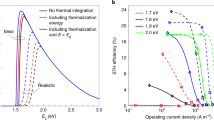
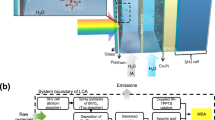
Abstract
In photochemical production of hydrogen from water, the hole-mediated oxidation reaction is the rate-determining step. A poor solar-to-hydrogen efficiency is usually related to a mismatch between the internal quantum efficiency of photon-induced hole generation and the apparent quantum yield of hydrogen. This waste of photogenerated holes is unwanted yet unavoidable. Although great progress has been made, we are still far away from the required level of dexterity to deal with the associated challenges of wasted holes and its consequential chemical effects that have placed one of the greatest bottlenecks in attaining high solar-to-hydrogen efficiency. A critical assessment of the hole and its related phenomena in solar hydrogen production would, therefore, pave the way moving forward. In this regard, we focus on the contextual and conceptual understanding of the dynamics and kinetics of photogenerated holes and its critical role in driving redox reactions, with the objective of guiding future research. The main reasons behind and consequences of unused holes are examined and different approaches to improve overall efficiency are outlined. We also highlight yet unsolved research questions related to holes in solar fuel production.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rahman, M. Z., Kibria, M. G. & Mullins, C. B. Metal-free photocatalysts for hydrogen evolution. Chem. Soc. Rev. 49, 1887–1931 (2020).
Global warming of 1.5°C (IPCC, 2018).
Timothy, M. L. et al. Climate tipping points — too risky to bet against. Nature 575, 592–595 (2019).
Bard, A. J. Photoelectrochemistry. Science 207, 139–144 (1980).
Voiry, D., Shin, H. S., Loh, K. P. & Chhowalla, M. Low-dimensional catalysts for hydrogen evolution and CO2 reduction. Nat. Rev. Chem. 2, 0105 (2018).
Davis, S. J. et al. Net-zero emissions energy systems. Science 360, eaas9793 (2018).
Dresselhaus, M. S. & Thomas, I. L. Alternative energy technologies. Nature 414, 332–337 (2001).
Creutzig, F. et al. The underestimated potential of solar energy to mitigate climate change. Nat. Energy 2, 1714 (2017).
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl Acad. Sci. USA 103, 15729–15735 (2006).
Ciamician, G. The photochemistry of the future. Science 36, 385–394 (1912).
Chen, S., Takata, T. & Domen, K. Particulate photocatalysts for overall water splitting. Nat. Rev. Mater. 2, 17050 (2017).
Serpone, N., Emeline, A. V., Horikoshi, S., Kuznetsov, V. N. & Ryabchuk, V. K. On the genesis of heterogeneous photocatalysis: a brief historical perspective in the period 1910 to the mid-1980s. Photochem. Photobiol. Sci. 11, 1121–1150 (2012). This article reports a brief historical perspective on the origins of photocatalysis and enumerates the timescale contribution from the pioneer researchers in developing this field.
Kisch, H. Semiconductor photocatalysis — mechanistic and synthetic aspects. Angew. Chem. Int. Ed. 52, 812–847 (2013).
Roth, H. D. The beginnings of organic photochemistry. Angew. Chem. Int. Ed. 28, 1193–1207 (1989).
Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972). This article demonstrates the first practical realization of water splitting using simulated solar energy.
Wang, Q. & Domen, K. Particulate photocatalysts for light-driven water splitting: mechanisms, challenges, and design strategies. Chem. Rev. 120, 919–985 (2020).
Wang, Z., Li, C. & Domen, K. Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem. Soc. Rev. 48, 2109–2125 (2019).
Mesa, C. A. et al. Multihole water oxidation catalysis on haematite photoanodes revealed by operando spectroelectrochemistry and DFT. Nat. Chem. 12, 82–89 (2020). This article reports a new mechanism for hole-mediated heterogeneous water oxidation on a metal oxide surface.
Dau, H. et al. The mechanism of water oxidation: from electrolysis via homogeneous to biological catalysis. ChemCatChem 2, 724–761 (2010).
Renger, G., Christen, G., Karge, M., Eckert, H. J. & Irrgang, K. D. Application of the Marcus theory for analysis of the temperature dependence of the reactions leading to photosynthetic water oxidation: results and implications. J. Biol. Inorg. Chem. 3, 360–366 (1998).
