Abstract
Dilated cardiomyopathy (DCM) is a clinical diagnosis characterized by left ventricular or biventricular dilation and impaired contraction that is not explained by abnormal loading conditions (for example, hypertension and valvular heart disease) or coronary artery disease. Mutations in several genes can cause DCM, including genes encoding structural components of the sarcomere and desmosome. Nongenetic forms of DCM can result from different aetiologies, including inflammation of the myocardium due to an infection (mostly viral); exposure to drugs, toxins or allergens; and systemic endocrine or autoimmune diseases. The heterogeneous aetiology and clinical presentation of DCM make a correct and timely diagnosis challenging. Echocardiography and other imaging techniques are required to assess ventricular dysfunction and adverse myocardial remodelling, and immunological and histological analyses of an endomyocardial biopsy sample are indicated when inflammation or infection is suspected. As DCM eventually leads to impaired contractility, standard approaches to prevent or treat heart failure are the first-line treatment for patients with DCM. Cardiac resynchronization therapy and implantable cardioverter–defibrillators may be required to prevent life-threatening arrhythmias. In addition, identifying the probable cause of DCM helps tailor specific therapies to improve prognosis. An improved aetiology-driven personalized approach to clinical care will benefit patients with DCM, as will new diagnostic tools, such as serum biomarkers, that enable early diagnosis and treatment.
Similar content being viewed by others
Introduction
Cardiomyopathies are disorders of the cardiac muscle that cause mechanical and/or electrical dysfunction that result in dilated, hypertrophic or restrictive pathophysiology1. Dilated cardiomyopathy (DCM) is a non-ischaemic heart muscle disease with structural and functional myocardial abnormalities. The clinical picture of DCM is defined by left or biventricular dilatation and systolic dysfunction in the absence of coronary artery disease, hypertension, valvular disease or congenital heart disease2. The American Heart Association classifies DCM as genetic, mixed or acquired, whereas the European Society of Cardiology (ESC) groups cardiomyopathy into familial (that is, genetic) or nonfamilial (that is, nongenetic) forms3,4. The WHO defines DCM as a serious cardiac disorder in which structural or functional abnormalities of the heart muscle can lead to substantial morbidity and mortality owing to complications such as heart failure and arrhythmia5.
Decades of research have revealed diverse aetiologies for DCM, including genetic mutations, infections, inflammation, autoimmune diseases, exposure to toxins and endocrine or neuromuscular causes5 (Box 1). Idiopathic and familial disease are the most commonly reported causes of DCM. To accurately identify the aetiology of DCM, a wide array of non-invasive and invasive examinations are needed6,7, but wide variability exists globally in the availability of advanced diagnostic tools (such as genetic testing for inborn metabolic errors, for example, and comprehensive cardiac imaging). Thus, many cases of DCM could be classified as idiopathic when in fact a thorough evaluation would enable a precise diagnosis of the underlying aetiology. Ongoing advances in genetic sequencing and molecular methods are increasing the resources available to clinicians to determine the disease aetiology and provide a tailored risk stratification, resources which improve patient care and could potentially reverse disease progression. Further complicating the clinically heterogeneous presentation of DCM are differences that exist according to sex, age of onset (which can have an acute or progressive presentation), rate of progression, risk of development of overt heart failure and probability of sudden cardiac death8,9. Finally, both inherited predisposition and environmental factors play an important part in the natural history of disease.
Epidemiology
In a small (45 patients) population-based study from Minnesota, USA, the estimated age-adjusted and sex-adjusted incidence of idiopathic DCM between 1975 and 1984 was 6 per 100,000 person-years10. The prevalence of idiopathic DCM in this study as of 1985 was estimated at 36.5 per 100,000 individuals. In a 2013 review, the prevalence of DCM was estimated to be >1 per 250 individuals on the basis of recent clinical trial and related data11; this result is particularly relevant as reliable population-based studies of the prevalence of hypertrophic cardiomyopathy have estimated it to be at least 1 per 500 individuals12, and the review article estimates that DCM is at least as common in the population as hypertrophic cardiomyopathy11. In 2015, the Global Burden of Disease study estimated the global prevalence of cardiomyopathy at 2.5 million cases — an increase of 27% in just 10 years13.
Mortality
In 2010, the estimated mortality associated with cardiomyopathy was 5.9 per 100,000 global population, corresponding to ~403,000 deaths — an increase from 5.4 per 100,000 in 1990 (ref.14). These estimates may be affected by bias due to misclassification and missing or incomplete data, among others. In addition, there is a need for international consortia to group cohorts of patients so as to overcome the issue of the low number of events during follow-up. The annual change in cardiomyopathy as a percentage of all deaths from 1990 to 2017 is shown in Fig. 1.
The map shows the annual percentage change in number of deaths (per 100,000 individuals) due to cardiomyopathy or myocarditis. Data include all ages and both sexes between 1990 and 2017. A decrease in deaths from cardiomyopathy is probably due to improvements in medication and health care; the reasons for the increase in deaths are less clear. Data from https://vizhub.healthdata.org/gbd-compare/. Accessed 21 February 2019.
Most causes of heart failure arising from DCM occur from pump failure (70%) due to dilatation, whereas sudden cardiac death from arrhythmias accounts for the remaining 30%15,16,17,18,19,20. Survival of patients with DCM who develop heart failure has improved over time in developed countries. Improvements in survival have been attributed in large part to the increased availability and use of angiotensin-converting enzyme (ACE) inhibitors, β-blockers, implantable cardioverter–defibrillators (ICDs) and heart transplantation21,22,23. However, these interventions are not available to most of the world’s population. Furthermore, the effect of medications, ventricular devices and heart transplantation on the natural history of DCM is not well characterized, and despite advances in the management of heart failure in patients with DCM, there is still substantial mortality.
Risk factors
The risk of developing congestive heart failure is ~30% higher in black than white individuals, a finding that is not explained by the confounding variables of hypertension or socio-economic factors24. Several studies report a relative risk of mortality from DCM of 1.2–1.5 for black compared with age-matched white people24,25. Black ethnicity is also associated with poor outcome in acute idiopathic DCM26,27.
The EuroHeart Failure Survey II examined data according to sex and found that DCM occurred more often in men than in women28. However, most epidemiological studies do not report data according to sex, and studies comparing sex differences in DCM phenotype, severity and outcome are lacking18,19,20,28,29. The population-based epidemiological study of idiopathic DCM in Minnesota reported a male to female ratio of 3:1 (refs10,18). In the multicentre IMAC (Intervention in Myocarditis and Acute Cardiomyopathy)-2 study, men with a left ventricular (LV) ejection fraction (LVEF; the fraction of the volume of fluid ejected from the left ventricle with each contraction; physiological values are ≥50%) ≤40%, <6 months of heart failure symptom duration and myocarditis (inflammation of the myocardium) or acute onset of idiopathic DCM (idiopathic DCM of <6 months symptom duration) had worse recovery (assessed by the New York Heart Association (NYHA) classification) and reduced transplant-free survival than women26, a finding that is similar to data on patients with heart failure in general20.
Paediatric DCM
In a multisite study in the USA and Canada, DCM was the most common form of cardiomyopathy among children (individuals of <18 years of age): 66% had idiopathic DCM, whereas of those with DCM due to known causes, 46% had myocarditis and 26% had neuromuscular disease30. The annual incidence of DCM in children ranges from 0.18 to 0.73 per 100,000 person-years19,31,32,33,34,35. The annual incidence was higher in boys than in girls, in black individuals than in white and in infants (<1 year of age) than in older children19. In North America, 34% of children with DCM progress to heart failure or receive a heart transplant within 1 year of diagnosis, a figure that increases to 49% 5 years after diagnosis36. Risk factors for increased risk of death or heart transplantation include congestive heart failure, increased LV end-systolic dimension z-score (a parameter often used in children as a surrogate of LVEF) at diagnosis and older age at diagnosis34,36.
Mechanisms/pathophysiology
Animal models have demonstrated that LV dilatation results from remodelling and fibrosis. Similarly, in patients with DCM, there is evidence of scar tissue, with the left ventricle assuming a spherical shape. Pathophysiological changes include a decrease in stroke volume (blood volume pumped by the left ventricle at each contraction) and cardiac output (the amount of blood pumped by the heart per minute), impaired ventricular filling and an increase in end-diastolic pressure (the amount of blood in the ventricle when relaxed, which is a measure of dilation). Compensatory changes in the vascular system include an increase in systemic vascular resistance, a decrease in arterial compliance and an increase in venous pressure and circulating blood volume. Both cardiac preload (stretching of the heart wall before contraction) and afterload (the force needed for contraction to eject blood from the heart) are increased, with increased afterload resulting in elevated wall stress. Diastole involves both active relaxation (early diastole) and passive compliance (mid-to-late diastole). In DCM, diastolic dysfunction that affects both components can accompany a reduction in systolic function. Impaired ventricular relaxation results in reduced rapid ventricular filling.
The wide spectrum of potential causes of DCM and the often-slow progression from initiation of cardiac damage to eventual signs and symptoms of heart failure make intervening in the disease process early enough to prevent irreversible damage particularly challenging.
DCM genetics
The development of next-generation sequencing has made possible a new era in human genetics and genomics for clinical and research purposes37. Several genes that, when mutated, are associated with the development of DCM have been identified (Table 1). However, gene and gene variant curation (that is, distinguishing between pathogenetic and non-pathogenetic genes and specific gene variants) remains a crucial unresolved issue. Moreover, the extent of genetic aetiology in sporadic DCM remains unresolved11. Finally, the interrelationship between environmental and genetic causes of DCM remains minimally studied38, although data show that alcohol exposure facilitates the development of DCM in individuals with rare variants in established DCM-associated genes39.
Familial DCM is commonly defined when at least two closely related (first-degree or second-degree) family members meet diagnostic criteria for idiopathic DCM11. Substantial evidence now is available to indicate that familial DCM can be considered to have a genetic basis11,40,41, and clinical genetic testing services of large panels of genes implicated in DCM are now routinely available for patients with DCM and their family members42. Family-based studies have established that 15–30% of patients with DCM may be diagnosed with familial DCM if their family members undergo clinical screening11,43. Large multigenerational familial DCM pedigrees historically have been the starting point for most DCM-associated gene discovery. Such multigenerational pedigrees have provided robust statistical genetic evidence for variant causality in DCM-associated genes. The genes most commonly known to cause DCM, which include LMNA44, MYH7 (ref.45), TNNT2 (ref.45), TTN46, RBM20 (ref.47), BAG3 (ref.48) and others, were identified initially in large DCM pedigrees (Fig. 2). TTN truncating mutations are a common cause of DCM, occurring in ~25% of familial cases of DCM and in 18% of sporadic cases49. Readers are encouraged to view the extensive DCM-associated gene lists in review articles11,40,50,51,52.
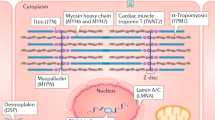
The ‘defective force transmission’ hypothesis postulates that the cytoskeleton provides an intracellular scaffolding that is important for transmission of force from the sarcomere to the extracellular matrix and for protection of the myocyte from external mechanical stress. Thus, defects in cytoskeletal proteins could predispose to dilated cardiomyopathy (DCM) by reducing force transmission and/or resistance to mechanical stress. Contractile dysfunction of myofibrils plays a central part in initiation and progression of DCM. The sarcomere is composed of numerous proteins, and mutations in several of them have been associated with DCM, including actin, α-cardiac muscle 1 (encoded by ACTC1), myosin-binding protein C, cardiac type (encoded by MYBPC3), myosin chains (encoded by MYL2, MYL3, MYH6 and MYH7) and tropomyosin-α1 chain (encoded by TPM1) (see Table 1). Mutations in genes encoding cardiac troponins (encoded by TNNT2, TNNC1 and TNNI3) are also linked directly to disordered force generation221,222. MYH7 mutations have been predicted to disrupt the actin–myosin binding and crossbridge function, whereas mutations in TTN change viscoelasticity properties223. Mutations in other non-contractile proteins (for example, a co-chaperone for heat shock protein 70 (HSP70) and heat shock cognate 70 chaperone proteins, encoded by BAG3) may induce defects in cell signalling pathways that modify cardiac response48,224. Mutations in phospholamban (encoded by PLN), a key calcium signalling protein, have been directly linked to abnormal contractility225. Variants in desmosomal proteins including desmin (encoded by DES) desmocollin 2 (encoded by DSC2), desmoglein 2 (encoded by DSG2), desmoplakin (encoded by DSP) and plakophilin 2 (encoded by PKP2) are most commonly associated with arrhythmogenic right ventricular cardiomyopathy, but mutations in these genes have also been implicated in DCM226. In some patients with genetic DCM, a particular gene defect may be suggested by cardiac conduction abnormalities. For example, variants of lamin A/C (LMNA; which is part of a protein structure associated with the inner nuclear membrane) are associated with high rates of conduction system disease, ventricular arrhythmias and sudden cardiac death41. However, in most cases of DCM, there are no specific distinguishing phenotype features. SERCA2a, sarcoplasmic/endoplasmic reticulum calcium ATPase 2a. Adapted from ref.11, Springer Nature Limited.
