Abstract
Urticaria is an inflammatory skin disorder that affects up to 20% of the world population at some point during their life. It presents with wheals, angioedema or both due to activation and degranulation of skin mast cells and the release of histamine and other mediators. Most cases of urticaria are acute urticaria, which lasts ≤6 weeks and can be associated with infections or intake of drugs or foods. Chronic urticaria (CU) is either spontaneous or inducible, lasts >6 weeks and persists for >1 year in most patients. CU greatly affects patient quality of life, and is linked to psychiatric comorbidities and high healthcare costs. In contrast to chronic spontaneous urticaria (CSU), chronic inducible urticaria (CIndU) has definite and subtype-specific triggers that induce signs and symptoms. The pathogenesis of CSU consists of several interlinked events involving autoantibodies, complement and coagulation. The diagnosis of urticaria is clinical, but several tests can be performed to exclude differential diagnoses and identify underlying causes in CSU or triggers in CIndU. Current urticaria treatment aims at complete response, with a stepwise approach using second-generation H1 antihistamines, omalizumab and cyclosporine. Novel treatment approaches centre on targeting mediators, signalling pathways and receptors of mast cells and other immune cells. Further research should focus on defining disease endotypes and their biomarkers, identifying new treatment targets and developing improved therapies.
Similar content being viewed by others
Introduction
Urticaria is a common and heterogeneous inflammatory skin disorder, with a lifetime prevalence of up to 20% worldwide1,2. The disease results from the activation and degranulation of skin mast cells, followed by the release of histamine and other mediators that lead to sensory nerve activation, vasodilatation, plasma extravasation and cellular recruitment3,4. This process causes the development of the disease-defining signs and symptoms, itchy wheals (hives) and angioedema or both (Fig. 1).
Urticaria is classified, first, based on its duration as acute urticaria (AU), lasting ≤6 weeks, or chronic urticaria (CU), lasting >6 weeks4. Urticaria is further divided into inducible and spontaneous forms. In inducible urticaria, the signs and symptoms are induced by a subtype-specific and definite trigger, for example cold in cold urticaria (ColdU). In spontaneous urticaria, the signs and symptoms appear unprompted, and there are no definite triggers, although stress, infections and other aggravators can increase disease activity in some patients. Spontaneous urticaria is more common than inducible urticaria and both can coexist in the same patient4,5,6 (Tables 1 and 2).
Most cases of AU resolve within 1 week, and <40% of cases become chronic (see Supplementary Table 1). CU often lasts for several years before spontaneous remission occurs (see Supplementary Tables 2 and 3). AU is mostly spontaneous and of unknown cause, although infections and the intake of drugs or foods are thought to be relevant in some patients (see Supplementary Table 4). The underlying causes of chronic inducible urticaria (CIndU) remain unknown, whereas two causes of chronic spontaneous urticaria (CSU) (previously known as chronic idiopathic urticaria) are recognized: autoallergy (also called type I autoimmunity) with IgE autoantibody involvement, and type IIb autoimmunity with IgG autoantibody involvement7,8. These mast cell-activating autoantibodies along with cell infiltration, coagulation and complement activation are thought to be key drivers of the pathogenesis of CSU9,10.
The burden of CU for patients and society is substantial. CU manifestations have a strong effect on health-related quality of life, including sleep impairment, diminished physical and emotional well-being and poor performance at school and work11,12. Current treatment options are still limited and are not effective in around one-third of patients with CU. Novel targeted treatments are needed, and several promising drugs are already under development13.
In this Primer, we discuss the current knowledge of urticaria epidemiology, diagnosis, screening, management and quality of life, and highlight recent advances in the understanding of disease pathogenesis and targeted treatment. Other pathophysiologically and clinically distinct conditions that present with wheals and/or angioedema, for example urticarial vasculitis, autoinflammatory syndromes and bradykinin-mediated angioedema, are briefly discussed as differential diagnoses. Detailed tables of study findings relating to epidemiology and diagnosis, with accompanying references, can be found in the Supplementary information.
Epidemiology
Prevalence and incidence
For 2017, the prevalence of urticaria was estimated at 86 million cases and the annual incidence at 160 million cases globally14. However, each urticaria subtype has its own prevalence within different populations. The prevalence of AU is highest in children <5 years of age14,15,16, whereas CU, especially CSU, is most prevalent in women >30 years old6,17,18,19,20,21. Adult patients with CSU are older than adult patients with CIndU (average age ~30–70 years versus ~20–40 years) and have older age of disease onset (~30–50 years versus ~20–35 years). In adults, all types of urticaria are more prevalent in women than in men, except for cholinergic urticaria (CholU), which is more prominent in both male adults and children; female predominance is absent or is less prominent in younger children21 (Table 1; see Supplementary Tables 2 and 3).
All ethnicities are affected; however, the prevalence of AU and CU was higher in non-white patients in some studies15,19,22 but not all23. The lifetime prevalence of all types of urticaria and AU is 3–22% and 6–19%, respectively (see Supplementary Table 5). The overall lifetime prevalence of CU is 4.4%22, and the point prevalence of CU (1-year prevalence in most studies) ranges from ≤1.5% in the USA and Europe to 3–4% in Mexico, Korea and China (Fig. 2; see Supplementary Table 5).
A point prevalence of chronic urticaria (CU) in children, adults and/or both is presented where available. The lowest prevalence rates were observed in the USA and Europe, whereas the highest rates were reported in Mexico, China and Korea. Detailed information can be found in Supplementary Table 5. CIndU, chronic inducible urticaria; CSU, chronic spontaneous urticaria.
No significant changes in the global prevalence, incidence and the years of life lived with disability were seen for urticaria between 1990 and 2017 (ref.14). The consistent increase in CU prevalence in South Korea21,24, Italy25,26 and Taiwan27 may be linked to country-specific demographic, environmental and behavioural factors and changes.
CIndU is less prevalent than CSU6,28. The pooled rate of all CIndU subtypes was 13%29 and that of CSU was ~60–90% across all cases of CU (see Supplementary Table 6). The most prevalent types of CIndU are symptomatic dermographism, CholU and ColdU, in both adults and children21,30, whereas aquagenic urticaria, solar urticaria, heat urticaria, vibratory angioedema and contact urticaria are rare, that is, are seen in <2–3% of all CU cases (Table 2; see Supplementary Table 6). Delayed pressure urticaria is rarely seen as an isolated disorder but present in combination with CSU in up to 36% of patients with CU (Table 2; see Supplementary Table 6).
Natural course
The average duration of AU is ≤1 week. The rates of progression of AU to CU are between 5% and 39% in most studies (see Supplementary Table 1). CSU is of shorter duration than CIndU, with a mean or median disease duration of ~1–4 years in most studies (see Supplementary Table 2), and the cumulative weighted average estimates for spontaneous remission at 1, 5 and 20 years are 17%, 45% and 73%, respectively31. CSU relapses in 6–31% of patients (see Supplementary Table 2). The mean or median duration of CIndU and its three most common subtypes (symptomatic dermographism, CholU and ColdU) was 2–12 years, 2–5 years, 3–8 years and 2–9 years, respectively. Remission of CIndU within 5 years occurs in only around one-third of patients, with the highest rates in symptomatic dermographism and the lowest in CholU and ColdU30 (see Supplementary Table 3). Various factors and markers have been found to be associated with urticaria natural course, phenotypes, endotypes, clinical and laboratory characteristics and response to treatment (see Supplementary Box 1).
Risk factors
Risk factors reported for AU include high population density15 and personal32 and parental history of allergic diseases2,33. Higher prevalence and/or higher risk of having AU may be associated with poverty and lower socio-economic status14,15, whereas risk for CU was linked to high income and socio-economic status in some studies2,23,34 but not all35,36. In studies involving twins, genetic factors could partly explain susceptibility to urticaria32. The role of polymorphisms of several genes, including TNFRS11A, TBXA2R and PLA2G4A37, has been suggested in susceptibility to AU and/or angioedema induced by multiple NSAIDs.
Genetic predisposition to CU has been associated with gene polymorphisms of IFNγ, IL-6, IL-17RA, IL-10, TGFβ, IL-6, tumour necrosis factor (TNF), PTPN22, IL-1, IL-2 and HLA class I and II alleles38. Some of these genes, such as PTPN22, and HLA alleles, for example HLA-DR4 allele, are also responsible for susceptibility to various autoimmune disorders. For example, HLA-DR4 was strongly associated with autoimmune CSU defined by a positive basophil histamine release assay (BHRA)39 and other autoimmune diseases, for example rheumatoid arthritis and type 1 diabetes mellitus. CSU, especially in middle-aged women with autoimmune CSU40, is associated with an increased risk of developing autoimmune disease within 10 years after the diagnosis of CSU41. For example, women with CSU were 23 and 20 times more likely to also have hypothyroidism and rheumatoid arthritis, respectively, compared with the control group41. Diagnosis of CSU was made in ~80% and ~20% of patients before and after diagnosis of autoimmune diseases, respectively, including rheumatoid arthritis, systemic lupus erythematosus, type I diabetes mellitus and coeliac disease41. Patients with autoimmune thyroid diseases, especially female patients, had a considerably higher risk of CSU development42. Up to 25% of patients with CSU, especially those with positive markers of autoimmune urticaria43,44,45, had a family history of CSU (see Supplementary Table 7). In addition, female patients with peptic ulcer disease46 and abnormal uterine bleeding47 have been shown to have a higher CU risk.
Geographical differences in frequency of CholU and ColdU were reported suggesting that some environmental factors, for example temperature and altitude, might increase the risk of developing CIndU48.
Healthcare use and annual treatment costs
Urticaria, especially CU, is associated with considerable healthcare utilization and economic burden, including both regular and unanticipated healthcare visits, costs due to laboratory expenses and indirect costs due to the absence from work or reduced efficiency11,49. Patients with CU, especially those with angioedema, more frequently visit physicians, mostly family doctors, allergists and dermatologists, and emergency rooms, and are more frequently hospitalized than individuals without urticaria12,49,50. Patients with CIndU were more often hospitalized than patients with CSU (paediatric, 15.5% versus 9.9%; adult, 7.8% versus 4.6%; respectively)6. Average annual economic costs for a patient with CSU ranged from PPP$907 (purchasing power parity dollars) in Italy to PPP$2,984 in France, mostly due to therapies and inpatient visits12.
Mechanisms/pathophysiology
Skin mast cells have a central role in urticaria pathogenesis and are found in the upper papillary dermis as well as the deep dermis and subcutis, mostly around cutaneous blood vessels and sensory nerves. Their activation with subsequent degranulation drives the development of itchy wheals and/or angioedema3.
Acute urticaria
The pathogenesis of AU is poorly investigated. Acute spontaneous urticaria, as well as wheals and/or angioedema in patients with anaphylaxis, have been described to result from type I hypersensitivity reactions to foods, drugs and other allergens51. Type I hypersensitivity, also known as an immediate IgE-mediated reaction, describes interaction between exoallergen and a pre-existing complex of an IgE antibody bound to its high-affinity receptor, FcεRI, on mast cells and basophils that leads to cell activation and degranulation52. NSAID‐induced urticaria and/or angioedema is IgE-mediated or T cell-mediated, or due to the pharmacological inhibition of cyclooxygenase 1 (COX1) and increased levels of cysteinyl leukotrienes53. Acute contact urticaria can develop in response to direct contact with allergens with previous sensitization54 and urticariogenic substances without prior sensitization, for example after touching stinging nettles4.
Chronic spontaneous urticaria
Mast cells
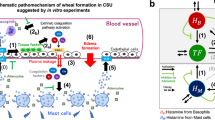
The development of wheals and angioedema in CSU is dependent on mast cell-activating signals and receptors, signalling pathways, inhibitory receptors and mediators, which are targets of current and future therapy3,10,13 (Fig. 3). Mast cells express many activating receptors including FcεRI, MRGPRX2, C5aR, PAR1, PAR2, chemoattractant receptor-homologous molecule expressed on T helper 2 cells (TH2 cells) (CRTh2) and cytokine receptors, which can be activated by various signals13. Interaction of stem cell factor (SCF), produced by fibroblasts, endothelial cells and mast cells, with its receptor KIT (CD117) on mast cells is a major driver of mast cell differentiation, migration, proliferation, survival and apoptosis55. Activation of FcεRI involves several cytoplasmic signalling proteins, for example. LYN, spleen tyrosine kinase (SYK) and Bruton’s tyrosine kinase (BTK), which phosphorylate downstream signalling targets and induce mast cell activation and degranulation56 The first step in FcεRI-mediated signalling is the phosphorylation of the FcεRI β-chain and γ-chain by LYN followed by activation of SYK and BTK. The cytosolic tyrosine kinase BTK is the central positive regulator of FcεRI-mediated mast activation and cytokine production56,57. In addition to its role in mast cell activation, BTK is also required for B cell receptor (BCR) signalling56,57. In addition to activating receptors, mast cells express a few inhibitory receptors, such as sialic acid-binding immunoglobulin-like lectin 8 (Siglec 8), CD200R, CD300a and FcγRIIb58, which can silence mast cells and block mast cell activation upon interaction with their ligands.
Activation and degranulation of mast cells leads to the development of signs and symptoms of urticaria due to the release of histamine and other mediators, which activate sensory skin nerves (itch), dilate skin blood vessels (erythema) and induce plasma extravasation (oedema and influx of other immune cells). In allergic urticaria, release of alarmins, TSLP, IL-33 and IL-25, by the epithelium, activation of skin-resident group 2 innate lymphoid cells (ILC2), polarization of T cells (mostly T helper 2 cells (TH2 cells)) with release of TH2 cytokines, for example IL-4, IL-5 and IL-13, and allergen-specific IgE production by B cells enable subsequent cross-linking of IgE–FcεRI complexes on the surface of mast cells by allergens, which triggers mast cell activation. Chronic spontaneous urticaria (CSU) can appear due to a chain of complex multistep interlinked events including cell infiltration (mostly eosinophils, basophils, neutrophils and T cells), autoimmunity (for example, IgE/IgG histamine-releasing autoantibodies), neurogenic inflammation (via histamine-dependent and histamine-independent itch signalling pathways mediated by cutaneous pruriceptive sensory nerves), activation of the complement cascade (for example, via production of anaphylatoxin C5a) and activation of tissue factor-initiated extrinsic pathway of the coagulation cascade. Here, receptors (for example, FcεRI, C5aR, MRGPRX2, Siglec 8, KIT and IL-4Rα), signalling pathways (for example, BTK and SYK) and mediators (for example, histamine, tryptase, IL-5, IL-17 and IL-31) of mast cells, eosinophils, basophils and/or other immune cells involved in urticaria, as well as activating signals (for example, IgE anti-autoallergens), are the targets for current therapy and drugs in development. In chronic inducible urticaria (CIndU), similar mechanisms of mast cell activation including autoallergy and/or autoimmunity may have a role. AR, alarmin receptors; BCR, B cell receptor; BTK, Bruton’s tyrosine kinase; CGRP, calcitonin gene-related peptide; CRTh2, chemoattractant receptor-homologous molecule expressed on TH2 cells; EPO, eosinophil peroxidase; FDP, fibrin degradation products; FXa, factor Xa; GM-CSF, granulocyte–macrophage colony-stimulating factor; HR, histamine receptors; MBP, major basic protein; MPO, myeloperoxidase; MRGPRX2, Mas-related G-protein-coupled receptor X2; PAF, platelet-activating factor; PAR, protease-activated receptor; PGD2, prostaglandin D2; SCF, stem cell factor; Siglec 8, sialic acid-binding immunoglobulin-like lectin 8; SYK, spleen tyrosine kinase; TNF, tumour necrosis factor; TSLP, thymic stromal lymphopoietin; VEGF, vascular endothelial growth factor.