Matheu, R. et al. The development of molecular water oxidation catalysts. Nat. Rev. Chem. 3, 331–341 (2019).
Fountaine, K. T., Lewerenz, H. J. & Atwater, H. A. Efficiency limits for photoelectrochemical water-splitting. Nat. Commun. 7, 13706 (2016).
Xiang, C. et al. Modeling, simulation, and implementation of solar-driven water-splitting devices. Angew. Chem. Int. Ed. 55, 12974–12988 (2016).
Akimov, A. V., Neukirch, A. J. & Prezhdo, O. V. Theoretical insights into photoinduced charge transfer and catalysis at oxide interfaces. Chem. Rev. 113, 4496–4565 (2013).
Le Formal, F. et al. Rate law analysis of water oxidation on a hematite surface. J. Am. Chem. Soc. 137, 6629–6637 (2015). This article reports a rate law analysis of the order of water oxidation as a function of surface hole density and provides direct evidence for the multihole catalysis of water oxidation by hematite.
Cadiau, A. et al. A titanium metal–organic framework with visible-light-responsive photocatalytic activity. Angew. Chem. Int. Ed. 59, 13468–13472 (2020).
Rahman, M. Z., Davey, K. & Mullins, C. B. Tuning the intrinsic properties of carbon nitride for high quantum yield photocatalytic hydrogen production. Adv. Sci. 5, 1800820 (2018).
Schneider, J. & Bahnemann, D. W. Undesired role of sacrificial reagents in photocatalysis. J. Phys. Chem. Lett. 4, 3479–3483 (2013).
Segev, G., Beeman, J. W., Greenblatt, J. B. & Sharp, I. D. Hybrid photoelectrochemical and photovoltaic cells for simultaneous production of chemical fuels and electrical power. Nat. Mater. 17, 1115–1121 (2018). This article demonstrates the conceptual design of a hybrid photovoltaic–photoelectrochemical device for the extraction of unused holes as electric current.
Luo, N. et al. Visible-light-driven coproduction of diesel precursors and hydrogen from lignocellulose-derived methylfurans. Nat. Energy 4, 575–584 (2019).
Rahman, M. Z., Moffatt, J. & Spooner, N. Topological carbon nitride: localized photon absorption and delocalized charge carrier separation at intertwined photocatalyst interfaces. Mater. Horiz. 5, 553–559 (2018).
Navarro-Jaén, S. et al. Highlights and challenges in the selective reduction of carbon dioxide to methanol. Nat. Rev. Chem. 5, 564–579 (2021).
Wagner, A., Sahm, C. D. & Reisner, E. Towards molecular understanding of local chemical environment effects in electro- and photocatalytic CO2 reduction. Nat. Catal. 3, 775–786 (2020).
Nosaka, Y. & Nosaka, A. Y. Generation and detection of reactive oxygen species in photocatalysis. Chem. Rev. 117, 11302–11336 (2017).
Studer, A. & Curran, D. P. The electron is a catalyst. Nat. Chem. 6, 765–773 (2014).
Luca, O. R., Gustafson, J. L., Maddox, S. M., Fenwick, A. Q. & Smith, D. C. Catalysis by electrons and holes: formal potential scales and preparative organic electrochemistry. Org. Chem. Front. 2, 823–848 (2015).
Hazlehurst, T. H. Acid-base reactions. Their analogy to oxidation-reduction reactions in solution. J. Chem. Educ. 17, 466 (1940).
Rahman, M. Z., Batmunkh, M., Bat-Erdene, M., Shapter, J. G. & Mullins, C. B. p-Type BP nanosheet photocatalyst with AQE of 3.9% in the absence of a noble metal cocatalyst: investigation and elucidation of photophysical properties. J. Mater. Chem. A 6, 18403–18408 (2018).
Smith, W. A., Sharp, I. D., Strandwitz, N. C. & Bisquert, J. Interfacial band-edge energetics for solar fuels production. Energy Environ. Sci. 8, 2851–2862 (2015).
Voznyy, O., Sutherland, B. R., Ip, A. H., Zhitomirsky, D. & Sargent, E. H. Engineering charge transport by heterostructuring solution-processed semiconductors. Nat. Rev. Mater. 2, 17026 (2017).