Although a genetic basis for familial DCM is well established, most cases of DCM seem to be sporadic; that is, even when the family members of patients newly diagnosed with idiopathic DCM are clinically screened11,43, most family members have no evidence of DCM, and, therefore, patients are eventually diagnosed with nonfamilial (sporadic) DCM. To date, there is no published large multicentre study of families whose members have been systematically clinically screened for DCM and have also undergone exome or genome sequencing to identify a possible genetic cause. A multisite study funded by the US National Heart, Lung and Blood Institute is now underway to test the hypothesis that DCM, whether familial or nonfamilial, has mostly a rare variant genetic basis53.
Gene curation
Curating the strength of evidence suggesting that a gene can (when mutated) cause DCM is fundamental to clinical care, as one cannot adjudicate a variant as pathogenetic or probably pathogenetic if the relevance of the gene as a cause of DCM is uncertain. Gene curation is also of fundamental importance for the discovery of DCM-associated genes. What level of evidence is required to change the classification of a gene from a gene of uncertain significance for DCM to a gene of confirmed relevance and one that is routinely included in clinical testing panels is unclear. The ideal gene discovery scenario is having one or more large multigenerational DCM pedigrees with numerous affected family members, or several smaller families, each family having the same very rare gene variant, and unaffected individuals who do not carry that variant. Additional supporting evidence would be genome-wide sequencing that has excluded the presence of rare variants in other established DCM-associated genes. However, this scenario is rarely achieved as DCM is usually a lethal condition, and few large families exist. For this reason, most studies include cohorts of individual patients with DCM, whose DCM-associated candidate genes are sequenced and rare coding variants are identified, and the numbers of individuals with rare variants in a gene are compared with the numbers of individuals in large, publicly accessible reference data sets who also carry rare coding variants of the same type37,54. The largest of these public databases is the Genome Aggregation Database (gnomAD)55, an expansion of the Exome Aggregation Consortium (ExAC) database, now having exome or genome sequences of >140,000 individuals from the general population, presumably with no known DCM. In response to the need for a systematic approach to gene curation56, the ClinGen consortium57, a large collaborative effort sponsored by the US National Human Genome Research Institute57, has published a systematic approach to integrate genetic and experimental evidence for gene–disease relationships. This approach uses a semi-quantitative scoring system that has been validated across several Mendelian diseases, and as a result, the evidence supporting the association between a specific gene and a disease is classified as definitive, strong, moderate, limited, absent or conflicting. This approach has been used by the ClinGen Cardiovascular Disease Working Group57 for gene curation for hypertrophic cardiomyopathy58, and curation of genes associated with arrhythmogenic right ventricular cardiomyopathy and DCM is now underway.
Variant curation
Like gene curation, variant curation is also challenging even in well-established DCM-associated genes. In patients with DCM, the sequencing of established or candidate DCM-associated genes can lead to the identification of rare variations (for example, allele frequencies <0.1% to <0.001%), and commonly >50% of all such very rare variants are unique37. However, the presence of a unique or very rare variant does not necessarily assign causality of DCM to that variant; furthermore, the plethora of rare variants identified with exome or genome sequencing has challenged prior variant adjudication approaches37.
The availability of very large genetic data sets from diverse ethnic groups has brought this new challenge into focus59. Some variants that had been considered rare by comparison to control DNA samples from a few hundred individuals are more common than previously thought and unlikely to be disease causing. Studies have leveraged the ExAC database of >60,000 exome sequences55 and have shown that the frequency of rare variants in control populations mirrors that of some genes proposed as relevant for hypertrophic cardiomyopathy54, thereby questioning the relevance attributed to such variants. This paradigm is also applicable to DCM. In 2015, the American College of Medical Genetics substantially revised the approach to variant interpretation60 for clinical sequencing, making the approach and application of standards much more rigorous, with an overall effect of making interpretation more conservative to adjudicate variants as pathogenetic or probably pathogenetic. A variant is considered to be probably pathogenetic if there is a ≥90% probability that the variant is pathogenetic. All variants not meeting these rigorous standards are noted as variants of uncertain significance. Furthermore, the recently developed but rapidly expanding database ClinVar61 now has assertions of variant pathology in a publicly accessible database. ClinGen is interacting with ClinVar to populate ClinVar with curated variants in medically relevant genes, including those associated with cardiomyopathies.
The final frontier for DCM genetics may be the deciphering of the interrelationships, if any, of genetic disease pathways with other established nongenetic disease mechanisms, including, for example, immunological or infectious aetiologies62. Large studies linking genetic and nongenetic causes in a unified research approach have yet to be accomplished.
Inflammation
Damage to the myocardium, whether from a genetic or environmental cause, triggers inflammation and recruits immune cells to the heart to repair the myocardium; the most common causes of inflammatory DCM are infections and autoimmunity. Pathological examination of myocardial biopsy samples (or autopsies) of patients with DCM frequently uncovers evidence of an inflammatory cell infiltrate and gene expression patterns compatible with immune cell activation5,63. Immune cells that contribute to remodelling include mast cells, macrophages (M2 macrophages, also known as alternatively activated macrophages, and myeloid-derived suppressor cells), T helper 2 (TH2) and TH17 cells and, in the case of autoimmune aetiologies, B cells that produce autoantibodies that form immune complexes with self-antigens and complement components64,65. Immune cells release cytokines, such as transforming growth factor-β1 (TGFβ1), IL-4, IL-1β, IL-17A, IL-33 and tumour necrosis factor (TNF), among other mediators, that promote remodelling, collagen deposition and fibrosis66,67,68,69,70,71. Fibrosis is a consequence of inflammation at the site of tissue damage and is the characteristic pathological feature of DCM aside from dilatation8,62. Regional dysfunction and/or volume overload lead to increased cardiac workload and wall stress, activation of fetal genes and myocyte reversion to fetal programming and myofibroblasts63; these mechanisms are both a consequence and a cause of fibrosis. With time, fibrotic scar tissue eventually replaces the damaged tissue, thereby stiffening the heart and further amplifying the progression to dilation and heart failure.
Infection
Infections are believed to account for ~30% of the aetiology of DCM and are typically associated with myocarditis, as it has been demonstrated in animal models and patients5,60,61,62,63,64,65,66.
One of the most common groups of viruses associated with DCM are the enteroviruses (including coxsackievirus group A and B in particular)72 (Box 1). Adenoviruses and herpesviruses are also commonly found in patients with DCM73. Less well established is the role of parvoviruses, which are associated with DCM in some patients74. Analysis of myocardial biopsy samples from patients with DCM has identified the molecular evidence of viral genome or evidence of viral proliferation75. The positive identification of viral genome with biopsy is associated with a more-rapid progression to DCM and worse clinical outcomes76,77,78.
Autoimmunity
Several lines of evidence indicate that DCM can result from an autoimmune disease according to the Rose-Witebsky criteria. This evidence includes the presence of cardiac immune cell infiltrates in ~50% of biopsy samples, abnormal expression of HLA class II and/or adhesion molecules (which are essential to initiate an immune response mediated by HLA class II and, therefore, can be used as biomarkers to identify autoimmune disease) on the cardiac microvascular endothelium in the absence of viral genomes detected from biopsy samples in index patients and family members79,80, increased serum levels of cardiac autoantibodies in patients and relatives8,81,82,83,84,85, experimentally induced animal models of DCM from immunization with recognized autoantigens that can also be found in patients with DCM8,86,87,88,89,90,91 and response to immunosuppression or immunomodulation in patients with suspected autoimmune DCM8,92,93,94. Furthermore, autoimmune diseases such as systemic lupus erythematosus, systemic sclerosis and rheumatoid arthritis are rare causes of DCM, occurring in ~5–10% of cases95,96,97.
Cardiac-specific autoantibodies occur in up to 60% of patients with DCM and their relatives and can directly influence myocyte function and the prognosis of disease82,84,98. In patients, inflammatory DCM may be familial81,84,85 and has been associated with HLA antigens, as described by a landmark multicentre European genome-wide association study99. The study reports that HLA is a risk locus for DCM, consistent with an autoimmune aetiology99. DCM probably represents the end result of more than one aetiological agent in each individual patient, in a multiple hit cascade involving gene–environment interactions62, which is best depicted by the application of a new complex classification — the MOGES classification100,101. This gene–environment model fits perfectly with the autoimmune DCM phenotype.
Many distinct cardiac autoantibodies have been found in patients with DCM8,98, with cardiac α-myosin and β-myosin heavy-chain isoforms being frequently recognized autoantigens83. Some of these autoantibodies seem to have a direct functional pathogenetic role102,103,104; furthermore, immunization of animals with autoantigens that have been identified in patients with DCM, such as β1-adrenergic receptor, muscarinic acetylcholine receptor M2, cardiac myosin heavy chain and cardiac troponins79,86,87,88,89,90,91,105,106,107, leads to cardiac abnormalities that reproduce the DCM phenotype. Of note, myocardial pathology has been produced by transfer of immune components from one experimental animal to another8,105,108. Passive transfer of antibodies purified from rats immunized with cardiac myosin leads to deposition of antibodies in the myocardium and myocyte apoptosis, producing cardiomyopathy in recipient animals88. In a mouse model of autoimmune myocarditis and DCM induced by immunization with α-myosin heavy chain, TGFβ-activated kinase 1 (TAK1; also known as mitogen-activated protein kinase kinase kinase 7) mediated rapid Wnt protein secretion, resulting in TGFβ-mediated myofibroblast differentiation and myocardial fibrosis progression, suggesting a possible mechanism for autoimmunity in promoting remodelling that leads to DCM109. Because myosin heavy chain is a relevant autoantigen in patients with autoimmune DCM83, pharmacological targeting of Wnt proteins might represent a promising therapeutic approach in the future109.
Chemical and toxin exposure
Chronic alcohol abuse is an important cause of DCM, occurring most often in men of 30–55 years of age who have been heavy consumers of alcohol for at least 10 years110. The proportion of alcoholic cardiomyopathy among all cardiomyopathy deaths was estimated at 6.9% globally111, and alcoholic DCM occurred more often in men (8.9%) than in women (2.9%). Studies in animals have demonstrated that acute and chronic ethanol administration impairs cardiac contractility112 and decreases the contractile protein α-myosin heavy chain110. The metabolite of alcohol, acetaldehyde, can alter cellular calcium, magnesium and phosphate homeostasis and impair mitochondrial respiration, for example113. Damage to the myocardium from alcohol also recruits inflammatory cells to the heart114.
Long-term abuse of cocaine is also known to cause DCM and lethal arrhythmias115. Depressed LV function has been reported in 4–9% of individuals who used cocaine and had cardiac symptoms116,117. Cocaine is believed to cause LV dysfunction by increasing the release of catecholamines, which are directly toxic to cardiomyocytes, leading to necrosis. Similar to the effects of alcohol, cocaine also causes myocyte death by damaging mitochondria118. Myocarditis has been reported in 20% of patients who died from suspected cocaine overdoses, suggesting that myocardial inflammation may lead to DCM at least in some cases119.
DCM can occur as an adverse effect from cancer chemotherapeutic agents such as anthracyclines120. Anthracyclines that frequently cause DCM include doxorubicin, epirubicin and idarubicin. Doxorubicin is a widely used and effective chemotherapy treatment for many adult and paediatric cancers; however, one of its adverse effects is cumulative, dose-related, progressive myocardial damage that may lead to DCM and congestive heart failure121. Doxorubicin binds both DNA and type II topoisomerase, leading to DNA double-stranded breaks and transcriptome changes resulting in mitochondrial dysfunction, reactive oxygen species generation and cardiac cell death122. More than 50% of children with cancer receive an anthracycline, resulting in >80% survival, but up to 60% develop at least some form of cardiac dysfunction that can lead to cardiomyocyte necrosis, apoptosis, hypertrophy, fibrosis, LV dysfunction, DCM and heart failure123,124. Endomyocardial biopsy (EMB) samples reveal features of cardiotoxicity, such as cytosolic vacuolization, lysis of myofibrils and cellular swelling. Troponin I, a marker of myocardial injury, is a useful serum biomarker of doxorubicin cardiotoxicity predicting progression to DCM125.
Sex differences
Male sex is an important risk factor for developing heart failure following a number of cardiovascular conditions including DCM; however, few clinical studies have examined sex differences in incidence or pathogenetic mechanisms in DCM specifically. In one of the few studies to examine sex differences in the pathogenetic mechanism of DCM, men with acute DCM were found to have higher expression of proteins associated with apoptosis than women, and higher expression levels correlated with a lower LVEF126.
In animal models of myocarditis, progression to DCM is dependent on sex and race (that is, mouse strain): 100% of males of susceptible mouse strains develop fibrosis and DCM after acute myocarditis, whereas only 20% of females develop mild DCM, based on echocardiography127. A similar finding has been observed in patients with severe, clinically symptomatic myocarditis, as men were twice as likely as women to present with evidence of myocardial fibrosis, assessed by cardiac MRI128. Animal studies have revealed that the pathogenetic mechanisms leading to cardiac remodelling and DCM are similar regardless of the cause of damage to the myocardium and are promoted by testosterone in male animals127,129,130,131,132. Testosterone has been found to increase the number of M2 macrophages that express CD11b (also known as αM integrin), Toll-like receptor 4 and IL-1β in animal models, leading to increased cardiac inflammation, remodelling and DCM127,133,134.
Sex hormones and cardiac function
Sex hormones alter cardiac function by binding to androgen and oestrogen receptors on cardiac vascular endothelial cells, smooth muscle cells, fibroblasts and myocytes. Additionally, the binding of sex hormones to their receptors directly alters the functions of immune cells and platelets influencing the type of cardiac inflammation, remodelling and thrombosis involved in DCM135.