CSU symptoms occur mainly due to the release of histamine, but also by a broad range of other produced and secreted mediators, including tryptase, prostaglandin D2 (PGD2), TNF, IL-4, IL-5, IL-13, IL-17 and IL-31, which can exert effects on resident skin cells and other recruited target cells, for example T cells, eosinophils and basophils3,13.
Cell infiltrate
In CSU, perivascular and interstitial inflammatory cellular infiltrates, resembling an allergic late-phase reaction, include eosinophils, neutrophils, lymphocytes and basophils9,59,60,61. Infiltrating cells are thought to migrate from the blood into the skin in response to chemotactic factors, for example eotaxins, MCP3, RANTES, IL-5, C3a, C5a, TNF, IL-17 and platelet-activating factor (PAF), released by mast cells, activated endothelial cells, TH2 cells, dermal fibroblasts and other cells9. Cell adhesion molecules, for example P-selectin, E-selectin, ICAM, VCAM and PECAM, are upregulated on the surface of endothelial cells in the lesional skin of patients with CSU due to action of histamine, thrombin, TNF and other factors9.
Blood basopenia and eosinopenia that are observed in ~10–15% of patients with CSU may reflect the transport of cells into the skin, and are associated with CSU activity, presence of autoantibodies and poor response to H1 antihistamines (H1-AH) and omalizumab62,63. Perivascular eosinophils and neutrophils appeared in wheals 30 min after intradermal injection of autologous serum and, along with T lymphocytes, increased in numbers over the following 2 h with a decrease by 48 h (neutrophils) or later (eosinophils and lymphocytes)64. Blood and skin-migrated basophils can contribute to the CSU pathogenesis by releasing histamine, leukotrienes and cytokines via activation of FcεRI and C5aR65,66. Eosinophil granule proteins such as major basic protein (MBP) were detected in the wheals of patients with CSU67,68. MBP was able to induce mast cell activation and degranulation, representing a putative mechanism of crosstalk between eosinophils and mast cells in CSU67. Activation of eosinophils can occur due to mast cell mediators, namely IL-5, TNF, PAF and eotaxin, and also IgG autoantibodies to the low-affinity IgE receptor69. Activated eosinophils can also release SCF, a growth factor for mast cells. Finally, eosinophils might be key players in activation of the coagulation cascade and of Mas-related G-protein-coupled receptor X2 (MRGPRX2) on mast cells via expression of tissue factor and release of MRGPRX2 agonists, respectively67,69.
TH2 cells are a predominant type of lymphocyte in CSU skin biopsy samples but TH1 cells and TH17 cells are also present9. TH2 cells are highly involved in allergic diseases, releasing a broad range of cytokines, stimulating IgE production and mast cell, basophil and eosinophil activation. In patients with CSU, cytokine levels and/or expression were increased in the blood and/or lesional skin, for example IFNγ, TNF, TGFβ, IL-1β, IL-3, IL-4, IL-5, IL-6, IL-13, IL-17, IL-23, IL-24, IL-31 and IL-33, and correlated with disease activity, for example TNF, IL-6, IL-17, IL-23 and IL-24, and autologous serum skin test (ASST) positivity, for example IL-17 (ref.9).
Coagulation cascade and complement
Eosinophils and dermal microvascular endothelial cells can express a large amount of tissue factor on their surface in response to tissue factor inducers, for example histamine, vascular endothelial growth factor (VEGF), LPS, TNF, IL-6, IL-33 and IL-1β70. Tissue factor can activate the extrinsic coagulation cascade leading to the production of activated coagulation factors, such as factor Xa (FXa) and FIIa (thrombin)71,72.
Coagulation factors, histamine, VEGF73, bradykinin, PAF and/or other molecules can lead to gap formation between vascular endothelial cells via protease-activated receptor 1 (PAR1), other specific receptors and/or a direct action on endothelial cells9. This results in the leakage of plasma that can contain autoantibodies to IgE or FcεRI, and/or autoantigens for specific IgE bound to mast cells in the skin with subsequent activation of mast cells and wheal and flare formation9. In addition, thrombin and FXa may induce mast cell degranulation via action on PAR1 and PAR2, respectively10,74. Furthermore, complement component C5a is thought to be produced following the activation of extrinsic coagulation, fibrinolysis and/or binding of IgG anti-FcεRI to FcεRI on mast cells and basophils10. Activated coagulation factors (FXa and FIIa) and plasmin can produce C5a and C5b from C5, and/or C3a and C3b from C3. C3a and C5a in leaked plasma can activate mast cells and basophils via C3aR and C5aR, respectively10,75. Finally, functional specific IgE antibodies to tissue factor were reported to release leukotriene C4 by tissue factor-stimulated peripheral basophils76.
Activation of coagulation and fibrinolysis in patients with CSU is reflected in increased mean platelet volume, levels of D-dimer, fibrin and fibrinogen degradation products, prothrombin fragment 1 + 2, FVIIa and other molecules41,71. An increase in blood markers of thrombin generation and fibrinolysis, for example D-dimer, was linked to severe CSU and a decrease was observed during CSU remission77. Although activation of the coagulation cascade happens in CSU, it is considered mostly a local process with active fibrinolysis without increased risk for thrombotic events71.
In patients with CSU, increased C-reactive protein (CRP) levels correlated with increased D-dimer, IL-6, C3 and C4 levels as well as CSU activity and ASST positivity78,79. This finding supports a close link between autoimmunity, inflammation, complement and coagulation activation pathways in CSU pathogenesis that may lead to maintenance and amplification of urticarial inflammation.
Autoantibodies
Antibodies of IgE, IgA, IgM and IgG classes are thought to be critically involved in the pathogenesis of CSU80,81,82. IgE binds to the α-subunit of its high-affinity receptor, FcɛRI, on mast cells and basophils and, if present at high concentrations, can activate these cells regardless of the specific antigen83. In addition, the cross-linking of IgE by their respective allergens, autoallergens and IgG anti-IgE antibodies can lead to mast cell and basophil activation and mediator release in allergic, autoallergic and autoimmune urticaria, respectively8,82,84,85.
Clinically relevant allergy as a cause of CSU is rare84,86. IgE antibodies to autoantigens, for example thyroid peroxidase (TPO), eosinophil peroxidase (EPO), double-stranded DNA, tissue factor, ECP, FcεRI, thyroglobulin and IL-24, are found in up to two-thirds of patients with CSU and some of these antibodies, for example IgE anti-IL-24 and IgE anti-TPO, were shown to activate mast cells and/or basophils in vitro8,87. Furthermore, evidence in support of IgE autoantibodies as a driver of CSU includes high rates of positive skin prick tests with TPO in patients with CSU with increased levels of IgE anti-TPO and successful experiments of passive transfer of IgE anti-TPO88. Cross-reactivity between proteins, for example between TPO (not present in the skin) and EPO (present in the skin)89, and expression of autoallergens in the skin, for example IL-24, might explain why IgE–autoallergen interaction leads to activation of mast cells in skin rather than in other organs90.
IgG autoantibodies to IgE and FcɛRI were the first autoantibodies described in the context of autoimmune CSU. Most studies report that 20–50% of patients with CSU have these autoantibodies91. Since 2013, when a taskforce position paper was published, autoimmune CSU associated with these autoantibodies is defined by triple positivity for presence of IgG autoantibodies by immunoassay, functionality of autoantibodies assessed by basophil tests (basophil activation test and/or BHRA) and skin autoreactivity assessed by ASST92. If these strict criteria are applied, only 8% of patients with CSU are diagnosed with autoimmune CSU7.
It is still a matter of debate whether clearly defined and distinct auto-IgE and auto-IgG endotypes exist. Although real overlap rates are still unknown, there is growing evidence of co-expression of IgG autoantibodies and other types of autoantibodies, specifically IgE, IgM and IgA autoantibodies, in the same patient80,81. IgE autoantibodies might appear in blood first, followed by other types of autoantibodies over the course of the disease.
Neurogenic inflammation
In CSU, the bidirectional interaction of mast cells, other immune cells and sensory nerves is thought to exist through release of histamine, IL-31, neuropeptides and other mediators67,93,94. This vicious cycle causes vasodilatation, plasma extravasation, neurogenic inflammation, pruritus and other urticaria symptoms, and might be supported via MRGPRX2-mediated activation of mast cells95. MRGPRX2 is a receptor for a broad range of exogenous and endogenous substances and is responsible for IgE-independent activation of mast cells96. MBP and EPO induced histamine release from human skin mast cells via MRGPRX2 (ref.67). Levels of substance P, a neuropeptide and MRGPRX2 agonist, were elevated in CSU, correlated with disease activity in some studies97, and provocation with MRGPRX2 agonists revealed an increased skin reactivity in patients with CSU98. Lastly, the number of MRGPRX2-expressing mast cells has been found to be increased in patients with CSU67.
Chronic inducible urticaria
The pathogenesis of CIndU is largely unknown. Autoallergic IgE-mediated mast cell activation was suggested in symptomatic dermographism, ColdU, solar urticaria and CholU by passive transfer experiments and/or the efficacy of omalizumab99. IgE autoantibodies might be produced to a skin-derived protein released upon stimulation, for example by cold100. In solar urticaria, molecular alteration of chromophores by solar electromagnetic radiation and their binding to surface-bound IgE on mast cells, leading to their activation, have been suggested101. In CholU, blockade of the sweat gland duct might lead to sweat reflux and leakage into the dermis inducing the symptoms of CholU due to production of a sweat antigen. Specific IgE to sweat antigen MGL_1304 have been identified in sera of some patients with CholU102. Another proposed mechanism in CholU is reduced expression of cholinergic receptor M3 (CHRM3) in eccrine sweat gland epithelial cells, which leads to escape of acetylcholine with acetylcholine-mediated degranulation of adjacent mast cells102. IgG and/or IgM directed to IgE were shown in ColdU100. In patients with heat urticaria, few cases of a positive reaction on intradermal testing with heated autologous serum, presumably containing denatured IgE and inactivated complement, were described103. Hereditary vibratory angioedema can present as an autosomal dominant variant due to a gain-of-function mutation in adhesion G-protein-coupled receptor E2 (ADGRE2), located on mast cells, which might decrease inhibitory interaction between the α-subunit and the β-subunit of this receptor leading to sensitization of mast cells to vibration-induced degranulation104. Delayed pressure urticaria is thought to occur via a non-immunologic mechanism with involvement of many pro-inflammatory mediators other than histamine, for example IL-1, IL-6, IL-3 and TNF105. In contrast to other CIndU, delayed pressure urticaria showed a substantial leukocyte infiltration in the dermis perhaps due to a sustained initiating stimulus105. Contact urticaria can be classified as an immunological reaction, that is IgE-mediated or T cell-mediated, or a non-immunological reaction106. Hypotheses explaining pathogenesis of aquagenic urticaria include the synthesis of a mast cell-degranulating substance due to interaction between water and a component in or on the skin or sebum; changes in osmotic pressure surrounding hair follicles and increased passive diffusion of water; the existence of water-soluble antigens in the epidermis; and a histamine-independent mechanism107.
Diagnosis, screening and prevention
Diagnosis
The clinical presentation of urticaria is very similar in all age groups, ethnicities and genders. Wheals and angioedema occur with the same anatomical distributions across all skin tones. However, erythema related to whealing is more difficult to detect in pigmented skin108. A detailed history and physical examination are the essential first steps in the diagnostic workup of all patients with urticaria. However, as wheals and angioedema are transient and may not be present at physical examination, physicians should also review patients’ pictures or their documentation of signs and symptoms (Fig. 4). Diagnosis of urticaria is usually straightforward irrespective of its type or subtype4 (see Supplementary Fig. 1).
a | Acute urticaria (AU). b | Chronic spontaneous urticaria (CSU) (wheals). c | CSU (angioedema). d | Cold urticaria (ColdU). e | Delayed pressure urticaria. f | Solar urticaria. g | Heat urticaria. h | Symptomatic dermographism. i | Cholinergic urticaria (CholU). j | Contact urticaria. k | Aquagenic urticaria. l | Vibratory angioedema. Part e, image courtesy of A. Kasperska-Zajac. Part g, image courtesy of L. F. C. Ensina. Part l, image courtesy of K. Brockow.
Acute urticaria
By definition, AU is self-limiting and does not require routine or extensive diagnostic tests unless strongly suggested by patient history4. For example, in patients with a history of type I food allergy or drug hypersensitivity, urticaria symptoms can appear immediately after a contact with respective allergens and allergy tests may help avoid re-exposure to relevant causative factors.
Chronic spontaneous urticaria
Most patients with CSU present with only wheals (~57%), whereas the occurrence of wheals and angioedema (~37%) or only angioedema (~6%) is less common (see Supplementary Table 8). The signs and symptoms of CSU can occur spontaneously at any time of the day but commonly during the evening and night109, which may reflect circadian variations in mast cell activation110 and differences in the underlying pathogenesis. For example, the presence of nocturnal symptoms has been associated with an autoimmune endotype of CSU111. Wheals can occur anywhere, but favour the arms and legs109. Angioedema appears most commonly on the face, for example lips and eyelids, but also on hands and feet and other body parts1. In most patients with moderate or severe CSU, wheals and/or angioedema occur on a daily or nearly daily basis or show an intermittent–recurrent course4.
In some patients with CSU, stress, foods, drugs (mostly NSAIDs) or infections can lead to exacerbation. NSAID hypersensitivity is observed in up to 30% of patients with CSU and is classified as NSAID‐exacerbated cutaneous disease53. CSU exacerbation occurs within minutes or hours after the intake of COX1 inhibitors (selective COX2 inhibitors are usually tolerated)112 and can be confirmed by oral drug provocation testing53.
The current international urticaria guideline has introduced the 7C concept, that is, seven aims of the diagnostic workup in patients with CSU4. The 7C concept includes ruling out differential diagnoses (confirm); looking for markers of autoimmune urticaria (cause); identifying potential triggers (cofactors); checking for autoimmunity, mental health disorders and other comorbidities; identifying problems with sleep, distress, sexual health and social performance (consequences); assessing potential biomarkers or predictors of treatment response (components); and monitoring CSU activity, impact and control (course).
Basic tests include a differential blood count and the erythrocyte sedimentation rate (ESR) and/or CRP in all patients with CSU, and measurements of IgG anti-TPO and total serum IgE levels in patients with CSU in specialist care. The latter two can help in the diagnosis of autoimmune CSU and elevated IgG anti-TPO levels point to concomitant autoimmune thyroiditis7,40,113. Further tests should only be performed if indicated by patient history and are rarely needed4.