Zhang, Z. & Yates, J. T. Jr Band bending in semiconductors: chemical and physical consequences at surfaces and interfaces. Chem. Rev. 112, 5520–5551 (2012).
Peter, L. M. in Photocatalysis: Fundamentals and Perspectives (eds Schneider, J. et al.) 1–28 (Royal Society of Chemistry, 2016).
Peter, L. Fundamental aspects of photoelectrochemical water splitting at semiconductor electrodes. Curr. Opin. Green Sustain. Chem. 31, 100505 (2021).
Wurfel, P. Physics of Solar Cells 2nd edn (Wiley, 2010).
Sato, N. Electrochemistry at Metal and Semiconductor Electrodes (Elsevier, 1998).
Peter, L. M. Energetics and kinetics of light-driven oxygen evolution at semiconductor electrodes: the example of hematite. J. Solid State Electrochem. 17, 315–326 (2012).
Emeline, A. V., Kuznetsov, V. N., Ryabchuk, V. K. & Serpone, N. in New and Future Developments in Catalysis (ed. Suib, S. L.) 1–47 (Elsevier, 2013).
Bard, A. J., Bocarsly, A. B., Fan, F. R. F., Walton, E. G. & Wrighton, M. S. The concept of Fermi level pinning at semiconductor/liquid junctions. Consequences for energy conversion efficiency and selection of useful solution redox couples in solar devices. J. Am. Chem. Soc. 102, 3671–3677 (1980).
Sze, S. M. Physics of Semiconductor Devices (Wiley, 1969).
Otero, R., Vázquez de Parga, A. L. & Gallego, J. M. Electronic, structural and chemical effects of charge-transfer at organic/inorganic interfaces. Surf. Sci. Rep. 72, 105–145 (2017).
Haneef, H. F., Zeidell, A. M. & Jurchescu, O. D. Charge carrier traps in organic semiconductors: a review on the underlying physics and impact on electronic devices. J. Mater. Chem. C 8, 759–787 (2020).
Rahman, M. Z. & Mullins, C. B. Understanding charge transport in carbon nitride for enhanced photocatalytic solar fuel production. Acc. Chem. Res. 52, 248–257 (2019).
Clarke, T. M. & Durrant, J. R. Charge photogeneration in organic solar cells. Chem. Rev. 110, 6736–6767 (2010).
Grave, D. A. et al. Extraction of mobile charge carrier photogeneration yield spectrum of ultrathin-film metal oxide photoanodes for solar water splitting. Nat. Mater. 20, 833–840 (2021).
Bakulin, A. A. et al. The role of driving energy and delocalized states for charge separation in organic semiconductors. Science 335, 1340–1344 (2012).
Hayes, D. et al. Electronic and nuclear contributions to time-resolved optical and X-ray absorption spectra of hematite and insights into photoelectrochemical performance. Energy Environ. Sci. 9, 3754–3769 (2016).
Nozik, A. J. Multiple exciton generation in semiconductor quantum dots. Chem. Phys. Lett. 457, 3–11 (2008).
Zhao, D. et al. Boron-doped nitrogen-deficient carbon nitride-based Z-scheme heterostructures for photocatalytic overall water splitting. Nat. Energy 6, 388–397 (2021).
Wang, Z. et al. Sequential cocatalyst decoration on BaTaO2N towards highly-active Z-scheme water splitting. Nat. Commun. 12, 1005 (2021).
Takata, T. et al. Photocatalytic water splitting with a quantum efficiency of almost unity. Nature 581, 411–414 (2020).
Rahman, M., Boschloo, G., Hagfeldt, A. & Edvinsson, T. On the mechanistic understanding of photovoltage loss in iron pyrite solar cells. Adv. Mater. 32, e1905653 (2020).
Rahman, M., Tian, H. & Edvinsson, T. Revisiting the limiting factors for overall water-splitting on organic photocatalysts. Angew. Chem. Int. Ed. 59, 16278–16293 (2020).
Merschjann, C. et al. Photophysics of polymeric carbon nitride: an optical quasimonomer. Phys. Rev. B 87, 205204 (2013).
Rao, A. et al. The role of spin in the kinetic control of recombination in organic photovoltaics. Nature 500, 435–439 (2013).
