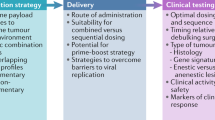
Abstract
Oncolytic viruses (OVs) are an emerging class of cancer therapeutics that offer the benefits of selective replication in tumour cells, delivery of multiple eukaryotic transgene payloads, induction of immunogenic cell death and promotion of antitumour immunity, and a tolerable safety profile that largely does not overlap with that of other cancer therapeutics. To date, four OVs and one non-oncolytic virus have been approved for the treatment of cancer globally although talimogene laherparepvec (T-VEC) remains the only widely approved therapy. T-VEC is indicated for the treatment of patients with recurrent melanoma after initial surgery and was initially approved in 2015. An expanding body of data on the clinical experience of patients receiving T-VEC is now becoming available as are data from clinical trials of various other OVs in a range of other cancers. Despite increasing research interest, a better understanding of the underlying biology and pharmacology of OVs is needed to enable the full therapeutic potential of these agents in patients with cancer. In this Review, we summarize the available data and provide guidance on optimizing the use of OVs in clinical practice, with a focus on the clinical experience with T-VEC. We describe data on selected novel OVs that are currently in clinical development, either as monotherapies or as part of combination regimens. We also discuss some of the preclinical, clinical and regulatory hurdles that have thus far limited the development of OVs.
Key points
-
Talimogene laherparepvec (T-VEC) for patients with melanoma is the first widely approved oncolytic virus, and real-world data from the past 7 years have optimized the role of T-VEC, including identifying patients who are most likely to derive benefit.
-
Research involving T-VEC has since been expanded to clinical trials involving patients with other cancers, earlier administration including in the neoadjuvant setting and combination with other therapeutic agents.
-
Three other oncolytic viruses have been approved in one or a few countries; one non-oncolytic virus was FDA approved for non-muscle invasive bladder cancer in December 2022 and other oncolytic viruses are in clinical development for a variety of cancer indications.
-
Novel oncolytic viruses typically have a tolerable safety profile and several have encouraging levels of activity in early phase clinical studies; nonetheless, challenges remain in optimizing appropriate clinical end points, regulatory pathways and clinical logistics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ilkow, C. S., Swift, S. L., Bell, J. C. & Diallo, J. S. From scourge to cure: tumour-selective viral pathogenesis as a new strategy against cancer. PLoS Pathog. 10, e1003836 (2014).
Macedo, N., Miller, D. M., Haq, R. & Kaufman, H. L. Clinical landscape of oncolytic virus research in 2020. J. Immunother. Cancer https://doi.org/10.1136/jitc-2020-001486 (2020).
Kaufman, H. L., Kohlhapp, F. J. & Zloza, A. Oncolytic viruses: a new class of immunotherapy drugs. Nat. Rev. Drug Discov. 14, 642–662 (2015).
Andtbacka, R. H. et al. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J. Clin. Oncol. 33, 2780–2788 (2015).
Russell, S. J., Peng, K. W. & Bell, J. C. Oncolytic virotherapy. Nat. Biotechnol. 30, 658–670 (2012).
Hu, J. C. et al. A phase I study of OncoVEXGM-CSF, a second-generation oncolytic herpes simplex virus expressing granulocyte macrophage colony-stimulating factor. Clin. Cancer Res. 12, 6737–6747 (2006).
Liu, B. L. et al. ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties. Gene Ther. 10, 292–303 (2003).
Roberts, M. S., Groene, W. S., Lorence, R. M. & Bamat, M. K. Naturally occurring viruses for the treatment of cancer. Discov. Med. 6, 217–222 (2006).
Bommareddy, P. K., Aspromonte, S., Zloza, A., Rabkin, S. D. & Kaufman, H. L. MEK inhibition enhances oncolytic virus immunotherapy through increased tumor cell killing and T cell activation. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aau0417 (2018).