Chronic inducible urticaria
In patients with CIndU, wheals are often of shorter duration (≤1 h) than in those with CSU (up to 24 h)4,106. High-frequency trigger exposure and a low trigger threshold are linked to high disease activity4,106. Even though the lesions are usually confined to areas of skin exposed to the trigger stimuli, systemic reactions including anaphylaxis may occur and adrenaline autoinjectors should be prescribed to high-risk patients114. During pregnancy, one-half of patients experience improvement of their CU, whereas exacerbation occurs in approximately one-third of patients115. In particular, patients with CIndU or both CSU and CIndU showed a twofold increase of disease exacerbation during pregnancy. Disease activity changes during pregnancy might be linked to changes in trigger exposure and/or hormonal and immunological changes that promote mast cell activation.
The diagnosis of CIndU is based on a thorough history and the results of provocation testing. The aims of provocation testing are to determine the relevant triggers and to assess trigger thresholds, which are useful for measuring disease activity and monitoring treatment responses. For most CIndU subtypes, validated tools for provocation testing are available (Table 2; see Supplementary Table 9 and Supplementary Fig. 2).
Comorbidities
Acute urticaria
Infection, mainly upper respiratory tract infection, is considered the most common comorbidity and aetiological factor of acute spontaneous urticaria (~30–40% of AU cases), although the prevalence of infectious aetiologies decreases as the age of patients increases (see Supplementary Table 4). Less frequently, reactions can be triggered by drugs, mostly NSAIDs and antibiotics, inhalant allergens (for example, pollen, dust mites), foods, stress, Hymenoptera stings and physical factors (see Supplementary Table 4). Drugs such as NSAIDs are often used when infection is present, and this makes it challenging to differentiate between medications or infection as the cause of urticaria. Inducible factors are considered to be a rare cause of AU in children but can lead to AU in <15% of all adult AU cases116 (see Supplementary Table 4). The cause of AU was more frequently detected in patients 0–6 years of age than in those 7–18 years of age51. Foods and infection caused AU more often in children <13 years of age than in children 13–18 years of age51. AU usually disappears after resolution or eradication of infection, or avoidance of drugs and other causative triggers.
Chronic spontaneous urticaria
CIndU is a frequent comorbidity of CSU, with most studies reporting rates >10% (see Supplementary Table 6). Among patients with CU, 29–93% had CSU only, 6–35% had CIndU only and 1–43% had both CSU and CIndU (see Supplementary Table 6). Of 245 patients with CSU, 36% had CIndU confirmed by a positive challenge test, mostly symptomatic dermographism (25%) and ColdU (13%)48. Multiple types of CIndU may coexist in the same patient with CSU117.
Autoimmune diseases are present in 28% of patients with CSU, most frequently autoimmune thyroid diseases (~25%, mostly Hashimoto’s thyroiditis with or without hypothyroidism), vitiligo, rheumatoid arthritis, autoimmune gastritis and diabetes mellitus (1–2%)40,118.
Bacterial infection including Helicobacter pylori and focal bacterial infections, for example dental infection, have been reported in up to 77% of patients with CSU5. However, the evidence for the causal link between bacterial infection and CSU development is still weak and conflicting5. The clinical relevance of viral infection, for example viral hepatitis and HIV infection, and fungal infection is still unknown and these infections are unlikely to contribute to the development of CSU5. In one study, CU did not affect the course of COVID-19, whereas COVID-19 induced CU exacerbation in one-third of patients, especially in patients with severe COVID-19 (ref.119). CSU improved in one-third of patients after the treatment of confirmed infection with helminths or protozoa with antiparasitic drugs, although the link between CSU and parasites is still ill-characterized5.
The evidence for the increased prevalence of allergic diseases in patients with CSU is inconsistent5. Three large studies suggest that individuals with CSU are considerably more likely to have allergic diseases compared with the general population or control subjects without urticaria34,120,121. Type I hypersensitivity to allergens as a cause of CSU should be ruled out in patients with intermittent symptoms, a temporal relationship to a particular allergen and possible symptoms due to other allergic diseases, for example asthma.
Cancer, mostly non-haematologic, was reported in 0–9% of patients with CSU5,26 and CSU resolved in some cases once patients were in remission122. CSU development in patients with cancer might be related to cancer-induced immune dysregulation including activation of complement and coagulation cascade123, and return to normal homeostatic conditions might parallel CSU improvement after cancer treatment. Mental health disorders, mainly depression and anxiety, are present in up to 60% of patients with CSU and are associated with considerably impaired quality of life5. Prevalence of one or several components of metabolic syndrome, that is, central obesity, dyslipidaemia, hyperglycaemia and hypertension, was considerably increased in patients with CU including CSU compared with control subjects without CU/CSU5.
Chronic inducible urticaria
CIndU can coexist with CSU and other CIndU forms. CSU was reported in 71%, 25% and 10% of patients with symptomatic dermographism124, CholU125 and ColdU114, respectively. Among patients with CU, symptomatic dermographism alone or in combination with CSU is more prevalent than other CIndU, followed by ColdU and CholU (see Supplementary Table 6). Allergic diseases were reported to be a frequent comorbidity (up to 26–48%) of CholU126, solar urticaria127, ColdU128 and symptomatic dermographism129.
Differential diagnosis
Patients with certain diseases other than urticaria can present with wheals and/or angioedema as associated or prodromic signs130 (Fig. 5; see Supplementary Table 10). The differential diagnoses of CSU are guided by the history and physical examination and supported by exploratory tests.
a | Urticarial vasculitis. b | Mastocytosis in the skin. c | Erythema annularis centrifugum. d | Cryopyrin-associated autoinflammatory syndrome e | Schnitzler syndrome. f | Hereditary angioedema. g | Erythema marginatum in a patient with hereditary angioedema. h | Melkersson–Rosenthal syndrome (oedema of the upper lip and fissured tongue are seen). i | Erythema multiforme. j | Hypereosinophilic syndrome.
In patients who exclusively develop wheals but not angioedema, autoinflammatory disorders, such as Schnitzler syndrome or cryopyrin-associated periodic syndromes, should be considered differential diagnoses4. Wheals in patients with autoinflammatory disorders are refractory to antihistamines and can present a distinct entity called neutrophilic urticarial dermatosis with a dense perivascular and interstitial infiltrate of neutrophils with leukocytoclasia but without vessel wall necrosis131.
In urticarial vasculitis, both long-lasting wheals (>24 h) and angioedema can occur and the diagnosis is confirmed by a combination of three histological criteria: leukocytoclasia, fibrin deposits and extravasated erythrocytes132,133. By contrast, CSU histopathology usually shows dermal oedema with an inflammatory infiltrate consisting of lymphocytes, eosinophils, neutrophils and nuclear dust, without evidence of vasculitis60,132.
When patients show recurrent angioedema without wheals, bradykinin-mediated angioedema including angiotensin-converting enzyme inhibitor-induced angioedema and hereditary angioedema should be excluded4.
Disease activity and control
Several patient-reported outcome measures, global and validated tools, were developed to assess CU activity and control and are used both in clinical care and in trials134 (Box 1). The Urticaria Activity Score (UAS) is a gold standard to assess disease activity (wheals number and itch intensity) in CSU and is usually used prospectively during seven consecutive days (UAS7)4,135. Similarly, the Angioedema Activity Score (AAS) is a prospective, diary-type tool that enables clinicians to measure angioedema activity in patients with all forms of recurrent angioedema136. In CIndU, the Cholinergic Urticaria Activity Score (CholUAS; not yet validated) and the Cold Urticaria Activity Score (ColdUAS), modified versions of the UAS, collect data on a daily basis assessing the intensity of wheals and pruritus as well as the intensity of exposure to specific triggers with a recall period of 24 h134,137,138. The Urticaria Control Test (UCT)139 and the Angioedema Control Test (AECT)140 are retrospective tools with a recall period of 4 weeks used to assess disease control in all forms of CU.
Screening
No screening methods are available for urticaria in the general population. Patients with autoimmune diseases, especially autoimmune thyroiditis, should be asked for signs and symptoms of CSU40,42,118. Similarly, patients with CSU should be regularly checked for autoimmune thyroid disease by palpation of the thyroid, thyroid function tests and anti-TPO and anti-thyroglobulin antibodies4,40,118.
In CSU, basic screening tests (differential blood count, ESR and/or CRP) and screening tests performed in specialist care (total IgE and IgG anti-TPO) can help in identifying comorbid and underlying diseases, for example chronic infection, autoimmune disease and allergy, as well as differential diagnoses, for example urticarial vasculitis or autoinflammatory disorder4. Mental health disorders, namely depression and anxiety, common comorbidities and consequences of CSU should be checked for in every patient with CSU4.
Prevention
No primary prevention exists for urticaria with the possible exception of allergic urticaria, in which general preventive measures for allergic diseases may apply (especially in children with high risk of allergy). A population-based cross-sectional study in China found that breastfeeding >6 months after birth can lower the risk of urticaria33. Secondary prevention measures include avoidance of exposure to a relevant trigger, for example wearing of tight-fitting clothes by a patient with delayed pressure urticaria or intake of particular food or drug by a patient with allergic urticaria4. Migration to different geographic regions with different ambient temperatures might decrease the risk of development of CIndU such as ColdU. Finally, tertiary prevention is possible with treatment of symptoms using guideline-recommended options, for example antihistamines and omalizumab4, and might be possible with induction of long-lasting remission with disease-modifying treatments, for example allergen-specific immune therapy in patients with allergic urticaria84, cyclosporine in patients with autoimmune CSU141,142 and novel mast cell-reducing therapies143.
Management
The management of urticaria has progressed substantially over the past two decades, with disease control possible for at least two-thirds of patients144,145,146. Increased understanding of disease pathogenesis underpins an expanding pipeline of targeted therapies with improved adverse effect profiles compared with traditional immunosuppressants (Table 3). Structured assembly of the evidence for pharmacological and non-pharmacological agents using GRADE methodologies (Grading of Recommendations Assessment, Development and Evaluation methodologies), together with publication of up-to-date international consensus guidelines, has led to the establishment and regular review of an international standard of care for urticaria4. The overall goal of treatment is safe and effective attainment of complete disease control (UAS7 = 0 and UCT = 16) with a normal quality of life. Figure 6 provides the current consensus algorithm for urticaria treatment4.
The 3A management approach of assess, act and adjust recognizes the importance of several comorbidities associated with urticaria; and the wide range and dynamic nature of disease severity across time, with the need to step up and step down therapies to achieve complete disease control while limiting cost and adverse effects. Only some biomarkers of response to treatment are shown246,259 and other biomarkers are listed in Supplementary Box 1. AAS7, Angioedema Activity Score used prospectively during 7 consecutive days; AECT, Angioedema Control Test; AE-QoL, Angioedema-Quality of Life Questionnaire; ASST, autologous serum skin test; BHRA, basophil histamine release assay; CholUAS, Cholinergic Urticaria Activity Score; CholU-QoL, Cholinergic Urticaria-Quality of Life Questionnaire; CIndU, chronic inducible urticaria; ColdUAS, Cold Urticaria Activity Score; CSU, chronic spontaneous urticaria; CU-Q2oL, Chronic Urticaria-Quality of Life Questionnaire; H1-AH, H1 antihistamines; UAS7, Urticaria Activity Score used prospectively during 7 consecutive days; UCT, Urticaria Control Test. Adapted with permission from ref.4, Wiley.
Elimination of causes and avoidance of triggers
AU or CSU with an intermittent–recurrent course with an identifiable exogenous trigger, for example an NSAID, is effectively managed with avoidance of the trigger and cross-reacting molecules4,147. Inducible urticaria, by definition, has an eliciting factor, or group of factors, and exposure reduction methods are a mainstay of therapy; however, avoidance might be hard to achieve, for example avoiding cold ambient temperatures, or might negatively affect quality of life4. Autoimmunity underlying CSU cannot be eliminated, although direct autoantibody removal through plasmapheresis has been successful, with high financial cost, in a limited number of patients with severe treatment resistance148,149. Removal of inciting antigens, either spontaneously (for example, resolution of an acute viral or bacterial infection) or through treatment of an underlying condition (for example, solid cancer surgery), can lead to resolution of urticaria122. Disappointingly, especially in CU, the treatment of underlying or associated factors often fails to alter the trajectory of CU. Eradication therapies for chronic infections, such as H. pylori, are a good example, and an analysis of studies based on the GRADE approach demonstrated weak and conflicting evidence between H. pylori eradication and improvement of CU150. Thyroid supplementation or anti-thyroid drugs in patients with CSU with Hashimoto’s thyroiditis or Graves’ disease, respectively, is another example when associated disease treatments have limited efficacy in reducing urticaria118,151. In most of these instances, no randomized controlled trial data are available and the evidence quality is low4,151. Thus, the intensive and costly general screening programmes for causes of CSU should not be performed, unless indicated by the patient’s clinical history, physical examination and/or initial CSU workup. In addition, the treatment of associated thyroid autoimmunity and other comorbidities should be individualized and based on disease-specific complications, for example hypothyroidism4.
Second-generation H1 antihistamines
Multiple effective and affordable second-generation antihistamines (sgAHs) that block the H1 receptor are available and are the standard and first step in urticaria treatment152,153. They do not antagonize the binding of histamine but act as inverse agonists and have the opposite effect on the receptor to histamine, therefore shifting the equilibrium towards the inactive state154. Most guidelines recommend against the use of first-generation H1-AH due to sedative and anticholinergic effects, and drug–drug interactions4,155. Most, but not all, sgAHs have been evaluated and shown effective in urticaria at standard doses4. Studies have also confirmed the safety and efficacy of using off-label high-dose sgAH therapy (up to fourfold the recommended daily dose), including for bilastine, cetirizine, desloratadine, ebastine, fexofenadine, levocetirizine and rupatadine145,156,157,158,159,160 However, the use of updosed sgAHs is associated with a higher risk of somnolence than the use of standard dose of sgAHs (relative risk 3.28; 95% confidence interval 1.55–6.95; p = 0.002)161. Of note, British urticaria guidelines from 2021 recommend against updosing on mizolastine162. High-dose sgAHs are now considered part of the first step in urticaria treatment, if the standard dose is not sufficient to control symptoms4. The optimal timing and approach to step up or step down antihistamine therapy are still based on expert opinion, and include both tapering or immediate discontinuation. Based on half-life considerations, a 2-week period is generally considered sufficient to observe the effect of antihistamine changes in CU163.
Around 61% of patients with CSU do not respond to the licensed doses of sgAHs and only ~63% of these non-responders benefit from updosed sgAHs145. The second-line therapy with omalizumab as an add-on to sgAHs is indicated in patients with CSU who do not respond to increased doses of sgAHs4.
Omalizumab
Omalizumab is the first in-class anti-IgE monoclonal antibody (mAb) and binds and lowers free IgE, with subsequent downregulation of FcεRI on basophils and mast cells164,165,166,167. Lowered FcεRI expression may render the cells less susceptible to activation by IgE and IgG anti-FcεRI and prevent histamine release and inflammation167. As the step-up treatment from antihistamines, omalizumab, as an add-on therapy, has the strongest evidence base and recommendations of all possible treatments for CSU in patients ≥12 years of age146. A meta-analysis of 67 real-life CSU studies reported complete and partial response rates of 72% and 18%, respectively, comparable with efficacy in clinical trials, with a mean adverse effect rate of 4%168. Omalizumab improves various aspects of quality of life in patients with CSU in both clinical trials and real-life studies169,170,171. It has also been shown to be effective (although has not yet been approved) in the management of CIndU, particularly symptomatic dermographism, ColdU, CholU and solar urticaria166,172,173,174,175. Poor and/or slow response to omalizumab was linked to several markers and features of autoimmune CSU, including BHRA positivity and low total IgE levels (see Supplementary Box 1).