Burke, T. M., Sweetnam, S., Vandewal, K. & McGehee, M. D. Beyond Langevin recombination: how equilibrium between free carriers and charge transfer states determines the open-circuit voltage of organic solar cells. Adv. Energy Mater. 5, 1500123 (2015).
Vandewal, K. et al. Efficient charge generation by relaxed charge-transfer states at organic interfaces. Nat. Mater. 13, 63–68 (2014).
Sachs, M. et al. Tracking charge transfer to residual metal clusters in conjugated polymers for photocatalytic hydrogen evolution. J. Am. Chem. Soc. 142, 14574–14587 (2020).
Rahman, M. Z., Tang, Y. & Kwong, P. Reduced recombination and low-resistive transport of electrons for photo-redox reactions in metal-free hybrid photocatalyst. Appl. Phys. Lett. 112, 253902 (2018).
Walter, M. G. et al. Solar water splitting cells. Chem. Rev. 110, 6446–6473 (2010).
Rahman, M. Z. & Alam, M. J. SRH recombination strength of interstitial iron in solar grade silicon. Photonics Lett. Poland 5, 118–120 (2013).
Marcus, R. A. On the theory of oxidation-reduction reactions involving electron transfer. I. J. Chem. Phys. 24, 966–978 (1956). In this article, a quantitative theory of the rates of oxidation–reduction reactions involving electron transfer in solution is presented.
Hush, N. S. Adiabatic theory of outer sphere electron-transfer reactions in solution. Trans. Faraday Soc. 57, 557–580 (1961).
Liu, L. & Corma, A. Structural transformations of solid electrocatalysts and photocatalysts. Nat. Rev. Chem. 5, 256–276 (2021).
Eberson, L. in Dislocation and the Reactivity of Organic Solids: Advances in Physical Organic Chemistry Vol. 18 (eds Gold, V. & Bethell, D.) 79–185 (Academic, 1982).
Tilley., R. J. D. Defects in Solids (Wiley, 2008).
Thomas, J. M. & Williams, J. O. Dislocations and the reactivity of organic solids. Prog. Solid State Chem. 6, 119–154 (1971).
Lohmann, F. & Mehl, W. Dark injection and radiative recombination of electrons and holes in naphthalene crystals. J. Chem. Phys. 50, 500–506 (1969).
Shluger, A. L., McKenna, K. P., Sushko, P. V., Ramo, D. M. & Kimmel, A. V. Modelling of electron and hole trapping in oxides. Model. Simul. Mater. Sci. Eng. 17, 084004 (2009).
Rettie, A. J. E., Chemelewski, W. D., Emin, D. & Mullins, C. B. Unravelling small-polaron transport in metal oxide photoelectrodes. J. Phys. Chem. Lett. 7, 471–479 (2016).
Franchini, C., Reticcioli, M., Setvin, M. & Diebold, U. Polarons in materials. Nat. Rev. Mater. 6, 560–586 (2021).
Carneiro, L. M. et al. Excitation-wavelength-dependent small polaron trapping of photoexcited carriers in alpha-Fe2O3. Nat. Mater. 16, 819–825 (2017).
Pastor, E. et al. In situ observation of picosecond polaron self-localisation in α-Fe2O3 photoelectrochemical cells. Nat. Commun. 10, 3962 (2019).
Lohaus, C., Klein, A. & Jaegermann, W. Limitation of Fermi level shifts by polaron defect states in hematite photoelectrodes. Nat. Commun. 9, 4309 (2018).
deQuilettes, D. W. et al. Charge-carrier recombination in halide perovskites. Chem. Rev. 119, 11007–11019 (2019).
Godin, R., Kafizas, A. & Durrant, J. R. Electron transfer dynamics in fuel producing photosystems. Curr. Opin. Electrochem. 2, 136–143 (2017).
Appavoo, K., Liu, M., Black, C. T. & Sfeir, M. Y. Quantifying bulk and surface recombination processes in nanostructured water splitting photocatalysts via in situ ultrafast spectroscopy. Nano Lett. 15, 1076–1082 (2015).
Pastor, E. et al. Spectroelectrochemical analysis of the mechanism of (photo)electrochemical hydrogen evolution at a catalytic interface. Nat. Commun. 8, 14280 (2017).
