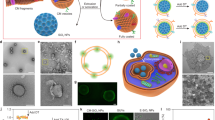
Abstract
Traditional cancer therapeutics, such as chemotherapies, are often limited by their non-specific nature, causing harm to non-malignant tissues. Over the past several decades, nanomedicine researchers have sought to address this challenge by developing nanoscale platforms capable of more precisely delivering drug payloads. Cell membrane-coated nanoparticles (CNPs) are an emerging class of nanocarriers that have demonstrated considerable promise for biomedical applications. Consisting of a synthetic nanoparticulate core camouflaged by a layer of naturally derived cell membranes, CNPs are adept at operating within complex biological environments; depending on the type of cell membrane utilized, the resulting biomimetic nanoformulation is conferred with several properties typically associated with the source cell, including improved biocompatibility, immune evasion and tumour targeting. In comparison with traditional functionalization approaches, cell membrane coating provides a streamlined method for creating multifunctional and multi-antigenic nanoparticles. In this Review, we discuss the history and development of CNPs as well as how these platforms have been used for cancer therapy. The application of CNPs for drug delivery, phototherapy and immunotherapy will be described in detail. Translational efforts are currently under way and further research to address key areas of need will ultimately be required to facilitate the successful clinical adoption of CNPs.
Key points
-
Cell membrane-coated nanoparticles (CNPs) are an emerging class of nanocarriers that are inherently multifunctional, combining the properties of synthetic nanoparticle cores with the bio-interfacing properties of cell membranes.
-
The type of membrane that is utilized is usually reflected in the biological properties of the resulting CNP, which can be further fine-tuned or augmented using various engineering approaches.
-
CNP technology has the potential to be applied in several therapeutic areas of oncology, including drug delivery, phototherapy and immunotherapy.
-
Efforts to translate promising CNPs into approved therapies are currently under way and will require the development of large-scale production methods and novel assays to facilitate the clinical adoption of CNPs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gangadhar, T. C. & Vonderheide, R. H. Mitigating the toxic effects of anticancer immunotherapy. Nat. Rev. Clin. Oncol. 11, 91–99 (2014).
Bower, J. E. Cancer-related fatigue—mechanisms, risk factors, and treatments. Nat. Rev. Clin. Oncol. 11, 597–609 (2014).
Stone, J. B. & DeAngelis, L. M. Cancer-treatment-induced neurotoxicity — focus on newer treatments. Nat. Rev. Clin. Oncol. 13, 92–105 (2016).
Holohan, C., Van Schaeybroeck, S., Longley, D. B. & Johnston, P. G. Cancer drug resistance: an evolving paradigm. Nat. Rev. Cancer 13, 714–726 (2013).
Vasan, N., Baselga, J. & Hyman, D. M. A view on drug resistance in cancer. Nature 575, 299–309 (2019).
Drago, J. Z., Modi, S. & Chandarlapaty, S. Unlocking the potential of antibody-drug conjugates for cancer therapy. Nat. Rev. Clin. Oncol. 18, 327–344 (2021).
Llovet, J. M., Montal, R., Sia, D. & Finn, R. S. Molecular therapies and precision medicine for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 15, 599–616 (2018).
Davis, M. E., Chen, Z. G. & Shin, D. M. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat. Rev. Drug Discov. 7, 771–782 (2008).
Wilhelm, S. et al. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 1, 16014 (2016).
Peer, D. et al. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2, 751–760 (2007).
Rosenblum, D., Joshi, N., Tao, W., Karp, J. M. & Peer, D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 9, 1410 (2018).
Mitchell, M. J. et al. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 20, 101–124 (2021).
Patra, J. K. et al. Nano based drug delivery systems: recent developments and future prospects. J. Nanobiotechnol. 16, 71 (2018).
Barenholz, Y. Doxil® — the first FDA-approved nano-drug: lessons learned. J. Control. Rel. 160, 117–134 (2012).