Many of the same inflammatory cells and cytokines that drive dilation — which occurs more often in men — also predispose patients with DCM to thromboembolic events, including complement and platelet activation136,137. Importantly, women have higher levels of oestrogen receptors on their arteries than men, and activation of these receptors by 17β-oestradiol prevents cardiomyocyte apoptosis, inhibits cardiac damage induced by reactive oxygen species and reduces cardiac hypertrophy and fibrosis in women138. In animal models, androgens promote hypertrophy139,140. Overall, underlying sex differences in cardiac pathophysiology and in the immune response to cardiac injury are believed to lead to the observed sex differences in DCM and heart failure.
Diagnosis, screening and prevention
Diagnosis
The clinical presentation of DCM is generally unrelated to the underlying aetiology and ranges from dyspnoea, swollen legs, ankles and stomach, fatigue and chest pain caused by reduced oxygen levels reaching the heart to arrhythmia, acute decompensation or cardiogenic shock. The signs and symptoms of DCM mainly relate to the degree of LV or biventricular systolic dysfunction leading to pump failure; heart failure signs and symptoms may be fulminant, acute, subacute or chronic. In addition, atypical chest pain and palpitation may be present.
DCM is typically diagnosed between 20 and 50 years of age20,141. Dilated chambers are readily identified using echocardiography; the diagnostic criteria are LV end-diastolic volumes or diameters >2 s.d. from normal according to normograms (z-scores >2 s.d.) corrected for age and body surface area and ejection fraction <50%62. Cardiac catheterization may be performed to rule out coexisting coronary artery disease, and cardiac MRI may assist with imaging dilatation and is used to determine the presence of oedema and/or fibrosis, which are suggestive of inflammation. Electrocardiography (ECG) in patients with DCM may be remarkably normal, but abnormalities ranging from isolated T wave changes and left bundle branch block to prolongation of atrioventricular conduction can occur. Sinus tachycardia and supraventricular arrhythmias are common; ~20–30% of patients have non-sustained ventricular tachycardia. Ambulatory Holter monitoring captures premature ventricular complex and sustained or non-sustained ventricular tachycardia, both of which are important, especially in differential diagnosis with arrhythmogenic cardiomyopathy. The diagnostic accuracy is dependent on the combination of conventional non-invasive and invasive cardiological examination with molecular and non-cardiac parameters including genetic analyses; this comprehensive approach is also needed to determine the aetiology of DCM. As described (see section on autoimmunity above), different serum autoantibodies to cardiac-specific and muscle-specific autoantigens have been found in myocarditis and autoimmune DCM, and some of these autoantibodies might have a direct pathogenetic role8,98,102,108,142,143,144.
Some genetic forms of DCM can be suggested by the presence of clinical traits, sometimes known as ‘diagnostic red flags’. Such rare but important signs and symptoms that indicate a specific multisystem disease or genotype include abnormal skin pigmentation, skeletal myopathy and neurosensory disorders (for example, deafness and blindness)145.
In individuals who are diagnosed with idiopathic DCM, a genetic aetiology should be considered. Thus, the basic evaluation of these patients should include a detailed family history as well as clinical screening of first-degree relatives. Next-generation sequencing methods, such as whole-exome or whole-genome sequencing, are emerging as moderately inexpensive approaches for diagnosis that have both clinical and research implications. Inherited cardiomyopathies, including DCM, are genetically heterogeneous and are associated with rare mutations in a large number of genes; many mutations result in similar phenotypes, and, therefore, the same phenotype can have different underlying mutations. Even in the absence of an identified pathogenetic mutation, first-degree relatives should have periodic ECG and echocardiography to detect early signs of DCM. In addition to genetic variation, numerous factors affect gene expression and, therefore, determine how gene mutations are manifested146. The genotype frequently does not equate to phenotype because of environmental epigenetic influences on gene expression. For example, mutations that promote cardiomyocyte damage recruit a pro-inflammatory response promoting remodelling and DCM147. Thus, assessment of familial DCM would benefit from an improved understanding of the influence of environmental and epigenetic factors148,149.
Imaging techniques
Doppler and 2D echocardiography remains the primary method to analyse cardiac function and undergirds the diagnostic criteria for most heart muscle diseases. Thus, all patients with unexplained or suspected moderate to severe heart failure symptoms should undergo echocardiography at first presentation and for follow-up150. Echocardiogram features can indicate a specific aetiology; however, echocardiography cannot definitively establish the underlying cause of DCM. For example, wall motion abnormalities of a non-ischaemic origin are suggestive of myocarditis. In addition, LV remodelling and functional mitral regurgitation can be assessed efficiently by echocardiography. LV dilatation and concomitant displacement of the papillary muscles can cause tethering of the mitral leaflets and poor valve coaptation (closure)41.
Speckle-tracking echocardiography is an imaging technique that analyses the motion of tissues in the heart using the naturally occurring speckle pattern in the myocardium and is a promising tool to identify the presence of an inflammatory infiltrate in patients with DCM (Fig. 3). In patients with biopsy-proven myocarditis, global longitudinal strain (longitudinal shortening as a percentage), which provides an accurate assessment of regional contractility, is significantly impaired compared with patients without inflammation151.
a,b | Speckle-tracking echocardiographic images in a patient with active myocarditis show reduced global longitudinal strain at baseline (before treatment). Analysis of the left ventricular longitudinal strain from the left two chambers before immunosuppressive treatment. The value of the peak longitudinal systolic strain for each segment being examined is superimposed on the colour 2D image. The curves of longitudinal strain (%) as a function of time are also shown (part a). Peak systolic strain in each segment before immunosuppressive treatment (part b). c,d | Immunohistological staining of an endomyocardial biopsy sample from the same patient shows active myocarditis with evidence of massive enhanced CD3+ T cell infiltration at baseline (red-brown staining) (part c), myocytolysis and extensive infiltration of immunocompetent cells (purple staining) (part d). e,f | Speckle-tracking imaging of the same patient after 6 months of immunosuppressive treatment shows substantial increase in global longitudinal strain. g,h | After treatment, immunohistological staining showed reduced CD3+ T cell infiltration (absence of red-brown staining) (part g) and no active myocardial inflammation (reduced purple staining) (part h). ANT, anterior; ANT_SEPT, anteroseptal; INF, inferior; LAT, lateral; POST, posterior; SEPT, septal.
Cardiac MRI has become increasingly accessible in clinical practice, where it represents the best method for morphological and functional evaluation and characterization of myocardial tissue in patients with DCM (Fig. 4). The presence of myocardial fibrosis can be detected with late gadolinium enhancement (LGE). LGE distribution may indicate muscular dystrophy, and LGE presence, patterns and quantification may assist in determining the risk of malignant ventricular arrhythmias. MRI detects myocardial oedema suggestive of acute myocarditis or sarcoidosis (an inflammatory disease in which granulomas form in different organs). Alongside cardiac MRI, 18F-fluorodeoxyglucose (FDG)–PET has emerged as a valuable tool for diagnosing cardiac sarcoidosis. However, the sensitivity of cardiac MRI to detect inflammation in DCM is low because inflammation is scarce at this stage of the disease process. The best method to confirm the presence of an inflammatory infiltrate in DCM is histology and/or immunohistology151. In addition, cardiac MRI cannot detect the presence of virus in the myocardium151. Future multicentre prospective studies are needed to confirm the role of cardiac MRI in the prognostic stratification of DCM.
Endomyocardial biopsy
The current expert consensus statement of the ESC recommends that all patients with suspected inflammatory cardiomyopathy should undergo an EMB to identify the type of inflammatory infiltrate, to determine the underlying aetiology and form an aetiology-based treatment strategy8. Although many centres do not conduct EMB on the basis of a perception that the procedure carries a high risk of complications, several studies report that EMB can be performed with a very low rate of major complications and that LV biopsy is as safe as right ventricular biopsy152,153,154. Tissue obtained from EMB should be analysed using histology, immunohistochemistry and molecular biology techniques.
Histological examination of paraffin sections with various staining protocols is used to detect several morphological alterations (such as myocardial disarray (loss of the parallel alignment of cardiomyocytes), vacuole formation, cardiomyocyte hypertrophy, myocytolysis (myofibrillar degeneration and sublethal injury of cardiac muscle cells), necrosis and cell death, scar formation, collagen deposition and fibrosis, pathological vascular conditions, granulomas and giant cells (multinucleated cells formed by the fusion of cells)) and to characterize the major inflammatory subtypes (Fig. 5).
Endomyocardial biopsy is important to determine the underlying cause of dilated cardiomyopathy. a | Active myocarditis with immune cell infiltration and myocytolysis (arrows), histological azan staining. b | Giant cell myocarditis with massive immune cell infiltration around multinuclear giant cells (arrows), histological haematoxylin and eosin (H&E) staining. c | Eosinophilic myocarditis with immune cell infiltration and eosinophils (arrows), histological H&E staining. d | Immunohistochemical staining depicting CD3+ T cells (red-brown staining) in a focal pattern in borderline myocarditis. e | Immunohistochemical staining of increased perforin-positive cytotoxic cells (arrows) in inflammatory cardiomyopathy. f | Immunohistochemical staining of increased cell-adhesion molecule HLA1 (red-brown staining) in inflammatory cardiomyopathy.
The use of immunohistochemistry has markedly increased the ability to detect cardiac inflammation from biopsy sections; monoclonal antibodies enable precise characterization, quantification and localization of different immune cell types and cell-adhesion molecules. Determining the cell composition of inflammatory foci affects the prognosis of DCM, and establishing the proportion and type of immune cells is crucial in deciding the optimal type of treatment.
Microbial genomes are determined, quantified and sequenced using PCR-based methods, including nested reverse transcriptase–PCR and quantitative PCR. Active or latent infections in the myocardium can be differentiated by parallel blood tests and confirmation of transcriptional activity in EMB samples. Principally, all viruses that are responsible for inflammatory cardiomyopathy can be analysed8. Viral titres in the serum alone are not useful for diagnosis because they do not correlate to the actual phase of cardiomyopathy.
Finally, novel biomarkers such as microRNAs and gene expression profiling have been introduced in the molecular examination of EMB. The identification of distinct gene expression profiles in the myocardium enables clear discrimination of samples with giant cells, acute myocarditis or no inflammation even if there is no direct histological proof of giant cells or inflammation. Moreover, the disease-specific gene expression profiles change during effective treatment and can thereby also be useful to monitor therapy155.
Prognosis
The prognosis is poor for patients with DCM with an LVEF <35%, right ventricular involvement and NYHA functional class III or IV (Box 2). Several factors can worsen the prognosis. Adverse remodelling characteristics in DCM include functional mitral regurgitation (leakage of blood backward through the mitral valve), myocardial fibrosis, dyssynchronous ventricular contraction and enlargement of other chambers.
Adverse remodelling
Sudden cardiac death in DCM can be caused by electromechanical dissociation (pulseless electrical activity) or ventricular arrhythmias. The development of new LV branch block during follow-up is a strong independent prognostic predictor of all-cause mortality, and atrial fibrillation is a sign of structural disease progression and negatively affects the prognosis. Patients with DCM and haemodynamically relevant mitral regurgitation may need invasive therapies, such as percutaneous or open repair of the mitral valve, mechanical circulatory support or even heart transplantation. Whereas right ventricular function frequently recovers under therapy (typically within 6 months), the development of right ventricular dysfunction during long-term follow-up indicates structural disease progression and portends a negative outcome. Strategies to detect pre-symptomatic DCM have a clear rationale because early treatment can retard adverse remodelling, prevent heart failure symptoms and increase life expectancy. In addition, patients with DCM should be regularly re-assessed, particularly in the presence of cardiovascular risk factors. Worsening of LV function or an increased ventricular arrhythmic burden can be caused not only by DCM progression but also by the development of new co-pathologies. Thus, the possible presence of coronary artery disease, hypertensive heart disease, structural valve disease or acute myocarditis should be systematically ruled out during follow-up. However, clinical and echocardiographical parameters have limited ability to predict long-term prognosis, suggesting that other host–environmental factors are important in determining the outcome of DCM.
Additional factors affecting prognosis
The persistence of enterovirus in the myocardium has been associated with ventricular dysfunction, whereas viral genome clearance is associated with improved ventricular function and 10-year prognosis77,78. Because different viruses respond differently to antiviral drugs and sometimes are not completely eliminated, knowing the specific pathogenic virus is crucial for making a tailored treatment decision.
In clinical and experimental studies, immunohistological evidence of inflammation was identified as an independent predictor of survival156,157. Thus, precise characterization and quantification of intramyocardial immune cell subtypes is needed to inform the decision to start immunosuppressive therapy with the goal of improving prognosis158,159.
Management
The management of DCM aims at reducing symptoms of heart failure and improving cardiac function (Fig. 6). Several pharmacological options and devices are available. The pivotal randomized controlled clinical trials that provided the evidence for the guidelines on the treatment of heart failure with reduced ejection fraction included a large proportion of patients with a non-ischaemic aetiology (that is, individuals who probably developed heart failure as a result of DCM). Consequently, on the basis of subgroup analyses, it is reasonable to assume that the efficacy and safety of most treatments for heart failure extend to patients with DCM. Of course, investigator-reported aetiology in large clinical trials may not necessarily be accurate, and non-ischaemic aetiology may include several causes of heart failure in addition to DCM, such as heart failure caused by hypertension.