Ferry, V. E., Polman, A. & Atwater, H. A. Modeling light trapping in nanostructured solar cells. ACS Nano 5, 10055–10064 (2011).
Godin, R., Wang, Y., Zwijnenburg, M. A., Tang, J. & Durrant, J. R. Time-resolved spectroscopic investigation of charge trapping in carbon nitrides photocatalysts for hydrogen generation. J. Am. Chem. Soc. 139, 5216–5224 (2017).
Martindale, B. C. M. et al. Enhancing light absorption and charge transfer efficiency in carbon dots through graphitization and core nitrogen doping. Angew. Chem. Int. Ed. 56, 6459–6463 (2017).
Kasap, H. et al. Solar-driven reduction of aqueous protons coupled to selective alcohol oxidation with a carbon nitride–molecular Ni catalyst system. J. Am. Chem. Soc. 138, 9183–9192 (2016).
Mefford, J. T. et al. Correlative operando microscopy of oxygen evolution electrocatalysts. Nature 593, 67–73 (2021). This report establishes a link between the oxygen evolution activity and the local operational chemical, physical and electronic nanoscale structure of the catalyst.
Dral, P. O. & Barbatti, M. Molecular excited states through a machine learning lens. Nat. Rev. Chem. 5, 388–405 (2021).
Villarreal, T. L., Gómez, R., Neumann-Spallart, M., Alonso-Vante, N. & Salvador, P. Semiconductor photooxidation of pollutants dissolved in water: a kinetic model for distinguishing between direct and indirect interfacial hole transfer. I. photoelectrochemical experiments with polycrystalline anatase electrodes under current doubling and absence of recombination. J. Phys. Chem. B 108, 15172–15181 (2004).
Bahnemann, D. W., Hilgendorff, M. & Memming, R. Charge carrier dynamics at TiO2 particles: reactivity of free and trapped holes. J. Phys. Chem. B 101, 4265–4275 (1997).
Ji, Y., Wang, B. & Luo, Y. A comparative theoretical study of proton-coupled hole transfer for H2O and small organic molecules (CH3OH, HCOOH, H2CO) on the anatase TiO2(101) surface. J. Phys. Chem. C 118, 21457–21462 (2014).
Chu, W. et al. Ultrafast dynamics of photongenerated holes at a CH3OH/TiO2 rutile interface. J. Am. Chem. Soc. 138, 13740–13749 (2016).
Mesa, C. A. et al. Kinetics of photoelectrochemical oxidation of methanol on hematite photoanodes. J. Am. Chem. Soc. 139, 11537–11543 (2017).
Chen, Z., Zhang, Q. & Luo, Y. Experimental identification of ultrafast reverse hole transfer at the interface of the photoexcited methanol/graphitic carbon nitride system. Angew. Chem. Int. Ed. 57, 5320–5324 (2018).
Kolesov, G., Vinichenko, D., Tritsaris, G. A., Friend, C. M. & Kaxiras, E. Anatomy of the photochemical reaction: excited-state dynamics reveals the C–H acidity mechanism of methoxy photo-oxidation on titania. J. Phys. Chem. Lett. 6, 1624–1627 (2015).
Shen, M. & Henderson, M. A. Identification of the active species in photochemical hole scavenging reactions of methanol on TiO2. J. Phys. Chem. Lett. 2, 2707–2710 (2011).
Phillips, K. R., Jensen, S. C., Baron, M., Li, S. C. & Friend, C. M. Sequential photo-oxidation of methanol to methyl formate on TiO2(110). J. Am. Chem. Soc. 135, 574–577 (2013).
Fu, C. et al. Site sensitivity of interfacial charge transfer and photocatalytic efficiency in photocatalysis: methanol oxidation on anatase TiO2 nanocrystals. Angew. Chem. Int. Ed. 60, 6160–6169 (2021).
Grela, M. A. & Colussi, A. J. Photon energy and photon intermittence effects on the quantum efficiency of photoinduced oxidations in crystalline and metastable TiO2 colloidal nanoparticles. J. Phys. Chem. B 103, 2614–2619 (1999).
Grela, M. A., Brusa, M. A. & Colussi, A. J. Harnessing excess photon energy in photoinduced surface electron transfer between salicylate and illuminated titanium dioxide nanoparticles. J. Phys. Chem. B 101, 10986–10989 (1997).