Filley, A. C. & Dey, M. Immune system, friend or foe of oncolytic virotherapy. Front. Oncol. 7, 106 (2017).
Hu, J. C. et al. A novel HSV-1 virus, JS1/34.5-/47-, purges contaminating breast cancer cells from bone marrow. Clin. Cancer Res. 12, 6853–6862 (2006).
Postow, M. A. et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 366, 925–931 (2012).
Senzer, N. N. et al. Phase II clinical trial of a granulocyte-macrophage colony-stimulating factor-encoding, second-generation oncolytic herpesvirus in patients with unresectable metastatic melanoma. J. Clin. Oncol. 27, 5763–5771 (2009).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Spitler, L. E. et al. Recombinant human granulocyte-macrophage colony-stimulating factor (GM-CSF, sargramostim) administered for 3 years as adjuvant therapy of stages II(T4), III, and IV melanoma. J. Immunother. 32, 632–637 (2009).
Kaufman, H. L. et al. Durable response rate as an endpoint in cancer immunotherapy: insights from oncolytic virus clinical trials. J. Immunother. Cancer 5, 72 (2017).
Andtbacka, R. H. I. et al. Final analyses of OPTiM: a randomized phase III trial of talimogene laherparepvec versus granulocyte-macrophage colony-stimulating factor in unresectable stage III-IV melanoma. J. Immunother. Cancer 7, 145 (2019).
Louie, R. J. et al. Real-world outcomes of talimogene laherparepvec therapy: a multi-institutional experience. J. Am. Coll. Surg. 228, 644–649 (2019).
Perez, M. C. et al. Observational study of talimogene laherparepvec use for melanoma in clinical practice in the United States (COSMUS-1). Melanoma Manag. 6, MMT19 (2019).
Louie, K. S. et al. Real-world use of talimogene laherparepvec in Germany: a retrospective observational study using a prescription database. Future Oncol. 16, 317–328 (2020).
Sun, J. et al. Observational study of talimogene laherparepvec use in the anti-PD-1 era for melanoma in the US (COSMUS-2). Melanoma Manag. 7, MMT41 (2020).
Kleemann, J. et al. Real-world experience of talimogene laherparepvec (T-VEC) in old and oldest-old patients with melanoma: a retrospective single center study. Cancer Manag. Res. 13, 5699–5709 (2021).
van Akkooi, A. C. J. et al. A retrospective chart review study of real-world use of talimogene laherparepvec in unresectable stage IIIB-IVM1a melanoma in four European countries. Adv. Ther. 38, 1245–1262 (2021).
Carr, M. J. et al. Talimogene laherparepvec (T-VEC) for the treatment of advanced locoregional melanoma after failure of immunotherapy: an international multi-institutional experience. Ann. Surg. Oncol. 29, 791–801 (2022).
Stahlie, E. H. A. et al. Single agent talimogene laherparepvec for stage IIIB-IVM1c melanoma patients: a systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 175, 103705 (2022).
Andtbacka, R. H. et al. Cutaneous head and neck melanoma in OPTiM, a randomized phase 3 trial of talimogene laherparepvec versus granulocyte-macrophage colony-stimulating factor for the treatment of unresected stage IIIB/IIIC/IV melanoma. Head Neck 38, 1752–1758 (2016).
Harrington, K. J. et al. Phase I/II study of oncolytic HSV GM-CSF in combination with radiotherapy and cisplatin in untreated stage III/IV squamous cell cancer of the head and neck. Clin. Cancer Res. 16, 4005–4015 (2010).
Chang, K. J., Senzer, N. N., Binmoeller, K., Goldsweig, H. & Coffin, R. Phase I dose-escalation study of talimogene laherparepvec (T-VEC) for advanced pancreatic cancer (ca). J. Clin. Oncol. 30 (Suppl. 15), e14546 (2012).
Topalian, S. L., Taube, J. M. & Pardoll, D. M. Neoadjuvant checkpoint blockade for cancer immunotherapy. Science https://doi.org/10.1126/science.aax0182 (2020).