Yardley, D. A. nab-Paclitaxel mechanisms of action and delivery. J. Control. Rel. 170, 365–372 (2013).
Lancet, J. E. et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J. Clin. Oncol. 36, 2684–2692 (2018).
Shi, J., Kantoff, P. W., Wooster, R. & Farokhzad, O. C. Cancer nanomedicine: progress, challenges and opportunities. Nat. Rev. Cancer 17, 20–37 (2017).
Suk, J. S., Xu, Q. G., Kim, N., Hanes, J. & Ensign, L. M. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv. Drug Deliv. Rev. 99, 28–51 (2016).
Abu Lila, A. S., Kiwada, H. & Ishida, T. The accelerated blood clearance (ABC) phenomenon: clinical challenge and approaches to manage. J. Control. Rel. 172, 38–47 (2013).
Torchilin, V. Tumor delivery of macromolecular drugs based on the EPR effect. Adv. Drug Deliv. Rev. 63, 131–135 (2011).
Wu, C. H., Liu, I. J., Lu, R. M. & Wu, H. C. Advancement and applications of peptide phage display technology in biomedical science. J. Biomed. Sci. 23, 8 (2016).
Little, M., Kipriyanov, S. M., Le Gall, F. & Moldenhauer, G. Of mice and men: hybridoma and recombinant antibodies. Immunol. Today 21, 364–370 (2000).
Darmostuk, M., Rimpelova, S., Gbelcova, H. & Ruml, T. Current approaches in SELEX: an update to aptamer selection technology. Biotechnol. Adv. 33, 1141–1161 (2015).
Yoo, J. W., Irvine, D. J., Discher, D. E. & Mitragotri, S. Bio-inspired, bioengineered and biomimetic drug delivery carriers. Nat. Rev. Drug Discov. 10, 521–535 (2011).
Fang, R. H., Kroll, A. V., Gao, W. & Zhang, L. Cell membrane coating nanotechnology. Adv. Mater. 30, 1706759 (2018).
Fang, R. H., Jiang, Y., Fang, J. C. & Zhang, L. Cell membrane-derived nanomaterials for biomedical applications. Biomaterials 128, 69–83 (2017).
Molinaro, R. et al. Biomimetic proteolipid vesicles for targeting inflamed tissues. Nat. Mater. 15, 1037–1046 (2016).
Toledano Furman, N. E. et al. Reconstructed stem cell nanoghosts: a natural tumor targeting platform. Nano Lett. 13, 3248–3255 (2013).
Jiang, Y., Chekuri, S., Fang, R. H. & Zhang, L. Engineering biological interactions on the nanoscale. Curr. Opin. Biotechnol. 58, 1–8 (2019).
Krishnan, N., Fang, R. H. & Zhang, L. Engineering of stimuli-responsive self-assembled biomimetic nanoparticles. Adv. Drug Deliv. Rev. 179, 114006 (2021).
Oldenborg, P. A. et al. Role of CD47 as a marker of self on red blood cells. Science 288, 2051–2054 (2000).
Hu, C.-M. J. et al. ‘Marker-of-self’ functionalization of nanoscale particles through a top-down cellular membrane coating approach. Nanoscale 5, 2664–2668 (2013).
Fang, R. H. et al. Cancer cell membrane-coated nanoparticles for anticancer vaccination and drug delivery. Nano Lett. 14, 2181–2188 (2014).
Hu, C. M. et al. Nanoparticle biointerfacing by platelet membrane cloaking. Nature 526, 118–121 (2015).
Zhou, J. et al. Physical disruption of solid tumors by immunostimulatory microrobots enhances antitumor immunity. Adv. Mater. 33, 2103505 (2021).
Jiang, Y. et al. Engineered cell-membrane-coated nanoparticles directly present tumor antigens to promote anticancer immunity. Adv. Mater. 32, 2001808 (2020).
Hu, C. M. et al. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc. Natl Acad. Sci. USA 108, 10980–10985 (2011).
