with sickle cell anemia during hydroxyurea therapy. Am. J. Hematol. 92, 1333–1339 (2017).
Powars, D. R., Weiss, J. N., Chan, L. S. & Schroeder, W. A. Is there a threshold level of fetal hemoglobin that ameliorates morbidity in sickle cell anemia? Blood 63, 921–926 (1984).
Charache, S. et al. Treatment of sickle cell anemia with 5-azacytidine results in increased fetal hemoglobin production and is associated with nonrandom hypomethylation of DNA around the γ-δ-β-globin gene complex. Proc. Natl Acad. Sci. USA 80, 4842–4846 (1983).
DeSimone, J., Heller, P., Hall, L. & Zwiers, D. 5-Azacytidine stimulates fetal hemoglobin synthesis in anemic baboons. Proc. Natl Acad. Sci. USA 79, 4428–4431 (1982).
Ley, T. J. et al. 5-Azacytidine selectively increases γ-globin synthesis in a patient with β+ thalassemia. N. Engl. J. Med. 307, 1469–1475 (1982).
McGann, P. T. & Ware, R. E. Hydroxyurea therapy for sickle cell anemia. Expert Opin. Drug Saf. 14, 1749–1758 (2015).
Molokie, R. et al. Oral tetrahydrouridine and decitabine for non-cytotoxic epigenetic gene regulation in sickle cell disease: a randomized phase 1 study. PLOS Med. 14, e1002382 (2017).
Atweh, G. F. et al. Sustained induction of fetal hemoglobin by pulse butyrate therapy in sickle cell disease. Blood 93, 1790–1797 (1999).
Kutlar, A. et al. A dose-escalation phase IIa study of 2,2-dimethylbutyrate (HQK-1001), an oral fetal globin inducer, in sickle cell disease. Am. J. Hematol. 88, E255–E260 (2013).
Reid, M. E. et al. A double-blind, placebo-controlled phase II study of the efficacy and safety of 2,2-dimethylbutyrate (HQK-1001), an oral fetal globin inducer, in sickle cell disease. Am. J. Hematol. 89, 709–713 (2014).
Bradner, J. E. et al. Chemical genetic strategy identifies histone deacetylase 1 (HDAC1) and HDAC2 as therapeutic targets in sickle cell disease. Proc. Natl Acad. Sci. USA 107, 12617–12622 (2010).
Shi, L., Cui, S., Engel, J. D. & Tanabe, O. Lysine-specific demethylase 1 is a therapeutic target for fetal hemoglobin induction. Nat. Med. 19, 291–294 (2013).
Rivers, A. et al. RN-1, a potent and selective lysine-specific demethylase 1 inhibitor, increases γ-globin expression, F reticulocytes, and F cells in a sickle cell disease mouse model. Exp. Hematol. 43, 546–553 (2015).
Cui, S. et al. The LSD1 inhibitor RN-1 induces fetal hemoglobin synthesis and reduces disease pathology in sickle cell mice. Blood 126, 386–396 (2015).
Dai, Y., Chen, T., Ijaz, H., Cho, E. H. & Steinberg, M. H. SIRT1 activates the expression of fetal hemoglobin genes. Am. J. Hematol. 92, 1177–1186 (2017).
Lanaro, C. et al. A thalidomide-hydroxyurea hybrid increases HbF production in sickle cell mice and reduces the release of proinflammatory cytokines in cultured monocytes. Exp. Hematol. 58, 35–38 (2018).
Theodorou, A. et al. The investigation of resveratrol and analogs as potential inducers of fetal hemoglobin. Blood Cells Mol. Dis. 58, 6–12 (2016).
Krishnamoorthy, S. et al. Dimethyl fumarate increases fetal hemoglobin, provides heme detoxification, and corrects anemia in sickle cell disease. JCI Insight 2, 96409 (2017).
Gomperts, E. et al. The role of carbon monoxide and heme oxygenase in the prevention of sickle cell disease vaso-occlusive crises. Am. J. Hematol. 92, 569–582 (2017).
Belcher, J. D. et al. MP4CO, a pegylated hemoglobin saturated with carbon monoxide, is a modulator of HO-1, inflammation, and vaso-occlusion in transgenic sickle mice. Blood 122, 2757–2764 (2013).
Howard, J. et al. Safety and tolerability of MP4CO: A dose escalation study In stable patients with sickle cell disease. Blood 122, 2205 (2013).
Misra, H., Lickliter, J., Kazo, F. & Abuchowski, A. PEGylated carboxyhemoglobin bovine (SANGUINATE): results of a phase I clinical trial. Artif. Organs 38, 702–707 (2014).
Abuchowski, A. PEGylated bovine carboxyhemoglobin (SANGUINATE): results of clinical safety testing and use in patients. Adv. Exp. Med. Biol. 876, 461–467 (2016).
Swift, R. et al. SCD-101: a new anti-sickling drug reduces pain and fatigue and improves red blood cell shape in peripheral blood of patients with sickle cell disease. Blood 128, 121–121 (2016).
Iyamu, E. W., Turner, E. A. & Asakura, T. In vitro effects of NIPRISAN (Nix-0699): a naturally occurring, potent antisickling agent. Br. J. Haematol. 118, 337–343 (2002).
Wambebe, C. et al. Double-blind, placebo-controlled, randomised cross-over clinical trial of NIPRISAN in patients with sickle cell disorder. Phytomedicine 8, 252–261 (2001).
Lehrer-Graiwer, J. et al. Long-term dosing in sickle cell disease subjects with GBT440, a novel HbS polymerization inhibitor. Blood 128, 2488 (2016).
Hebbel, R. P. & Hedlund, B. E. Sickle hemoglobin oxygen affinity-shifting strategies have unequal cerebrovascular risks. Am. J. Hematol. 93, 321–325 (2018).
Morris, C. R. et al. Erythrocyte glutamine depletion, altered redox environment, and pulmonary hypertension in sickle cell disease. Blood 111, 402–410 (2008).