Cercek, A. et al. PD-1 blockade in mismatch repair-deficient, locally advanced rectal cancer. N. Engl. J. Med. 386, 2363–2376 (2022).
Chalabi, M. et al. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat. Med. 26, 566–576 (2020).
Gross, N. D. et al. Neoadjuvant cemiplimab for stage II to IV cutaneous squamous-cell carcinoma. N. Engl. J. Med. 387, 1557–1568 (2022).
Forde, P. M. et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N. Engl. J. Med. 386, 1973–1985 (2022).
Patel, S. et al. Neoadjuvant versus adjuvant pembrolizumab for resected stage III-IV melanoma (SWOG 1801). Ann. Oncol. 33 (Suppl. 7), S808–S869 (2022).
Dummer, R. et al. Neoadjuvant talimogene laherparepvec plus surgery versus surgery alone for resectable stage IIIB-IVM1a melanoma: a randomized, open-label, phase 2 trial. Nat. Med. 27, 1789–1796 (2021).
Aghi, M., Rabkin, S. & Martuza, R. L. Effect of chemotherapy-induced DNA repair on oncolytic herpes simplex viral replication. J. Natl Cancer Inst. 98, 38–50 (2006).
Muthana, M. et al. Macrophage delivery of an oncolytic virus abolishes tumor regrowth and metastasis after chemotherapy or irradiation. Cancer Res. 73, 490–495 (2013).
Toyoizumi, T. et al. Combined therapy with chemotherapeutic agents and herpes simplex virus type 1 ICP34.5 mutant (HSV-1716) in human non-small cell lung cancer. Hum. Gene Ther. 10, 3013–3029 (1999).
Puzanov, I. et al. Talimogene laherparepvec in combination with ipilimumab in previously untreated, unresectable stage IIIB-IV melanoma. J. Clin. Oncol. 34, 2619–2626 (2016).
Bommareddy, P. K., Shettigar, M. & Kaufman, H. L. Integrating oncolytic viruses in combination cancer immunotherapy. Nat. Rev. Immunol. 18, 498–513 (2018).
Chesney, J. et al. Randomized, open-label phase II study evaluating the efficacy and safety of talimogene laherparepvec in combination with ipilimumab versus ipilimumab alone in patients with advanced, unresectable melanoma. J. Clin. Oncol. 36, 1658–1667 (2018).
Ribas, A. et al. Oncolytic virotherapy promotes intratumoral T cell infiltration and improves anti-PD-1 immunotherapy. Cell 170, 1109–1119.e10 (2017).
Chesney, J. A. et al. Randomized, double-blind, placebo-controlled, global phase III trial of talimogene laherparepvec combined with pembrolizumab for advanced melanoma. J. Clin. Oncol. https://doi.org/10.1200/JCO.22.00343 (2022).
Chiocca, E. A. et al. Combined immunotherapy with controlled interleukin-12 gene therapy and immune checkpoint blockade in recurrent glioblastoma: an open-label, multi-institutional phase I trial. Neuro-Oncology 24, 951–963 (2022).
Passaro, C. et al. Arming an oncolytic herpes simplex virus type 1 with a single-chain fragment variable antibody against PD-1 for experimental glioblastoma therapy. Clin. Cancer Res. 25, 290–299 (2019).
Watanabe, N., McKenna, M. K., Rosewell Shaw, A. & Suzuki, M. Clinical CAR-T cell and oncolytic virotherapy for cancer treatment. Mol. Ther. 29, 505–520 (2021).
Huang, J. et al. Interleukin-7-loaded oncolytic adenovirus improves CAR-T cell therapy for glioblastoma. Cancer Immunol. Immunother. 70, 2453–2465 (2021).
Li, Y. et al. Oncolytic adenovirus targeting TGF-β enhances anti-tumor responses of mesothelin-targeted chimeric antigen receptor T cell therapy against breast cancer. Cell Immunol. 348, 104041 (2020).