Noris, M. & Remuzzi, G. Overview of complement activation and regulation. Semin. Nephrol. 33, 479–492 (2013).
Fang, R. H., Hu, C. M. J. & Zhang, L. F. Nanoparticles disguised as red blood cells to evade the immune system. Expert Opin. Biol. Ther. 12, 385–389 (2012).
Rao, L. et al. Red blood cell membrane as a biomimetic nanocoating for prolonged circulation time and reduced accelerated blood clearance. Small 11, 6225–6236 (2015).
Olsson, M., Bruhns, P., Frazier, W. A., Ravetch, J. V. & Oldenborg, P. A. Platelet homeostasis is regulated by platelet expression of CD47 under normal conditions and in passive immune thrombocytopenia. Blood 105, 3577–3582 (2005).
Gay, L. J. & Felding-Habermann, B. Contribution of platelets to tumour metastasis. Nat. Rev. Cancer 11, 123–134 (2011).
Schlesinger, M. Role of platelets and platelet receptors in cancer metastasis. J. Hematol. Oncol. 11, 125 (2018).
Zamarron, B. F. & Chen, W. J. Dual roles of immune cells and their factors in cancer development and progression. Int. J. Biol. Sci. 7, 651–658 (2011).
Gonzalez, H., Hagerling, C. & Werb, Z. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 32, 1267–1284 (2018).
Harris, J. C., Scully, M. A. & Day, E. S. Cancer cell membrane-coated nanoparticles for cancer management. Cancers 11, 1836 (2019).
Rao, L. et al. Cancer cell membrane-coated upconversion nanoprobes for highly specific tumor imaging. Adv. Mater. 28, 3460–3466 (2016).
Glinsky, V. V. et al. Intravascular metastatic cancer cell homotypic aggregation at the sites of primary attachment to the endothelium. Cancer Res. 63, 3805–3811 (2003).
Kroll, A. V. et al. Nanoparticulate delivery of cancer cell membrane elicits multiantigenic antitumor immunity. Adv. Mater. 29, 1703969 (2017).
Ye, X. et al. Surgical tumor-derived personalized photothermal vaccine formulation for cancer immunotherapy. ACS Nano 13, 2956–2968 (2019).
Wang, M. et al. Recent advances in mesenchymal stem cell membrane-coated nanoparticles for enhanced drug delivery. Biomater. Sci. 9, 1088–1103 (2021).
Zhang, W. & Huang, X. Stem cell membrane-camouflaged targeted delivery system in tumor. Mater. Today Bio 16, 100377 (2022).
Li, J. et al. Cell membrane coated semiconducting polymer nanoparticles for enhanced multimodal cancer phototheranostics. ACS Nano 12, 8520–8530 (2018).
Krishnan, N. et al. Bacterial membrane vesicles for vaccine applications. Adv. Drug Deliv. Rev. 185, 114294 (2022).
Gao, W. W. et al. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles. Nano Lett. 15, 1403–1409 (2015).
Mogensen, T. H. Pathogen recognition and inflammatory signaling in innate immune defenses. Clin. Microbiol. Rev. 22, 240–273 (2009).
Zemek, R. M. et al. Sensitizing the tumor microenvironment to immune checkpoint therapy. Front. Immunol. 11, 223 (2020).
Drake, C. G. Combination immunotherapy approaches. Ann. Oncol. 23, viii41–viii46 (2012).
McCarthy, E. F. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop. J. 26, 154–158 (2006).
Holay, M. et al. Bacteria-inspired nanomedicine. ACS Appl. Bio Mater. 4, 3830–3848 (2021).
Zariri, A. et al. Meningococcal outer membrane vesicle composition-dependent activation of the innate immune response. Infect. Immun. 84, 3024–3033 (2016).
Moore, G. E. & Ulrich, K. Suspension cultures of mammalian cells: a review. J. Surg. Res. 5, 270–282 (1965).
Baek, E. J. et al. In vitro clinical-grade generation of red blood cells from human umbilical cord blood CD34+ cells. Transfusion 48, 2235–2245 (2008).