Real-world practice continues to raise several questions around omalizumab use as monotherapy (without antihistamines): the initial treatment duration, updosing or a shortened dosing interval and discontinuation strategies176. Alternative approaches include combination therapy with other systemic immunosuppressives (cyclosporine, dapsone and colchine), although evidence is very limited, or a complete switch to cyclosporine, a third-line treatment of CU4,164,177,178.
Cyclosporine
Cyclosporine is a T cell immunosuppressive agent that also inhibits mediator release from basophils and mast cells179. It has been used for more than three decades for the treatment of CU180. Current guidelines recommend off-label use of cyclosporine as an add-on to sgAHs for patients with CSU with severe disease that is refractory to the combination of high-dose antihistamine and omalizumab4. Efficacy has been demonstrated in placebo-controlled randomized control trials180,181,182 and open-label studies183, with meta-analysis data suggesting response rates up to 73%182. Adverse effects can be severe, particularly renal impairment, and occur in up to 50% of patients; hence, most guidelines do not recommend cyclosporine as standard treatment4. However, in many countries without access to omalizumab, cyclosprine is used as a step-up therapy from antihistamines, owing to a more favourable risk–benefit profile than long-term corticosteroid use4.
Alternative and specific treatments
Intravenous or oral systemic corticosteroids are effective for AU, and for rapid control of severe disease flares in CSU4. Guidelines and experts recommend limiting oral steroid therapy to a maximum of 10 days using the lowest effective dose, due to severe adverse effects associated with long therapy duration4. Other immunosuppressant or immunomodulatory therapies were used in CSU with various success rates, including dapsone, colchicine, sulfasalazine, methotrexate, interferon, phototherapy, intravenous immunoglobulin and plasmapheresis4,184. Few of these approaches have been studied in well-designed randomized control trials, with most data published as case series; hence, all have a low-quality evidence base4. Some immune therapies have shown efficacy including anti-TNF in CSU and delayed pressure urticaria185,186, and UVA, UVB and psoralen plus UVA (commonly termed PUVA) treatment in CSU, ColdU, CholU and symptomatic dermographism187,188,189.
Evidence for other therapies, such as H2 antihistamines, leukotriene receptor antagonists, diets, such as a pseudo-allergen-free diet, and mast cell stabilizers also remains low4. However, subgroups of patients with CSU might benefit from specific approaches, for example the use of leukotriene receptor antagonists in patients who are aspirin-intolerant4,190. Tolerance induction can be useful in selected patients with ColdU, CholU or solar urticaria191, but tolerance is lost without continuous daily exposure, making the therapy impractical for many patients, for example the need for daily cold baths or showers in patients with ColdU4.
Treatment of special populations
Pregnant women, breastfeeding women, children and geriatric populations require special consideration in CSU. The over-the-counter use of first-generation antihistamines and sgAHs for allergic disease makes it likely that, at least, early pregnancy exposure to these drugs is common4. However, safety in pregnancy has been shown for cetirizine and loratadine only, and these drugs are therefore preferred192,193. The use of sgAHs such as loratadine and cetirizine is also advised in breastfeeding women4, as nursing infants can develop sedation from first-generation antihistamines secreted into breast milk194. Omalizumab is reported safe in pregnancy115,195,196,197,198 and in younger children, although current licensing is for individuals aged 12 years and older146.
In the paediatric population, several sgAHs have proven efficacy and safety: bilastine, cetirizine, desloratadine, fexofenadine, levocetirizine, loratadine and rupatadine4. The individual choice should consider country-specific availability of suitable preparations, such as syrups.
The older population is particularly sensitive to adverse effects from first-generation antihistamines, which should be avoided199. Data have shown efficacy and safety of standard doses of sgAHs and omalizumab in older patients199,200 (≥65 years old). However, older populations may be particularly susceptible to the sedative action of some sgAHs, such as cetirizine and loratadine, when recommended doses are exceeded199. Also, sgAHs updosing might cause risks in some older patients with renal, hepatic and/or cardiac disorders.
Risk–benefit profiles for immunosuppressive therapies, including corticosteroids and cyclosporine, need to be carefully considered given the increased toxic effects in all three of these patient populations. Detailed reviews of all aspects of CSU management in these groups are available115,199,201,202,203.
Therapies in development
Various therapies, targeting mediators, receptors and signalling pathways of mast and other immune cells, are in preclinical and clinical development13,164,204,205,206 (Fig. 3; all ongoing studies are listed in Table 3). Most therapies are focused on patients with antihistamine-refractory and/or omalizumab-refractory CSU.
Ligelizumab, a humanized anti-IgE mAb, had a greater affinity to IgE and showed higher efficacy than omalizumab in a phase IIb trial207 but not in the phase III PEARL1 and PEARL2 trials208.
Fenebrutinib, a selective, reversible, oral BTK inhibitor, improved CSU within the first week and demonstrated a substantial benefit in patients with antihistamine-resistant CSU including patients with circulating anti-FcεRI autoantibodies; however, reversible grade 3 liver enzyme abnormalities were observed209. Another BTK inhibitor, remibrutinib, provided improvements in UAS7 versus placebo, increased patient quality of life210 and had a favourable safety profile211; a phase III trial is ongoing.
An anti-KIT mAb, barzolvolimab, was tested in CSU and CIndU212. In 95% of patients with antihistamine-resistant ColdU and symptomatic dermographism, a complete response (negative provocation testing) was observed after a single dose of barzolvolimab. Serum tryptase and skin mast cell depletion mirrored barzolvolimab clinical activity. Similar effects of multiple-dose treatment with barzolvolimab were seen in patients with CSU213 and a phase II study is ongoing. Barzolvolimab was generally well tolerated and most adverse effects were mild or moderate in severity including urinary tract infections, headache, neutropenia and back pain in patients with CSU214 and hair colour changes, mild infusion reactions and transient changes in taste in patients with CIndU212.
Benralizumab, an anti-IL-5Rα mAb, demonstrated sustained mean changes in UAS7 from baseline to week 24 in patients with CSU in a single-centre study: both components of the UAS7, pruritus severity and wheal size, decreased to a similar extent215. In a phase III trial, dupilumab, an anti-IL-4Rα mAb, reduced itch and wheals in patients with CSU who were omalizumab-naïve sgAH-resistant, but failed to meet primary end points in patients with poor response to omalizumab216.
Lirentelimab, an anti-Siglec 8 mAb, increased UCT scores across cohorts of patients with CSU and CIndU, with complete response rates of 92%, 36%, 82% and 40% in patients who are omalizumab-naïve, patients who are omalizumab-refractory, patients with CholU and patients with symptomatic dermographism, respectively217. Secukinumab, an anti-IL-17 mAb, was effective in eight patients with CSU who were omalizumab-refractory, but onset of action was slow218. Other promising targets include tryptase, SYK, C5aR, MRGPRX2, IL-33, thymic stromal lymphopoietin (TSLP) and H4R; for some of them, drugs are already in development (Table 3 and Fig. 3).
Quality of life
The quality of life of patients with urticaria, especially CU, is often severely impaired, with marked physical, psychological, social and emotional effects11. Many different instruments are available for the assessment of quality of life impairment in patients with urticaria, ranging from generic questionnaires, such as the Short Form-36 (SF-36)219 or the Nottingham Health Profile (NHP)220, to organ-specific quality of life questionnaires, such as the Skindex-29 (ref.221) or the Dermatology Life Quality Index (DLQI)222, to disease-specific instruments such as the Chronic Urticaria-Quality of Life Questionnaire (CU-Q2oL)223, Cholinergic Urticaria-Quality of Life Questionnaire (CholU-QoL)224 and Angioedema-Quality of Life Questionnaire (AE-QoL)225 (Box 1). These questionnaires are available in validated form for many countries and languages, and the disease-specific questionnaires are recommended by the current international guideline on the diagnosis and treatment of urticaria for all patients with CU4.
Owing to its short duration, AU has only a limited effect on patient quality of life, showing mean DLQI scores <1 for questions regarding social or leisure activities and personal relationships226. Patients with AU expressed greater satisfaction with life compared with patients with CU227. Patients with CIndU have a slightly better quality of life than patients with CSU, probably due to the transient nature of stimulus, whereas a combination of CSU and CIndU showed the highest DLQI scores, indicating the largest impact on quality of life228.
The two most important drivers of quality of life impairment in CSU are the unpredictable course of the disease with the sudden onset of wheals and angioedema, and the severe pruritus. Consequences of pruritus in patients with CSU include lack of sleep, fatigue and lack of concentration12,18,109,229,230. Many patients with CSU suffer from daily or almost daily occurrence of the symptoms4,231, which can lead to patients feeling loss of control over their lives229. The signs and symptoms of urticaria also result in embarrassment, frustration, sadness and anxiety in many patients18,223,229. These feelings are further exacerbated by the fact that the importance of urticaria is underestimated by others, including treating physicians232,233. In addition to reduced quality of life, CSU also leads to limitations in social interactions, work performance and functioning in daily life, including impairments in interpersonal relationships and sexual life12,18,229,234,235.
Few direct comparisons of quality of life impairment in CU with other skin diseases exist. Independent studies with large numbers of patients in Europe and the USA have shown that quality of life impairment in CU is comparable with that in patients with moderate to severe psoriasis18,236,237 and atopic dermatitis236. Compared with non-dermatologic conditions, social quality of life impairments in patients with CU were shown to be similar to those in patients with coronary artery disease who were about to undergo arterial bypass surgery229 and worse than in patients with type I diabetes mellitus238.
Untreated CU has substantial negative effects on patient quality of life, but response to therapy is associated with a corresponding improvement in quality of life: the more effective the therapy, the better the quality of life239,240,241,242.
Outlook
Personalized treatment
AU is a disease with transient manifestations and without long-standing impairment of quality of life. However, for CU, no cure exists and the burden of disease is substantial. Future research should focus on the aetiology and pathogenesis of CU, which are currently poorly investigated and understood. Patients with CU often present with the same type of skin rash but differ in genotype, endotype and phenotype38,91,243.
Genetic contributions to urticaria pathogenesis should be elucidated in high-throughput genetic studies, including whole-genome sequencing. Presence of comorbid diseases along with the phenotypes of CU subtypes are factors that contribute to the heterogeneity of the disease and can influence the variable therapeutic response and adverse effects to available drugs. Differences in CU phenotypes have been reported, including better response to antihistamines in patients with exclusive CIndU and less psychiatric comorbidities in patients with isolated CIndU or recurrent CSU243. Defining disease endotypes and their biomarkers is a major unmet need and will enable the identification of novel therapeutic targets, refine disease management via optimization of prevention and treatment with the available drugs, and assign newly developed drugs to those patients who will have the best benefit to risk ratio. Ultimately, this will also contribute to reducing healthcare costs, with improved quality of life for patients with CU. In this regard, mast cell, basophil and eosinophil activating factors have to be identified to better characterize the aetiology and pathogenesis of CU. For example, commercially available in vitro tests for detecting serum autoantibodies need to be developed.
A multi-omics approach, using data from genomics, transcriptomics and proteomics, can help identify relevant biomarkers to objectively measure characteristics of the disease. Reliable biomarkers, either alone or in combination, can help stratify patients with CU according to phenotypes and endotypes of disease, predict progression from AU to CU and measure disease activity, relapse and response to treatment, all of which would lead to a more precise treatment approach. Markers of CSU activity are already emerging, including increased levels of CRP, IL-6 and D-dimer78,79,244. The autoimmune endotype of CSU is associated with basopenia, eosinopenia, elevated IgG anti-TPO and low total IgE levels, which were linked to poor response to treatment with antihistamines and/or omalizumab. By contrast, higher baseline levels of total IgE were associated with better clinical responses to omalizumab245, whereas low total IgE levels and a positive BHRA were linked to good response to cyclosporine246. Further promising markers exist (see Supplementary Box 1). Nevertheless, prospective multicentre studies are needed to further investigate these markers and identify new biomarkers, as well as to study pathogenesis in antihistamine-resistant urticaria and angioedema.
Drugs that failed to meet the primary end points in phase II/III trials in the classic one-size-fits-all approach may still be promising candidates in certain subpopulations of patients. Finally, drugs that have the potential to modify the disease course and induce sustained remission, especially in patients with CSU who are non-responders to sgAHs and omalizumab, are urgently needed.
Multidisciplinary approach including patients
A multidisciplinary approach should involve patients, dermatologists, allergists, rheumatologists, geneticists, pharmacologists, immunologists and researchers in these fields. Patient-reported outcome measures require more explicit recognition both in trials and in clinical practice. Specific quality of life instruments for ColdU, symptomatic dermographism and CholU already exist or are under development, but tools for the other forms of CIndU are missing and should be developed.
Patients with CU are interested in using mobile applications to monitor their disease activity and control247. For example, several mobile applications for the self-evaluation of CU and angioedema are available including UrCare, Urticaria, UrticariApp-Control Urticaria, SymTrac HIVES and TARGET My Hives248. However, existing mobile applications are limited in function and geographical reach248. The Chronic Urticaria Self Evaluation App (CRUSE) was developed in 2022 by the CRUSE core team and CRUSE advisory board, an international team of expert dermatologists and allergists, based on validated patient-reported outcome measures to help patients assess CSU activity and control and effects of CSU on quality of life. Patient’s data can be sent directly to the treating physician and included in the Chronic Urticaria Registry (CURE). Initially launched in Germany, the CRUSE team aims to cover patients with CSU worldwide in the near future. Patients with CU are increasingly seeking information from information and communications technologies with web browsers being the preferred platform to obtain general health information249, followed by email to contact physicians and WhatsApp for communicating with other patients250. The educational needs of patients can be addressed by urticaria-related websites and groups on social media, webinars and urticaria seminars.
Global approach
All urticaria stakeholders, including those in basic science, medical specialists, the pharmaceutical industry and health authorities together with patients, should contribute to creating solid evidence that enables identifying, managing and treating urticaria early with the aim of minimizing its effects on individuals and society. Awareness of up-to-date urticaria management should be increased worldwide to decrease diagnostic and therapeutic delays (Box 2). Several global initiatives have been launched to address these objectives.
First, international guidelines for diagnosis and treatment of urticaria endorsed by 50 national and international societies from 31 countries are revised every 4 years4. Second, networks of Urticaria and Angioedema Centers of Reference and Excellence (UCARE and ACARE, respectively)251,252 have been launched by the Global Allergy and Asthma European Network (GA2LEN) with the aim to provide excellence in urticaria and angioedema management and to increase the knowledge of urticaria and angioedema through research and education. Third, the CURE253 is the first international academia-driven registry to collect high-quality, real-life data on CU including patient characteristics, course of disease including cause, comorbidities, treatment response, quality of life impairment and healthcare data. Fourth, UCARE initiatives (UCARE LevelUp for physicians and UCARE 4U for patients) present up-to-date urticaria-related educational webinars. Finally, the Global Burden of Disease initiative can help assess urticaria prevalence, incidence and disability-adjusted life years14. However, as yet, these data do not differentiate between AU and CU, and therefore CU prevalence data for some countries and continents, for example Africa, are still lacking14,22.