Feist, M. et al. Oncolytic virus promotes tumor-reactive infiltrating lymphocytes for adoptive cell therapy. Cancer Gene Ther. 28, 98–111 (2021).
O’Cathail, S. M. et al. Combining oncolytic adenovirus with radiation — a paradigm for the future of radiosensitization. Front. Oncol. 7, 153 (2017).
Kyula, J. N. et al. Synergistic cytotoxicity of radiation and oncolytic Lister strain vaccinia in (V600D/E)BRAF mutant melanoma depends on JNK and TNF-α signaling. Oncogene 33, 1700–1712 (2014).
Gao, H. et al. Synergistic suppression effect on tumor growth of colorectal cancer by combining radiotherapy with a TRAIL-armed oncolytic adenovirus. Technol. Cancer Res. Treat. 18, 1533033819853290 (2019).
Shirakawa, Y. et al. Phase I dose-escalation study of endoscopic intratumoral injection of OBP-301 (Telomelysin) with radiotherapy in oesophageal cancer patients unfit for standard treatments. Eur. J. Cancer 153, 98–108 (2021).
Mao, L. J. et al. Oncolytic adenovirus harboring Interleukin-24 improves chemotherapy for advanced prostate cancer. J. Cancer 9, 4391–4397 (2018).
Dhar, D., Spencer, J. F., Toth, K. & Wold, W. S. Effect of preexisting immunity on oncolytic adenovirus vector INGN 007 antitumor efficacy in immunocompetent and immunosuppressed Syrian hamsters. J. Virol. 83, 2130–2139 (2009).
Qiao, J. et al. Cyclophosphamide facilitates antitumor efficacy against subcutaneous tumors following intravenous delivery of reovirus. Clin. Cancer Res. 14, 259–269 (2008).
Peng, K. W. et al. Using clinically approved cyclophosphamide regimens to control the humoral immune response to oncolytic viruses. Gene Ther. 20, 255–261 (2013).
Rizvi, N. A. et al. Nivolumab in combination with platinum-based doublet chemotherapy for first-line treatment of advanced non-small-cell lung cancer. J. Clin. Oncol. 34, 2969–2979 (2016).
Long, G. V. et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N. Engl. J. Med. 371, 1877–1888 (2014).
Luke, J. J., Flaherty, K. T., Ribas, A. & Long, G. V. Targeted agents and immunotherapies: optimizing outcomes in melanoma. Nat. Rev. Clin. Oncol. 14, 463–482 (2017).
Crespo-Rodriguez, E. et al. Combining BRAF inhibition with oncolytic herpes simplex virus enhances the immune-mediated antitumor therapy of BRAF-mutant thyroid cancer. J. Immunother. Cancer https://doi.org/10.1136/jitc-2020-000698 (2020).
Gartrell, R. D. et al. Combination immunotherapy including OncoVEX(mGMCSF) creates a favorable tumor immune micro-environment in transgenic BRAF murine melanoma. Cancer Immunol. Immunother. 71, 1837–1849 (2022).
Yamamura, K. et al. Combination treatment of human pancreatic cancer xenograft models with the epidermal growth factor receptor tyrosine kinase inhibitor erlotinib and oncolytic herpes simplex virus HF10. Ann. Surg. Oncol. 21, 691–698 (2014).
Wu, Z. et al. Combination of cetuximab and oncolytic virus canerpaturev synergistically inhibits human colorectal cancer growth. Mol. Ther. Oncolytics 13, 107–115 (2019).
Liang, M. et al. Targeting matrix metalloproteinase MMP3 greatly enhances oncolytic virus mediated tumor therapy. Transl. Oncol. 14, 101221 (2021).
Zou, J. et al. Establishment and genomic characterizations of patient-derived esophageal squamous cell carcinoma xenograft models using biopsies for treatment optimization. J. Transl. Med. 16, 15 (2018).