Ito, Y. et al. Turbulence activates platelet biogenesis to enable clinical scale ex vivo production. Cell 174, 636–648.e18 (2018).
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19, 213–228 (2018).
Holay, M. et al. Organotropic targeting of biomimetic nanoparticles to treat lung disease. Bioconjug. Chem. 33, 586–593 (2022).
Copp, J. A. et al. Clearance of pathological antibodies using biomimetic nanoparticles. Proc. Natl Acad. Sci. USA 111, 13481–13486 (2014).
Rao, L. et al. Microfluidic electroporation-facilitated synthesis of erythrocyte membrane-coated magnetic nanoparticles for enhanced imaging-guided cancer therapy. ACS Nano 11, 3496–3505 (2017).
Sherwood, J. et al. Cell-membrane coated iron oxide nanoparticles for isolation and specific identification of drug leads from complex matrices. Nanoscale 11, 6352–6359 (2019).
Luk, B. T. et al. Interfacial interactions between natural RBC membranes and synthetic polymeric nanoparticles. Nanoscale 6, 2730–2737 (2014).
Gao, W. et al. Surface functionalization of gold nanoparticles with red blood cell membranes. Adv. Mater. 25, 3549–3553 (2013).
Fang, R. H. et al. Lipid-insertion enables targeting functionalization of erythrocyte membrane-cloaked nanoparticles. Nanoscale 5, 8884–8888 (2013).
Dehaini, D. et al. Erythrocyte–platelet hybrid membrane coating for enhanced nanoparticle functionalization. Adv. Mater. 29, 1606209 (2017).
Park, J. H. et al. Genetically engineered cell membrane-coated nanoparticles for targeted delivery of dexamethasone to inflamed lungs. Sci. Adv. 7, eabf7820 (2021).
Park, J. H. et al. Virus-mimicking cell membrane-coated nanoparticles for cytosolic delivery of mRNA. Angew. Chem. Int. Ed. Engl. 61, e202113671 (2022).
Ai, X. et al. Surface glycan modification of cellular nanosponges to promote SARS-CoV-2 inhibition. J. Am. Chem. Soc. 143, 17615–17621 (2021).
Aryal, S. et al. Erythrocyte membrane-cloaked polymeric nanoparticles for controlled drug loading and release. Nanomedicine 8, 1271–1280 (2013).
Fu, Q. et al. Programmed co-delivery of paclitaxel and doxorubicin boosted by camouflaging with erythrocyte membrane. Nanoscale 7, 4020–4030 (2015).
Luk, B. T. et al. Safe and immunocompatible nanocarriers cloaked in RBC membranes for drug delivery to treat solid tumors. Theranostics 6, 1004–1011 (2016).
Su, J. et al. Enhanced blood suspensibility and laser-activated tumor-specific drug release of theranostic mesoporous silica nanoparticles by functionalizing with erythrocyte membranes. Theranostics 7, 523–537 (2017).
Zhang, L. et al. Erythrocyte membrane cloaked metal–organic framework nanoparticle as biomimetic nanoreactor for starvation-activated colon cancer therapy. ACS Nano 12, 10201–10211 (2018).
Zhuang, J. et al. Targeted gene silencing in vivo by platelet membrane–coated metal-organic framework nanoparticles. Sci. Adv. 6, eaaz6108 (2020).
Zhang, Y. et al. Erythrocyte membrane-coated nanogel for combinatorial antivirulence and responsive antimicrobial delivery against Staphylococcus aureus infection. J. Control. Rel. 263, 185–191 (2017).
Gao, C. et al. Stem cell membrane-coated nanogels for highly efficient in vivo tumor targeted drug delivery. Small 12, 4056–4062 (2016).
Zhang, X. et al. Remote loading of small-molecule therapeutics into cholesterol-enriched cell-membrane-derived vesicles. Angew. Chem. Int. Ed. Engl. 56, 14075–14079 (2017).
