Abstract
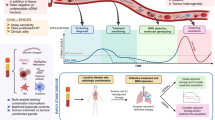
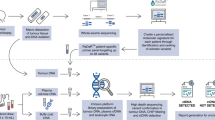
Circulating tumour DNA (ctDNA) refers to the fraction of cell-free DNA in a patient’s blood that originates from a tumour. Advances in DNA sequencing technologies and our understanding of the molecular biology of tumours have resulted in increased interest in exploiting ctDNA as a tool to facilitate earlier detection of cancer and thereby improve therapeutic outcomes by enabling early intervention. ctDNA analysis might also have utility in the adjuvant therapeutic setting by enabling the identification of patients at a high risk of disease recurrence on the basis of the detection of post-surgical minimal (or molecular) residual disease (MRD). This approach could provide the capability to adapt clinical trials in the adjuvant setting in order to optimize risk stratification, and we argue that this objective is achievable with current technologies. Herein, we evaluate contemporary next-generation sequencing (NGS) approaches to ctDNA detection with a focus on non-small-cell lung cancer. We explain the technical and analytical challenges to low-frequency mutation detection using NGS-based ctDNA profiling and evaluate the feasibility of ctDNA profiling in both screening and MRD assessment contexts.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Figure adapted from ref.8, Macmillan Publishers Limited.
Similar content being viewed by others
References
Goldstraw, P. et al. The IASLC Lung Cancer Staging Project: proposals for revision of the TNM stage groupings in the forthcoming (eighth) edition of the TNM classification for lung cancer. J. Thorac. Oncol. 11, 39–51 (2016).
Pignon, J. P. et al. Lung adjuvant cisplatin evaluation: a pooled analysis by the LACE Collaborative Group. J. Clin. Oncol. 26, 3552–3559 (2008).
Non-Small Cell Lung Cancer Collaborative Group. Chemotherapy for non-small cell lung cancer. Cochrane Database Syst. Rev. 2, CD002139 (2000).
Phallen, J. et al. Direct detection of early-stage cancers using circulating tumor DNA. Sci. Transl Med. 9, eaan2415 (2017).
Cohen, J. D. et al. Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 359, 926–930 (2018).
Newman, A. M. et al. An ultrasensitive method for quantitating circulating tumor DNA with broad patient coverage. Nat. Med. 20, 548–554 (2014).
Chaudhuri, A. A. et al. Early detection of molecular residual disease in localized lung cancer by circulating tumor DNA profiling. Cancer Discov. 7, 1394–1403 (2017).
Abbosh, C. et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature 545, 446–451 (2017).
Tie, J. et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci. Transl Med. 8, 346ra392 (2016).
Garcia-Murillas, I. et al. Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Sci. Transl Med. 7, 302ra133 (2015).
Beaver, J. A. et al. Detection of cancer DNA in plasma of patients with early-stage breast cancer. Clin. Cancer Res. 20, 2643–2650 (2014).
Pantel, K. & Alix-Panabières, C. Tumour microenvironment: informing on minimal residual disease in solid tumours. Nat. Rev. Clin. Oncol. 14, 325–326 (2017).
Burgener, J. M., Rostami, A., De Carvalho, D. D. & Bratman, S. V. Cell-free DNA as a post-treatment surveillance strategy: current status. Semin. Oncol. 44, 330–346 (2017).
Newman, A. M. et al. Integrated digital error suppression for improved detection of circulating tumor DNA. Nat. Biotechnol. 34, 547–555 (2016).
Forshew, T. et al. Noninvasive identification and monitoring of cancer mutations by targeted deep sequencing of plasma DNA. Sci. Transl Med. 4, 136ra168 (2012).
Dawson, S.-J. et al. Analysis of circulating tumor DNA to monitor metastatic breast cancer. N. Engl. J. Med. 368, 1199–1209 (2013).
Adalsteinsson, V. A. et al. Scalable whole-exome sequencing of cell-free DNA reveals high concordance with metastatic tumors. Nat. Commun. 8, 1324 (2017).
Guibert, N. et al. Amplicon-based next-generation sequencing of plasma cell-free DNA for detection of driver and resistance mutations in advanced non-small cell lung cancer. Ann. Oncol. 29, 1049–1055 (2018).
Parkinson, C. A. et al. Exploratory analysis of TP53 mutations in circulating tumour DNA as biomarkers of treatment response for patients with relapsed high-grade serous ovarian carcinoma: a retrospective study. PLOS Med. 13, e1002198 (2016).
Kinde, I., Wu, J., Papadopoulos, N., Kinzler, K. W. & Vogelstein, B. Detection and quantification of rare mutations with massively parallel sequencing. Proc. Natl Acad. Sci. USA 108, 9530–9535 (2011).
Schmitt, M. W. et al. Detection of ultra-rare mutations by next-generation sequencing. Proc. Natl Acad. Sci. USA 109, 14508–14513 (2012).