Ott, P. A., Hodi, F. S., Kaufman, H. L., Wigginton, J. M. & Wolchok, J. D. Combination immunotherapy: a road map. J. Immunother. Cancer 5, 16 (2017).
Krysko, D. V. et al. Immunogenic cell death and DAMPs in cancer therapy. Nat. Rev. Cancer 12, 860–875 (2012).
Riera Romo, M. Cell death as part of innate immunity: cause or consequence. Immunology 163, 399–415 (2021).
Casares, N. et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J. Exp. Med. 202, 1691–1701 (2005).
Matzinger, P. Tolerance, danger, and the extended family. Annu. Rev. Immunol. 12, 991–1045 (1994).
Wakimoto, H. et al. The complement response against an oncolytic virus is species-specific in its activation pathways. Mol. Ther. 5, 275–282 (2002).
Ikeda, K. et al. Complement depletion facilitates the infection of multiple brain tumors by an intravascular, replication-conditional herpes simplex virus mutant. J. Virol. 74, 4765–4775 (2000).
Nosaki, K. et al. A novel, polymer-coated oncolytic measles virus overcomes immune suppression and induces robust antitumor activity. Mol. Ther. Oncolytics 3, 16022 (2016).
Xia, M. et al. Graphene oxide arms oncolytic measles virus for improved effectiveness of cancer therapy. J. Exp. Clin. Cancer Res. 38, 408 (2019).
Lv, P. et al. Genetically engineered cell membrane nanovesicles for oncolytic adenovirus delivery: a versatile platform for cancer virotherapy. Nano Lett. 19, 2993–3001 (2019).
Muthana, M. et al. Use of macrophages to target therapeutic adenovirus to human prostate tumors. Cancer Res. 71, 1805–1815 (2011).
Mader, E. K. et al. Mesenchymal stem cell carriers protect oncolytic measles viruses from antibody neutralization in an orthotopic ovarian cancer therapy model. Clin. Cancer Res. 15, 7246–7255 (2009).
Power, A. T. et al. Carrier cell-based delivery of an oncolytic virus circumvents antiviral immunity. Mol. Ther. 15, 123–130 (2007).
Martinez-Quintanilla, J., He, D., Wakimoto, H., Alemany, R. & Shah, K. Encapsulated stem cells loaded with hyaluronidase-expressing oncolytic virus for brain tumor therapy. Mol. Ther. 23, 108–118 (2015).
Chen, S. R., Chen, M. M., Ene, C., Lang, F. F. & Kan, P. Perfusion-guided endovascular super-selective intra-arterial infusion for treatment of malignant brain tumors. J. Neurointerv. Surg. 14, 533–538 (2022).
Nguyen, T. T. et al. Mutations in the IFNgamma-JAK-STAT pathway causing resistance to immune checkpoint inhibitors in melanoma increase sensitivity to oncolytic virus treatment. Clin. Cancer Res. 27, 3432–3442 (2021).
Bommareddy, P. K., Zloza, A., Rabkin, S. D. & Kaufman, H. L. Oncolytic virus immunotherapy induces immunogenic cell death and overcomes STING deficiency in melanoma. Oncoimmunology 8, 1591875 (2019).
Kaufman, H. L. Can biomarkers guide oncolytic virus immunotherapy. Clin. Cancer Res. 27, 3278–3279 (2021).
Harrington, K. J. et al. A practical guide to the handling and administration of talimogene laherparepvec in Europe. OncoTargets Ther. 10, 3867–3880 (2017).
McBride, A., Valgus, J., Parsad, S., Sommermann, E. M. & Nunan, R. Pharmacy operationalization of the intralesional oncolytic immunotherapy talimogene laherparepvec. Hosp. Pharm. 53, 296–302 (2018).
Hom, V., Karonis, E., Sigidi, T. & Cawley, K. Development of a nursing policy for the administration of an oncolytic virus in the outpatient setting. Semin. Oncol. Nurs. 35, 150928 (2019).