A global approach together with mobile health and personalized medicine should help implement prevention and control of urticaria in a cost-effective manner with each patient getting the best available treatment.
References
Zuberbier, T., Balke, M., Worm, M., Edenharter, G. & Maurer, M. Epidemiology of urticaria: a representative cross-sectional population survey. Clin. Exp. Dermatol. 35, 869–873 (2010).
Lee, S. J. et al. Prevalence and risk factors of urticaria with a focus on chronic urticaria in children. Allergy Asthma Immunol. Res. 9, 212–219 (2017).
Church, M. K., Kolkhir, P., Metz, M. & Maurer, M. The role and relevance of mast cells in urticaria. Immunol. Rev. 282, 232–247 (2018).
Zuberbier, T. et al. The international EAACI/GA²LEN/EuroGuiDerm/APAAACI guideline for the definition, classification, diagnosis, and management of urticaria. Allergy 77, 734–766 (2022). This update and revision of the international guideline for urticaria developed by 64 experts from 31 countries covers the definition and classification of urticaria and outlines expert-guided and evidence-based diagnostic and therapeutic approaches for the different subtypes of urticaria.
Kolkhir, P. & Maurer, M. in Urticaria and Angioedema (eds Zuberbier, T., Grattan, C. & Maurer, M.) 77–107 (Springer International, 2021).
Weller, K. et al. Epidemiology, comorbidities, and healthcare utilization of patients with chronic urticaria in Germany. J. Eur. Acad. Dermatol. Venereol. 36, 91–99 (2022).
Schoepke, N. et al. Biomarkers and clinical characteristics of autoimmune chronic spontaneous urticaria: results of the PURIST study. Allergy 74, 2427–2436 (2019). This international multicentre study characterizes for the first time the subpopulation of patients with autoimmune CSU diagnosed by triple positivity of the ASST, basophil tests and immunoassay.
Schmetzer, O. et al. IL-24 is a common and specific autoantigen of IgE in patients with chronic spontaneous urticaria. J. Allergy Clin. Immunol. 142, 876–882 (2018). In this study, IL-24 is a common, specific and functional autoantigen of IgE antibodies in patients with CSU, suggesting autoallergic mechanism in many patients with CSU.
Giménez-Arnau, A. M. et al. The pathogenesis of chronic spontaneous urticaria: the role of infiltrating cells. J. Allergy Clin. Immunol. Pract. 9, 2195–2208 (2021).
Yanase, Y., Takahagi, S., Ozawa, K. & Hide, M. The role of coagulation and complement factors for mast cell activation in the pathogenesis of chronic spontaneous urticaria. Cells 10, 1759 (2021).
Goncalo, M. et al. The global burden of chronic urticaria for the patient and society. Br. J. Dermatol. 184, 226–236 (2021).
Maurer, M. et al. The burden of chronic spontaneous urticaria is substantial: real-world evidence from ASSURE-CSU. Allergy 72, 2005–2016 (2017).
Kolkhir, P., Elieh-Ali-Komi, D., Metz, M., Siebenhaar, F. & Maurer, M. Understanding human mast cells: lesson from therapies for allergic and non-allergic diseases. Nat. Rev. Immunol. 22, 294–308 (2022).
Peck, G. et al. Global epidemiology of urticaria: increasing burden among children, females and low-income regions. Acta Derm. Venereol. 101, adv00433 (2021).
Jadhav, R., Alcala, E., Sirota, S. & Capitman, J. Risk factors for acute urticaria in central California. Int. J. Environ. Res. Public Health 18, 3728 (2021).
Eun, S. J., Lee, J. Y., Kim, D.-Y. & Yoon, H.-S. Natural course of new-onset urticaria: results of a 10-year follow-up, nationwide, population-based study. Allergol. Int. 68, 52–58 (2019).
Parisi, C. A., Ritchie, C., Petriz, N., Torres, C. M. & Gimenez-Arnau, A. Chronic urticaria in a health maintenance organization of Buenos Aires, Argentina — new data that increase global knowledge of this disease. Bras. Dermatol. 93, 76–79 (2018).
Balp, M. M. et al. Burden of chronic urticaria relative to psoriasis in five European countries. J. Eur. Acad. Dermatol. Venereol. 32, 282–290 (2018).
Wertenteil, S., Strunk, A. & Garg, A. Prevalence estimates for chronic urticaria in the United States: a sex- and age-adjusted population analysis. J. Am. Acad. Dermatol. 81, 152–156 (2019).
Tayefi, M. et al. Chronic urticaria: a swedish registry-based cohort study on population, comorbidities and treatment characteristics. Acta Derm. Venereol. 102, adv00624 (2022).
Seo, J.-H. & Kwon, J.-W. Epidemiology of urticaria including physical urticaria and angioedema in Korea. Korean J. Intern. Med. 34, 418–425 (2019).
Fricke, J. et al. Prevalence of chronic urticaria in children and adults across the globe: systematic review with meta-analysis. Allergy 75, 423–432 (2020). This systematic review and meta-analysis reports prevalence of CU on a global level and shows considerable regional differences.
Xiao, Y. et al. The prevalence of atopic dermatitis and chronic spontaneous urticaria are associated with parental socioeconomic status in adolescents in China. Acta Derm. Venereol. 99, 321–326 (2019).
Kim, Y. S. et al. Prevalence and incidence of chronic spontaneous urticaria in the entire Korean adult population. Br. J. Dermatol. 178, 976–977 (2018).
Cantarutti, A. et al. Epidemiology of frequently occurring skin diseases in Italian children from 2006 to 2012: a retrospective, population-based study. Pediatr. Dermatol. 32, 668–678 (2015).
Lapi, F. et al. Epidemiology of chronic spontaneous urticaria: results from a nationwide, population-based study in Italy. Br. J. Dermatol. 174, 996–1004 (2016).
Chu, C.-Y., Cho, Y.-T., Jiang, J.-H., Lin, E. I.-C. & Tang, C.-H. Epidemiology and comorbidities of patients with chronic urticaria in Taiwan: a nationwide population-based study. J. Dermatol. Sci. 88, 192–198 (2017).
Balp, M.-M. et al. Prevalence and clinical characteristics of chronic spontaneous urticaria in pediatric patients. Pediatr. Allergy Immunol. 29, 630–636 (2018).
Trevisonno, J., Balram, B., Netchiporouk, E. & Ben-Shoshan, M. Physical urticaria: review on classification, triggers and management with special focus on prevalence including a meta-analysis. Postgrad. Med. 127, 565–570 (2015).
Bal, F., Kahveci, M., Soyer, O., Sekerel, B. E. & Sahiner, U. M. Chronic inducible urticaria subtypes in children: clinical features and prognosis. Pediatr. Allergy Immunol. 32, 146–152 (2021).
Balp, M.-M. et al. Clinical remission of chronic spontaneous urticaria (CSU): a targeted literature review. Derm. Ther. 12, 15–27 (2022).
Thomsen, S. F., van der Sluis, S., Kyvik, K. O. & Backer, V. Urticaria in monozygotic and dizygotic twins. J. Allergy 2012, 125367 (2012).
Yabin, H. et al. Breastfeeding duration modified the effects of neonatal and familial risk factors on childhood asthma and allergy: a population-based study. Respir. Res. 22, 41 (2021).
Rosman, Y. et al. Characterization of chronic urticaria and associated conditions in a large population of adolescents. J. Am. Acad. Dermatol. 81, 129–135 (2019).
Gaig, P. et al. Epidemiology of urticaria in Spain. J. Investig. Allergol. Clin. Immunol. 14, 214–220 (2004).
Ferrer, M. Epidemiology, healthcare, resources, use and clinical features of different types of urticaria. Alergológica 2005. J. Investig. Allergol. Clin. Immunol. 19, 21–26 (2009).
Jurado-Escobar, R. et al. Genetic variants in cytosolic phospholipase A2 associated with nonsteroidal anti-inflammatory drug-induced acute urticaria/angioedema. Front. Pharmacol. 12, 667824 (2021).
Losol, P., Yoo, H.-S. & Park, H.-S. Molecular genetic mechanisms of chronic urticaria. Allergy Asthma Immunol. Res. 6, 13 (2014).
O’Donnell, B. F. et al. Human leucocyte antigen class II associations in chronic idiopathic urticaria. Br. J. Dermatol. 140, 853–858 (1999).
Kolkhir, P. et al. Autoimmune diseases are linked to type IIb autoimmune chronic spontaneous urticaria. Allergy Asthma Immunol. Res. 13, 545–559 (2021).
Confino-Cohen, R. et al. Chronic urticaria and autoimmunity: associations found in a large population study. J. Allergy Clin. Immunol. 129, 1307–1313 (2012). This study involving more than 12,000 patients with CU points to a strong association between CU and major autoimmune diseases.
Kim, Y. S. et al. Increased risk of chronic spontaneous urticaria in patients with autoimmune thyroid diseases: a nationwide, population-based study. Allergy Asthma Immunol. Res. 9, 373–377 (2017).
Asero, R. Chronic idiopathic urticaria: a family study. Ann. Allergy Asthma Immunol. 89, 195–196 (2002).
Sahiner, U. M. et al. Chronic urticaria: etiology and natural course in children. Int. Arch. Allergy Immunol. 156, 224–230 (2011).
Irinyi, B. et al. Clinical and laboratory examinations in the subgroups of chronic urticaria. Int. Arch. Allergy Immunol. 144, 217–225 (2007).
Chen, C.-M., Huang, W.-T., Chang, L.-J., Hsu, C.-C. & Hsu, Y.-H. Peptic ulcer disease is associated with increased risk of chronic urticaria independent of Helicobacter pylori infection: a population-based cohort study. Am. J. Clin. Dermatol. 22, 129–137 (2021).
Chen, T.-L. et al. Risk of chronic spontaneous urticaria in reproductive-aged women with abnormal uterine bleeding: a population-based cohort study. J. Dermatol. 48, 1754–1762 (2021).
Sánchez, J. et al. Prevalence of inducible urticaria in patients with chronic spontaneous urticaria: associated risk factors. J. Allergy Clin. Immunol. Pract. 5, 464–470 (2017).
Vietri, J. et al. Effect of chronic urticaria on US patients: analysis of the National Health and Wellness Survey. Ann. Allergy Asthma Immunol. 115, 306–311 (2015).
Balp, M.-M., Vietri, J., Tian, H. & Isherwood, G. The impact of chronic urticaria from the patient’s perspective: a survey in five European countries. Patient 8, 551–558 (2015).
Techasatian, L., Phungoen, P., Chaiyarit, J. & Uppala, R. Etiological and predictive factors of pediatric urticaria in an emergency context. BMC Pediatrics 21, 92 (2021).
Galli, S. J. & Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 18, 693 (2022).
Doña, I. et al. Progress in understanding hypersensitivity reactions to nonsteroidal anti-inflammatory drugs. Allergy 75, 561–575 (2020).
Asero, R. Peach-induced contact urticaria is associated with lipid transfer protein sensitization. Int. Arch. Allergy Immunol. 154, 345–348 (2011).
Okayama, Y. & Kawakami, T. Development, migration, and survival of mast cells. Immunol. Res. 34, 97–115 (2006).
Gilfillan, A. M. & Tkaczyk, C. Integrated signalling pathways for mast-cell activation. Nat. Rev. Immunol. 6, 218–230 (2006).
Mendes-Bastos, P. et al. Bruton’s tyrosine kinase inhibition — an emerging therapeutic strategy in immune-mediated dermatological conditions. Allergy https://doi.org/10.1111/all.15261 (2022).
Karra, L., Berent-Maoz, B., Ben-Zimra, M. & Levi-Schaffer, F. Are we ready to downregulate mast cells? Curr. Opin. Immunol. 21, 708–714 (2009).
Kay, A. B. et al. Elevations in vascular markers and eosinophils in chronic spontaneous urticarial weals with low-level persistence in uninvolved skin. Br. J. Dermatol. 171, 505–511 (2014).
Batista, M. et al. Histopathology of chronic spontaneous urticaria with occasional bruising lesions is not significantly different from urticaria with typical wheals. J. Cutan. Pathol. 48, 1020–1026 (2021).
Ying, S., Kikuchi, Y., Meng, Q., Kay, A. B. & Kaplan, A. P. TH1/TH2 cytokines and inflammatory cells in skin biopsy specimens from patients with chronic idiopathic urticaria: comparison with the allergen-induced late-phase cutaneous reaction. J. Allergy Clin. Immunol. 109, 694–700 (2002).
Kolkhir, P. et al. Eosinopenia, in chronic spontaneous urticaria, is associated with high disease activity, autoimmunity, and poor response to treatment. J. Allergy Clin. Immunol. Pract. 8, 318–325 (2020).
MacGlashan, D. Jr, Saini, S. & Schroeder, J. T. Response of peripheral blood basophils in subjects with chronic spontaneous urticaria during treatment with omalizumab. J. Allergy Clin. Immunol. 147, 2295–2304 (2021).
Grattan, C. E., Boon, A. P., Eady, R. A. & Winkelmann, R. K. The pathology of the autologous serum skin test response in chronic urticaria resembles IgE-mediated late-phase reactions. Int. Arch. Allergy Appl. Immunol. 93, 198–204 (1990).
Saini, S. S. Basophil responsiveness in chronic urticaria. Curr. Allergy Asthma Rep. 9, 286 (2009).
Ferrer, M. Immunological events in chronic spontaneous urticaria. Clin. Transl Allergy 5, 30 (2015).
Fujisawa, D. et al. Expression of Mas-related gene X2 on mast cells is upregulated in the skin of patients with severe chronic urticaria. J. Allergy Clin. Immunol. 134, 622–6339 (2014).
Caproni, M. et al. Infiltrating cells and related cytokines in lesional skin of patients with chronic idiopathic urticaria and positive autologous serum skin test. Exp. Dermatol. 12, 621–628 (2003).
Altrichter, S. et al. The role of eosinophils in chronic spontaneous urticaria. J. Allergy Clin. Immunol. 145, 1510–1516 (2020).
Cugno, M. et al. Expression of tissue factor by eosinophils in patients with chronic urticaria. Int. Arch. Allergy Immunol. 148, 170–174 (2009).
Tedeschi, A. et al. Chronic urticaria and coagulation: pathophysiological and clinical aspects. Allergy 69, 683–691 (2014).
Cugno, M., Borghi, A., Garcovich, S. & Marzano, A. V. Coagulation and skin autoimmunity. Front. Immunol. 10, 1407 (2019).
Tedeschi, A. et al. Plasma levels and skin-eosinophil-expression of vascular endothelial growth factor in patients with chronic urticaria. Allergy 64, 1616–1622 (2009).
Dugina, T. N., Kiseleva, E. V., Chistov, I. V., Umarova, B. A. & Strukova, S. M. Receptors of the PAR family as a link between blood coagulation and inflammation. Biochemistry 67, 65–74 (2002).
Yanase, Y. et al. Coagulation factors induce human skin mast cell and basophil degranulation via activation of complement 5 and the C5a receptor. J. Allergy Clin. Immunol. 147, 1101–1104 (2021).