Rahman, M. Z. et al. A benchmark quantum yield for water photoreduction on amorphous carbon nitride. Adv. Funct. Mater. 27, 1702384 (2017).
Boudjemaa, A., Bouarab, R., Saadi, S., Bouguelia, A. & Trari, M. Photoelectrochemical H2-generation over Spinel FeCr2O4 in X2− solutions (X2− = S2− and \({{\rm{SO}}}_{3}^{2-}\)). Appl. Energy 86, 1080–1086 (2009).
Tsuji, I., Kato, H., Kobayashi, H. & Kudo, A. Photocatalytic H2 evolution reaction from aqueous solutions over band structure-controlled (AgIn)xZn2(1−x)S2 solid solution photocatalysts with visible-light response and their surface nanostructures. J. Am. Chem. Soc. 126, 13406–13413 (2004).
Kawai, T. & Sakata, T. Photocatalytic hydrogen production from liquid methanol and water. J. Chem. Soc. Chem. Commun. 1980, 694–695 (1980).
Rahman, M. Z., Kwong, C. W., Davey, K. & Qiao, S. Z. 2D phosphorene as a water splitting photocatalyst: fundamentals to applications. Energy Environ. Sci. 9, 709–728 (2016).
Hykaway, N., Sears, W. M., Morisaki, H. & Morrison, S. R. Current-doubling reactions on titanium dioxide photoanodes. J. Phys. Chem. 90, 6663–6667 (1986).
Memming, R. in Electron Transfer I (ed. Mattay, J.) 105–181 (Springer, 1994).
Segev, G. et al. Quantification of the loss mechanisms in emerging water splitting photoanodes through empirical extraction of the spatial charge collection efficiency. Energy Environ. Sci. 11, 904–913 (2018).
Segev, G. et al. The spatial collection efficiency of charge carriers in photovoltaic and photoelectrochemical cells. Joule 2, 210–224 (2018).
Wakerley, D. W. et al. Solar-driven reforming of lignocellulose to H2 with a CdS/CdOx photocatalyst. Nat. Energy 2, 17021 (2017).
Puga, A. V. Photocatalytic production of hydrogen from biomass-derived feedstocks. Coord. Chem. Rev. 315, 1–66 (2016).
Kampouri, S. & Stylianou, K. C. Dual-functional photocatalysis for simultaneous hydrogen production and oxidation of organic substances. ACS Catal. 9, 4247–4270 (2019).
Meng, Q. Y. et al. A cascade cross-coupling hydrogen evolution reaction by visible light catalysis. J. Am. Chem. Soc. 135, 19052–19055 (2013).
Zhao, F. et al. Photocatalytic hydrogen-evolving cross-coupling of arenes with primary amines. Org. Lett. 20, 7753–7757 (2018).
Zheng, Y. W. et al. Photocatalytic hydrogen-evolution cross-couplings: benzene C–H amination and hydroxylation. J. Am. Chem. Soc. 138, 10080–10083 (2016).
Chen, B., Wu, L.-Z. & Tung, C.-H. Photocatalytic activation of less reactive bonds and their functionalization via hydrogen-evolution cross-couplings. Acc. Chem. Res. 51, 2512–2523 (2018).
Wang, H., Gao, X., Lv, Z., Abdelilah, T. & Lei, A. Recent advances in oxidative R1-H/R2-H cross-coupling with hydrogen evolution via photo-/electrochemistry. Chem. Rev. 119, 6769–6787 (2019).
Tang, S. et al. Catalytic oxidative deamination by water with H2 liberation. J. Am. Chem. Soc. 142, 20875–20882 (2020).
Tang, S., Ben-David, Y. & Milstein, D. Oxidation of alkenes by water with H2 liberation. J. Am. Chem. Soc. 142, 5980–5984 (2020).
Robertson, J. C., Coote, M. L. & Bissember, A. C. Synthetic applications of light, electricity, mechanical force and flow. Nat. Rev. Chem. 3, 290–304 (2019).
Tang, S., Zeng, L. & Lei, A. Oxidative R1–H/R2-H cross-coupling with hydrogen evolution. J. Am. Chem. Soc. 140, 13128–13135 (2018).