Clinical management of a patient with symptomatic dilated cardiomyopathy starts with standard heart failure medications. If there is haemodynamic improvement, the treatment will be continued with careful follow-up to monitor for progressive left ventricular dysfunction. If left ventricular dysfunction is noted or there is a lack of haemodynamic improvement, an endomyocardial biopsy should be performed. If viral infection is detected by reverse transcriptase–PCR or immunohistochemistry staining, the patient may receive antiviral therapy. If certain inflammatory cells are discovered, a tailored immunosuppressive therapy may be administered. If there is no haemodynamic improvement, additional treatment options for heart failure should be explored.
In patients with advanced disease, pharmacological and device therapy may be insufficient to maintain adequate cardiac function and surgery might be required. The two major options are heart transplantation and implantation of long-term mechanical circulatory support, either as a temporary measure while awaiting transplantation or permanently. Additional surgical approaches include correction of mitral valve regurgitation.
Finally, additional aetiology-based therapies might be appropriate in patients with EMB-confirmed myocarditis or infection-associated DCM. These include immunosuppression and antiviral therapy; immunoadsorption therapy to remove circulating autoantibodies is still under investigation.
Exercise
Evidence suggests that exercise is beneficial in heart failure, although elderly patients were not enrolled in most studies and the optimum level and intensity of exercise prescribed are uncertain160. Furthermore, the only large trial on exercise in patients with heart failure, HF-ACTION, showed a modest treatment effect that was obtained only with a very intensive training regimen that may not be practical to deliver in every health-care centre. Importantly, exercise is contraindicated in the active phase of inflammatory cardiomyopathy, both in athletes and non-athletes, and in DCM due to lamin A/C mutations62.
Pharmacological therapy for heart failure
ACE inhibitors (or angiotensin receptor antagonists in patients who develop cough as an adverse effect of ACE inhibitors) and β-blockers are the standard drug therapy for heart failure in DCM (Table 2). Combined angiotensin receptor–neprilysin inhibitors reduce total mortality and hospital admissions compared with ACE inhibitors and could replace ACE inhibitors as one of the cornerstones of drug therapy in chronic heart failure161. Mineralocorticoid antagonists and If inhibitors provide incremental benefit in survival or hospital admission when combined with ACE inhibitors and β-blockers162.
All pharmacological therapies should be used in accordance with international guidelines, taking into account blood pressure, heart rate and rhythm, renal function and blood chemistry.
Device therapy
Cardiac resynchronization therapy (CRT) is a modality of cardiac pacing used in patients with LV systolic dysfunction and dyssynchronous ventricular activation that provides simultaneous or nearly simultaneous electrical activation of the left ventricle and right ventricle via stimulation of both (biventricular pacing) or the left ventricle alone. This therapy is performed by either a CRT-pacemaker (CRT-P) or a combined CRT–ICD (which, in addition to the pacemaking function, can deliver an electric shock to reset the sinus rhythm in case of arrhythmia). CRT devices include a transvenous pacing lead placed in a branch of the coronary sinus (or, less commonly, an epicardial or endocardial LV lead) for LV pacing in addition to leads in the right ventricle and right atrium.
CRT is indicated for patients with heart failure and LVEF ≤35% who have a life expectancy with good functional status of >1 year if they are in sinus rhythm and have a markedly prolonged QRS duration (≥130 ms) and an ECG that shows left bundle branch block, irrespective of symptom severity. The benefits of CRT for patients with right bundle branch block or interventricular conduction delay are unclear (subgroup analyses suggest little benefit or even harm)163.
Two randomized placebo-controlled trials, the Comparison of Medical Therapy, Pacing, and Defibrillation in Heart Failure (COMPANION) and Cardiac Resynchronization in Heart Failure (CARE-HF) study, randomized 2,333 patients with moderate to severe symptomatic heart failure (NYHA class III or IV) to either optimal medical therapy or optimal medical therapy plus CRT164,165,166. In each trial, CRT reduced the risk of death from any cause and hospital admission for worsening heart failure with a relative risk ratio of death of 24% with a CRT-P and of 36% with a CRT-ICD (CRT-D) in the COMPANION study and of 36% with CRT-P in the CARE-HF study. In the CARE-HF study, the relative risk ratio of hospitalization with CRT-P was 52%. These trials also showed that CRT improves symptoms, quality of life (QoL) and ventricular function.
Similarly, two randomized placebo-controlled trials evaluated the effects of CRT in 3,618 patients with mild symptoms (85% had NYHA class I heart failure in the MADIT-CRT trial) or moderate symptoms (80% had NYHA class II heart failure in the Resynchronization/Defibrillation for Ambulatory Heart Failure Trial (RAFT)); patients received either optimal medical therapy plus an ICD (while maintaining spontaneous sinus rhythm) or optimal medical therapy plus CRT-D (in which the pacing was controlled by the ICD)167,168. In each trial, CRT reduced the risk of the primary composite end point of death or heart failure hospitalization, with a 34% relative risk ratio in MADIT-CRT and a 25% relative risk ratio in RAFT. All-cause mortality was significantly reduced in RAFT but not in MADIT-CRT. CRT improves symptoms, QoL and ventricular function in these patient populations as well. Both MADIT-CRT and RAFT showed a significant treatment-by-subgroup interaction whereby QRS duration modified the treatment effect (CRT seemed more effective in patients with a QRS ≥150 ms), and CRT was more beneficial for patients with left bundle branch block than for those with right bundle branch block or an interventricular conduction defect. In a meta-analysis, the mortality benefit of CRT was similar in individuals with ischaemic disease and non-ischaemic cardiomyopathy169. Thus, for all these trials, CRT reduced risk of death and improved symptoms, QoL and ventricular function.
Aetiology-based therapies
A definitive diagnosis of the pathophysiology that underlies DCM is a prerequisite for a specific, personalized therapy based on aetiology170. However, treatment guidelines for causes of inflammatory DCM are missing at this time. Thus, multicentre, prospective, randomized studies to test immunosuppressive and antiviral therapies in inflammatory DCM are needed.
A retrospective analysis showed that immunosuppressive treatment in patients with EMB-proven, virus-positive cardiomyopathy is deleterious94. Consequently, a prospective, randomized, placebo-controlled study (TIMIC-trial) showed that in patients with EMB-proven, chronic, virus-negative myocarditis who were refractory to standard therapy, immunosuppressive therapy resulted in significant improvement in LVEF compared with placebo93, indicating that immunosuppressive therapies can be effective in patients with inflammatory DCM. These results were subsequently confirmed in another trial94. Ideally, immunosuppressive therapy should be started as early as possible before irreversible remodelling occurs8.
Viral infections are a frequent cause of myocarditis that can lead to inflammatory DCM in some individuals77. In a biopsy-based analysis of patients with myocarditis, the presence of virus in the myocardium was associated with a progressive impairment of LV function, whereas spontaneous viral clearance was associated with significant improvement of LV function171,172. Consequently, in a nonrandomized study in patients with enterovirus-associated or adenovirus-associated cardiomyopathy, the administration of IFNβ significantly reduced virus load, reduced myocardial injury and improved long-term survival171,172. In the Beta-Interferon in a Chronic Viral Cardiomyopathy (BICC) trial, virus load reduction or clearance was significantly higher in the IFNβ group than in the placebo group. However, IFNβ treatment was statistically significantly less effective at clearing parvovirus B19 (ref.173).
Finally, small open-labelled studies have found that removal of circulating autoantibodies by immunoadsorption improved cardiac function and decreased myocardial inflammation108,174,175. However, at present, immunoadsorption is still an experimental treatment option because there are conflicting results regarding its effectiveness; a placebo-controlled multicentre study is underway to test this therapy in patients with cardiomyopathy176 (Table 2).
Prognostic stratification
The current risk stratification scheme for the prediction of life-threatening arrhythmias in patients with DCM is based mainly on the degree of systolic dysfunction of the left ventricle, with an LVEF <35% set as the threshold for a high risk of sudden death on the basis of the results of randomized clinical trials177. Although data supporting the use of ICDs are robust for patients with ischaemic DCM, the evidence from randomized trials for patients with non-ischaemic DCM has been debated over time. The current European and American College of Cardiology/American Heart Association guidelines for ICD implantation in patients with non-ischaemic DCM are based on a 2004 meta-analysis that included trials in which ICD was implanted for primary prevention of sudden cardiac death and trials in which ICD was implanted as a secondary prevention measure (that is, in patients who had survived an episode of ventricular fibrillation), which demonstrated a 31% reduction in all-cause mortality with ICD use178.
In the DANISH trial (Defibrillator Implantation in Patients with Nonischemic Systolic Heart Failure), >1,100 patients with non-ischaemic DCM on optimal medical therapy and CRT were randomly assigned to ICD versus no ICD for primary prevention of sudden cardiac death; no difference in all-cause mortality (the primary end point) was observed between the two groups at 5-year follow-up179.
The results of the DANISH study might be explained by the overall low mortality in the control group, possibly justified by the high use of CRT, and by the high adherence to optimal medical therapy in both groups that may have reduced the power of the study to prove the efficacy of ICD. Nevertheless, it is remarkable that the ICD group showed not only a 50% reduction in incidence of sudden cardiac death but also an overall survival benefit in the subgroup of patients of <60 years of age.
An updated meta-analysis of six randomized trials, assessing the effect of ICD on all-cause mortality in 2,970 patients with non-ischaemic DCM who were randomized to ICD or to optimal medical therapy for the primary prevention of sudden cardiac death, found a significant 23% risk reduction in all-cause mortality in the ICD arm180. Interestingly, the results remained relevant and statistically significant after eliminating the contribution of trials that involved patients with a CRT-D, with a 24% reduction in all-cause mortality with ICD.
The problem of using LVEF as the pivotal risk marker for patients with DCM is that this single parameter does not recapitulate the complexity of the disease that arises from a number of different aetiologies. The availability of genotype–phenotype studies in patients with DCM has enabled physicians to identify populations at increased risk of sudden cardiac death, such as patients with mutations in LMNA or FNLC. With the increasing availability of personalized aetiological diagnosis for patients with DCM, it will be possible to provide more-refined risk stratification schemes that, in addition to LVEF, take into account genetic information and additional clinical parameters, possibly combining them in multiparametric scores. Promising approaches for risk stratification include the assessment of LGE during cardiac MRI of cardiac sympathetic nerve function through metaiodobenzylguanidine labelled with iodine-123 (MIBG)181,182.
Quality of life
The health-related QoL in patients with DCM is poorly analysed. Many patients with DCM progress to chronic heart failure, and the patient’s awareness of their risk of dying, the adverse effects of therapy and frequent hospitalizations have considerable negative effects on a patient’s QoL183. A recent study184 identified three factors that predicted positive self-care with compliance to treatment: clear information about the medication, a belief in the illness having serious consequences and the effect of medication use on lifestyle. Moreover, previous studies showed a significant improvement in all aspects of QoL after comprehensive cardiac rehabilitation with daily light exercise, a weight management programme and nurse-led patient education185,186. Thus, the most important way to improve QoL in patients with DCM is providing an early, pathophysiological-based diagnosis, better risk stratification and, ultimately, personalized therapy based on aetiological assessment of disease.
Outlook
Many advances have been made to improve diagnosis and better characterize DCM on the basis of the underlying aetiology. Advances in our understanding of the pathogenesis of disease are derived largely from preclinical animal models. However, many gaps in our understanding remain. Clinical care could be improved by harnessing novel advances in imaging modalities for DCM; by identifying and utilizing novel serum biomarkers; by improving our understanding of the relationship between infection, inflammation and autoimmunity and cardiac damage and remodelling in the pathogenesis of DCM; and by taking advantage of the new therapeutic opportunities of cardiac regeneration.
Improved diagnostic tools
The refinement of current diagnostic tools and imaging modalities could revolutionize our understanding of the pathogenesis of disease. FDG–PET is an important tool to better understand the relationship between cardiac inflammation and the development of DCM187 and is used in preclinical mouse models of myocarditis to advance the detection of cardiac inflammation134,188. Once new imaging markers from animal studies can successfully detect inflammation in patients with myocarditis, they could be refined to detect inflammation in patients with DCM, where inflammatory cells are often scarce.
Echocardiography is the most established method for assessing systolic dysfunction. Advances in echocardiography techniques and measurements (such as global longitudinal strain, tissue Doppler strain and speckle-tracking echocardiography) have the potential to enable much earlier detection of systolic dysfunction leading to DCM, especially if echocardiography results can be paired to serum biomarkers of heart failure such as BNP and pro-NT-BNP, soluble interleukin-1 receptor-like 1 (IL1R1; also known as sST2) and C-reactive protein (CRP)189,190.
Serum biomarkers
Advances in genetic sequencing and molecular analysis of the genes and proteins involved in the pathogenesis of DCM, both in preclinical animal models and in human studies, have unveiled promising new serum biomarkers that can be used not only to indicate the risk of heart failure and prognosis but also to reveal the underlying pathology and thereby inform treatment.
Elevated serum levels of sST2 correlate with the severity of heart failure (NYHA class) in patients with myocarditis and DCM as well as other cardiovascular conditions191,192,193,194. sST2 is a cytokine induced by biomechanical strain in cardiac fibroblasts, cardiomyocytes and vascular endothelial cells195,196. In a small study, sST2 was significantly higher in patients with DCM than in controls197. sST2 has been found to predict the risk of developing heart failure in patients with DCM in several recent studies193,198,199,200. When combined with NT-proBNP, which is released under conditions of pressure overload and, therefore, is a marker for heart failure, serum sST2 levels provide additive value in predicting sudden death in patients with heart failure and an LVEF ≤45%201.