Gutzmer, R. et al. Practical clinical guide on the use of talimogene laherparepvec monotherapy in patients with unresectable melanoma in Europe. Eur. J. Dermatol. 28, 736–749 (2018).
Kaufman, H. L., Shalhout, S. Z. & Iodice, G. Talimogene laherparepvec: moving from first-in-class to best-in-class. Front. Mol. Biosci. 9, 834841 (2022).
Robilotti, E. V., Kumar, A., Glickman, M. S. & Kamboj, M. Viral oncolytic immunotherapy in the war on cancer: Infection control considerations. Infect. Control Hosp. Epidemiol. 40, 350–354 (2019).
Garcia-Carbonero, R. et al. Phase 1 study of intravenous administration of the chimeric adenovirus enadenotucirev in patients undergoing primary tumor resection. J. Immunother. Cancer 5, 71 (2017).
Samson, A. et al. Neoadjuvant intravenous oncolytic vaccinia virus therapy promotes anticancer immunity in patients. Cancer Immunol. Res. 10, 745–756 (2022).
Goldmacher, G. V. et al. Response criteria for intratumoral immunotherapy in solid tumors: itRECIST. J. Clin. Oncol. 38, 2667–2676 (2020).
Ramelyte, E. et al. Oncolytic virotherapy-mediated anti-tumor response: a single-cell perspective. Cancer Cell 39, 394–406.e4 (2021).
Harrington, K., Freeman, D. J., Kelly, B., Harper, J. & Soria, J. C. Optimizing oncolytic virotherapy in cancer treatment. Nat. Rev. Drug Discov. 18, 689–706 (2019).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Fukuhara, H., Ino, Y. & Todo, T. Oncolytic virus therapy: a new era of cancer treatment at dawn. Cancer Sci. 107, 1373–1379 (2016).
Todo, T. et al. Intratumoral oncolytic herpes virus G47 for residual or recurrent glioblastoma: a phase 2 trial. Nat. Med. 28, 1630–1639 (2022).
Karrasch, M. et al. Treatment of recurrent malignant glioma with G207, a genetically engineered herpes simplex virus-1, followed by irradiation: phase I study results. J. Clin. Oncol. 27 (Suppl. 15), 2042 (2009).
Markert, J. M. et al. A phase 1 trial of oncolytic HSV-1, G207, given in combination with radiation for recurrent GBM demonstrates safety and radiographic responses. Mol. Ther. 22, 1048–1055 (2014).
Friedman, G. K. et al. Oncolytic HSV-1 G207 immunovirotherapy for pediatric high-grade gliomas. N. Engl. J. Med. 384, 1613–1622 (2021).
Otani, Y. et al. NOTCH-induced MDSC recruitment after oHSV virotherapy in CNS cancer models modulates antitumor immunotherapy. Clin. Cancer Res. 28, 1460–1473 (2022).
Gallego Perez-Larraya, J. et al. Oncolytic DNX-2401 virus for pediatric diffuse intrinsic pontine glioma. N. Engl. J. Med. 386, 2471–2481 (2022).
Lang, F. F. et al. Phase I study of DNX-2401 (Delta-24-RGD) oncolytic adenovirus: replication and immunotherapeutic effects in recurrent malignant glioma. J. Clin. Oncol. 36, 1419–1427 (2018).
Desjardins, A. et al. Recurrent glioblastoma treated with recombinant poliovirus. N. Engl. J. Med. 379, 150–161 (2018).
Merrill, M. K. & Gromeier, M. The double-stranded RNA binding protein 76:NF45 heterodimer inhibits translation initiation at the rhinovirus type 2 internal ribosome entry site. J. Virol. 80, 6936–6942 (2006).
Gromeier, M., Lachmann, S., Rosenfeld, M. R., Gutin, P. H. & Wimmer, E. Intergeneric poliovirus recombinants for the treatment of malignant glioma. Proc. Natl Acad. Sci. USA 97, 6803–6808 (2000).