Gunanathan, C. & Milstein, D. Applications of acceptorless dehydrogenation and related transformations in chemical synthesis. Science 341, 1229712 (2013).
Kohl, S. W. et al. Consecutive thermal H2 and light-induced O2 evolution from water promoted by a metal complex. Science 324, 74–77 (2009).
Liu, J. et al. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science 347, 970–974 (2015).
Jiao, Y., Zheng, Y., Davey, K. & Qiao, S.-Z. Activity origin and catalyst design principles for electrocatalytic hydrogen evolution on heteroatom-doped graphene. Nat. Energy 1, 16130 (2016).
Zheng, Y., Jiao, Y., Jaroniec, M. & Qiao, S. Z. Advancing the electrochemistry of the hydrogen-evolution reaction through combining experiment and theory. Angew. Chem. Int. Ed. 54, 52–65 (2015).
Jiao, Y., Zheng, Y., Jaroniec, M. & Qiao, S. Z. Design of electrocatalysts for oxygen- and hydrogen-involving energy conversion reactions. Chem. Soc. Rev. 44, 2060–2086 (2015).
Bai, Y. et al. Accelerated discovery of organic polymer photocatalysts for hydrogen evolution from water through the integration of experiment and theory. J. Am. Chem. Soc. 141, 9063–9071 (2019).
Masood, H., Toe, C. Y., Teoh, W. Y., Sethu, V. & Amal, R. Machine learning for accelerated discovery of solar photocatalysts. ACS Catal. 9, 11774–11787 (2019).
Pihosh, Y. et al. Ta3N5-Nanorods enabling highly efficient water oxidation via advantageous light harvesting and charge collection. Energy Environ. Sci. 13, 1519–1530 (2020).
Nandal, V., Suzuki, Y., Kobayashi, H., Domen, K. & Seki, K. Theoretical perspective of performance-limiting parameters of Cu(In1−xGax)Se2-based photocathodes. J. Mater. Chem. A 8, 9194–9201 (2020).
Nandal, V. et al. Probing fundamental losses in nanostructured Ta3N5 photoanodes: design principles for efficient water oxidation. Energy Environ. Sci. 14, 4038–4047 (2021).
Jacobsson, T. J., Fjällström, V., Edoff, M. & Edvinsson, T. Sustainable solar hydrogen production: from photoelectrochemical cells to PV-electrolyzers and back again. Energy Environ. Sci. 7, 2056–2070 (2014).
Nozik, A. J. Photoelectrolysis of water using semiconducting TiO2 crystals. Nature 257, 383–386 (1975).
Wang, D., Sheng, T., Chen, J., Wang, H.-F. & Hu, P. Identifying the key obstacle in photocatalytic oxygen evolution on rutile TiO2. Nat. Catal. 1, 291–299 (2018). This article demonstrates unambiguously the influence of the concentration of surface-reaching photoholes as the rate-determining factor of the oxygen evolution reaction.
Zou, N. et al. Cooperative communication within and between single nanocatalysts. Nat. Chem. 10, 607–614 (2018).
Chen, P. et al. Spatiotemporal catalytic dynamics within single nanocatalysts revealed by single-molecule microscopy. Chem. Soc. Rev. 43, 1107–1117 (2014).
Weckhuysen, B. M. Catalysts live and up close. Nature 439, 548–548 (2006).
Roeffaers, M. B. et al. Spatially resolved observation of crystal-face-dependent catalysis by single turnover counting. Nature 439, 572–575 (2006).
Wu, C. Y. et al. High-spatial-resolution mapping of catalytic reactions on single particles. Nature 541, 511–515 (2017).
Sambur, J. B. et al. Sub-particle reaction and photocurrent mapping to optimize catalyst-modified photoanodes. Nature 530, 77–80 (2016). This article shows that most active sites for water oxidation are also the most important sites for charge carrier recombination.
Zou, N. et al. Imaging catalytic hotspots on single plasmonic nanostructures via correlated super-resolution and electron microscopy. ACS Nano 12, 5570–5579 (2018).
Chen, G. et al. Bimetallic effect of single nanocatalysts visualized by super-resolution catalysis imaging. ACS Cent. Sci. 3, 1189–1197 (2017).