Park, G. et al. Characterization of background noise in capture-based targeted sequencing data. Genome Biol. 18, 136 (2017).
Hestand, M. S., Houdt, J. V., Cristofoli, F. & Vermeesch, J. R. Polymerase specific error rates and profiles identified by single molecule sequencing. Mutat. Res. 784–785, 39–45 (2016).
Chen, G., Mosier, S., Gocke, C. D., Lin, M. T. & Eshleman, J. R. Cytosine deamination is a major cause of baseline noise in next-generation sequencing. Mol. Diagn. Ther. 18, 587–593 (2014).
Ewing, B. & Green, P. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome Res. 8, 186–194 (1998).
Manley, L. J., Ma, D. & Levine, S. S. Monitoring error rates in Illumina sequencing. J. Biomol. Tech. 27, 125–128 (2016).
Wan, J. C. M. et al. Liquid biopsies come of age: towards implementation of circulating tumour DNA. Nat. Rev. Cancer 17, 223–238 (2017).
Szpechcinski, A. et al. Cell-free DNA levels in plasma of patients with non-small-cell lung cancer and inflammatory lung disease. Br. J. Cancer 113, 476–483 (2015).
Snyder, M. W., Kircher, M., Hill, A. J., Daza, R. M. & Shendure, J. Cell-free DNA comprises an in vivo nucleosome footprint that informs its tissues-of-origin. Cell 164, 57–68 (2016).
Lui, Y. Y. et al. Predominant hematopoietic origin of cell-free DNA in plasma and serum after sex-mismatched bone marrow transplantation. Clin. Chem. 48, 421–427 (2002).
Xie, M. et al. Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat. Med. 20, 1472–1478 (2014).
Welch, J. S. et al. The origin and evolution of mutations in acute myeloid leukemia. Cell 150, 264–278 (2012).
Steensma, D. P. et al. Clonal hematopoiesis of indeterminate potential and its distinction from myelodysplastic syndromes. Blood 126, 9–16 (2015).
Razavi, P. et al. Cell-free DNA (cfDNA) mutations from clonal hematopoiesis: implications for interpretation of liquid biopsy tests. J. Clin. Oncol. 35, 11526 (2017).
Hu, Y. et al. False positive plasma genotyping due to clonal hematopoiesis. Clin. Cancer Res. https://doi.org/10.1158/1078-0432.CCR-18-0143 (2018).
Higgins, K. A. et al. Lymphovascular invasion in non-small-cell lung cancer: implications for staging and adjuvant therapy. J. Thorac. Oncol. 7, 1141–1147 (2012).
Warth, A. et al. Tumour cell proliferation (Ki-67) in non-small cell lung cancer: a critical reappraisal of its prognostic role. Br. J. Cancer 111, 1222 (2014).
Duhaylongsod, F. G. et al. Lung tumor growth correlates with glucose metabolism measured by fluoride-18 fluorodeoxyglucose positron emission tomography. Ann. Thorac. Surg. 60, 1348–1352 (1995).
Higashi, K. et al. 18F-FDG uptake as a biologic prognostic factor for recurrence in patients with surgically resected non-small cell lung cancer. J. Nucl. Med. 43, 39–45 (2002).
Gkogkou, C., Frangia, K., Saif, M. W., Trigidou, R. & Syrigos, K. Necrosis and apoptotic index as prognostic factors in non-small cell lung carcinoma: a review. SpringerPlus 3, 120 (2014).
Diaz, L. A. Jr et al. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature 486, 537–540 (2012).
Winther-Larsen, A. et al. Correlation between circulating mutant DNA and metabolic tumour burden in advanced non-small cell lung cancer patients. Br. J. Cancer 117, 704–709 (2017).
Pécuchet, N. et al. Base-position error rate analysis of next-generation sequencing applied to circulating tumor DNA in non-small cell lung cancer: a prospective study. PLOS Med. 13, e1002199 (2016).
Aberle, D. R. et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N. Engl. J. Med. 365, 395–409 (2011).
Haque, I. S. & Elemento, O. Challenges in using ctDNA to achieve early detection of cancer. Preprint at bioRxiv https://doi.org/10.1101/237578 (2017).
Krug, A. K. et al. Improved EGFR mutation detection using combined exosomal RNA and circulating tumor DNA in NSCLC patient plasma. Ann. Oncol. 29, 700–706 (2017).
Patz, E. F. Jr et al. Overdiagnosis in low-dose computed tomography screening for lung cancer. JAMA Intern. Med. 174, 269–274 (2014).
Winton, T. et al. Vinorelbine plus cisplatin vs. observation in resected non-small-cell lung cancer. N. Engl. J. Med. 352, 2589–2597 (2005).
Lou, F., Sima, C. S., Rusch, V. W., Jones, D. R. & Huang, J. Differences in patterns of recurrence in early-stage versus locally advanced non-small cell lung cancer. Ann. Thorac. Surg. 98, 1755–1761 (2014).