Whillier, S., Garcia, B., Chapman, B. E., Kuchel, P. W. & Raftos, J. E. Glutamine and α-ketoglutarate as glutamate sources for glutathione synthesis in human erythrocytes. FEBS J. 278, 3152–3163 (2011).
Niihara, Y. et al. L-Glutamine therapy reduces endothelial adhesion of sickle red blood cells to human umbilical vein endothelial cells. BMC Blood Disord. 5, 4 (2005).
Niihara, Y. et al. A Phase 3 study of L-glutamine therapy for sickle cell anemia and sickle ß-thalassemia. Blood 124, 86–86 (2014).
Wilmore, D. W. Food and Drug Administration approval of glutamine for sickle cell disease: success and precautions in glutamine research. JPEN J. Parenter Enteral Nutr. 41, 912–917 (2017).
Wood, K. C., Hebbel, R. P. & Granger, D. N. Endothelial cell NADPH oxidase mediates the cerebral microvascular dysfunction in sickle cell transgenic mice. FASEB J. 19, 989–991 (2005).
Kaul, D. K. et al. Anti-inflammatory therapy ameliorates leukocyte adhesion and microvascular flow abnormalities in transgenic sickle mice. American journal of physiology. Heart Circulatory Physiol. 287, H293–301 (2004).
Nur, E. et al. N-Acetylcysteine reduces oxidative stress in sickle cell patients. Ann. Hematol. 91, 1097–1105 (2012).
Ozpolat, H. T. et al. A pilot study of high-dose N-acetylcysteine infusion in patients with sickle cell disease. Blood 128, 1299–1299 (2016).
Sins, J. W. R. et al. N-acetylcysteine in patients with sickle cell disease: a randomized controlled trial. Blood 128, 123–123 (2016).
Wallace, K. L. & Linden, J. Adenosine A2A receptors induced on iNKT and NK cells reduce pulmonary inflammation and injury in mice with sickle cell disease. Blood 116, 5010–5020 (2010).
Reilly, E. C., Wands, J. R. & Brossay, L. Cytokine dependent and independent iNKT cell activation. Cytokine 51, 227–231 (2010).
Field, J. J. et al. Sickle cell vaso-occlusion causes activation of iNKT cells that is decreased by the adenosine A2A receptor agonist regadenoson. Blood 121, 3329–3334 (2013).
Field, J. J. et al. Randomized phase 2 trial of regadenoson for treatment of acute vaso-occlusive crises in sickle cell disease. Blood Adv. 1, 1645–1649 (2017).
Solovey, A. et al. A monocyte-TNF-endothelial activation axis in sickle transgenic mice: therapeutic benefit from TNF blockade. Am. J. Hemat. 12292, 1119–1130 (2017).
Adelowo, O. & Edunjobi, A. S. Juvenile idiopathic arthritis coexisting with sickle cell disease: two case reports. BMJ Case Rep. 2011, bcr1020114889 (2011).
Gorsuch, W. B., Chrysanthou, E., Schwaeble, W. J. & Stahl, G. L. The complement system in ischemia-reperfusion injuries. Immunobiology 217, 1026–1033 (2012).
Wang, R. H., Phillips, G. Jr., Medof, M. E. & Mold, C. Activation of the alternative complement pathway by exposure of phosphatidylethanolamine and phosphatidylserine on erythrocytes from sickle cell disease patients. J. Clin. Invest. 92, 1326–1335 (1993).
Zwaal, R. F. & Schroit, A. J. Pathophysiologic implications of membrane phospholipid asymmetry in blood cells. Blood 89, 1121–1132 (1997).
Krisinger, M. J. et al. Thrombin generates previously unidentified C5 products that support the terminal complement activation pathway. Blood 120, 1717–1725 (2012).
Schaid, T. R. et al. Complement activation in a murine model of sickle cell disease: inhibition of vaso-occlusion by blocking C5 activation. Blood 128, 158 (2016).
Hoppe, C. et al. A pilot study of the short-term use of simvastatin in sickle cell disease: effects on markers of vascular dysfunction. Br. J. Haematol. 153, 655–663 (2011).
Hoppe, C. et al. Simvastatin reduces vaso-occlusive pain in sickle cell anaemia: a pilot efficacy trial. Br. J. Haematol. 177, 620–629 (2017).
Rybicki, A. C. & Benjamin, L. J. Increased levels of endothelin-1 in plasma of sickle cell anemia patients. Blood 92, 2594–2596 (1998).
Sabaa, N. Endothelin receptor antagonism prevents hypoxia-induced mortality and morbidity in a mouse model of sickle-cell disease. J. Clin. Invest. 118, 1924–1933 (2008).
Prado, G. N., Romero, J. R. & Rivera, A. Endothelin-1 receptor antagonists regulate cell surface-associated protein disulfide isomerase in sickle cell disease. FASEB J. 27, 4619–4629 (2013).
Koehl, B. et al. The endothelin B receptor plays a crucial role for the adhesion of neutrophils to the endothelium in sickle cell disease. Haematologica 1028, 1161–1172 (2017).
Elisa, T. et al. Endothelin receptors expressed by immune cells are involved in modulation of inflammation and in fibrosis: relevance to the pathogenesis of systemic sclerosis. J. Immunol. Res. 2015, 147616 (2015).
Chang, J., Shi, P. A., Chiang, E. Y. & Frenette, P. S. Intravenous immunoglobulins reverse acute vaso-occlusive crises in sickle cell mice through rapid inhibition of neutrophil adhesion. Blood 111, 915–923 (2008).
Jang, J. E., Hidalgo, A. & Frenette, P. S. Intravenous immunoglobulins modulate neutrophil activation and vascular injury through FcγRIII and SHP-1. Circul. Res. 110, 1057–1066 (2012).
Manwani, D. et al. Single-dose intravenous γ-globulin can stabilize neutrophil Mac-1 activation in sickle cell pain crisis. Am. J. Hematol. 90, 381–385 (2015).