Zou, M. Z. et al. Artificial natural killer cells for specific tumor inhibition and renegade macrophage re-education. Adv. Mater. 31, 1904495 (2019).
Zhuang, J. et al. Biomimetic nanoemulsions for oxygen delivery in vivo. Adv. Mater. 30, 1804693 (2018).
Miao, Y. et al. Cell membrane-camouflaged nanocarriers with biomimetic deformability of erythrocytes for ultralong circulation and enhanced cancer therapy. ACS Nano 16, 6527–6540 (2022).
Zou, Y. et al. Effective and targeted human orthotopic glioblastoma xenograft therapy via a multifunctional biomimetic nanomedicine. Adv. Mater. 30, 1803717 (2018).
Chai, Z. et al. Ligand-modified cell membrane enables the targeted delivery of drug nanocrystals to glioma. ACS Nano 13, 5591–5601 (2019).
Zhai, Y. et al. Traceable bioinspired nanoparticle for the treatment of metastatic breast cancer via NIR-trigged intracellular delivery of methylene blue and cisplatin. Adv. Mater. 30, 1802378 (2018).
Sung, S. Y. et al. Graphene quantum dots-mediated theranostic penetrative delivery of drug and photolytics in deep tumors by targeted biomimetic nanosponges. Nano Lett. 19, 69–81 (2019).
Gao, L. et al. Erythrocyte membrane-wrapped pH sensitive polymeric nanoparticles for non-small cell lung cancer therapy. Bioconjug. Chem. 28, 2591–2598 (2017).
Song, Q. et al. Tumor microenvironment responsive nanogel for the combinatorial antitumor effect of chemotherapy and immunotherapy. Nano Lett. 17, 6366–6375 (2017).
Hu, Q. et al. Anticancer platelet-mimicking nanovehicles. Adv. Mater. 27, 7043–7050 (2015).
Li, J. et al. Targeted drug delivery to circulating tumor cells via platelet membrane-functionalized particles. Biomaterials 76, 52–65 (2016).
Hu, Q. et al. Engineered nanoplatelets for enhanced treatment of multiple myeloma and thrombus. Adv. Mater. 28, 9573–9580 (2016).
Zhang, M. et al. Platelet-mimicking biotaxis targeting vasculature-disrupted tumors for cascade amplification of hypoxia-sensitive therapy. ACS Nano 13, 14230–14240 (2019).
Ying, M. et al. Remote-loaded platelet vesicles for disease-targeted delivery of therapeutics. Adv. Funct. Mater. 28, 1801032 (2018).
Cao, H. Q. et al. Liposomes coated with isolated macrophage membrane can target lung metastasis of breast cancer. ACS Nano 10, 7738–7748 (2016).
Zhang, Y. et al. Macrophage-membrane-coated nanoparticles for tumor-targeted chemotherapy. Nano Lett. 18, 1908–1915 (2018).
Kang, T. et al. Nanoparticles coated with neutrophil membranes can effectively treat cancer metastasis. ACS Nano 11, 1397–1411 (2017).
Zhang, C. et al. Artificial super neutrophils for inflammation targeting and HClO generation against tumors and infections. Adv. Mater. 31, 1901179 (2019).
Sun, H. et al. Cancer-cell-biomimetic nanoparticles for targeted therapy of homotypic tumors. Adv. Mater. 28, 9581–9588 (2016).
Zhu, J. Y. et al. Preferential cancer cell self-recognition and tumor self-targeting by coating nanoparticles with homotypic cancer cell membranes. Nano Lett. 16, 5895–5901 (2016).
Shao, D. et al. Bioinspired diselenide-bridged mesoporous silica nanoparticles for dual-responsive protein delivery. Adv. Mater. 30, 1801198 (2018).
Shao, D. et al. Biomimetic diselenide-bridged mesoporous organosilica nanoparticles as an X-ray-responsive biodegradable carrier for chemo-immunotherapy. Adv. Mater. 32, 2004385 (2020).