Kelsey, C. R. et al. Local recurrence after surgery for early stage lung cancer: an 11-year experience with 975 patients. Cancer 115, 5218–5227 (2009).
Sugimura, H. et al. Survival after recurrent nonsmall-cell lung cancer after complete pulmonary resection. Ann. Thorac. Surg. 83, 409–417 (2007).
Dziedzic, D. A., Rudzinski, P., Langfort, R. & Orlowski, T. Risk factors for local and distant recurrence after surgical treatment in patients with non-small-cell lung cancer. Clin. Lung Cancer 17, e157–e167 (2016).
Artal Cortés, Á., Calera Urquizu, L. & Hernando Cubero, J. Adjuvant chemotherapy in non-small cell lung cancer: state-of-the-art. Transl Lung Cancer Res. 4, 191–197 (2015).
Ng, S. B. et al. Individualised multiplexed circulating tumour DNA assays for monitoring of tumour presence in patients after colorectal cancer surgery. Sci. Rep. 7, 40737 (2017).
Govindan, R. et al. Genomic landscape of non-small cell lung cancer in smokers and never smokers. Cell 150, 1121–1134 (2012).
Salazar, M. C. et al. Association of delayed adjuvant chemotherapy with survival after lung cancer surgery. JAMA Oncol. 3, 610–619 (2017).
Zehir, A. et al. Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat. Med. 23, 703–713 (2017).
Cheng, D. T. et al. Memorial Sloan Kettering-Integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J. Mol. Diagn. 17, 251–264 (2015).
Merker, J. D. et al. Circulating tumor DNA analysis in patients with cancer: American Society of Clinical Oncology and College of American Pathologists joint review. J. Clin. Oncol. 36, 1631–1641 (2018).
Denis, J. A., Guillerm, E., Coulet, F., Larsen, A. K. & Lacorte, J.-M. The role of BEAMing and digital PCR for multiplexed analysis in molecular oncology in the era of next-generation sequencing. Mol. Diagn. Ther. 21, 587–600 (2017).
Acknowledgements
The work of C.A. is supported by a Stand Up To Cancer–LUNGevity–American Lung Association Lung Cancer Interception Dream Team translational research grant and the Rosetrees Trust. The work of C.S. has been supported by the Breast Cancer Research Foundation, Cancer Research UK (CRUK; Tracking Cancer Evolution Through Therapy (TRACERx) and CRUK Cancer Immunotherapy Catalyst Network), the CRUK Lung Cancer Centre of Excellence, the CRUK University College London Experimental Cancer Medicine Centre, a Stand Up To Cancer–LUNGevity–American Lung Association Lung Cancer Interception Dream Team Translational Research Grant, the European Research Council (THESEUS project), the Medical Research Council, the UK National Institute for Health Research, the Novo Nordisk Foundation (ID 16584), the Prostate Cancer Foundation, the Rosetrees Trust, the University College London Hospitals Biomedical Research Centre, and the Wellcome Trust.
Reviewer information
Nature Reviews Clinical Oncology thanks B. Li, C. Rolfo, and Y.-L. Wu for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.A. and N.J.B. researched the data for the article, and C.A. and C.S. wrote the manuscript. All authors contributed to discussions of content and reviewed and/or edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.A. and C.S. submitted a patent with University College London (UCL) business PLC (provisional patent number 1618485.5) based on a phylogenetic approach to analysis of circulating tumour DNA. C.S. has received grant support from AstraZeneca; personal fees from Boehringer Ingelheim, Celgene, Eli Lilly, GlaxoSmithKline, Novartis, Pfizer, and Roche; has stock options in Achilles Therapeutics, ApoGen Biotechnologies, EPIC Bioscience, and GRAIL; and is a co-founder of Achilles Therapeutics. N.J.B. declares no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
ClinicalTrials.gov database: https://clinicaltrials.gov/
Supplementary information
Rights and permissions
About this article
Cite this article
Abbosh, C., Birkbak, N.J. & Swanton, C. Early stage NSCLC — challenges to implementing ctDNA-based screening and MRD detection. Nat Rev Clin Oncol 15, 577–586 (2018). https://doi.org/10.1038/s41571-018-0058-3
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-018-0058-3
This article is cited by
-
Analytical evaluation of circulating tumor DNA sequencing assays
Scientific Reports (2024)
-
Bridging horizons beyond CIRCULATE-Japan: a new paradigm in molecular residual disease detection via whole genome sequencing-based circulating tumor DNA assay
International Journal of Clinical Oncology (2024)
-
Prognostic value of circulating tumor DNA in operable non-small cell lung cancer: a systematic review and reconstructed individual patient-data based meta-analysis
BMC Medicine (2023)
-
Accuracy of minimal residual disease detection by circulating tumor DNA profiling in lung cancer: a meta-analysis
BMC Medicine (2023)
-
Associations between detectable circulating tumor DNA and tumor glucose uptake measured by 18F-FDG PET/CT in early-stage non-small cell lung cancer
BMC Cancer (2023)