Almeida, C. B. et al. Hydroxyurea and a cGMP-amplifying agent have immediate benefits on acute vaso-occlusive events in sickle cell disease mice. Blood 120, 2879–2888 (2012).
Barbosa, M. C. et al. The effect of a selective inhibitor of phosphodiesterase-9 on oxidative stress, inflammation and cytotoxicity in neutrophils from patients with sickle cell anaemia. Bas. Clin. Pharmacol. Toxicol. 118, 271–278 (2016).
McArthur, J. C. et al. Novel highly potent and selective PDE9 inhibitor for the treament of sickle cell disease. Blood 128, 268 (2016).
Morris, C. R. Alterations of the arginine metabolome in sickle cell disease: a growing rationale for arginine therapy. Hematol. Oncol. Clin. North Am. 28, 301–321 (2014).
Morris, C. R. et al. A randomized, placebo-controlled trial of arginine therapy for the treatment of children with sickle cell disease hospitalized with vaso-occlusive pain episodes. Haematologica 98, 1375–1382 (2013).
Almeida, C. B. et al. Acute hemolytic vascular inflammatory processes are prevented by nitric oxide replacement or a single dose of hydroxyurea. Blood 126, 711–720 (2015).
King, S. B. Nitric oxide production from hydroxyurea. Free Radic. Biol. Med. 37, 737–744 (2004).
Belcher, J. D. et al. Haptoglobin and hemopexin inhibit vaso-occlusion and inflammation in murine sickle cell disease: role of heme oxygenase-1 induction. PLOS ONE 13, e0196455 (2018).
Vercellotti, G. M. et al. Hepatic overexpression of hemopexin inhibits inflammation and vascular stasis in murine models of sickle cell disease. Mol. Med. 22, 437–451 (2016).
Belcher, J. D. et al. Haptoglobin and hemopexin infusion efficiently activates the Nrf2/HO-1 axis and inhibits inflammation and vaso-occlusion in murine sickle cell disease. Blood 128, 2477–2477 (2016).
Sparkenbaugh, E. & Pawlinski, R. Interplay between coagulation and vascular inflammation in sickle cell disease. Br. J. Haematol. 162, 3–14 (2013).
Zhang, D., Xu, C., Manwani, D. & Frenette, P. S. Neutrophils, platelets, and inflammatory pathways at the nexus of sickle cell disease pathophysiology. Blood 127, 801–809 (2016).
Setty, B. N. & Stuart, M. J. Vascular cell adhesion molecule-1 is involved in mediating hypoxia-induced sickle red blood cell adherence to endothelium: potential role in sickle cell disease. Blood 88, 2311–2320 (1996).
Lim, M. Y., Ataga, K. I. & Key, N. S. Hemostatic abnormalities in sickle cell disease. Curr. Opin. Hematol. 20, 472–477 (2013).
Noubouossie, D., Key, N. S. & Ataga, K. I. Coagulation abnormalities of sickle cell disease: relationship with clinical outcomes and the effect of disease modifying therapies. Blood Rev. 30, 245–256 (2016).
Whelihan, M. F. et al. Thrombin generation and cell-dependent hypercoagulability in sickle cell disease. J. Thromb. Haemost. 14, 1941–1952 (2016).
Gordon, E. M. et al. Reduction of contact factors in sickle cell disease. J. Pediatr. 106, 427–430 (1985).
LourenCo, D., Sampaio, M. U., Kerbauy, J. & Sampaio, C. A. Estimation of plasma kallikrein in sickle-cell anemia, and its relation to the coagulation and fibrinolytic systems. Adv. Exp. Med. Biol. 247B, 553–557 (1989).
Miller, R. L., Verma, P. S. & Adams, R. G. Studies of the kallikrein-kinin system in patients with sickle cell anemia. J. Natl Med. Associ. 75, 551–556 (1983).
Solovey, A., Gui, L., Key, N. S. & Hebbel, R. P. Tissue factor expression by endothelial cells in sickle cell anemia. J. Clin. Invest. 101, 1899–1904 (1998).
Solovey, A. et al. Endothelial cell expression of tissue factor in sickle mice is augmented by hypoxia/reoxygenation and inhibited by lovastatin. Blood 104, 840–846 (2004).
Salvaggio, J. E., Arnold, C. A. & Banov, C. H. Long-term anti-coagulation in sickle-cell disease. A clinical study. N. Engl. J. Med. 269, 182–186 (1963).
Adelson, H. T. Long-term dicumarol administration as a therapeutic trial in sicklemia; report of a case. N. Engl. J. Med. 256, 353–354 (1957).
Chaplin, H. Jr. et al. Preliminary trial of minidose heparin prophylaxis for painful sickle cell crises. East Afr. Med. J. 66, 574–584 (1989).
Qari, M. H. et al. Reduction of painful vaso-occlusive crisis of sickle cell anaemia by tinzaparin in a double-blind randomized trial. Thromb. Haemostasis 98, 392–396 (2007).
Wun, T. et al. Platelet activation in patients with sickle cell disease. Br. J. Haematol. 100, 741–749 (1998).
Kataoka, H. et al. Protease-activated receptors 1 and 4 mediate thrombin signaling in endothelial cells. Blood 102, 3224–3231 (2003).
Coughlin, S. R. Protease-activated receptors in hemostasis, thrombosis and vascular biology. J. Thromb. Haemost. 3, 1800–1814 (2005).
Loscalzo, J. Nitric oxide insufficiency, platelet activation, and arterial thrombosis. Circ. Res. 88, 756–762 (2001).
Kim, K. et al. Neutrophil Akt2 plays a critical role in heterotypic neutrophil-platelet interactions during vascular inflammation. Blood 122, 321–321 (2013).
Li, J. et al. Neutrophil AKT2 regulates heterotypic cell-cell interactions during vascular inflammation. J. Clin. Invest. 124, 1483–1496 (2014).