Two important processes in heart failure are inflammation and renal impairment, both of which can increase serum immunoglobulin free light chain (FLC) κ and λ concentrations. The sum of circulating FLC κ and λ is increased in patients with autoimmune diseases202 and DCM (A.Mat., unpublished results). A community-based study of 15,000 individuals of the general population showed that high FLC concentration was associated with an increased risk of all-cause mortality203. FLC, especially λ, is increased in patients with myocarditis and is associated with mortality (A.Mat., unpublished results). Combined FLC, the sum of κ and λ FLC, has shown an association between high combined FLC and prognosis204 and provided the incremental prognostic value of multiple biomarkers205 in patients recently hospitalized with decompensated heart failure. The mechanism for FLC increase in DCM and heart failure remains to be clarified, but FLC may be a promising new biomarker of inflammation in DCM.
Inflammation
Infection
Future studies should also aim to better understand the relationship between viral and parasitic infections (such as Trypanosoma cruzi infection) in the development of DCM. These infections remain endemic in many countries and probably contribute to the emerging public health burden of DCM. Of particular importance will be determining the relationship of infections to host immune responses in the context of host genetics.
In addition to hepatitis, hepatitis C virus (HCV) infection frequently causes cardiac and renal abnormalities, including hypertrophic cardiomyopathy and DCM. In Egypt, in patients with HCV infection, echocardiographic abnormalities were frequently found, such as dilated left atrium, dilatation and hypertrophy of the left ventricle and disturbed diastolic and systolic function of the left ventricle206. In China, NT-proBNP levels are higher in patients with HCV-infection-associated hepatitis than in those with hepatitis B virus infection or in healthy controls207. An association between major histocompatibility complex (MHC) antigens and HCV clearance and susceptibility has been identified208,209. The molecular mechanisms behind the development of cardiomyopathies with HCV infection related to MHC class II molecules were found to differ between DCM and hypertrophic cardiomyopathy210,211. Antiviral treatment for HCV infection has been reported to improve renal and cardiovascular outcomes in patients with diabetes mellitus212. The annual number of deaths associated with HCV is now higher than all other nationally notifiable infectious conditions in the USA213, suggesting that this viral infection could make an important contribution to DCM cases in developed countries. The mechanism for how HCV causes DCM remains to be elucidated.
Autoimmunity
Preclinical and clinical evidence supports that some forms of DCM are the result of a pathogenetic autoimmune response8,214. Mice lacking the T cell receptor co-stimulatory receptor programmed cell death protein 1 (PD-1) spontaneously develop autoimmune DCM with autoantibodies that bind cardiac troponin I106. Passive transfer of monoclonal antibodies directed against cardiac troponin I induce cardiac dysfunction107. An antibody against cardiac troponin I was found in patients with myocarditis and DCM215, and the presence of this biomarker probably reflects damage to cardiac muscle107. Future experimental and clinical studies are needed to clarify the role of autoimmunity and autoantibodies in the pathogenesis of DCM because serum autoantibodies may form an inexpensive early screening method to determine a subset of patients at an increased risk of developing DCM. Clinical and animal studies are also needed to find novel therapies with improved efficacy and safety for cases of EMB-proven virus-negative autoimmune DCM with or without serum cardiac-specific autoantibodies.
Regenerative medicine
A rapidly emerging and promising new area of medicine is the potential to regenerate damaged and scarred heart tissue in patients with DCM. As care has improved in cardiovascular medicine, with improved reperfusion therapies, implantable defibrillators, novel medications and cardiac transplantation, mortality has decreased. Advances in this area are particularly important for DCM because the disease is often detected only at an end stage when the scar tissue is well established and is refractory to medication to reverse the process. Promising new tools include mesenchymal stem cells, purified platelet exosome products, fat-derived extracellular vesicles and others. Extracellular vesicles and the more-purified form termed exosomes derived from cells such as adipocytes or platelets of healthy individuals are important in cell-to-cell communication and have been found to contain many regulatory factors such as proteins, RNAs and lipids that inhibit pro-inflammatory and profibrotic pathology216. Although many of these therapies have yet to be applied to patients with DCM in the clinical setting, they hold the promise of a therapy that could prevent or reverse remodelling and promote cardiac regeneration217,218.
References
Hershberger, R. E., Morales, A. & Siegfried, J. D. Clinical and genetic issues in dilated cardiomyopathy: a review for genetics professionals. Genet. Med. 12, 655–667 (2010). This review article provides a wide and detailed overview of clinical and genetic issues in specific types of genetic DCM.
McKenna, W. J., Maron, B. J. & Thiene, G. Classification, epidemiology, and global burden of cardiomyopathies. Circ. Res. 121, 722–730 (2017).
Maron, B. J. Contemporary definitions and classification of the cardiomyopathies: an American Heart Association scientific statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113, 1807–1816 (2006).
Elliott, P. et al. Classification of the cardiomyopathies: a position statement from the european society of cardiology working group on myocardial and pericardial diseases. Eur. Heart J. 29, 270–276 (2007).
Richardson, P. et al. Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the definition and classification of cardiomyopathies. Circulation 93, 841–842 (1996).
Pollack, A., Kontorovich, A. R., Fuster, V. & Dec, G. W. Viral myocarditis — diagnosis, treatment options and current controversies. Nat. Rev. Cardiol. 12, 670–680 (2015).
Sagar, S., Liu, P. P. & Cooper, L. T. Myocarditis. Lancet 379, 738–747 (2012).
Caforio, A. L. P. et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 34, 2636–2648 (2013). In this position statement of the ESC Working Group on Myocardial and Pericardial Diseases, an expert consensus group reviews the knowledge on clinical presentation, diagnosis and treatment of myocarditis and proposes diagnostic criteria for clinically suspected myocarditis and its distinct biopsy-proven pathogenetic forms.
Braunwald, E. Cardiomyopathies. Circ. Res. 121, 711–721 (2017).
Codd, M. B., Sugrue, D. D., Gersh, B. J. & Melton, L. J. Epidemiology of idiopathic dilated and hypertrophic cardiomyopathy. A population-based study in Olmsted County, Minnesota, 1975–1984. Circulation 80, 564–572 (1989).
Hershberger, R. E., Hedges, D. J. & Morales, A. Dilated cardiomyopathy: the complexity of a diverse genetic architecture. Nat. Rev. Cardiol. 10, 531–547 (2013).
Maron, B. J. et al. Prevalence of hypertrophic cardiomyopathy in a general population of young adults. Echocardiographic analysis of 4111 subjects in the CARDIA Study. Coronary artery risk development in (young) adults. Circulation 92, 785–789 (1995).
Vos, T. et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1545–1602 (2016).
Lozano, R. et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2095–2128 (2012).
Ehlert, F. A. et al. Comparison of dilated cardiomyopathy and coronary artery disease in patients with life-threatening ventricular arrhythmias: differences in presentation and outcome in the AVID registry. Am. Heart J. 142, 816–822 (2001).
Saxon, L. A. et al. Predicting death from progressive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am. J. Cardiol. 72, 62–65 (1993).
Saxon, L. A. & De Marco, T. Arrhythmias associated with dilated cardiomyopathy. Card. Electrophysiol. Rev. 6, 18–25 (2002).
Fairweather, D., Cooper, L. T. & Blauwet, L. A. Sex and gender differences in myocarditis and dilated cardiomyopathy. Curr. Probl. Cardiol. 38, 7–46 (2013). This article provides an overview on sex and gender differences in myocarditis and DCM. The authors highlight the gaps in our knowledge regarding the management of women with acute DCM and discuss emerging therapies.
McGoon, M. D. & Miller, D. P. REVEAL: a contemporary US pulmonary arterial hypertension registry. Eur. Respir. Rev. 21, 8–18 (2012).
Halliday, B. P. et al. Sex- and age-based differences in the natural history and outcome of dilated cardiomyopathy. Eur. J. Heart Fail. 20, 1392–1400 (2018).
Kubo, T. et al. Improvement in prognosis of dilated cardiomyopathy in the elderly over the past 20 years. J. Cardiol. 52, 111–117 (2008).
Binkley, P. F. et al. Recovery of normal ventricular function in patients with dilated cardiomyopathy: predictors of an increasingly prevalent clinical event. Am. Heart J. 155, 69–74 (2008).
Castelli, G. et al. Improving survival rates of patients with idiopathic dilated cardiomyopathy in Tuscany over 3 decades. Circ. Heart Fail. 6, 913–921 (2013).
Dries, D. L. et al. Racial differences in the outcome of left ventricular dysfunction. N. Engl. J. Med. 340, 609–616 (1999).
Coughlin, S. S. et al. Black-white differences in mortality in idiopathic dilated cardiomyopathy: the Washington, DC, dilated cardiomyopathy study. J. Natl Med. Assoc. 86, 583–591 (1994).
McNamara, D. M. et al. Clinical and demographic predictors of outcomes in recent onset dilated cardiomyopathy. J. Am. Coll. Cardiol. 58, 1112–1118 (2011).
Dec, G. W. The natural history of acute dilated cardiomyopathy. Trans. Am. Clin. Climatol. Assoc. 125, 76–86 (2014).
Nieminen, M. S. et al. Gender related differences in patients presenting with acute heart failure. Results from EuroHeart Failure Survey II. Eur. J. Heart Fail. 10, 140–148 (2008).
Benjamin, E. J. et al. Heart disease and stroke statistics—2017 update: a report from the American Heart Association. Circulation 135, e146–e603 (2017).
Towbin, J. A. et al. Incidence, causes, and outcomes of dilated cardiomyopathy in children. JAMA 296, 1867 (2006).
Nugent, A. W. et al. The epidemiology of childhood cardiomyopathy in Australia. N. Engl. J. Med. 348, 1639–1646 (2003).
Lipshultz, S. E. et al. The incidence of pediatric cardiomyopathy in two regions of the United States. N. Engl. J. Med. 348, 1647–1655 (2003). The authors of this article estimate that the incidence of paediatric cardiomyopathy is 1.13 cases per 100,000 children. Most cases are identified at an early age, and the incidence appears to vary according to region, sex and racial or ethnic origin.
Lipshultz, S. E. et al. Pediatric cardiomyopathies: causes, epidemiology, clinical course, preventive strategies and therapies. Future Cardiol. 9, 817–848 (2013).
Alvarez, J. A. et al. Competing risks for death and cardiac transplantation in children with dilated cardiomyopathy: results from the Pediatric Cardiomyopathy Registry. Circulation 124, 814–823 (2011).
Arola, A. et al. Epidemiology of idiopathic cardiomyopathies in children and adolescents: a nationwide study in Finland. Am. J. Epidemiol. 146, 385–393 (1997).
Rusconi, P. et al. Differences in presentation and outcomes between children with familial dilated cardiomyopathy and children with idiopathic dilated cardiomyopathy. Circ. Heart Fail. 10, e002637 (2017).
Ashley, E. A. Towards precision medicine. Nat. Rev. Genet. 17, 507–522 (2016).
Piran, S., Liu, P., Morales, A. & Hershberger, R. E. Where genome meets phenome: rationale for integrating genetic and protein biomarkers in the diagnosis and management of dilated cardiomyopathy and heart failure. J. Am. Coll. Cardiol. 60, 283–289 (2012).
Ware, J. S. et al. Genetic etiology for alcohol-induced cardiac toxicity. J. Am. Coll. Cardiol. 71, 2293–2302 (2018).
McNally, E. M., Golbus, J. R. & Puckelwartz, M. J. Genetic mutations and mechanisms in dilated cardiomyopathy. J. Clin. Invest. 123, 19–26 (2013).
Japp, A. G., Gulati, A., Cook, S. A., Cowie, M. R. & Prasad, S. K. The diagnosis and evaluation of dilated cardiomyopathy. J. Am. Coll. Cardiol. 67, 2996–3010 (2016).
Morales, A. & Hershberger, R. E. The rationale and timing of molecular genetic testing for dilated cardiomyopathy. Can. J. Cardiol. 31, 1309–1312 (2015).
Burkett, E. L. & Hershberger, R. E. Clinical and genetic issues in familial dilated cardiomyopathy. J. Am. Coll. Cardiol. 45, 969–981 (2005).
Fatkin, D. et al. Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N. Engl. J. Med. 341, 1715–1724 (1999).
Kamisago, M. et al. Mutations in sarcomere protein genes as a cause of dilated cardiomyopathy. N. Engl. J. Med. 343, 1688–1696 (2000).
Gerull, B. et al. Mutations of TTN, encoding the giant muscle filament titin, cause familial dilated cardiomyopathy. Nat. Genet. 30, 201–204 (2002).
Brauch, K. M. et al. Mutations in ribonucleic acid binding protein gene cause familial dilated cardiomyopathy. J. Am. Coll. Cardiol. 54, 930–941 (2009).
Norton, N. et al. Genome-wide studies of copy number variation and exome sequencing identify rare variants in BAG3 as a cause of dilated cardiomyopathy. Am. J. Hum. Genet. 88, 273–282 (2011).
Herman, D. S. et al. Truncations of titin causing dilated cardiomyopathy. N. Engl. J. Med. 366, 619–628 (2012).
Hershberger, R. E. & Siegfried, J. D. Update 2011: clinical and genetic issues in familial dilated cardiomyopathy. J. Am. Coll. Cardiol. 57, 1641–1649 (2011).