Bose, R. J. C. et al. Tumor cell-derived extracellular vesicle-coated nanocarriers: an efficient theranostic platform for the cancer-specific delivery of anti-miR-21 and imaging agents. ACS Nano 12, 10817–10832 (2018).
Xie, W. et al. Cancer cell membrane camouflaged nanoparticles to realize starvation therapy together with checkpoint blockades for enhancing cancer therapy. ACS Nano 13, 2849–2857 (2019).
Chitgupi, U., Qin, Y. & Lovell, J. F. Targeted nanomaterials for phototherapy. Nanotheranostics 1, 38–58 (2017).
Li, X., Lovell, J. F., Yoon, J. & Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 17, 657–674 (2020).
Piao, J. G. et al. Erythrocyte membrane is an alternative coating to polyethylene glycol for prolonging the circulation lifetime of gold nanocages for photothermal therapy. ACS Nano 8, 10414–10425 (2014).
Ren, X. et al. Red blood cell membrane camouflaged magnetic nanoclusters for imaging-guided photothermal therapy. Biomaterials 92, 13–24 (2016).
Liu, Z. et al. Fabrication of red blood cell membrane-camouflaged Cu2-xSe nanoparticles for phototherapy in the second near-infrared window. Chem. Commun. 55, 6523–6526 (2019).
Rao, L. et al. Antitumor platelet-mimicking magnetic nanoparticles. Adv. Funct. Mater. 27, 1604774 (2017).
Chen, Z. et al. Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dual-modal imaging and photothermal therapy. ACS Nano 10, 10049–10057 (2016).
Han, Y. et al. T cell membrane mimicking nanoparticles with bioorthogonal targeting and immune recognition for enhanced photothermal therapy. Adv. Sci. 6, 1900251 (2019).
Wang, D. et al. Erythrocyte–cancer hybrid membrane camouflaged hollow copper sulfide nanoparticles for prolonged circulation life and homotypic-targeting photothermal/chemotherapy of melanoma. ACS Nano 12, 5241–5252 (2018).
Gao, C. et al. In vivo hitchhiking of immune cells by intracellular self-assembly of bacteria-mimetic nanomedicine for targeted therapy of melanoma. Sci. Adv. 8, eabn1805 (2022).
Li, S. Y. et al. Cancer cell membrane camouflaged cascade bioreactor for cancer targeted starvation and photodynamic therapy. ACS Nano 11, 7006–7018 (2017).
Yang, X. et al. Biomimetic hybrid nanozymes with self-supplied H+ and accelerated O2 generation for enhanced starvation and photodynamic therapy against hypoxic tumors. Nano Lett. 19, 4334–4342 (2019).
Wang, J. P. et al. A porous Au@Rh bimetallic core–shell nanostructure as an H2O2-driven oxygenerator to alleviate tumor hypoxia for simultaneous bimodal imaging and enhanced photodynamic therapy. Adv. Mater. 32, 2001862 (2020).
Liu, C. et al. Biodegradable biomimic copper/manganese silicate nanospheres for chemodynamic/photodynamic synergistic therapy with simultaneous glutathione depletion and hypoxia relief. ACS Nano 13, 4267–4277 (2019).
Wang, Z. et al. Janus nanobullets combine photodynamic therapy and magnetic hyperthermia to potentiate synergetic anti-metastatic immunotherapy. Adv. Sci. 6, 1901690 (2019).
Min, H. et al. Biomimetic metal–organic framework nanoparticles for cooperative combination of antiangiogenesis and photodynamic therapy for enhanced efficacy. Adv. Mater. 31, 1808200 (2019).
Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
Esfahani, K. et al. A review of cancer immunotherapy: from the past, to the present, to the future. Curr. Oncol. 27, S87–S97 (2020).
Zhang, F. et al. Engineering magnetosomes for ferroptosis/immunomodulation synergism in cancer. ACS Nano 13, 5662–5673 (2019).