Barazia, A., Li, J., Kim, K., Shabrani, N. & Cho, J. Hydroxyurea with AKT2 inhibition decreases vaso-occlusive events in sickle cell disease mice. Blood 126, 2511–2517 (2015).
Hoppe, C. C. et al. Design of the DOVE (Determining Effects of Platelet Inhibition on Vaso-Occlusive Events) trial: a global Phase 3 double-blind, randomized, placebo-controlled, multicenter study of the efficacy and safety of prasugrel in pediatric patients with sickle cell anemia utilizing a dose titration strategy. Pediatr. Blood Cancer 63, 299–305 (2016).
Heeney, M. M. et al. A multinational trial of prasugrel for sickle cell vaso-occlusive events. N. Engl. J. Med. 374, 625–635 (2016).
Styles, L. et al. Prasugrel in children with sickle cell disease: pharmacokinetic and pharmacodynamic data from an open-label, adaptive-design, dose-ranging study. J. Pediatr. Hematol. Oncol. 37, 1–9 (2015).
Jakubowski, J. A. et al. A phase 1 study of prasugrel in patients with sickle cell disease: effects on biomarkers of platelet activation and coagulation. Thromb. Res. 133, 190–195 (2014).
Wun, T. et al. A double-blind, randomized, multicenter phase 2 study of prasugrel versus placebo in adult patients with sickle cell disease. J. Hematol. Oncol. 6, 17 (2013).
Chang, J. et al. GMI-1070, a novel pan-selectin antagonist, reverses acute vascular occlusions in sickle cell mice. Blood 116, 1779–1786 (2010).
Telen, M. J. et al. Randomized phase 2 study of GMI-1070 in SCD: reduction in time to resolution of vaso-occlusive events and decreased opioid use. Blood 125, 2656–2664 (2015).
Ataga, K. I. et al. Crizanlizumab for the prevention of pain crises in sickle cell disease. N. Engl. J. Med. 376, 429–439 (2017).
Telen, M. J. et al. Sevuparin binds to multiple adhesive ligands and reduces sickle red blood cell-induced vaso-occlusion. Br. J. Haematol. 175, 935–948 (2016).
Leitgeb, A. M. et al. Low anticoagulant heparin disrupts Plasmodium falciparum rosettes in fresh clinical isolates. Am. J. Trop. Med. Hyg. 84, 390–396 (2011).
Alshaiban, A., Muralidharan-Chari, V., Nepo, A. & Mousa, S. A. Modulation of sickle red blood cell adhesion and its associated changes in biomarkers by sulfated nonanticoagulant heparin derivative. Clin. Appl. Thromb. Hemost. 22, 230–238 (2015).
Kaul, D. K. et al. Monoclonal antibodies to αVβ3 (7E3 and LM609) inhibit sickle red blood cell-endothelium interactions induced by platelet-activating factor. Blood 95, 368–374 (2000).
Zennadi, R. et al. Epinephrine-induced activation of LW-mediated sickle cell adhesion and vaso-occlusion in vivo. Blood 110, 2708–2717 (2007).
Zennadi, R. et al. Role and regulation of sickle red cell interactions with other cells: ICAM-4 and other adhesion receptors. Transfus. Clin. Biol. 15, 23–28 (2008).
De Castro, L. M., Zennadi, R., Jonassaint, J. C., Batchvarova, M. & Telen, M. J. Effect of propranolol as antiadhesive therapy in sickle cell disease. Clin. Transl Sci. 5, 437–444 (2012).
De Castro, L. M. Propranolol: anti-adhesive scd treatment. (a phase II study of propranolol as anti-adhesive therapy for sickle cell disease). J. Sick. Cell Dis. Hemoglobinopathies 15, 00039 (2015).
Soderblom, E. J. et al. Proteomic analysis of ERK1/2-mediated human sickle red blood cell membrane protein phosphorylation. Clin. Proteomics 10, 1 (2013).
Zennadi, R. MEK inhibitors, novel anti-adhesive molecules, reduce sickle red blood cell adhesion in vitro and in vivo, and vasoocclusion in vivo. PLOS ONE 9, e110306 (2014).
Allareddy, V. et al. Outcomes of acute chest syndrome in adult patients with sickle cell disease: predictors of mortality. PLOS ONE 9, e94387 (2014).
Ataga, K. I. et al. Pulmonary hypertension in patients with sickle cell disease: a longitudinal study. Br. J. Haematol. 134, 109–115 (2006).
Gladwin, M. T. et al. Risk factors for death in 632 patients with sickle cell disease in the United States and United Kingdom. PLOS ONE 9, e99489 (2014).
Lima, A. R., Ribeiro, V. S. & Nicolau, D. I. Trends in mortality and hospital admissions of sickle cell disease patients before and after the newborn screening program in Maranhao. Brazil. Revista Brasileira Hematol. Hemoterapia 37, 12–16 (2015).
Sabarense, A. P., Lima, G. O., Silva, L. M. & Viana, M. B. Characterization of mortality in children with sickle cell disease diagnosed through the Newborn Screening Program. J. Pediatr. 91, 242–247 (2014).
Wang, Y. et al. Mortality of New York children with sickle cell disease identified through newborn screening. Genet. Med. 174, 452–459 (2014).
Stuart, M. J. & Setty, B. N. Sickle cell acute chest syndrome: pathogenesis and rationale for treatment. Blood 94, 1555–1560 (1999).
US Food and Frug Administration. FDA approved L-glutamine powder for the treatment of sickle cell disease. https://www.fda.gov/drugs/informationondrugs/approveddrugs/ucm566097.htm (FDA, 2017).
Styles, L. et al. Refining the value of secretory phospholipase A2 as a predictor of acute chest syndrome in sickle cell disease: results of a feasibility study (PROACTIVE). Br. J. Haematol. 157, 627–636 (2012).
Maitre, B. et al. Inhaled nitric oxide for acute chest syndrome in adult sickle cell patients: a randomized controlled study. Intensive Care Med. 41, 2121–2129 (2015).