Harakalova, M. et al. A systematic analysis of genetic dilated cardiomyopathy reveals numerous ubiquitously expressed and muscle-specific genes. Eur. J. Heart Fail. 17, 484–493 (2015).
Hershberger, R. E. & Morales, A. Dilated cardiomyopathy overview. GeneReviews https://www.ncbi.nlm.nih.gov/books/NBK1309 (updated 23 Aug 2018).
Kinnamon, D. D. et al. Toward genetics-driven early intervention in dilated cardiomyopathy. Circ. Cardiovasc. Genet. 10, e001826 (2017).
Walsh, R. et al. Defining the genetic architecture of hypertrophic cardiomyopathy: re-evaluating the role of non-sarcomeric genes. Eur. Heart J. 38, 3461–3468 (2017).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
Strande, N. T. et al. Evaluating the clinical validity of gene-disease associations: an evidence-based framework developed by the clinical genome resource. Am. J. Hum. Genet. 100, 895–906 (2017).
Rehm, H. L. et al. ClinGen — the clinical genome resource. N. Engl. J. Med. 372, 2235–2242 (2015).
Ingles, J. et al. Evaluating the clinical validity of hypertrophic cardiomyopathy genes. Circ. Genom. Precis. Med. 12, e002460 (2019).
Shendure, J. A deep dive into genetic variation. Nature 536, 277–278 (2016).
Richards, S. et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17, 405–423 (2015).
Landrum, M. J. et al. ClinVar: public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 44, D862–D868 (2015).
Pinto, Y. M. et al. Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur. Heart J. 37, 1850–1858 (2016).
Noutsias, M. et al. Expression of functional T cell markers and T cell receptor Vbeta repertoire in endomyocardial biopsies from patients presenting with acute myocarditis and dilated cardiomyopathy. Eur. J. Heart Fail. 13, 611–618 (2011).
Liu, P. et al. The tyrosine kinase p56lck is essential in coxsackievirus B3-mediated heart disease. Nat. Med. 6, 429–434 (2000). Viral infections have been linked to chronic DCM. The authors of this article show that the sarcoma (Src) family kinase Lck (p56-LCK) is required for efficient CVB3 replication in T cell lines and for viral replication and persistence in vivo.
Irie-Sasaki, J. et al. CD45 is a JAK phosphatase and negatively regulates cytokine receptor signalling. Nature 409, 349–354 (2001).
Fairweather, D. et al. Mast cells and innate cytokines are associated with susceptibility to autoimmune heart disease following coxsackievirus B3 infection. Autoimmunity 37, 131–145 (2004).
Frisancho-Kiss, S. et al. Cutting edge: cross-regulation by TLR4 and T cell Ig mucin-3 determines sex differences in inflammatory heart disease. J. Immunol. 178, 6710–6714 (2007).
Tschöpe, C. et al. NOD2 (nucleotide-binding oligomerization domain 2) is a major pathogenic mediator of coxsackievirus B3-induced myocarditis. Circ. Heart Fail. 10, e003870 (2017).
Fuse, K. Myeloid differentiation factor-88 plays a crucial role in the pathogenesis of coxsackievirus B3-induced myocarditis and influences type I interferon production. Circulation 112, 2276–2285 (2005).
Epelman, S., Liu, P. P. & Mann, D. L. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat. Rev. Immunol. 15, 117–129 (2015).
Li, A.-H., Liu, P. P., Villarreal, F. J. & Garcia, R. A. Dynamic changes in myocardial matrix and relevance to disease: translational perspectives. Circ. Res. 114, 916–927 (2014).
Cabrerizo, M. et al. Molecular epidemiology of enterovirus and parechovirus infections according to patient age over a 4-year period in Spain. J. Med. Virol. 89, 435–442 (2016).
Pauschinger, M. et al. Detection of adenoviral genome in the myocardium of adult patients with idiopathic left ventricular dysfunction. Circulation 99, 1348–1354 (1999).
Tschope, C. High prevalence of cardiac parvovirus B19 infection in patients with isolated left ventricular diastolic dysfunction. Circulation 111, 879–886 (2005).
Pauschinger, M. et al. Enteroviral RNA replication in the myocardium of patients with left ventricular dysfunction and clinically suspected myocarditis. Circulation 99, 889–895 (1999).
Caforio, A. L. P. et al. A prospective study of biopsy-proven myocarditis: prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur. Heart J. 28, 1326–1333 (2007).
Kuhl, U. Viral persistence in the myocardium is associated with progressive cardiac dysfunction. Circulation 112, 1965–1970 (2005).
Maekawa, Y., Ouzounian, M., Opavsky, M. A. & Liu, P. P. Connecting the missing link between dilated cardiomyopathy and viral myocarditis: virus, cytoskeleton, and innate immunity. Circulation 115, 5–8 (2006).
Kindermann, I. et al. Predictors of outcome in patients with suspected myocarditis. Circulation 118, 639–648 (2008).
Mahon, N. G. et al. Immunohistologic evidence of myocardial disease in apparently healthy relatives of patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 39, 455–462 (2002).
Caforio, A. L. et al. Evidence from family studies for autoimmunity in dilated cardiomyopathy. Lancet 344, 773–777 (1994).
Caforio, A. L. et al. Novel organ-specific circulating cardiac autoantibodies in dilated cardiomyopathy. J. Am. Coll. Cardiol. 15, 1527–1534 (1990).
Caforio, A. L. et al. Identification of alpha- and beta-cardiac myosin heavy chain isoforms as major autoantigens in dilated cardiomyopathy. Circulation 85, 1734–1742 (1992).
Caforio, A. L. P. et al. Prospective familial assessment in dilated cardiomyopathy: cardiac autoantibodies predict disease development in asymptomatic relatives. Circulation 115, 76–83 (2006).
Mestroni, L. et al. Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. Heart Muscle Disease Study Group. J. Am. Coll. Cardiol. 34, 181–190 (1999).
Neu, N. et al. Cardiac myosin induces myocarditis in genetically predisposed mice. J. Immunol. 139, 3630–3636 (1987).
Smith, S. C. & Allen, P. M. Myosin-induced acute myocarditis is a T cell-mediated disease. J. Immunol. 147, 2141–2147 (1991).
Li, Y., Heuser, J. S., Cunningham, L. C., Kosanke, S. D. & Cunningham, M. W. Mimicry and antibody-mediated cell signaling in autoimmune myocarditis. J. Immunol. 177, 8234–8240 (2006).
Kodama, M. et al. Rat dilated cardiomyopathy after autoimmune giant cell myocarditis. Circ. Res. 75, 278–284 (1994).
Elliott, J. F. et al. Autoimmune cardiomyopathy and heart block develop spontaneously in HLA-DQ8 transgenic IAbeta knockout NOD mice. Proc. Natl Acad. Sci. USA 100, 13447–13452 (2003).
Guler, M. L. et al. Two autoimmune diabetes loci influencing T cell apoptosis control susceptibility to experimental autoimmune myocarditis. J. Immunol. 174, 2167–2173 (2005).
Frustaci, A. Immunosuppressive therapy for active lymphocytic myocarditis: virological and immunologic profile of responders versus nonresponders. Circulation 107, 857–863 (2003).
Frustaci, A., Russo, M. A. & Chimenti, C. Randomized study on the efficacy of immunosuppressive therapy in patients with virus-negative inflammatory cardiomyopathy: the TIMIC study. Eur. Heart J. 30, 1995–2002 (2009).
Escher, F. et al. Long-term outcome of patients with virus-negative chronic myocarditis or inflammatory cardiomyopathy after immunosuppressive therapy. Clin. Res. Cardiol. 105, 1011–1020 (2016).
Kazzam, E. et al. Non-invasive assessmenty of systolic left ventricular function in systemic sclerosis. Eur. Heart J. 12, 151–156 (1991).
Goldenberg, J. et al. Symptomatic cardiac involvement in juvenile rheumatoid arthritis. Int. J. Cardiol. 34, 57–62 (1992).
Paradiso, M. et al. Evaluation of myocarial involvement in systemic lupus erythematosus by signal-averaged electrocardiography and echocardiography. Acta Cardiol. 56, 381–386 (2001).
Caforio, A. L. P. et al. Clinical implications of anti-heart autoantibodies in myocarditis and dilated cardiomyopathy. Autoimmunity 41, 35–45 (2008).
Meder, B. et al. A genome-wide association study identifies 6p21 as novel risk locus for dilated cardiomyopathy. Eur. Heart J. 35, 1069–1077 (2014).
Arbustini, E. et al. The MOGE(S) classification for a phenotype–genotype nomenclature of cardiomyopathy. J. Am. Coll. Cardiol. 62, 2046–2072 (2013).
Hazebroek, M. R. et al. Prognostic relevance of gene-environment interactions in patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 66, 1313–1323 (2015).
Schulze, K., Becker, B. F. & Schultheiss, H. P. Antibodies to the ADP/ATP carrier, an autoantigen in myocarditis and dilated cardiomyopathy, penetrate into myocardial cells and disturb energy metabolism in vivo. Circ. Res. 64, 179–192 (1989).
Caforio, A. L. P. et al. Passive transfer of affinity-purified anti-heart autoantibodies (AHA) from sera of patients with myocarditis induces experimental myocarditis in mice. Int. J. Cardiol. 179, 166–177 (2015).
Nikolaev, V. O. et al. A novel fluorescence method for the rapid detection of functional β1-adrenergic receptor autoantibodies in heart failure. J. Am. Coll. Cardiol. 50, 423–431 (2007).
Jahns, R. et al. Direct evidence for a β1-adrenergic receptor–directed autoimmune attack as a cause of idiopathic dilated cardiomyopathy. J. Clin. Invest. 113, 1419–1429 (2004).
Nishimura, H. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science 291, 319–322 (2001).
Okazaki, T. et al. Autoantibodies against cardiac troponin I are responsible for dilated cardiomyopathy in PD-1-deficient mice. Nat. Med. 9, 1477–1483 (2003).
Kaya, Z., Leib, C. & Katus, H. A. Autoantibodies in heart failure and cardiac dysfunction. Circ. Res. 110, 145–158 (2012).
Blyszczuk, P. et al. Transforming growth factor-β-dependent Wnt secretion controls myofibroblast formation and myocardial fibrosis progression in experimental autoimmune myocarditis. Eur. Heart J. 38, 1413–1425 (2016).
Piano, M. R. Alcoholic cardiomyopathy. Chest 121, 1638–1650 (2002).
Manthey, J., Imtiaz, S., Neufeld, M., Rylett, M. & Rehm, J. Quantifying the global contribution of alcohol consumption to cardiomyopathy. Popul. Health Metr. 15, 20 (2017).
Lang, R. M. Adverse cardiac effects of acute alcohol ingestion in young adults. Ann. Intern. Med. 102, 742 (1985).
Hantson, P. Mechanisms of toxic cardiomyopathy. Clin. Toxicol. (Phila.) 57, 1–9 (2018).
Waszkiewicz, N., Szulc, A. & Zwierz, K. Binge drinking-induced subtle myocardial injury. Alcohol. Clin. Exp. Res. 37, 1261–1263 (2013).
Havakuk, O., Rezkalla, S. H. & Kloner, R. A. The cardiovascular effects of cocaine. J. Am. Coll. Cardiol. 70, 101–113 (2017).
Om, A., Warner, M., Sabri, N., Cecich, L. & Vetrovec, G. Frequency of coronary artery disease and left ventricular dysfunction in cocaine users. Am. J. Cardiol. 69, 1549–1552 (1992).
Chakko, S. & Myerburg, R. J. Cardiac complications of cocaine abuse. Clin. Cardiol. 18, 67–72 (1995).
Varga, Z. V., Ferdinandy, P., Liaudet, L. & Pacher, P. Drug-induced mitochondrial dysfunction and cardiotoxicity. Am. J. Physiol. Heart Circ. Physiol. 309, H1453–H1467 (2015).
Virmani, R., Robinowitz, M., Smialek, J. E. & Smyth, D. F. Cardiovascular effects of cocaine: an autopsy study of 40 patients. Am. Heart J. 115, 1068–1076 (1988).
Bellinger, A. M. et al. Cardio-oncology: how new targeted cancer therapies and precision medicine can inform cardiovascular discovery. Circulation 132, 2248–2258 (2015).
Chatterjee, K., Zhang, J., Honbo, N. & Karliner, J. S. Doxorubicin cardiomyopathy. Cardiology 115, 155–162 (2010).
Zhang, S. et al. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat. Med. 18, 1639–1642 (2012).
Lipshultz, S. E. et al. Chronic progressive cardiac dysfunction years after doxorubicin therapy for childhood acute lymphoblastic leukemia. J. Clin. Oncol. 23, 2629–2636 (2005).
Lipshultz, S. E. & Adams, M. J. Cardiotoxicity after childhood cancer: beginning with the end in mind. J. Clin. Oncol. 28, 1276–1281 (2010).
Cardinale, D. et al. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation 114, 2474–2481 (2006).
Sheppard, R. et al. Myocardial expression of fas and recovery of left ventricular function in patients with recent-onset cardiomyopathy. J. Am. Coll. Cardiol. 46, 1036–1042 (2005).
Coronado, M. J. et al. Testosterone and interleukin-1β increase cardiac remodeling during coxsackievirus B3 myocarditis via serpin A 3n. Am. J. Physiol. Heart Circ. Physiol. 302, H1726–H1736 (2012).