Deng, G. et al. Cell-membrane immunotherapy based on natural killer cell membrane coated nanoparticles for the effective inhibition of primary and abscopal tumor growth. ACS Nano 12, 12096–12108 (2018).
Zhang, J. et al. Immunostimulant hydrogel for the inhibition of malignant glioma relapse post-resection. Nat. Nanotechnol. 16, 538–548 (2021).
Rao, L. et al. Activating macrophage-mediated cancer immunotherapy by genetically edited nanoparticles. Adv. Mater. 32, 2004853 (2020).
Zhang, X. et al. PD-1 blockade cellular vesicles for cancer immunotherapy. Adv. Mater. 30, 1707112 (2018).
Zhai, Y. et al. T lymphocyte membrane-decorated epigenetic nanoinducer of interferons for cancer immunotherapy. Nat. Nanotechnol. 16, 1271–1280 (2021).
Zhang, Y., Liao, Y., Tang, Q., Lin, J. & Huang, P. Biomimetic nanoemulsion for synergistic photodynamic-immunotherapy against hypoxic breast tumor. Angew. Chem. Int. Ed. Engl. 60, 10647–10653 (2021).
Bahmani, B. et al. Intratumoral immunotherapy using platelet-cloaked nanoparticles enhances antitumor immunity in solid tumors. Nat. Commun. 12, 1999 (2021).
Pollard, A. J. & Bijker, E. M. A guide to vaccinology: from basic principles to new developments. Nat. Rev. Immunol. 21, 83–100 (2021).
Higano, C. S. et al. Sipuleucel-T. Nat. Rev. Drug Discov. 9, 513–514 (2010).
van der Burg, S. H., Arens, R., Ossendorp, F., van Hall, T. & Melief, C. J. Vaccines for established cancer: overcoming the challenges posed by immune evasion. Nat. Rev. Cancer 16, 219–233 (2016).
Zhou, J., Kroll, A. V., Holay, M., Fang, R. H. & Zhang, L. Biomimetic nanotechnology toward personalized vaccines. Adv. Mater. 32, 1901255 (2020).
Guo, Y. Y. et al. Erythrocyte membrane-enveloped polymeric nanoparticles as nanovaccine for induction of antitumor immunity against melanoma. ACS Nano 9, 6918–6933 (2015).
Reuven, E. M. et al. Biomimetic glyconanoparticle vaccine for cancer immunotherapy. ACS Nano 13, 2936–2947 (2019).
Fontana, F. et al. Multistaged nanovaccines based on porous silicon@acetalated dextran@cancer cell membrane for cancer immunotherapy. Adv. Mater. 29, 1603239 (2017).
Yang, R. et al. Cancer cell membrane-coated adjuvant nanoparticles with mannose modification for effective anticancer vaccination. ACS Nano 12, 5121–5129 (2018).
Chen, Q. et al. A hybrid eukaryotic-prokaryotic nanoplatform with photothermal modality for enhanced antitumor vaccination. Adv. Mater. 32, 1908185 (2020).
Zhai, Y. W. et al. A splenic-targeted versatile antigen courier: iPSC wrapped in coalescent erythrocyte-liposome as tumor nanovaccine. Sci. Adv. 7, eabi6326 (2021).
Hammerich, L., Binder, A. & Brody, J. D. In situ vaccination: cancer immunotherapy both personalized and off-the-shelf. Mol. Oncol. 9, 1966–1981 (2015).
Patel, R. B. et al. Development of an in situ cancer vaccine via combinational radiation and bacterial-membrane-coated nanoparticles. Adv. Mater. 31, 1902626 (2019).
Eggermont, L. J., Paulis, L. E., Tel, J. & Figdor, C. G. Towards efficient cancer immunotherapy: advances in developing artificial antigen-presenting cells. Trends Biotechnol. 32, 456–465 (2014).
Zhang, Q. B. et al. Biomimetic magnetosomes as versatile artificial antigen-presenting cells to potentiate T-cell-based anticancer therapy. ACS Nano 11, 10724–10732 (2017).