Barr, F. E. et al. Pharmacokinetics and safety of intravenously administered citrulline in children undergoing congenital heart surgery: potential therapy for postoperative pulmonary hypertension. J. Thorac. Cardiovasc. Surg. 134, 319–326 (2007).
Quinn, C. T. et al. Tapered oral dexamethasone for the acute chest syndrome of sickle cell disease. Br. J. Haematol. 155, 263–267 (2011).
Kumar, R., Qureshi, S., Mohanty, P., Rao, S. P. & Miller, S. T. A short course of prednisone in the management of acute chest syndrome of sickle cell disease. J. Pediatr. Hematol. Oncol. 32, e91–94 (2010).
Strouse, J. J., Takemoto, C. M., Keefer, J. R., Kato, G. J. & Casella, J. F. Corticosteroids and increased risk of readmission after acute chest syndrome in children with sickle cell disease. Pediatr. Blood Cancer 50, 1006–1012 (2008).
Sabaa, N. et al. Endothelin receptor antagonism prevents hypoxia-induced mortality and morbidity in a mouse model of sickle-cell disease. J. Clin. Invest. 118, 1924–1933 (2008).
Minniti, C. P. et al. Endothelin receptor antagonists for pulmonary hypertension in adult patients with sickle cell disease. Br. J. Haematol. 147, 737–743 (2009).
Barst, R. J. et al. Exercise capacity and haemodynamics in patients with sickle cell disease with pulmonary hypertension treated with bosentan: results of the ASSET studies. Br. J. Haematol. 149, 426–435 (2010).
Ashley-Koch, A. E. et al. MYH9 and APOL1 are both associated with sickle cell disease nephropathy. Br. J. Haematol. 155, 386–394 (2011).
De Castro, L. M., Jonassaint, J. C., Graham, F. L., Ashley-Koch, A. & Telen, M. J. Pulmonary hypertension associated with sickle cell disease: clinical and laboratory endpoints and disease outcomes. Am. J. Hematol. 83, 19–25 (2008).
Falk, R. J. et al. Prevalence and pathologic features of sickle cell nephropathy and response to inhibition of angiotensin-converting enzyme. N. Engl. J. Med. 326, 910–915 (1992).
Sasongko, T. H., Nagalla, S. & Ballas, S. K. Angiotensin-converting enzyme (ACE) inhibitors for proteinuria and microalbuminuria in people with sickle cell disease. Cochrane Database Syst. Rev. 6, CD009191 (2015).
Ataga, K. I., Derebail, V. K. & Archer, D. R. The glomerulopathy of sickle cell disease. Am. J. Hematol. 89, 907–914 (2014).
Quinn, C. T. et al. Losartan for the nephropathy of sickle cell anemia: a phase-2, multi-center trial. Am. J. Hematol. 92, E520–E528 (2017).
Walters, M. C. et al. Stable mixed hematopoietic chimerism after bone marrow transplantation for sickle cell anemia. Biol. Blood Marrow Transplant. 7, 665–673 (2001).
Perumbeti, A. et al. A novel human γ-globin gene vector for genetic correction of sickle cell anemia in a humanized sickle mouse model: critical determinants for successful correction. Blood 114, 1174–1185 (2009).
Naldini, L. Lentiviruses as gene transfer agents for delivery to non-dividing cells. Curr. Opin. Biotechnol. 9, 457–463 (1998).
Case, S. S. et al. Stable transduction of quiescent CD34+CD38– human hematopoietic cells by HIV-1-based lentiviral vectors. Proc. Natl Acad. Sci. USA 96, 2988–2993 (1999).
Sessa, M. et al. Lentiviral haemopoietic stem-cell gene therapy in early-onset metachromatic leukodystrophy: an ad-hoc analysis of a non-randomised, open-label, phase 1/2 trial. Lancet 388, 476–487 (2016).
Aiuti, A. et al. Lentiviral hematopoietic stem cell gene therapy in patients with Wiskott-Aldrich syndrome. Science 341, 1233151 (2013).
Eichler, F. et al. Hematopoietic stem-cell gene therapy for cerebral adrenoleukodystrophy. N. Engl. J. Med. 377, 1630–1638 (2017).
Cartier, N. et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science 326, 818–823 (2009).
Ribeil, J. A. et al. Gene therapy in a patient with sickle cell disease. N. Engl. J. Med. 376, 848–855 (2017).
Cavazzana-Calvo, M. et al. Transfusion independence and HMGA2 activation after gene therapy of human β-thalassaemia. Nature 467, 318–322 (2010).
Thompson, A. A. et al. Gene therapy in patients with transfusion-dependent β-thalassemia. N. Engl. J. Med. 378, 1479–1493 (2018).
Pawliuk, R. et al. Correction of sickle cell disease in transgenic mouse models by gene therapy. Science 294, 2368–2371 (2001).
Hoban, M. D. et al. Correction of the sickle cell disease mutation in human hematopoietic stem/progenitor cells. Blood 125, 2597–2604 (2015).
Levasseur, D. N., Ryan, T. M., Pawlik, K. M. & Townes, T. M. Correction of a mouse model of sickle cell disease: lentiviral/antisickling β-globin gene transduction of unmobilized, purified hematopoietic stem cells. Blood 102, 4312–4319 (2003).
Levasseur, D. N. et al. A recombinant human hemoglobin with anti-sickling properties greater than fetal hemoglobin. J. Biol. Chem. 279, 27518–27524 (2004).
Dever, D. P. et al. CRISPR/Cas9 β-globin gene targeting in human haematopoietic stem cells. Nature 539, 384–389 (2016).
De Ravin, S. S. et al. CRISPR-Cas9 gene repair of hematopoietic stem cells from patients with X-linked chronic granulomatous disease. Sci. Transl Med. 9, eaah3480 (2017).
Cornu, T. I., Mussolino, C. & Cathomen, T. Refining strategies to translate genome editing to the clinic. Nat. Med. 23, (415–423 (2017).