Cugno, M. et al. Elevated IgE to tissue factor and thyroglobulin are abated by omalizumab in chronic spontaneous urticaria. Allergy 73, 2408–2411 (2018).
Farres, M. N. et al. Activation of coagulation in chronic urticaria in relation to disease severity and activity. Allergol. Immunopathol. 43, 162–167 (2015).
Kolkhir, P., Altrichter, S., Hawro, T. & Maurer, M. C-reactive protein is linked to disease activity, impact, and response to treatment in patients with chronic spontaneous urticaria. Allergy 73, 940–948 (2018).
Kasperska-Zajac, A., Sztylc, J., Machura, E. & Jop, G. Plasma IL-6 concentration correlates with clinical disease activity and serum C-reactive protein concentration in chronic urticaria patients. Clin. Exp. Allergy 41, 1386–1391 (2011).
Asero, R. et al. Co-occurrence of IgE and IgG autoantibodies in patients with chronic spontaneous urticaria. Clin. Exp. Immunol. 200, 242–249 (2020).
Altrichter, S. et al. IgM and IgA in addition to IgG autoantibodies against FcɛRIα are frequent and associated with disease markers of chronic spontaneous urticaria. Allergy 75, 3208–3215 (2020).
Hide, M. et al. Autoantibodies against the high-affinity IgE receptor as a cause of histamine release in chronic urticaria. N. Engl. J. Med. 328, 1599–1604 (1993). In this report, functional histamine-releasing IgG autoantibodies to the α-subunit of the high-affinity IgE receptor are detected in the circulation of some patients with CU, suggesting an involvement of these autoantibodies in the pathogenesis of CU.
Kawakami, T. & Kitaura, J. Mast cell survival and activation by IgE in the absence of antigen: a consideration of the biologic mechanisms and relevance. J. Immunol. 175, 4167–4173 (2005).
Shi, C.-R. et al. IgE-mediated allergy: a rare cause of chronic spontaneous urticarial with allergen-specific immunotherapy as treatment option — a systematic review with meta-analysis from China. J. Eur. Acad. Dermatol. Venereol. 26, 533–544 (2012).
Sabroe, R. A. et al. Chronic idiopathic urticaria: comparison of the clinical features of patients with and without anti-FcεRI or anti-IgE autoantibodies. J. Am. Acad. Dermatol. 40, 443–450 (1999).
Augey, F. et al. Chronic spontaneous urticaria is not an allergic disease. Eur. J. Dermatol. 21, 349–353 (2011).
Shin, Y. S., Suh, D.-H., Yang, E.-M., Ye, Y.-M. & Park, H.-S. Serum specific IgE to thyroid peroxidase activates basophils in aspirin intolerant urticaria. J. Korean Med. Sci. 30, 705–709 (2015).
Sánchez, J., Sánchez, A. & Cardona, R. Causal relationship between anti-TPO IgE and chronic urticaria by in vitro and in vivo tests. Allergy Asthma Immunol. Res. 11, 29–42 (2019).
Sánchez, J. et al. Presence of IgE autoantibodies against eosinophil peroxidase and eosinophil cationic protein in severe chronic spontaneous urticaria and atopic dermatitis. Allergy Asthma Immunol. Res. 13, 746–761 (2021).
de Montjoye, L. et al. Increased expression of IL-24 in chronic spontaneous urticaria. Allergy 74, 1811–1813 (2019).
Kolkhir, P. et al. Autoimmune chronic spontaneous urticaria. J. Allergy Clin. Immunol. 149, 1819–1831 (2022).
Konstantinou, G. N. et al. EAACI taskforce position paper: evidence for autoimmune urticaria and proposal for defining diagnostic criteria. Allergy 68, 27–36 (2013).
Siiskonen, H. & Harvima, I. Mast cells and sensory nerves contribute to neurogenic inflammation and pruritus in chronic skin inflammation. Front. Cell. Neurosci. 13, 422 (2019).
Raap, U. et al. Increased levels of serum IL-31 in chronic spontaneous urticaria. Exp. Dermatol. 19, 464–466 (2010).
Meixiong, J. et al. Activation of mast-cell-expressed Mas-related G-protein-coupled receptors drives non-histaminergic itch. Immunity 50, 1163–11715 (2019).
McNeil, B. D. et al. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 519, 237–241 (2015).
Kühn, H. et al. Mas-related G protein-coupled receptor X2 and its activators in dermatologic allergies. J. Allergy Clin. Immunol. 147, 456–469 (2021).
Shtessel, M. et al. MRGPRX2 activation causes increased skin reactivity in patients with chronic spontaneous urticaria. J. Invest. Dermatol. 141, 678–681.e2 (2021).
Newcomb, R. W. & Nelson, H. Dermographia mediated by immunoglobulin E. Am. J. Med. 54, 174–180 (1973).
Maltseva, N. et al. Cold urticaria — what we know and what we do not know. Allergy 76, 1077–1094 (2021).
McSweeney, S. M., Sarkany, R., Fassihi, H., Tziotzios, C. & McGrath, J. A. Pathogenesis of solar urticaria: classic perspectives and emerging concepts. Exp. Dermatol. 31, 586–593 (2022).
Fukunaga, A. et al. Cholinergic urticaria: epidemiology, physiopathology, new categorization, and management. Clin. Auton. Res. 28, 103–113 (2018).
Pezzolo, E., Peroni, A., Schena, D. & Girolomoni, G. Preheated autologous serum skin test in localized heat urticaria. Clin. Exp. Dermatol. 39, 921–923 (2014).
Kulthanan, K. et al. Vibratory angioedema subgroups, features, and treatment: results of a systematic review. J. Allergy Clin. Immunol. Pract. 9, 971–984 (2021).
Cassano, N., Mastrandrea, V., Vestita, M. & Vena, G. A. An overview of delayed pressure urticaria with special emphasis on pathogenesis and treatment. Dermatol. Ther. 22, 1 (2009).
Magerl, M. et al. The definition, diagnostic testing, and management of chronic inducible urticarias — the EAACI/GA(2) LEN/EDF/UNEV consensus recommendations 2016 update and revision. Allergy 71, 780–802 (2016).
Rujitharanawong, C. et al. A systematic review of aquagenic urticaria — subgroups and treatment options. J. Allergy Clin. Immunol. Pract. https://doi.org/10.1016/j.jaip.2022.04.033 (2022).
Lehloenya, R. J., Phillips, E. J., Pasieka, H. B. & Peter, J. Recognizing drug hypersensitivity in pigmented skin. Immunol. Allergy Clin. North. Am. 42, 219–238 (2022).
Maurer, M., Ortonne, J. P. & Zuberbier, T. Chronic urticaria: an internet survey of health behaviours, symptom patterns and treatment needs in European adult patients. Br. J. Dermatol. 160, 633–641 (2009).
Nakao, A. & Nakamura, Y. Time will tell about mast cells: circadian control of mast cell activation. Allergol. Int. https://doi.org/10.1016/j.alit.2022.06.008 (2022).
Marcelino, J. et al. What basophil testing tells us about CSU patients — results of the CORSA study. Front. Immunol. 12, 742470 (2021).
Sánchez-Borges, M., Caballero-Fonseca, F. & Capriles-Hulett, A. Tolerance of nonsteroidal anti-inflammatory drug-sensitive patients to the highly specific cyclooxygenase 2 inhibitors rofecoxib and valdecoxib. Ann. Allergy Asthma Immunol. 94, 34–38 (2005).
Kolkhir, P. et al. Autoimmune chronic spontaneous urticaria detection with IgG anti-TPO and total IgE. J. Allergy Clin. Immunol. Pract. 9, 4138–41468 (2021).
Bizjak, M. et al. Risk factors for systemic reactions in typical cold urticaria: results from the COLD-CE study. Allergy 77, 2185–2219 (2022).
Kocatürk, E. et al. Effects of pregnancy on chronic urticaria: results of the PREG-CU UCARE study. Allergy 76, 3133–3144 (2021).
Sánchez-Borges, M., Capriles-Hulett, A. & Caballero-Fonseca, F. Demographic and clinical profiles in patients with acute urticaria. Allergol. Immunopathol. 43, 409–415 (2015).
Skander, D., Allenova, A., Maurer, M. & Kolkhir, P. Omalizumab is effective in patients with chronic spontaneous urticaria plus multiple chronic inducible urticaria. Eur. Ann. Allergy Clin. Immunol. 53, 91–93 (2021).
Kolkhir, P., Metz, M., Altrichter, S. & Maurer, M. Comorbidity of chronic spontaneous urticaria and autoimmune thyroid diseases: a systematic review. Allergy 72, 1440–1460 (2017).
Kocatürk, E. et al. The global impact of the COVID-19 pandemic on the management and course of chronic urticaria. Allergy 76, 816–830 (2021).
Shalom, G. et al. Chronic urticaria and atopic disorders: a cross-sectional study of 11 271 patients. Br. J. Dermatol. 177, 96–97 (2017).
Kim, B. R., Yang, S., Choi, J. W., Choi, C. W. & Youn, S. W. Epidemiology and comorbidities of patients with chronic urticaria in Korea: a nationwide population-based study. J. Dermatol. 45, 10–16 (2018).
Larenas-Linnemann, D., Saini, S. S., Azamar-Jácome, A. A. & Maurer, M. Chronic urticaria can be caused by cancer and resolves with its cure. Allergy 73, 1562–1566 (2018).
Bauer, A. T., Gorzelanny, C., Gebhardt, C., Pantel, K. & Schneider, S. W. Interplay between coagulation and inflammation in cancer: limitations and therapeutic opportunities. Cancer Treat. Rev. 102, 102322 (2022).
Liu, L., Wang, X., Wang, W., Wang, B. & Li, L. Symptomatic dermographism in Chinese population: an epidemiological study of hospital-based multicenter questionnaire survey. Curr. Med. Res. Opin. 38, 131–137 (2022).
Rujitharanawong, C., Tuchinda, P., Chularojanamontri, L., Chanchaemsri, N. & Kulthanan, K. Cholinergic urticaria: clinical presentation and natural history in a tropical country. Biomed. Res. Int. 2020, 7301652 (2020).
Asady, A. et al. Cholinergic urticaria patients of different age groups have distinct features. Clin. Exp. Allergy 47, 1609–1614 (2017).
Monfrecola, G. et al. Solar urticaria: a report on 57 cases. Am. J. Contact Dermat. 11, 89–94 (2000).
Möller, A., Henning, M., Zuberbier, T. & Czarnetzki-Henz, B. M. Epidemiology and clinical aspects of cold urticaria [German]. Hautarzt 47, 510–514 (1996).
Schoepke, N., Młynek, A., Weller, K., Church, M. K. & Maurer, M. Symptomatic dermographism: an inadequately described disease. J. Eur. Acad. Dermatol. Venereol. 29, 708–712 (2015).
Peter, J., Krause, K., Staubach, P., Wu, M. A. & Davis, M. Chronic urticaria and recurrent angioedema: clues to the mimics. J. Allergy Clin. Immunol. Pract. 9, 2220–2228 (2021).
Gusdorf, L. & Lipsker, D. Neutrophilic urticarial dermatosis: an entity bridging monogenic and polygenic autoinflammatory disorders, and beyond. J. Eur. Acad. Dermatol. Venereol. 34, 685–690 (2020).
Puhl, V. et al. A novel histopathological scoring system to distinguish urticarial vasculitis from chronic spontaneous urticaria. Clin. Transl Allergy 11, e12031 (2021).
Marzano, A. V. et al. Urticarial vasculitis: clinical and laboratory findings with a particular emphasis on differential diagnosis. J. Allergy Clin. Immunol. 149, 1137–1149 (2022).
Weller, K. et al. Clinical measures of chronic urticaria. Immunol. Allergy Clin. North. Am. 37, 35–49 (2017).
Młynek, A. et al. How to assess disease activity in patients with chronic urticaria? Allergy 63, 777–780 (2008).
Weller, K. et al. Development, validation, and initial results of the Angioedema Activity Score. Allergy 68, 1185–1192 (2013).
Ahsan, D. M. et al. Development of the Cold Urticaria Activity Score. Allergy 77, 2509–2519 (2022).
Koch, K., Weller, K., Werner, A., Maurer, M. & Altrichter, S. Antihistamine updosing reduces disease activity in patients with difficult-to-treat cholinergic urticaria. J. Allergy Clin. Immunol. 138, 1483–1485 (2016).
Weller, K. et al. Development and validation of the Urticaria Control Test: a patient-reported outcome instrument for assessing urticaria control. J. Allergy Clin. Immunol. 133, 1365–1372 (2014).
Weller, K. et al. Validation of the Angioedema Control Test (AECT) — a patient-reported outcome instrument for assessing angioedema control. J. Allergy Clin. Immunol. Pract. 8, 2050–20574 (2020).
Alves, F., Calado, R., Relvas, M., Gomes, T. & Gonçalo, M. Short courses of ciclosporin can induce long remissions in chronic spontaneous urticaria. J. Eur. Acad. Dermatol. Venereol. 36, 645–646 (2022).
Kolkhir, P., Pogorelov, D. & Kochergin, N. Chronic spontaneous urticaria associated with vitiligo and thyroiditis (autoimmune polyglandular syndrome IIIC): case series. Int. J. Dermatol. 56, 89–90 (2017).
Alvarado, D. et al. Anti-KIT monoclonal antibody CDX-0159 induces profound and durable mast cell suppression in a healthy volunteer study. Allergy https://doi.org/10.1111/all.15262 (2022). This publication describes systemic mast cell depletion associated with administration of an anti-KIT mAb that may represent a safe and novel approach to treat mast cell-driven disorders.
Maurer, M. et al. Unmet clinical needs in chronic spontaneous urticaria. A GA²LEN task force report. Allergy 66, 317–330 (2011).
Guillen-Aguinaga, S., Jauregui Presa, I., Aguinaga-Ontoso, E., Guillen-Grima, F. & Ferrer, M. Updosing nonsedating antihistamines in patients with chronic spontaneous urticaria: a systematic review and meta-analysis. Br. J. Dermatol. 175, 1153–1165 (2016).
Agache, I. et al. EAACI biologicals guidelines — omalizumab for the treatment of chronic spontaneous urticaria in adults and in the paediatric population 12–17 years old. Allergy 77, 17–38 (2022).
Doña, I. et al. NSAID-induced urticaria/angioedema does not evolve into chronic urticaria: a 12-year follow-up study. Allergy 69, 438–444 (2014).
Grattan, C. E., Francis, D. M., Slater, N. G., Barlow, R. J. & Greaves, M. W. Plasmapheresis for severe, unremitting, chronic urticaria. Lancet 339, 1078–1080 (1992).
Aleksandraviciute, L., Malinauskiene, L., Cerniauskas, K. & Chomiciene, A. Plasmapheresis: is it a potential alternative treatment for chronic urticaria? Open Med. (Wars.) 17, 113–118 (2022).
Shakouri, A., Compalati, E., Lang, D. M. & Khan, D. A. Effectiveness of Helicobacter pylori eradication in chronic urticaria: evidence-based analysis using the grading of recommendations assessment, development, and evaluation system. Curr. Opin. Allergy Clin. Immunol. 10, 362–369 (2010).