Cocker, M. S., Abdel-Aty, H., Strohm, O. & Friedrich, M. G. Age and gender effects on the extent of myocardial involvement in acute myocarditis: a cardiovascular magnetic resonance study. Heart 95, 1925–1930 (2009).
Fairweather, D. et al. Interferon-gamma protects against chronic viral myocarditis by reducing mast cell degranulation, fibrosis, and the profibrotic cytokines transforming growth factor-beta 1, interleukin-1 beta, and interleukin-4 in the heart. Am. J. Pathol. 165, 1883–1894 (2004).
Baldeviano, G. C. et al. Interleukin-17A is dispensable for myocarditis but essential for the progression to dilated cardiomyopathy. Circ. Res. 106, 1646–1655 (2010).
Myers, J. M. et al. Cardiac myosin-Th17 responses promote heart failure in human myocarditis. JCI Insight 1, 85851 (2016).
Diny, N. L. et al. Eosinophil-derived IL-4 drives progression of myocarditis to inflammatory dilated cardiomyopathy. J. Exp. Med. 214, 943–957 (2017).
Frisancho-Kiss, S. et al. Gonadectomy of male BALB/c mice increases Tim-3(+) alternatively activated M2 macrophages, Tim-3(+) T cells, Th2 cells and Treg in the heart during acute coxsackievirus-induced myocarditis. Brain Behav. Immun. 23, 649–657 (2009).
Fairweather, D. et al. Sex differences in translocator protein 18 kDa (TSPO) in the heart: implications for imaging myocardial inflammation. J. Cardiovasc. Transl Res. 7, 192–202 (2014).
Regitz-Zagrosek, V. & Kararigas, G. Mechanistic pathways of sex differences in cardiovascular disease. Physiol. Rev. 97, 1–37 (2017).
Abston, E. D. et al. IL-33 independently induces eosinophilic pericarditis and cardiac dilation: ST2 improves cardiac function. Circ. Heart Fail. 5, 366–375 (2012).
Fairweather, D. et al. Complement receptor 1 and 2 deficiency increases coxsackievirus B3-induced myocarditis, dilated cardiomyopathy, and heart failure by increasing macrophages, IL-1beta, and immune complex deposition in the heart. J. Immunol. 176, 3516–3524 (2006).
Vitale, C., Mendelsohn, M. E. & Rosano, G. M. C. Gender differences in the cardiovascular effect of sex hormones. Nat. Rev. Cardiol. 6, 532–542 (2009).
Melchert, R. B. & Welder, A. A. Cardiovascular effects of androgenic-anabolic steroids. Med. Sci. Sports Exerc. 27, 1252–1262 (1995).
Scheuer, J., Malhotra, A., Schaible, T. F. & Capasso, J. Effects of gonadectomy and hormonal replacement on rat hearts. Circ. Res. 61, 12–19 (1987).
Dec, G. W. & Fuster, V. Idiopathic dilated cardiomyopathy. N. Engl. J. Med. 331, 1564–1575 (1994).
Caforio, A. L. P. et al. Diagnosis and management of myocardial involvement in systemic immune-mediated diseases: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Disease. Eur. Heart J. 38, 2649–2662 (2017).
Kuhl, U., Melzner, B., Schafer, B., Schultheiss, H.-P. & Strauer, B. E. The Ca-channel as cardiac autoantigen. Eur. Heart J. 12, 99–104 (1991).
Lauer, B., Schannwell, M., Kühl, U., Strauer, B.-E. & Schultheiss, H.-P. Antimyosin autoantibodies are associated with deterioration of systolic and diastolic left ventricular function in patients with chronic myocarditis. J. Am. Coll. Cardiol. 35, 11–18 (2000).
Rapezzi, C. et al. Diagnostic work-up in cardiomyopathies: bridging the gap between clinical phenotypes and final diagnosis. A position statement from the ESC Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 34, 1448–1458 (2012).
Fatkin, D., Seidman, C. E. & Seidman, J. G. Genetics and disease of ventricular muscle. Cold Spring Harb. Perspect. Med. 4, a021063 (2014).
Lynch, T. L. et al. Cardiac inflammation in genetic dilated cardiomyopathy caused by MYBPC3 mutation. J. Mol. Cell. Cardiol. 102, 83–93 (2017).
Knowlton, K. U. Myocarditis. J. Am. Coll. Cardiol. 69, 1666–1668 (2017).
Poller, W. et al. Genome-environment interactions in the molecular pathogenesis of dilated cardiomyopathy. J. Mol. Med. 83, 579–586 (2005).
Jan, M. F. & Tajik, A. J. Modern imaging techniques in cardiomyopathies. Circ. Res. 121, 874–891 (2017).
Kasner, M. et al. Multimodality imaging approach in the diagnosis of chronic myocarditis with preserved left ventricular ejection fraction (MCpEF): the role of 2D speckle-tracking echocardiography. Int. J. Cardiol. 243, 374–378 (2017).
Chimenti, C. & Frustaci, A. Contribution and risks of left ventricular endomyocardial biopsy in patients with cardiomyopathies: a retrospective study over a 28-year period. Circulation 128, 1531–1541 (2013).
Holzmann, M. et al. Complication rate of right ventricular endomyocardial biopsy via the femoral approach: a retrospective and prospective study analyzing 3048 diagnostic procedures over an 11-year period. Circulation 118, 1722–1728 (2008).
Escher, F. et al. Analysis of endomyocardial biopsies in suspected myocarditis — diagnostic value of left versus right ventricular biopsy. Int. J. Cardiol. 177, 76–78 (2014).
Lassner, D. et al. Improved diagnosis of idiopathic giant cell myocarditis and cardiac sarcoidosis by myocardial gene expression profiling. Eur. Heart J. 35, 2186–2195 (2014).
Escher, F. et al. Presence of perforin in endomyocardial biopsies of patients with inflammatory cardiomyopathy predicts poor outcome. Eur. J. Heart Fail. 16, 1066–1072 (2014). In this EMB-based analysis of the long-term disease course, perforin, a key predictor of poor outcome, was detected in the myocardium of a large cohort of patients with inflammatory cardiomyopathy.
Marchant, D. J. et al. Inflammation in myocardial diseases. Circ. Res. 110, 126–144 (2012).
Heymans, S. et al. Inflammation as a therapeutic target in heart failure? A scientific statement from the Translational Research Committee of the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 11, 119–129 (2009).
Glezeva, N. & Baugh, J. A. Role of inflammation in the pathogenesis of heart failure with preserved ejection fraction and its potential as a therapeutic target. Heart Fail. Rev. 19, 681–694 (2013).
O’Connor, C. M. et al. Efficacy and safety of exercise training in patients with chronic heart failure. JAMA 301, 1439 (2009).
McMurray, J. J. V. et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 371, 993–1004 (2014). This study compares the angiotensin receptor–neprilysin inhibitor LCZ696 with enalapril in patients who had heart failure with a reduced ejection fraction. LCZ696 was superior to enalapril in reducing the risks of death and of hospitalization for heart failure.
Zannad, F. et al. Eplerenone in patients with systolic heart failure and mild symptoms. N. Engl. J. Med. 364, 11–21 (2011).
Abraham, W. T. et al. Cardiac resynchronization in chronic heart failure. N. Engl. J. Med. 346, 1845–1853 (2002).
Packer, M. Effect of carvedilol on the morbidity of patients with severe chronic heart failure: results of the carvedilol prospective randomized cumulative survival (COPERNICUS) study. Circulation 106, 2194–2199 (2002).
Cleland, J. G. F. et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N. Engl. J. Med. 352, 1539–1549 (2005).
Cleland, J. G. F. et al. Longer-term effects of cardiac resynchronization therapy on mortality in heart failure [the CArdiac REsynchronization-Heart Failure (CARE-HF) trial extension phase]. Eur. Heart J. 27, 1928–1932 (2006).
Moss, A. J. et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N. Engl. J. Med. 361, 1329–1338 (2009).
Tang, A. S. L. et al. Cardiac-resynchronization therapy for mild-to-moderate heart failure. N. Engl. J. Med. 363, 2385–2395 (2010).
Chen, Y. et al. Impact of etiology on the outcomes in heart failure patients treated with cardiac resynchronization therapy: a meta-analysis. PLOS ONE 9, e94614 (2014).
Schultheiss, H.-P., Kuhl, U. & Cooper, L. T. The management of myocarditis. Eur. Heart J. 32, 2616–2625 (2011). This review article aims to help bridge the widening gap between recent mechanistic insights and their potential impact on disease burden. The article provides an overview of the entire management of myocarditis, particularly with respect to aetiology-based therapy.
Kuhl, U. et al. Interferon-beta treatment eliminates cardiotropic viruses and improves left ventricular function in patients with myocardial persistence of viral genomes and left ventricular dysfunction. Circulation 107, 2793–2798 (2003).
Kühl, U., Lassner, D., von Schlippenbach, J., Poller, W. & Schultheiss, H.-P. Interferon-beta improves survival in enterovirus-associated cardiomyopathy. J. Am. Coll. Cardiol. 60, 1295–1296 (2012).
Schultheiss, H.-P. et al. Betaferon in chronic viral cardiomyopathy (BICC) trial: effects of interferon-β treatment in patients with chronic viral cardiomyopathy. Clin. Res. Cardiol. 105, 763–773 (2016).
Felix, S. B. et al. Hemodynamic effects of immunoadsorption and subsequent immunoglobulin substitution in dilated cardiomyopathy. J. Am. Coll. Cardiol. 35, 1590–1598 (2000).
Mobini, R. et al. Hemodynamic improvement and removal of autoantibodies against beta1-adrenergic receptor by immunoadsorption therapy in dilated cardiomyopathy. J. Autoimmun. 20, 345–350 (2003).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00558584 (2018).
Priori, S. G. et al. 2015 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur. Heart J. 36, 2793–2867 (2015). This article includes the 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: the Task Force for the Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death of the ESC.
Desai, A. S., Fang, J. C., Maisel, W. H. & Baughman, K. L. Implantable defibrillators for the prevention of mortality in patients with nonischemic cardiomyopathy. JAMA 292, 2874 (2004).
Køber, L. et al. Defibrillator implantation in patients with nonischemic systolic heart failure. N. Engl. J. Med. 375, 1221–1230 (2016).
Golwala, H., Bajaj, N. S., Arora, G. & Arora, P. Implantable cardioverter-defibrillator for nonischemic cardiomyopathy: an updated meta-analysis. Circulation 135, 201–203 (2017).
Shi, H.-W. et al. Prognostic value of late gadolinium enhancement in dilated cardiomyopathy patients. A meta-analysis. Saudi Med. J. 34, 719–726 (2013).
Matsuo, S., Nakajima, K. & Nakata, T. Prognostic value of cardiac sympathetic nerve imaging using long-term follow-up data. Circulation 80, 435–441 (2016).
Solomou, S., Stavrou, M. & Marley, J. Diagnosis of dilated cardiomyopathy: patient reaction and adaptation — case study and review of the literature. Case Rep. Psychiatry 2016, 1756510 (2016).
MacInnes, J. & Williams, L. A review of integrated heart failure care. Prim. Health Care Res. Dev. https://doi.org/10.1017/S1463423618000312 (2018).
Rice, H., Say, R. & Betihavas, V. The effect of nurse-led education on hospitalisation, readmission, quality of life and cost in adults with heart failure. A systematic review. Patient Educ. Couns. 101, 363–374 (2018).
Mehani, S. H. M. Correlation between changes in diastolic dysfunction and health-related quality of life after cardiac rehabilitation program in dilated cardiomyopathy. J. Adv. Res. 4, 189–200 (2013).
Ohira, H. et al. Comparison of 18F-fluorodeoxyglucose positron emission tomography (FDG PET) and cardiac magnetic resonance (CMR) in corticosteroid-naive patients with conduction system disease due to cardiac sarcoidosis. Eur. J. Nucl. Med. Mol. Imaging 43, 259–269 (2015).
Werner, R. A. et al. Longitudinal 18F-FDG PET imaging in a rat model of autoimmune myocarditis. Eur. Heart J. Cardiovasc. Imaging 20, 467–474 (2018).
Klaeboe, L. G. & Edvardsen, T. Echocardiographic assessment of left ventricular systolic function. J. Echocardiogr. 17, 10–16 (2018).
Luis, S. A., Chan, J. & Pellikka, P. A. Echocardiographic assessment of left ventricular systolic function: an overview of contemporary techniques, including speckle-tracking echocardiography. Mayo Clin. Proc. 94, 125–138 (2019).
Weinberg, E. O. et al. Identification of serum soluble ST2 receptor as a novel heart failure biomarker. Circulation 107, 721–726 (2003).
Rehman, S. U., Mueller, T. & Januzzi, J. L. Characteristics of the novel interleukin family biomarker ST2 in patients with acute heart failure. J. Am. Coll. Cardiol. 52, 1458–1465 (2008).
Binas, D. et al. The prognostic value of sST2 and galectin-3 considering different aetiologies in non-ischaemic heart failure. Open Heart 5, e000750 (2018).
Coronado, M. J. et al. Elevated sera sST 2 is associated with heart failure in men ≤50 years old with myocarditis. J. Am. Heart Assoc. 8, e008968 (2019).
Weinberg, E. O. et al. Expression and regulation of ST2, an interleukin-1 receptor family member, in cardiomyocytes and myocardial infarction. Circulation 106, 2961–2966 (2002).
Villacorta, H. & Maisel, A. S. Soluble ST2 testing: a promising biomarker in the management of heart failure. Arq. Bras. Cardiol. 106, 145–152 (2016).