Bak, R. O. & Porteus, M. H. CRISPR-mediated integration of large gene cassettes using AAV donor vectors. Cell Rep. 20, 750–756 (2017).
Wang, J. et al. Homology-driven genome editing in hematopoietic stem and progenitor cells using ZFN mRNA and AAV6 donors. Nat. Biotechnol. 33, 1256–1263 (2015).
Lin, S., Staahl, B. T., Alla, R. K. & Doudna, J. A. Enhanced homology-directed human genome engineering by controlled timing of CRISPR/Cas9 delivery. eLife 3, e04766 (2014).
Charpentier, M. et al. CtIP fusion to Cas9 enhances transgene integration by homology-dependent repair. Nat. Commun. 9, 1133 (2018).
Canny, M. D. et al. Inhibition of 53BP1 favors homology-dependent DNA repair and increases CRISPR-Cas9 genome-editing efficiency. Nat. Biotechnol. 36, 95–102 (2018).
Kuscu, C., Arslan, S., Singh, R., Thorpe, J. & Adli, M. Genome-wide analysis reveals characteristics of off-target sites bound by the Cas9 endonuclease. Nat. Biotechnol. 32, 677–683 (2014).
Fu, Y. et al. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat. Biotechnol. 31, 822–826 (2013).
Basak, A. et al. BCL11A deletions result in fetal hemoglobin persistence and neurodevelopmental alterations. J. Clin. Invest. 125, 2363–2368 (2015).
Brendel, C. et al. Lineage-specific BCL11A knockdown circumvents toxicities and reverses sickle phenotype. J. Clin. Invest. 126, 3868–3878 (2016).
Traxler, E. A. et al. A genome-editing strategy to treat β-hemoglobinopathies that recapitulates a mutation associated with a benign genetic condition. Nat. Med. 22, 987–990 (2016).
Hoban, M. D. et al. CRISPR/Cas9-mediated correction of the sickle mutation in human CD34+ cells. Mol. Ther. 24, 1561–1569 (2016).
DeWitt, M. A. et al. Selection-free genome editing of the sickle mutation in human adult hematopoietic stem/progenitor cells. Sci. Transl Med. 8, 360ra134 (2016).
Boulad, F. et al. Safety and efficacy of plerixafor dose escalation for the mobilization of CD34+ hematopoietic progenitor cells in patients with sickle cell disease: interim results. Haematologica 103, 770–777 (2018).
Hsieh, M. M. & Tisdale, J. F. Hematopoietic stem cell mobilization with plerixafor in sickle cell disease. Haematologica 103, 749–750 (2018).
Lagresle-Peyrou, C. et al. Plerixafor enables safe, rapid, efficient mobilization of hematopoietic stem cells in sickle cell disease patients after exchange transfusion. Haematologica 103, 778–786 (2018).
Hoppe, C. C. et al. Initial results from a cohort in a phase 2a study (GBT440-007) evaluating adolescents with sickle cell disease treated with multiple doses of GBT440, a HbS polymerization inhibitor. Blood 689, 689 (2017).
Paszty, C. et al. Transgenic knockout mice with exclusively human sickle hemoglobin and sickle cell disease. Science 278, 876–878 (1997).
Ryan, T. M. et al. Human sickle hemoglobin in transgenic mice. Science 247, 566–568 (1990).
Heeney, M. M., Hoppe, C. C. & Rees, D. C. Prasugrel for sickle cell vaso-occlusive events. N. Engl. J. Med. 375, 185–186 (2016).
Charache, S. et al. Hydroxyurea and sickle cell anemia. Clinical utility of a myelosuppressive “switching” agent. The multicenter study of hydroxyurea in sickle cell anemia. Medicine 75, 300–326 (1996).
Alayash, A. I. Oxidative pathways in the sickle cell and beyond. Blood Cells Mol. Dis. 70, 78–86 (2017).
Sangokoya, C., Telen, M. J. & Chi, J. T. microRNA miR-144 modulates oxidative stress tolerance and associates with anemia severity in sickle cell disease. Blood 116, 4338–4348 (2010).
Jagadeeswaran, R. et al. Pharmacological inhibition of LSD1 and mTOR reduces mitochondrial retention and associated ROS levels in the red blood cells of sickle cell disease. Exp. Hematol. 50, 46–52 (2017).
Kato, G. J., Steinberg, M. H. & Gladwin, M. T. Intravascular hemolysis and the pathophysiology of sickle cell disease. J. Clin. Invest. 127, 750–760 (2017).
Schaer, D. J., Buehler, P. W., Alayash, A. I., Belcher, J. D. & Vercellotti, G. M. Hemolysis and free hemoglobin revisited: exploring hemoglobin and hemin scavengers as a novel class of therapeutic proteins. Blood 121, 1276–1284 (2013).
Manwani, D. & Frenette, P. S. Vaso-occlusion in sickle cell disease: pathophysiology and novel targeted therapies. Blood 122, 3892–3898 (2013).
Dulmovits, B. M. et al. Pomalidomide reverses γ-globin silencing through the transcriptional reprogramming of adult hematopoietic progenitors. Blood 127, 1481–1492 (2016).
Abdulmalik, O. et al. 5-Hydroxymethyl-2-furfural modifies intracellular sickle haemoglobin and inhibits sickling of red blood cells. Br. J. Haematol. 128, 552–561 (2005).
Iyamu, E. W., Turner, E. A. & Asakura, T. Niprisan (Nix-0699) improves the survival rates of transgenic sickle cell mice under acute severe hypoxic conditions. Br. J. Haematol. 122, 1001–1008 (2003).
Abdulmalik, O. et al. Crystallographic analysis of human hemoglobin elucidates the structural basis of the potent and dual antisickling activity of pyridyl derivatives of vanillin. Acta Crystallogr. D Biol. Crystallogr. 67, 920–928 (2011).