Chandramohan, V. et al. Validation of an immunohistochemistry assay for detection of CD155, the poliovirus receptor, in malignant gliomas. Arch. Pathol. Lab. Med. 141, 1697–1704 (2017).
Beasley, G. M. et al. Phase I trial of intratumoral PVSRIPO in patients with unresectable, treatment-refractory melanoma. J. Immunother. Cancer https://doi.org/10.1136/jitc-2020-002203 (2021).
Burke, J. M. et al. A first in human phase 1 study of CG0070, a GM-CSF expressing oncolytic adenovirus, for the treatment of nonmuscle invasive bladder cancer. J. Urol. 188, 2391–2397 (2012).
Packiam, V. T. et al. An open label, single-arm, phase II multicenter study of the safety and efficacy of CG0070 oncolytic vector regimen in patients with BCG-unresponsive non-muscle-invasive bladder cancer: interim results. Urol. Oncol. 36, 440–447 (2018).
Li, R. et al. CORE1: phase 2, single-arm study of CG0070 combined with pembrolizumab in patients with nonmuscle-invasive bladder cancer (NMIBC) unresponsive to bacillus Calmette-Guerin (BCG). J. Clin. Oncol. 40 (Suppl. 16), 4597 (2022).
Boorjian, S. A. et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 22, 107–117 (2021).
Streby, K. A. et al. First-in-human intravenous seprehvir in young cancer patients: a phase 1 clinical trial. Mol. Ther. 27, 1930–1938 (2019).
Andtbacka, R. H. I. et al. Final results of a phase II multicenter trial of HF10, a replication-competent HSV-1 oncolytic virus, and ipilimumab combination treatment in patients with stage IIIB-IV unresectable or metastatic melanoma. J. Clin. Oncol. 35 (Suppl. 15), 9510 (2017).
Ferris, R. L. et al. Phase I trial of intratumoral therapy using HF10, an oncolytic HSV-1, demonstrates safety in HSV+/HSV- patients with refractory and superficial cancers. J. Clin. Oncol. 32 (Suppl. 15), 6082 (2014).
Middleton, M. R. et al. An open-label, single-arm, phase II clinical trial of RP1, an enhanced potency oncolytic herpes virus, combined with nivolumab in four solid tumor types: initial results from the skin cancer cohorts. J. Clin. Oncol. 38 (Suppl. 15), e22050 (2020).
Gong, J. & Mita, M. M. Activated ras signaling pathways and reovirus oncolysis: an update on the mechanism of preferential reovirus replication in cancer cells. Front. Oncol. 4, 167 (2014).
Maitra, R., Ghalib, M. H. & Goel, S. Reovirus: a targeted therapeutic–progress and potential. Mol. Cancer Res. 10, 1514–1525 (2012).
Harrington, K. J., Vile, R. G., Melcher, A., Chester, J. & Pandha, H. S. Clinical trials with oncolytic reovirus: moving beyond phase I into combinations with standard therapeutics. Cytokine Growth Factor. Rev. 21, 91–98 (2010).
Sborov, D. W. et al. A phase I trial of single-agent reolysin in patients with relapsed multiple myeloma. Clin. Cancer Res. 20, 5946–5955 (2014).
Kolb, E. A. et al. A phase I trial and viral clearance study of reovirus (Reolysin) in children with relapsed or refractory extra-cranial solid tumors: a Children’s Oncology Group Phase I Consortium report. Pediatr. Blood Cancer 62, 751–758 (2015).
Parakrama, R. et al. Immune characterization of metastatic colorectal cancer patients post reovirus administration. BMC Cancer 20, 569 (2020).
Morris, D. G. et al. REO-001: a phase I trial of percutaneous intralesional administration of reovirus type 3 dearing (Reolysin(R)) in patients with advanced solid tumors. Invest. New Drugs 31, 696–706 (2013).