Lichtenauer, M. et al. A comparative analysis of novel cardiovascular biomarkers in patients with chronic heart failure. Eur. J. Intern. Med. 44, 31–38 (2017).
Jirak, P. et al. Influences of Ivabradine treatment on serum levels of cardiac biomarkers sST2, GDF-15, suPAR and H-FABP in patients with chronic heart failure. Acta Pharmacol. Sin. 39, 1189–1196 (2018).
Nair, N. & Gongora, E. Correlations of GDF-15 with sST2, MMPs, and worsening functional capacity in idiopathic dilated cardiomyopathy. J. Circ. Biomark. 7, 184945441775173 (2018).
Stojkovic, S. et al. GDF-15 is a better complimentary marker for risk stratification of arrhythmic death in non-ischaemic, dilated cardiomyopathy than soluble ST2. J. Cell. Mol. Med. 22, 2422–2429 (2018).
Pascual-Figal, D. A. et al. Soluble ST2 for predicting sudden cardiac death in patients with chronic heart failure and left ventricular systolic dysfunction. J. Am. Coll. Cardiol. 54, 2174–2179 (2009).
Basile, U. et al. Free light chains: eclectic multipurpose biomarker. J. Immunol. Methods 451, 11–19 (2017).
Dispenzieri, A. et al. Use of nonclonal serum immunoglobulin free light chains to predict overall survival in the general population. Mayo Clin. Proc. 87, 517–523 (2012).
Jackson, C. E. et al. Combined free light chains are novel predictors of prognosis in heart failure. JACC Heart Fail. 3, 618–625 (2015).
Jackson, C. E. et al. The incremental prognostic and clinical value of multiple novel biomarkers in heart failure. Eur. J. Heart Fail. 18, 1491–1498 (2016).
Saleh, A. et al. Assessment of cardiac involvement of hepatitis C virus; tissue Doppler imaging and NTproBNP study. J. Saudi Heart Assoc. 23, 217–223 (2011).
Wang, L. et al. The biomarker N-terminal pro-brain natriuretic peptide and liver diseases. Clin. Invest. Med. 34, E30–E37 (2011).
Minton, E. J. et al. Association between MHC class II alleles and clearance of circulating hepatitis C virus. Members of the Trent Hepatitis C Virus Study Group. J. Infect. Dis 178, 39–44 (1998).
Höhler, T. et al. MHC class II genes influence the susceptibility to chronic active hepatitis C. J. Hepatol. 27, 259–264 (1997).
Matsumori, A. et al. in Cardiomyopathies and Heart Failure: Biomolecular, Infectious, and Immune Mechanisms (Kluwer Academic Publishers, 2003).
Shichi, D. et al. The haplotype block, NFKBIL1-ATP6V1G2-BAT1-MICB-MICA, within the class III - class I boundary region of the human major histocompatibility complex may control susceptibility to hepatitis C virus-associated dilated cardiomyopathy. Tissue Antigens 66, 200–208 (2005).
Hsu, Y.-C. et al. Antiviral treatment for hepatitis C virus infection is associated with improved renal and cardiovascular outcomes in diabetic patients. Hepatology 59, 1293–1302 (2014).
Ly, K. N., Hughes, E. M., Jiles, R. B. & Holmberg, S. D. Rising mortality associated with hepatitis C virus in the United States, 2003–2013. Clin. Infect. Dis 62, 1287–1288 (2016).
Kawai, C. & Matsumori, A. Dilated cardiomyopathy update: infectious-immune theory revisited. Heart Fail. Rev. 18, 703–714 (2013). This review article focuses on immune reactions in the myocardium, evoked by external triggers, that continue insidiously and lead to the process of cardiac remodelling with ventricular dilatation, systolic dysfunction and DCM.
Matsumori, A., Shimada, T., Chapman, N. M., Tracy, S. M. & Mason, J. W. Myocarditis and heart failure associated with hepatitis C virus infection. J. Card. Fail. 12, 293–298 (2006).
Davidson, S. M. et al. Circulating blood cells and extracellular vesicles in acute cardioprotection. Cardiovasc. Res. https://doi.org/10.1093/cvr/cvy314 (2018).
Micheu, M. M., Scarlatescu, A. I., Scafa-Udriste, A. & Dorobantu, M. The winding road of cardiac regeneration—stem cell omics in the spotlight. Cells 7, 255 (2018).
Isomi, M., Sadahiro, T. & Ieda, M. Progress and challenge of cardiac regeneration to treat heart failure. J. Cardiol. 73, 97–101 (2019).
Arbustini, E. et al. Cardiac phenotypes in hereditary muscle disorders. J. Am. Coll. Cardiol. 72, 2485–2506 (2018).
Stergiopoulos, K. & Lima, F. V. Peripartum cardiomyopathy-diagnosis, management, and long term implications. Trends Cardiovasc. Med. 29, 164–173 (2018).
Willott, R. H. et al. Mutations in Troponin that cause HCM, DCM AND RCM: what can we learn about thin filament function? J. Mol. Cell. Cardiol. 48, 882–892 (2010).
Hershberger, R. E. et al. Clinical and functional characterization of TNNT2 mutations identified in patients with dilated cardiomyopathy. Circ. Cardiovasc. Genet. 2, 306–313 (2009).
Ware, J. S. & Cook, S. A. Role of titin in cardiomyopathy: from DNA variants to patient stratification. Nat. Rev. Cardiol. 15, 241–252 (2018).
Knezevic, T. et al. BAG3: a new player in the heart failure paradigm. Heart Fail. Rev. 20, 423–434 (2015).
Liu, G.-S. et al. A novel human R25C-phospholamban mutation is associated with super-inhibition of calcium cycling and ventricular arrhythmia. Cardiovasc. Res. 107, 164–174 (2015).
Sen-Chowdhry, S. et al. Left-dominant arrhythmogenic cardiomyopathy: an under-recognized clinical entity. J. Am. Coll. Cardiol. 52, 2175–2187 (2008).
Gerull, B. et al. Identification of a novel frameshift mutation in the giant muscle filament titin in a large Australian family with dilated cardiomyopathy. J. Mol. Med. 84, 478–483 (2006).
Norton, N. et al. Exome sequencing and genome-wide linkage analysis in 17 families illustrate the complex contribution of TTN truncating variants to dilated cardiomyopathy. Circ. Cardiovasc. Genet. 6, 144–153 (2013).
Roberts, A. M. et al. Integrated allelic, transcriptional, and phenomic dissection of the cardiac effects of titin truncations in health and disease. Sci. Transl Med. 7, 270ra6 (2015).
Ware, J. S. et al. Shared genetic predisposition in peripartum and dilated cardiomyopathies. N. Engl. J. Med. 374, 233–241 (2016).
Schafer, S. et al. Titin-truncating variants affect heart function in disease cohorts and the general population. Nat. Genet. 49, 46–53 (2017).
Taylor, M. R. G. et al. Natural history of dilated cardiomyopathy due to lamin A/C gene mutations. J. Am. Coll. Cardiol. 41, 771–780 (2003).
Parks, S. B. et al. Lamin A/C mutation analysis in a cohort of 324 unrelated patients with idiopathic or familial dilated cardiomyopathy. Am. Heart J. 156, 161–169 (2008).
Villard, E. et al. Mutation screening in dilated cardiomyopathy: prominent role of the beta myosin heavy chain gene. Eur. Heart J. 26, 794–803 (2005).
Arimura, T., Ishikawa, T., Nunoda, S., Kawai, S. & Kimura, A. Dilated cardiomyopathy-associated BAG3 mutations impair Z-disc assembly and enhance sensitivity to apoptosis in cardiomyocytes. Hum. Mutat. 32, 1481–1491 (2011).
Begay, R. L. et al. FLNC gene splice mutations cause dilated cardiomyopathy. JACC Basic Transl Sci. 1, 344–359 (2016).
Ortiz-Genga, M. F. et al. Truncating FLNC mutations are associated with high-risk dilated and arrhythmogenic cardiomyopathies. J. Am. Coll. Cardiol. 68, 2440–2451 (2016).
Li, D. et al. Identification of novel mutations in RBM20 in patients with dilated cardiomyopathy. Clin. Transl Sci. 3, 90–97 (2010).
McNair, W. P. et al. SCN5A mutation associated with dilated cardiomyopathy, conduction disorder, and arrhythmia. Circulation 110, 2163–2167 (2004).
Olson, T. M. et al. Sodium channel mutations and susceptibility to heart failure and atrial fibrillation. JAMA 293, 447–454 (2005).
Schmitt, J. P. et al. Dilated cardiomyopathy and heart failure caused by a mutation in phospholamban. Science 299, 1410–1413 (2003).
DeWitt, M. M., MacLeod, H. M., Soliven, B. & McNally, E. M. Phospholamban R14 deletion results in late-onset, mild, hereditary dilated cardiomyopathy. Am. J. Coll. Cardiol. 48, 1396–1398 (2006).
Hershberger, R. E. et al. Coding sequence rare variants identified in MYBPC3, MYH6, TPM1, TNNC1, and TNNI3 from 312 patients with familial or idiopathic dilated cardiomyopathy. Circ. Cardiovasc. Genet. 3, 155–161 (2010).
Pinto, J. R. et al. Functional characterization of TNNC1 rare variants identified in dilated cardiomyopathy. J. Biol. Chem. 286, 34404–34412 (2011).
Murphy, R. T. et al. Novel mutation in cardiac troponin I in recessive idiopathic dilated cardiomyopathy. Lancet 363, 371–372 (2004).
Carballo, S. et al. Identification and functional characterization of cardiac troponin I as a novel disease gene in autosomal dominant dilated cardiomyopathy. Circ. Res. 105, 375–382 (2009).
Olson, T. M., Kishimoto, N. Y., Whitby, F. G. & Michels, V. V. Mutations that alter the surface charge of alpha-tropomyosin are associated with dilated cardiomyopathy. J. Mol. Cell. Cardiol. 33, 723–732 (2001).
Lakdawala, N. K. et al. Familial dilated cardiomyopathy caused by an alpha-tropomyosin mutation: the distinctive natural history of sarcomeric dilated cardiomyopathy. J. Am. Coll. Cardiol. 55, 320–329 (2010).
Garg, R. Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. Collaborative Group on ACE Inhibitor Trials. JAMA 273, 1450–1456 (1995).
Granger, C. B. et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: the CHARM-alternative trial. Lancet 362, 772–776 (2003).
Fauchier, L., Pierre, B., de Labriolle, A. & Babuty, D. Comparison of the beneficial effect of beta-blockers on mortality in patients with ischaemic or non-ischaemic systolic heart failure: a meta-analysis of randomised controlled trials. Eur. J. Heart Fail. 9, 1136–1139 (2007).
Pitt, B. et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N. Engl. J. Med. 341, 709–717 (1999).
Taylor, A. L. et al. Early and sustained benefit on event-free survival and heart failure hospitalization from fixed-dose combination of isosorbide dinitrate/hydralazine: consistency across subgroups in the African-American heart failure trial. Circulation 115, 1747–1753 (2007).
Swedberg, K. et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet 376, 875–885 (2010).
The Digitalis Investigation Group. The effect of digoxin on mortality and morbidity in patients with heart failure. N. Engl. J. Med. 336, 525–533 (1997).
Acknowledgements
This work was partly supported by NIH R01 HL111938, NIH R21 ES024414 and American Heart Association grant AHA 16GRNT30950007 to D.F., and by ERA-Net grant on Cardiovascular Diseases (ERA-CVD; JTC2016-40-158), and grants of the German Research Foundation (DFG), Transregional Collaborative Research Center (CRC TR19) to H.P.S and F.E.
Reviewer information
Nature Reviews Disease Primers thanks S. Morimoto, B. Pinamonti, Y. Sawa and the other anonymous reviewer(s), for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
Introduction (H.-P.S. and P.P.L.); Epidemiology (S.E.L. and D.F.); Mechanisms/pathophysiology (R.E.H., P.P.L., A.L.P.C. and D.F.); Diagnosis, screening and prevention (H.-P.S. and F.E.); Management (J.M., H.-P.S., A.Maz. and S.G.P.); Quality of life (H.-P.S. and F.E.); Outlook (A.Mat.); Overview of Primer (H.-P.S. and D.F.).
Corresponding authors
Ethics declarations
Competing interests
All authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schultheiss, HP., Fairweather, D., Caforio, A.L.P. et al. Dilated cardiomyopathy. Nat Rev Dis Primers 5, 32 (2019). https://doi.org/10.1038/s41572-019-0084-1
Published:
DOI: https://doi.org/10.1038/s41572-019-0084-1
This article is cited by
-
Familial dilated cardiomyopathy in a child: a case report
BMC Pediatrics (2024)
-
HTRA1-driven detachment of type I collagen from endoplasmic reticulum contributes to myocardial fibrosis in dilated cardiomyopathy
Journal of Translational Medicine (2024)
-
Intestinal microbiota and metabolome perturbations in ischemic and idiopathic dilated cardiomyopathy
Journal of Translational Medicine (2024)
-
Construction and evaluation of immune-related diagnostic model in patients with heart failure caused by idiopathic dilated cardiomyopathy
BMC Cardiovascular Disorders (2024)
-
The impact of type 2 diabetes mellitus on the clinical profile, myocardial fibrosis, and prognosis in non-ischemic dilated cardiomyopathy: a prospective cohort study
Cardiovascular Diabetology (2024)