Abraham, D. J. et al. Vanillin, a potential agent for the treatment of sickle cell anemia. Blood 77, 1334–1341 (1991).
Ouattara, B. et al. Antisickling properties of divanilloylquinic acids isolated from Fagara zanthoxyloides Lam. (Rutaceae). Phytomedicine 16, 125–129 (2009).
Safo, M. K. et al. Vzhe-039, a novel structurally-enhanced allosteric hemoglobin effector inhibits sickling of SS erythrocytes in vitro, and exhibits improved pharmacologic properties in vivo. Blood 128, 3645–3645 (2016).
Nakagawa, A. et al. Identification of a small molecule that increases hemoglobin oxygen affinity and reduces SS erythrocyte sickling. ACS Chem. Biol. 9, 2318–2325 (2014).
Zhang, Y. et al. Elevated sphingosine-1-phosphate promotes sickling and sickle cell disease progression. J. Clin. Invest. 124, 2750–2761 (2014).
De Franceschi, L., Brugnara, C., Rouyer-Fessard, P., Jouault, H. & Beuzard, Y. Formation of dense erythrocytes in SAD mice exposed to chronic hypoxia: evaluation of different therapeutic regimens and of a combination of oral clotrimazole and magnesium therapies. Blood 94, 4307–4313 (1999).
Brugnara, C. et al. Therapy with oral clotrimazole induces inhibition of the Gardos channel and reduction of erythrocyte dehydration in patients with sickle cell disease. J. Clin. Invest. 97, 1227–1234 (1996).
Ataga, K. I. et al. Improvements in haemolysis and indicators of erythrocyte survival do not correlate with acute vaso-occlusive crises in patients with sickle cell disease: a phase III randomized, placebo-controlled, double-blind study of the Gardos channel blocker senicapoc (ICA-17043). Br. J. Haematol. 153, 92–104 (2011).
Hebbel, R. P. et al. The HDAC inhibitors trichostatin A and suberoylanilide hydroxamic acid exhibit multiple modalities of benefit for the vascular pathobiology of sickle transgenic mice. Blood 115, 2483–2490 (2010).
Kaul, D. K. et al. Robust vascular protective effect of hydroxamic acid derivatives in a sickle mouse model of inflammation. Microcirculation 13, 489–497 (2006).
Zhao, Y., Schwartz, E. A., Palmer, G. M. & Zennadi, R. MEK1/2 inhibitors reverse acute vascular occlusion in mouse models of sickle cell disease. FASEB J. 30, 1171–1186 (2016).
Mahaseth, H. et al. Polynitroxyl albumin inhibits inflammation and vasoocclusion in transgenic sickle mice. J. Lab. Clin. Med. 145, 204–211 (2005).
Martins, V. D., Manfredini, V., Peralba, M. C. & Benfato, M. S. α-lipoic acid modifies oxidative stress parameters in sickle cell trait subjects and sickle cell patients. Clin. Nutr. 28, 192–197 (2009).
El-Beshlawy, A. et al. Diastolic dysfunction and pulmonary hypertension in sickle cell anemia: is there a role for L-carnitine treatment? Acta Haematol. 115, 91–96 (2006).
Serjeant, B. E., Harris, J., Thomas, P. & Serjeant, G. R. Propionyl-L-carnitine in chronic leg ulcers of homozygous sickle cell disease: a pilot study. J. Am. Acad. Dermatol. 37, 491–493 (1997).
Musicki, B., Liu, T., Sezen, S. F. & Burnett, A. L. Targeting NADPH oxidase decreases oxidative stress in the transgenic sickle cell mouse penis. J. Sex. Med. 9, 1980–1987 (2012).
Field, J. J. et al. A phase I single ascending dose study of NKTT120 in stable adult sickle cell patients. Blood 122, 977 (2013).
Kalish, B. T. et al. Dietary omega-3 fatty acids protect against vasculopathy in a transgenic mouse model of sickle cell disease. Haematologica 100, 870–880 (2015).
Daak, A. A. et al. Effect of omega-3 (n-3) fatty acid supplementation in patients with sickle cell anemia: randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 97, 37–44 (2013).
Beckman, J. D. et al. Inhaled carbon monoxide reduces leukocytosis in a murine model of sickle cell disease. Am. J. Physiol. Heart Circ. Physiol. 297, H1243–1253 (2009).
Misra, H. et al. A Phase Ib open label, randomized, safety study of SANGUINATE in patients with sickle cell anemia. Rev. Bras. Hematol. Hemoter 39, 20–27 (2017).
Haynes, J. et al. Zileuton induces hemoglobin F synthesis in erythroid progenitors: role of the L-arginine-nitric oxide signaling pathway. Blood 103, 3945–3950 (2004).
Eiymo Mwa Mpollo, M. S. et al. Placenta growth factor augments airway hyperresponsiveness via leukotrienes and IL-13. J. Clin. Invest. 126, 571–584 (2016).
Opene, M., Kurantsin-Mills, J., Husain, S. & Ibe, B. O. Sickle erythrocytes and platelets augment lung leukotriene synthesis with downregulation of anti-inflammatory proteins: relevance in the pathology of the acute chest syndrome. Pulmonary Circul. 4, 482–495 (2014).
Belcher, J. D. et al. The fucosylation inhibitor, 2-fluorofucose, inhibits vaso-occlusion, leukocyte-endothelium interactions and NF-κB activation in transgenic sickle mice. PLOS ONE 10, e0117772 (2015).
Zhang, D. et al. Neutrophil ageing is regulated by the microbiome. Nature 525, 528–532 (2015).
Valverde, Y., Benson, B., Gupta, M. & Gupta, K. Spinal glial activation and oxidative stress are alleviated by treatment with curcumin or coenzyme Q in sickle mice. Haematologica 101, e44–e47 (2016).
Semple, M. J., Al-Hasani, S. F., Kioy, P. & Savidge, G. F. A double-blind trial of ticlopidine in sickle cell disease. Thromb. Haemostasis 51, 303–306 (1984).