Villalona-Calero, M. A. et al. Oncolytic reovirus in combination with chemotherapy in metastatic or recurrent non-small cell lung cancer patients with KRAS-activated tumors. Cancer 122, 875–883 (2016).
Mahalingam, D. et al. A phase II study of pelareorep (REOLYSIN((R))) in combination with gemcitabine for patients with advanced pancreatic adenocarcinoma. Cancers https://doi.org/10.3390/cancers10060160 (2018).
Mahalingam, D. et al. Pembrolizumab in combination with the oncolytic virus pelareorep and chemotherapy in patients with advanced pancreatic adenocarcinoma: a phase Ib study. Clin. Cancer Res. 26, 71–81 (2020).
Hamid, O., Ismail, R. & Puzanov, I. Intratumoral immunotherapy — update 2019. Oncologist 25, e423–e438 (2020).
Xia, Z. J. et al. Phase III randomized clinical trial of intratumoral injection of E1B gene-deleted adenovirus (H101) combined with cisplatin-based chemotherapy in treating squamous cell cancer of head and neck or esophagus [Chinese]. Ai Zheng. 23, 1666–1670 (2004).
Liang, M. Oncorine, the world first oncolytic virus medicine and its update in China. Curr. Cancer Drug. Targets 18, 171–176 (2018).
Alberts, P., Tilgase, A., Rasa, A., Bandere, K. & Venskus, D. The advent of oncolytic virotherapy in oncology: the Rigvir(R) story. Eur. J. Pharmacol. 837, 117–126 (2018).
Taguchi, S., Fukuhara, H. & Todo, T. Oncolytic virus therapy in Japan: progress in clinical trials and future perspectives. Jpn. J. Clin. Oncol. 49, 201–209 (2019).
U.S. Department of Health and Human Services. Food and Drug Administration Center for Biologics Evaluation and Research. Design and Analysis of Shedding Studies for Virus or Bacteria-Based Gene Therapy and Oncolytic Products — Guidance for Industry, https://www.fda.gov/files/vaccines%2C%20blood%20%26%20biologics/published/Design-and-Analysis-of-Shedding-Studies-for-Virus-or-Bacteria-Based-Gene-Therapy-and-Oncolytic-Products--Guidance-for-Industry.pdf (2015).
Author information
Authors and Affiliations
Contributions
S.Z.S. and H.L.K. researched data for this manuscript and made a substantial contribution to discussions of content. All authors wrote and edited and/or reviewed this manuscript prior to submission.
Corresponding author
Ethics declarations
Competing interests
H.L.K. is an employee of Ankyra Therapeutics and has received honoraria for participating on advisory boards for Castle Biosciences and Marengo Therapeutics. D.M.M. has received honoraria for participating on advisory boards for Checkpoint Therapeutics, EMD Serono, Pfizer, Merck, Regeneron and Sanofi Genzyme. K.S.E. and S.Z.S. report no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks A. van Akkooi, J. Bell, E. A. Chiocca, G. Friedman and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shalhout, S.Z., Miller, D.M., Emerick, K.S. et al. Therapy with oncolytic viruses: progress and challenges. Nat Rev Clin Oncol 20, 160–177 (2023). https://doi.org/10.1038/s41571-022-00719-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-022-00719-w
This article is cited by
-
New immunotherapy approaches for colorectal cancer: focusing on CAR-T cell, BiTE, and oncolytic viruses
Cell Communication and Signaling (2024)
-
Intratumoural microbiota: a new frontier in cancer development and therapy
Signal Transduction and Targeted Therapy (2024)
-
Nucleic acid-based drugs for patients with solid tumours
Nature Reviews Clinical Oncology (2024)
-
Targeting immunogenic cell stress and death for cancer therapy
Nature Reviews Drug Discovery (2024)
-
SOCS3 inhibiting JAK-STAT pathway enhances oncolytic adenovirus efficacy by potentiating viral replication and T-cell activation
Cancer Gene Therapy (2024)