Gonzalez-Diaz, S. N. et al. Chronic urticaria and thyroid pathology. World Allergy Organ. J. 13, 100101 (2020).
Sharma, M., Bennett, C., Cohen, S. N. & Carter, B. H1-antihistamines for chronic spontaneous urticaria. Cochrane Database Syst. Rev. 11, Cd006137 (2014).
Zuberbier, T. & Maurer, M. Antihistamines in the treatment of urticaria. Adv. Exp. Med. Biol. 709, 67–72 (2010).
Leurs, R., Church, M. K. & Taglialatela, M. H1-antihistamines: inverse agonism, anti-inflammatory actions and cardiac effects. Clin. Exp. Allergy 32, 489–498 (2002).
Church, M. K. et al. Risk of first-generation H1-antihistamines: a GA(2)LEN position paper. Allergy 65, 459–466 (2010).
Zuberbier, T. et al. Double-blind crossover study of high-dose cetirizine in cholinergic urticaria. Dermatology 193, 324–327 (1996).
Staevska, M. et al. The effectiveness of levocetirizine and desloratadine in up to 4 times conventional doses in difficult-to-treat urticaria. J. Allergy Clin. Immunol. 125, 676–682 (2010).
Siebenhaar, F., Degener, F., Zuberbier, T., Martus, P. & Maurer, M. High-dose desloratadine decreases wheal volume and improves cold provocation thresholds compared with standard-dose treatment in patients with acquired cold urticaria: a randomized, placebo-controlled, crossover study. J. Allergy Clin. Immunol. 123, 672–679 (2009).
Gimenez-Arnau, A., Izquierdo, I. & Maurer, M. The use of a responder analysis to identify clinically meaningful differences in chronic urticaria patients following placebo-controlled treatment with rupatadine 10 and 20 mg. J. Eur. Acad. Dermatol. Venereol. 23, 1088–1091 (2009).
Cataldi, M., Maurer, M., Taglialatela, M. & Church, M. K. Cardiac safety of second-generation H1-antihistamines when updosed in chronic spontaneous urticaria. Clin. Exp. Allergy 49, 1615–1623 (2019).
Zhou, P., Zeng, S., Fu, L., Chen, H. & Li, L. Efficacy and safety of intensive nonsedating antihistamines for chronic spontaneous urticaria: a meta-analysis of randomized controlled trials. Int. Arch. Allergy Immunol. 183, 796–803 (2022).
Sabroe, R. A. et al. British Association of Dermatologists guidelines for the management of people with chronic urticaria 2021. Br. J. Dermatol. 186, 398–413 (2022).
Türk, M. et al. Experience-based advice on stepping up and stepping down the therapeutic management of chronic spontaneous urticaria: where is the guidance? Allergy 77, 1626–1630 (2022).
Gimenez-Arnau, A. M. & Salman, A. Targeted therapy for chronic spontaneous urticaria: rationale and recent progress. Drugs 80, 1617–1634 (2020).
Maurer, M. et al. Omalizumab for the treatment of chronic idiopathic or spontaneous urticaria. N. Engl. J. Med. 368, 924–935 (2013). This key study demonstrates omalizumab efficacy in patients with antihistamine-resistant CSU and highlights the importance of IgE–FcεRI-driven mast cell activation in this disease.
Maurer, M. et al. Omalizumab treatment in patients with chronic inducible urticaria: a systematic review of published evidence. J. Allergy Clin. Immunol. 141, 638–649 (2018).
Kaplan, A. P., Giménez-Arnau, A. M. & Saini, S. S. Mechanisms of action that contribute to efficacy of omalizumab in chronic spontaneous urticaria. Allergy 72, 519–533 (2017).
Tharp, M. D. et al. Benefits and harms of omalizumab treatment in adolescent and adult patients with chronic idiopathic (spontaneous) urticaria: a meta-analysis of “real-world” evidence. JAMA Dermatol. 155, 29–38 (2019).
Finlay, A. Y. et al. Omalizumab substantially improves dermatology-related quality of life in patients with chronic spontaneous urticaria. J. Eur. Acad. Dermatol. Venereol. 31, 1715–1721 (2017).
Buyukozturk, S. et al. Omalizumab markedly improves Urticaria Activity Scores and quality of life scores in chronic spontaneous urticaria patients: a real life survey. J. Dermatol. 39, 439–442 (2012).
Salman, A., Demir, G. & Bekiroglu, N. The impact of omalizumab on quality of life and its predictors in patients with chronic spontaneous urticaria: real-life data. Dermatol. Ther. 32, e12975 (2019).
Gastaminza, G. et al. Efficacy and safety of omalizumab (Xolair) for cholinergic urticaria in patients unresponsive to a double dose of antihistamines: a randomized mixed double-blind and open-label placebo-controlled clinical trial. J. Allergy Clin. Immunol. Pract. 7, 1599–1609.e1 (2019).
Dressler, C. et al. Chronic inducible urticaria: a systematic review of treatment options. J. Allergy Clin. Immunol. 141, 1726–1734 (2018).
Maurer, M. et al. Omalizumab is effective in symptomatic dermographism — results of a randomized placebo-controlled trial. J. Allergy Clin. Immunol. 140, 870–873 (2017).
Metz, M. et al. Omalizumab is effective in cold urticaria — results of a randomized placebo-controlled trial. J. Allergy Clin. Immunol. 140, 864–867 (2017).
Türk, M. et al. How to treat patients with chronic spontaneous urticaria with omalizumab: questions and answers. J. Allergy Clin. Immunol. Pract. 8, 113–124 (2020).
Salman, A., Ergun, T. & Gimenez-Arnau, A. M. Real-life data on the effectiveness and safety of omalizumab in monotherapy or combined for chronic spontaneous urticaria: a retrospective cohort study. J. Dermatol. Treat. 31, 204–209 (2020).
Rosenblum, J. D., Nassau, S., Fonacier, L. & Mawhirt, S. L. Concomitant treatment with omalizumab and cyclosporine for chronic spontaneous urticaria. Ann. Allergy Asthma Immunol. 125, 111–112 (2020).
Harrison, C. A., Bastan, R., Peirce, M. J., Munday, M. R. & Peachell, P. T. Role of calcineurin in the regulation of human lung mast cell and basophil function by cyclosporine and FK506. Br. J. Pharmacol. 150, 509–518 (2007).
Grattan, C. E. et al. Randomized double-blind study of cyclosporin in chronic ‘idiopathic’ urticaria. Br. J. Dermatol. 143, 365–372 (2000). This paper shows efficacy of cyclosporine in patients with CSU including autoimmune/autoreactive CSU.
Vena, G. A. et al. Cyclosporine in chronic idiopathic urticaria: a double-blind, randomized, placebo-controlled trial. J. Am. Acad. Dermatol. 55, 705–709 (2006).
Kulthanan, K. et al. Cyclosporine for chronic spontaneous urticaria: a meta-analysis and systematic review. J. Allergy Clin. Immunol. Pract. 6, 586–599 (2018).
Doshi, D. R. & Weinberger, M. M. Experience with cyclosporine in children with chronic idiopathic urticaria. Pediatr. Dermatol. 26, 409–413 (2009).
Rutkowski, K. & Grattan, C. E. H. How to manage chronic urticaria ‘beyond’ guidelines: a practical algorithm. Clin. Exp. Allergy 47, 710–718 (2017).
Magerl, M., Philipp, S., Manasterski, M., Friedrich, M. & Maurer, M. Successful treatment of delayed pressure urticaria with anti-TNF-α. J. Allergy Clin. Immunol. 119, 752–754 (2007).
Bangsgaard, N., Skov, L. & Zachariae, C. Treatment of refractory chronic spontaneous urticaria with adalimumab. Acta Derm. Venereol. 97, 524–525 (2017).
Hannuksela, M. & Kokkonen, E. L. Ultraviolet light therapy in chronic urticaria. Acta Derm. Venereol. 65, 449–450 (1985).
Borzova, E., Rutherford, A., Konstantinou, G. N., Leslie, K. S. & Grattan, C. E. Narrowband ultraviolet B phototherapy is beneficial in antihistamine-resistant symptomatic dermographism: a pilot study. J. Am. Acad. Dermatol. 59, 752–757 (2008).
Engin, B., Ozdemir, M., Balevi, A. & Mevlitoglu, I. Treatment of chronic urticaria with narrowband ultraviolet B phototherapy: a randomized controlled trial. Acta Derm. Venereol. 88, 247–251 (2008).
Pacor, M. L., Di Lorenzo, G. & Corrocher, R. Efficacy of leukotriene receptor antagonist in chronic urticaria. A double-blind, placebo-controlled comparison of treatment with montelukast and cetirizine in patients with chronic urticaria with intolerance to food additive and/or acetylsalicylic acid. Clin. Exp. Allergy 31, 1607–1614 (2001).
Beissert, S., Stander, H. & Schwarz, T. UVA rush hardening for the treatment of solar urticaria. J. Am. Acad. Dermatol. 42, 1030–1032 (2000).
Weber-Schoendorfer, C. & Schaefer, C. The safety of cetirizine during pregnancy. A prospective observational cohort study. Reprod. Toxicol. 26, 19–23 (2008).
Schwarz, E. B., Moretti, M. E., Nayak, S. & Koren, G. Risk of hypospadias in offspring of women using loratadine during pregnancy: a systematic review and meta-analysis. Drug Saf. 31, 775–788 (2008).
Ngo, E. et al. Antihistamine use during breastfeeding with focus on breast milk transfer and safety in humans: a systematic literature review. Basic Clin. Pharmacol. Toxicol. 130, 171–181 (2022).
Namazy, J. et al. The Xolair pregnancy registry (EXPECT): the safety of omalizumab use during pregnancy. J. Allergy Clin. Immunol. 135, 407–412 (2015).
Gonzalez-Medina, M., Curto-Barredo, L., Labrador-Horrillo, M. & Gimenez-Arnau, A. Omalizumab use during pregnancy for chronic spontaneous urticaria (CSU): report of two cases. J. Eur. Acad. Dermatol. Venereol. 31, e245–e246 (2017).
Ghazanfar, M. N. & Thomsen, S. F. Successful and safe treatment of chronic spontaneous urticaria with omalizumab in a woman during two consecutive pregnancies. Case Rep. Med. 2015, 368053 (2015).
Ensina, L. F., Cusato-Ensina, A. P., Camelo-Nunes, I. C. & Sole, D. Omalizumab as third-line therapy for urticaria during pregnancy. J. Investig. Allergol. Clin. Immunol. 27, 326–327 (2017).
Ventura, M. T. et al. Management of chronic spontaneous urticaria in the elderly. Drugs Aging 32, 271–282 (2015).
Nettis, E. et al. Omalizumab in elderly patients with chronic spontaneous urticaria: an Italian real-life experience. Ann. Allergy Asthma Immunol. 120, 318–323 (2018).
Saini, S., Shams, M., Bernstein, J. A. & Maurer, M. Urticaria and angioedema across the ages. J. Allergy Clin. Immunol. Pract. 8, 1866–1874 (2020).
Caffarelli, C. et al. Management of chronic urticaria in children: a clinical guideline. Ital. J. Pediatr. 45, 101 (2019).
Cornillier, H. et al. Chronic spontaneous urticaria in children — a systematic review on interventions and comorbidities. Pediatr. Allergy Immunol. 29, 303–310 (2018).
Kolkhir, P., Altrichter, S., Munoz, M., Hawro, T. & Maurer, M. New treatments for chronic urticaria. Ann. Allergy Asthma Immunol. 124, 2–12 (2020).
Hon, K. L., Li, J. T. S., Leung, A. K. C. & Lee, V. W. Y. Current and emerging pharmacotherapy for chronic spontaneous urticaria: a focus on non-biological therapeutics. Expert Opin. Pharmacother. 22, 497–509 (2021).
Wedi, B. Emerging treatments for chronic urticaria. Expert Opin. Investig. Drugs 31, 281–290 (2022).
Maurer, M. et al. Ligelizumab for chronic spontaneous urticaria. N. Engl. J. Med. 381, 1321–1332 (2019).
Novartis. Novartis provides an update on phase III ligelizumab (QGE031) studies in chronic spontaneous urticaria (CSU). Novartis https://www.novartis.com/news/media-releases/novartis-provides-update-phase-iii-ligelizumab-qge031-studies-chronic-spontaneous-urticaria-csu (2021).
Metz, M. et al. Fenebrutinib in H1 antihistamine-refractory chronic spontaneous urticaria: a randomized phase 2 trial. Nat. Med. 27, 1961–1969 (2021). The results of this study indicate that fenebrutinib, a BTK inhibitor, can be effective in reducing disease activity in patients with antihistamine-refractory CSU including patients with refractory type IIb autoimmunity.
Maurer, M. et al. Remibrutinib treatment improves quality of life in patients with chronic spontaneous urticaria. J. Allergy Clin. Immunol. 149, AB179 (2022).
Kaul, M. et al. Remibrutinib (LOU064): a selective potent oral BTK inhibitor with promising clinical safety and pharmacodynamics in a randomized phase I trial. Clin. Transl Sci. 14, 1756–1768 (2021).
Terhorst-Molawi, D. et al. The anti-KIT antibody, CDX-0159, reduces mast cell numbers and circulating tryptase and improves disease control in patients with chronic inducible urticaria (CIndU). J. Allergy Clin. Immunol. 149, AB178 (2022).
Bernstein, J. et al. Effects of multiple dose treatment with an anti-KIT antibody, CDX-0159, in chronic spontaneous urticaria [abstract 100097]. Presented at the EAACI Congress (2022).
Celldex. Celldex Therapeutics presents positive interim data from barzolvolimab phase 1b study in chronic spontaneous urticaria at EAACI 2022. GlobeNewswire https://www.globenewswire.com/news-release/2022/06/30/2472574/24180/en/Celldex-Therapeutics-Presents-Positive-Interim-Data-from-Barzolvolimab-Phase-1b-Study-in-Chronic-Spontaneous-Urticaria-at-EAACI-2022.html (2022).
Bernstein, J. A. et al. Benralizumab for chronic spontaneous urticaria. N. Engl. J. Med. 383, 1389–1391 (2020). This is the first clinical study showing that benralizumab can be effective in patients with antihistamine-resistant CSU, supporting further investigation of this treatment approach in urticaria and other mast cell-driven diseases.
Maurer, M. et al. Dupilumab significantly reduces itch and hives in patients with chronic spontaneous urticaria: results from a phase 3 trial (LIBERTY-CSU CUPID study A). J. Allergy Clin. Immunol. 149, AB312 (2022).
Altrichter, S. et al. An open-label, proof-of-concept study of lirentelimab for antihistamine-resistant chronic spontaneous and inducible urticaria. J. Allergy Clin. Immunol. 149, 1683–1690 (2022). This study shows efficacy of lirentelimab, an anti-sialic acid-binding immunoglobulin-like lectin 8 mAb, in patients with CSU and CIndU.
Sabag, D. A. et al. Interleukin-17 is a potential player and treatment target in severe chronic spontaneous urticaria. Clin. Exp. Allergy 50, 799–804 (2020). This report demonstrates efficacy of the anti-IL-17 mAb, secukinumab, in patients with antihistamine-resistant and omalizumab-resistant CSU, suggesting the role of IL-17 in disease pathogenesis.