Cabannes, R. et al. Clinical and biological double-blind-study of ticlopidine in preventive treatment of sickle-cell disease crises. Agents and actions. Supplements 15, 199–212 (1984).
Eke, F. U., Obamyonyi, A., Eke, N. N. & Oyewo, E. A. An open comparative study of dispersible piroxicam versus soluble acetylsalicylic acid for the treatment of osteoarticular painful attack during sickle cell crisis. Trop. Med. Int. Health 5, 81–84 (2000).
Desai, P. C. et al. A pilot study of eptifibatide for treatment of acute pain episodes in sickle cell disease. Thromb. Res. 132, 341–345 (2013).
Schnog, J. B. et al. Low adjusted-dose acenocoumarol therapy in sickle cell disease: a pilot study. Am. J. Hematol. 68, 179–183 (2001).
van Zuuren, E. J. & Fedorowicz, Z. Low-molecular-weight heparins for managing vaso-occlusive crises in people with sickle cell disease. Cochrane Database Syst. Rev. 12, CD010155 (2015).
Gladwin, M. T. et al. Nitric oxide for inhalation in the acute treatment of sickle cell pain crisis: a randomized controlled trial. JAMA 305, 893–902 (2011).
Silva, F. H. et al. Beneficial effect of the nitric oxide donor compound 3-(1,3-dioxoisoindolin-2-yl)benzyl nitrate on dysregulated phosphodiesterase 5, NADPH oxidase, and nitrosative stress in the sickle cell mouse penis: implication for priapism treatment. J. Pharmacol. Exp. Ther. 359, 230–237 (2016).
Katusic, Z. S., d’Uscio, L. V. & Nath, K. A. Vascular protection by tetrahydrobiopterin: progress and therapeutic prospects. Trends Pharmacol. Sci. 30, 48–54 (2009).
Machado, R. F. et al. Hospitalization for pain in patients with sickle cell disease treated with sildenafil for elevated TRV and low exercise capacity. Blood 118, 855–864 (2011).
Minniti, C. P. et al. Topical sodium nitrite for chronic leg ulcers in patients with sickle cell anaemia: a phase 1 dose-finding safety and tolerability trial. Lancet Haematol. 1, e95–e103 (2014).
Belcher, J. D. N. et al. Dimethyl fumarate induces cytoprotection and inhibits vaso-occlusion in transgenic sickle mice. Blood 124, 219 (2014).
Keleku-Lukwete, N. et al. Amelioration of inflammation and tissue damage in sickle cell model mice by Nrf2 activation. Proc. Natl Acad. Sci. USA 112, 12169–12174 (2015).
Ghosh, S. et al. Nonhematopoietic Nrf2 dominantly impedes adult progression of sickle cell anemia in mice. JCI Insight 1, e81090 (2016).
Belcher, J. D. et al. Heme oxygenase-1 gene delivery by Sleeping Beauty inhibits vascular stasis in a murine model of sickle cell disease. J. Mol. Med. 88, 665–675 (2010).
Shi, P. A. et al. Sustained treatment of sickle cell mice with haptoglobin increases HO-1 and H-ferritin expression and decreases iron deposition in the kidney without improvement in kidney function. Br. J. Haematol. 175, 714–723 (2016).
Vinchi, F. et al. Hemopexin therapy reverts heme-induced proinflammatory phenotypic switching of macrophages in a mouse model of sickle cell disease. Blood 127, 473–486 (2016).
Burnette, A. D. et al. RNA aptamer therapy for vaso-occlusion in sickle cell disease. Nucleic Acid. Ther. 21, 275–283 (2011).
Gutsaeva, D. R. et al. Inhibition of cell adhesion by anti-P-selectin aptamer: a new potential therapeutic agent for sickle cell disease. Blood 117, 727–735 (2011).
De Franceschi, L. et al. Protective effects of phosphodiesterase-4 (PDE-4) inhibition in the early phase of pulmonary arterial hypertension in transgenic sickle cell mice. FASEB J. 22, 1849–1860 (2008).
Aoki, R. Y. & Saad, S. T. Enalapril reduces the albuminuria of patients with sickle cell disease. Am. J. Med. 98, 432–435 (1995).
Foucan, L. et al. A randomized trial of captopril for microalbuminuria in normotensive adults with sickle cell anemia. Am. J. Med. 104, 339–342 (1998).
Acknowledgements
This work was partially supported by the following grant awards from the US National Institutes of Health: R21DK106509 (National Institute of Diabetes and Digestive and Kidney Diseases; to M.J.T.), U01-HL117709 and RO1-HL112603 (National Heart, Lung and Blood Institute (NHLBI); to P.M.) and R01HL114567 (NHLBI; to G.M.V.). M.J.T. and P.M. also received support from the Doris Duke Charitable Foundation.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the conception, writing and editing of this manuscript.
Corresponding author
Ethics declarations
Competing interests
M.J.T., P.M. and G.M.V. have been involved in clinical and/or basic research involving multiple compounds and therapeutic strategies discussed in this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Telen, M.J., Malik, P. & Vercellotti, G.M. Therapeutic strategies for sickle cell disease: towards a multi-agent approach. Nat Rev Drug Discov 18, 139–158 (2019). https://doi.org/10.1038/s41573-018-0003-2
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41573-018-0003-2
This article is cited by
-
Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management
Scientific Reports (2023)
-
The application of CRISPR-Cas in disease diagnosis and treatment
Science China Chemistry (2023)
-
A phase 1 study in healthy participants to characterize the safety and pharmacology of inclacumab, a fully human anti-P-selectin antibody, in development for treatment of sickle cell disease
European Journal of Clinical Pharmacology (2023)
-
VZHE-039, a novel antisickling agent that prevents erythrocyte sickling under both hypoxic and anoxic conditions
Scientific Reports (2020)
-
Therapies for rare diseases: therapeutic modalities, progress and challenges ahead
Nature Reviews Drug Discovery (2020)