Liu, W. L. et al. Expandable immunotherapeutic nanoplatforms engineered from cytomembranes of hybrid cells derived from cancer and dendritic cells. Adv. Mater. 31, 1900499 (2019).
Xiao, P. et al. Engineering nanoscale artificial antigen-presenting cells by metabolic dendritic cell labeling to potentiate cancer immunotherapy. Nano Lett. 21, 2094–2103 (2021).
Herrmann, I. K., Wood, M. J. A. & Fuhrmann, G. Extracellular vesicles as a next-generation drug delivery platform. Nat. Nanotechnol. 16, 748–759 (2021).
Anselmo, A. C. & Mitragotri, S. Nanoparticles in the clinic: an update. Bioeng. Transl. Med. 4, e10143 (2019).
Busatto, S. et al. Tangential flow filtration for highly efficient concentration of extracellular vesicles from large volumes of fluid. Cells 7, 273 (2018).
Ganesan, P., Karthivashan, G., Park, S. Y., Kim, J. & Choi, D. K. Microfluidization trends in the development of nanodelivery systems and applications in chronic disease treatments. Int. J. Nanomed. 13, 6109–6121 (2018).
Wortzel, I., Dror, S., Kenific, C. M. & Lyden, D. Exosome-mediated metastasis: communication from a distance. Dev. Cell 49, 347–360 (2019).
Jensen, C. & Teng, Y. Is it time to start transitioning from 2D to 3D cell culture? Front. Mol. Biosci. 7, 33 (2020).
Szczepiorkowski, Z. M. & Dunbar, N. M. Transfusion guidelines: when to transfuse. Hematol. Am. Soc. Hematol. Educ. Program 2013, 638–644 (2013).
Shlomchik, W. D. Graft-versus-host disease. Nat. Rev. Immunol. 7, 340–352 (2007).
Ren, J. et al. Multiplex genome editing to generate universal CAR T cells resistant to PD1 inhibition. Clin. Cancer Res. 23, 2255–2266 (2017).
Gornalusse, G. G. et al. HLA-E-expressing pluripotent stem cells escape allogeneic responses and lysis by NK cells. Nat. Biotechnol. 35, 765–772 (2017).
Acknowledgements
The work of the authors is supported by the Defense Threat Reduction Agency Joint Science and Technology Office for Chemical and Biological Defense under Grant Number HDTRA1‐21‐1‐0010, the National Institutes of Health under Award Number R01CA200574 and the National Science Foundation Grant DMR-1904702.
Author information
Authors and Affiliations
Contributions
All authors contributed to all aspects of the preparation of this manuscript prior to submission.
Corresponding author
Ethics declarations
Competing interests
L.Z. is the founder of Cellics Therapeutics and Cello Therapeutics and holds equity interests in both companies. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Clinical Oncology thanks X. Chen and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fang, R.H., Gao, W. & Zhang, L. Targeting drugs to tumours using cell membrane-coated nanoparticles. Nat Rev Clin Oncol 20, 33–48 (2023). https://doi.org/10.1038/s41571-022-00699-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-022-00699-x
This article is cited by
-
Development of rice bran-derived nanoparticles with excellent anti-cancer activity and their application for peritoneal dissemination
Journal of Nanobiotechnology (2024)
-
A modular approach to enhancing cell membrane-coated nanoparticle functionality using genetic engineering
Nature Nanotechnology (2024)
-
Engineering biomimetic nanosystem targeting multiple tumor radioresistance hallmarks for enhanced radiotherapy
Science China Life Sciences (2024)
-
Tyrosinase enzyme purification and immobilization from Pseudomonas sp. EG22 using cellulose coated magnetic nanoparticles: characterization and application in melanin production
World Journal of Microbiology and Biotechnology (2024)
-
Wnt/β-catenin-driven EMT regulation in human cancers
Cellular and Molecular Life Sciences (2024)