Chai, Z. et al. Efficient visible light-driven splitting of alcohols into hydrogen and corresponding carbonyl compounds over a Ni-modified CdS photocatalyst. J. Am. Chem. Soc. 138, 10128–10131 (2016).
O’Neil, M. P. et al. Picosecond optical switching based on biphotonic excitation of an electron donor-acceptor-donor molecule. Science 257, 63–65 (1992).
Karlsson, S. et al. Accumulative charge separation inspired by photosynthesis. J. Am. Chem. Soc. 132, 17977–17979 (2010).
Acknowledgements
Funding from King Abdullah University of Science and Technology through the CRG programme is gratefully acknowledged. T.E. acknowledges generous supports through funding from the Swedish Research Council (VR-2015-03814), the Swedish Research Council for Sustainable Development (grant no. 2016-00908) and the Swedish Energy Agency (grant no. 44648-1).
Author information
Authors and Affiliations
Contributions
M.Z.R. researched data, conceived the structure and wrote the first draft of the manuscript. T.E. and J.G. edited and revised the manuscript. All authors agreed on the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Chemistry thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Charge carrier: https://en.wikipedia.org/wiki/Charge_carrier
Gibbs free energy: https://www.sciencedirect.com/topics/chemistry/gibbs-free-energy
Photon: https://en.wikipedia.org/wiki/Photon
Valence band: https://www.sciencedirect.com/topics/chemistry/valence-band
Supplementary information
Glossary
- Water photolysis
-
Photon-driven chemical reaction that breaks a water molecule into hydrogen and oxygen.
- Co-catalyst
-
A metal or semiconductor that cooperatively improves the catalytic activity when it embeds into a catalyst without being consumed in the chemical reaction.
- Sacrificial reagents
-
Chemical substance that can either donate electrons or remove holes.
- Fermi levels
-
Measure of the highest energy level that an electron can occupy at absolute zero.
- Quasi-Fermi level
-
A displaced Fermi level from equilibrium due to occupancy of electrons/holes generated by external stimuli.
- Redox Fermi level
-
Fermi level of reductant and oxidant species in solution.
- Lattice
-
A crystal structure composed of a series of atoms in a distinct pattern.
- Internal photovoltage
-
The chemical potential of the separated electrons and holes.
- Dielectric constant
-
Coulomb electrostatic attraction between point charges is inversely proportional to the dielectric constant.
- Conduction band
-
The band of electron orbitals above the Fermi level where electrons can move freely or jump up into from the conduction band when excited.
- Emissive state
-
An excited state of electrons in the conduction band or in energy states below the conduction band.
- Valence band
-
The outermost electron orbital of an atom of any specific material that electrons actually occupy.
- Ground state
-
The lowest energy state where electrons are in equilibrium.
- Excitons
-
Excited electron–hole pairs that are bound by Coulomb electrostatic force.
- Impact ionization
-
Process by which one energetic charge carrier can lose energy by the creation of other charge carriers.
- Multiple electron generation
-
Phenomenon wherein the absorption of a single photon leads to the excitation of multiple electrons from the valence band to the conduction band.
- Shockley–Queisser limit
-
Maximum theoretical efficiency of a single p–n junction solar cell.
- Geminate recombination
-
A process where an electron recombines with the same hole that is created in the photogeneration process.
- Auger recombination
-
When the electron–hole recombine by transferring excess energy to a third electron (or hole).
- Shockley–Read–Hall recombination
-
Trap-assisted recombination of electron–hole pairs.
- Hole scavengers
-
A sacrificial chemical that consumes holes in the photocatalytic process.
- Nanostructuring
-
Scaling down of bulk materials into the nanoscale.
Rights and permissions
About this article
Cite this article
Rahman, M.Z., Edvinsson, T. & Gascon, J. Hole utilization in solar hydrogen production. Nat Rev Chem 6, 243–258 (2022). https://doi.org/10.1038/s41570-022-00366-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41570-022-00366-w
This article is cited by
-
Solar reforming as an emerging technology for circular chemical industries
Nature Reviews Chemistry (2024)
-
Triplet–triplet annihilation photon upconversion-mediated photochemical reactions
Nature Reviews Chemistry (2024)
-
Recent advances in efficient and scalable solar hydrogen production through water splitting
Carbon Neutrality (2023)