Ware, J. E. Jr & Sherbourne, C. D. The MOS 36-item short-form health survey (SF-36). I. Conceptual framework and item selection. Med. Care 30, 473–483 (1992).
Hunt, S. M., McEwen, J. & McKenna, S. P. Measuring health status: a new tool for clinicians and epidemiologists. J. R. Coll. Gen. Pract. 35, 185 (1985).
Chren, M. M., Lasek, R. J., Quinn, L. M., Mostow, E. N. & Zyzanski, S. J. Skindex, a quality-of-life measure for patients with skin disease: reliability, validity, and responsiveness. J. Invest. Dermatol. 107, 707–713 (1996).
Finlay, A. Y. & Khan, G. K. Dermatology Life Quality Index (DLQI) — a simple practical measure for routine clinical use. Clin. Exp. Dermatol. 19, 210–216 (1994).
Baiardini, I. et al. A new tool to evaluate the impact of chronic urticaria on quality of life: Chronic Urticaria Quality of Life Questionnaire (CU-QoL). Allergy 60, 1073–1078 (2005).
Ruft, J. et al. Development and validation of the Cholinergic Urticaria Quality-of-Life Questionnaire (CholU-QoL). Clin. Exp. Allergy 48, 433–444 (2018).
Weller, K. et al. Development and construct validation of the Angioedema Quality of Life Questionnaire. Allergy 67, 1289–1298 (2012).
Kulthanan, K., Chiawsirikajorn, Y. & Jiamton, S. Acute urticaria: etiologies, clinical course and quality of life. Asian Pac. J. Allergy Immunol. 26, 1–9 (2008).
Zelić, S. B., Rubeša, G., Brajac, I., Peitl, M. V. & Pavlović, E. Satisfaction with life and coping skills in the acute and chronic urticaria. Psychiatr. Danub. 28, 34–38 (2016).
Maurer, M. et al. Chronic urticaria treatment patterns and changes in quality of life: AWARE study 2-year results. World Allergy Organ. J. 13, 100460 (2020).
O’Donnell, B. F., Lawlor, F., Simpson, J., Morgan, M. & Greaves, M. W. The impact of chronic urticaria on the quality of life. Br. J. Dermatol. 136, 197–201 (1997).
Mann, C., Dreher, M., Weess, H. G. & Staubach, P. Sleep disturbance in patients with urticaria and atopic dermatitis: an underestimated burden. Acta Derm. Venereol. 100, adv00073 (2020).
Saini, S. S. et al. Efficacy and safety of omalizumab in patients with chronic idiopathic/spontaneous urticaria who remain symptomatic on H1 antihistamines: a randomized, placebo-controlled study. J. Invest. Dermatol. 135, 67–75 (2015).
Hoskin, B., Ortiz, B., Paknis, B. & Kavati, A. Exploring the real-world profile of refractory and non-refractory chronic idiopathic urticaria in the USA: clinical burden and healthcare resource use. Curr. Med. Res. Opin. 35, 1387–1395 (2019).
Sussman, G. et al. Angioedema in chronic spontaneous urticaria is underdiagnosed and has a substantial impact: analyses from ASSURE-CSU. Allergy 73, 1724–1734 (2018).
Baiardini, I. et al. Quality of life and patients’ satisfaction in chronic urticaria and respiratory allergy. Allergy 58, 621–623 (2003).
Ertas, R., Erol, K., Hawro, T., Yilmaz, H. & Maurer, M. Sexual functioning is frequently and markedly impaired in female patients with chronic spontaneous urticaria. J. Allergy Clin. Immunol. Pract. 8, 1074–1082 (2020).
Grob, J. J., Revuz, J., Ortonne, J. P., Auquier, P. & Lorette, G. Comparative study of the impact of chronic urticaria, psoriasis and atopic dermatitis on the quality of life. Br. J. Dermatol. 152, 289–295 (2005).
Mendelson, M. H. et al. Patient-reported impact of chronic urticaria compared with psoriasis in the United States. J. Dermatol. Treat. 28, 229–236 (2017).
Brzoza, Z. et al. Chronic spontaneous urticaria and type 1 diabetes mellitus — does quality of life impairment always reflect health danger? J. Clin. Med. 9, 2505 (2020).
Gimenez-Arnau, A. et al. Ligelizumab improves sleep interference and disease burden in patients with chronic spontaneous urticaria. Clin. Transl Allergy 12, e12121 (2022).
Maurer, M. et al. Antihistamine-resistant chronic spontaneous urticaria remains undertreated: 2-year data from the AWARE study. Clin. Exp. Allergy 50, 1166–1175 (2020).
Nochaiwong, S. et al. Impact of pharmacological treatments for chronic spontaneous urticaria with an inadequate response to H1-antihistamines on health-related quality of life: a systematic review and network meta-analysis. J. Allergy Clin. Immunol. Pract. 10, 297–308 (2022).
Di Agosta, E. et al. Quality of life in patients with allergic and immunologic skin diseases: in the eye of the beholder. Clin. Mol. Allergy 19, 26 (2021).
Curto-Barredo, L., Pujol, R. M., Roura-Vives, G. & Gimenez-Arnau, A. M. Chronic urticaria phenotypes: clinical differences regarding triggers, activity, prognosis and therapeutic response. Eur. J. Dermatol. 29, 627–635 (2019).
Asero, R. et al. Severe chronic urticaria is associated with elevated plasma levels of D-dimer. Allergy 63, 176–180 (2008).
Marzano, A. V. et al. Predictors of response to omalizumab and relapse in chronic spontaneous urticaria: a study of 470 patients. J. Eur. Acad. Dermatol. Venereol. 33, 918–924 (2019).
Fok, J. S., Kolkhir, P., Church, M. K. & Maurer, M. Predictors of treatment response in chronic spontaneous urticaria. Allergy 76, 2965–2981 (2021).
Cherrez-Ojeda, I. et al. Chronic urticaria patients are interested in apps to monitor their disease activity and control: A UCARE CURICT analysis. Clin. Transl Allergy 11, e12089 (2021).
Antó, A. et al. Automatic screening of self-evaluation apps for urticaria and angioedema shows a high unmet need. Allergy 76, 3810–3813 (2021).
Maurer, M. et al. The usage, quality and relevance of information and communications technologies in patients with chronic urticaria: a UCARE study. World Allergy Organ. J. 13, 100475 (2020).
Cherrez-Ojeda, I. et al. How are patients with chronic urticaria interested in using information and communication technologies to guide their healthcare? A UCARE study. World Allergy Organ. J. 14, 100542 (2021).
Maurer, M. et al. Definition, aims, and implementation of GA2LEN Urticaria Centers of Reference and Excellence. Allergy 71, 1210–1218 (2016). This publication describes the aims, the requirements and deliverables, the application process and the audit and accreditation protocol for Urticaria Centers of Reference and Excellence.
Maurer, M. et al. Definition, aims, and implementation of GA2LEN/HAEi Angioedema Centers of Reference and Excellence. Allergy 75, 2115–2123 (2020).
Weller, K. et al. The Chronic Urticaria Registry: rationale, methods and initial implementation. J. Eur. Acad. Dermatol. Venereol. 35, 721–729 (2021). This report describes the rationale, methods and implementation of the CURE.
Mao, M. et al. Clinical difference between single subtype and mixed subtype chronic urticaria: a retrospective study. Indian J. Dermatol. Venereol. Leprol. 88, 171–176 (2022).
Prosty, C. et al. Prevalence, management, and anaphylaxis risk of cold urticaria: a systematic review and meta-analysis. J. Allergy Clin. Immunol. Pract. 10, 586–5964 (2022).
Maul, J.-T. et al. Canakinumab lacks efficacy in treating adult patients with moderate to severe chronic spontaneous urticaria in a phase II randomized double-blind placebo-controlled single-center study. J. Allergy Clin. Immunol. Pract. 9, 463–4683 (2021).
Oliver, E. T. et al. Effects of an oral CRTh2 antagonist (AZD1981) on eosinophil activity and symptoms in chronic spontaneous urticaria. Int. Arch. Allergy Immunol. 179, 21–30 (2019).
Dickson, M. C. et al. Effects of a topical treatment with spleen tyrosine kinase inhibitor in healthy subjects and patients with cold urticaria or chronic spontaneous urticaria: results of a phase 1a/b randomised double-blind placebo-controlled study. Br. J. Clin. Pharmacol. 87, 4797–4808 (2021).
Gericke, J. et al. Serum autoreactivity predicts time to response to omalizumab therapy in chronic spontaneous urticaria. J. Allergy Clin. Immunol. 139, 1059–1061.e1 (2017).
Kolkhir, P. et al. Management of chronic spontaneous urticaria: a worldwide perspective. World Allergy Organ. J. 11, 14 (2018).
Maurer, M. et al. Differences in chronic spontaneous urticaria between Europe and Central/South America: results of the multi-center real world AWARE study. World Allergy Organ. J. 11, 32 (2018).
Wagner, N. et al. Patients with chronic urticaria remain largely undertreated: results from the DERMLINE online survey. Dermatol. Ther. 11, 1027–1039 (2021).
Cherrez, A. et al. Knowledge and management of chronic spontaneous urticaria in Latin America: a cross-sectional study in Ecuador. World Allergy Organ. J. 10, 21 (2017).
Zuberbier, T. & Bernstein, J. A. A comparison of the United States and international perspective on chronic urticaria guidelines. J. Allergy Clin. Immunol. Pract. 6, 1144–1151 (2018).
Wilches, P., Wilches, P., Calderon, J. C., Cherrez, A. & Cherrez Ojeda, I. Omalizumab for chronic urticaria in Latin America. World Allergy Organ. J. 9, 1–5 (2016).
Acknowledgements
The authors thank H. Bonnekoh, K. Brockow, T. Buttgereit, L. Ensina, M. Hide, T. Jacob, A. Kasperska-Zając, L. Kiefer, E. Kocatürk, O. M. E. Calderón LLosa and P. Pyatilova for providing clinical pictures during the writing of this publication.
Author information
Authors and Affiliations
Contributions
Introduction (P.K., A.M.G.-A., K.K., M. Maurer); Epidemiology (P.K., K.K., M. Maurer); Mechanisms/pathophysiology (P.K., M. Metz, M. Maurer); Diagnosis/screening/prevention (P.K., A.M.G.-A., K.K., M. Maurer); Management (P.K., A.M.G.-A., J.P., M. Maurer); Quality of life (P.K., M. Metz, M. Maurer); Outlook (P.K., A.M.G.-A., J.P., M. Maurer); Overview of the Primer (P.K., M. Maurer).
Corresponding authors
Ethics declarations
Competing interests
P.K. received speakers fees, honoraria or travel support from Novartis and Roche. A.M.G.-A. is/was a medical adviser for Uriach Pharma/Neucor, Genentech, Novartis, FAES, GSK, Sanofi–Regeneron, Amgen, Thermo Fisher Scientific, Almirall, Celldex and LEO Pharma; received research grants supported by Uriach Pharma, Novartis and Instituto Carlos III — FEDER; and participated in educational activities for Uriach Pharma, Novartis, Genentech, Menarini, LEO Pharma, GSK, MSD, Almirall, Sanofi and Avene. K.K. received grants/research support from Novartis and honoraria or consultation fees from Novartis, Menarini and Sanofi. J.P. received speakers fees, honoraria or travel support from Takeda, CSL Behring, Janssen, Novartis and Sanofi and Pharming, and educational research grants from Takeda and Novartis. M. Metz has received honoraria (advisory board, speaker) from Amgen, ArgenX, Celldex, Moxie, Novartis, Roche and Sanofi. M. Maurer is or recently was a speaker and/or adviser for and/or has received research funding from Astria, Allakos, Alnylam, Amgen, Aralez, ArgenX, AstraZeneca, BioCryst, Blueprint, Celldex, Centogene, CSL Behring, Dyax, FAES, Genentech, GIInnovation, GSK, Innate Pharma, Kalvista, Kyowa Kirin, LEO Pharma, Lilly, Menarini, Moxie, Novartis, Pfizer, Pharming, Pharvaris, Roche, Sanofi/Regeneron, Shire/Takeda, Third Harmonic Bio, UCB and Uriach.
Peer review
Peer review information
Nature Reviews Disease Primers thanks Y. M. Ye, Z. K. Brzoza, A. Marzano, K. V. Godse and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Informed consent
The authors affirm that human research participants provided written informed consent for publication of the images in Figs. 1 and 4.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Degranulation
-
The process by which cytoplasmic granules (originating from mast cells or other granulocytes) release their contents including preformed mediators such as histamine.
- Wheals
-
Intermittent, circumscribed, superficial, central swellings, often surrounded by reflex erythema, that cause itching or burning sensations.
- Angioedema
-
An intermittent, localized and self-limiting swelling of the subcutaneous or submucosal tissue, due to a temporary increase in vascular permeability, causing tingling, burning, tightness and, sometimes, pain.
- Mast cells
-
Immune cells of the myeloid lineage that contain many granules rich in histamine and other mediators and are present in connective tissues throughout the body, especially skin, near blood vessels, lungs and intestines.
- Cysteinyl leukotrienes
-
A family of inflammatory lipid mediators synthesized from arachidonic acid by various cells including mast cells, eosinophils and basophils.
- Eotaxins
-
A CC chemokine subfamily of potent eosinophil chemoattractants including CCL11 (eotaxin 1), CCL24 (eotaxin 2) and CCL26 (eotaxin 3).
- Autologous serum skin test
-
(ASST). Intradermal injection of autologous serum to assess autoreactivity associated with autoantibodies and other histamine-releasing factors; defined as a serum-induced wheal response with a diameter ≥1.5 mm compared with that of the saline-induced response.
- Tissue factor
-
The high-affinity receptor expressed on eosinophils, epithelial cells and other cell types, and cofactor for factor VII (FVII)/FVIIa important for initiation of blood coagulation.
- Oral drug provocation testing
-
The gold standard for diagnosis of NSAID-mediated reactions including identification of a culprit drug, confirming or excluding cross-reactivity, and finding the most likely tolerated alternative drug.
- Provocation testing
-
A method to confirm the diagnosis of chronic inducible urticaria (CIndU) using established protocols.
- GRADE methodologies
-
(Grading of Recommendations Assessment, Development and Evaluation methodologies). A transparent framework for rating the quality of evidence and grading the strength of recommendations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kolkhir, P., Giménez-Arnau, A.M., Kulthanan, K. et al. Urticaria. Nat Rev Dis Primers 8, 61 (2022). https://doi.org/10.1038/s41572-022-00389-z
Accepted:
Published:
DOI: https://doi.org/10.1038/s41572-022-00389-z
This article is cited by
-
Gut microbiota facilitate chronic spontaneous urticaria
Nature Communications (2024)
-
Pressure and Skin: A Review of Disease Entities Driven or Influenced by Mechanical Pressure
American Journal of Clinical Dermatology (2024)
-
Welche Augmentations- und Triggerfaktoren sind bei der Urtikaria relevant?
Die Dermatologie (2024)
-
Spontanremission der Urtikaria: Gibt es das, und wenn ja, wann?
Die Dermatologie (2024)
-
Erkennen und Management relevanter Komorbiditäten bei chronischer spontaner Urtikaria
Die Dermatologie (2024)