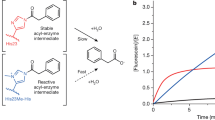
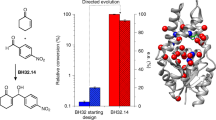
Abstract
The ability of one enzyme to catalyse multiple, mechanistically distinct transformations likely played a crucial role in organisms’ abilities to adapt to changing external stimuli in the past and can still be observed in extant enzymes. Given the importance of catalytic promiscuity in nature, enzyme designers have recently begun to create catalytically promiscuous enzymes in order to expand the canon of transformations catalysed by proteins. This article aims to both critically review different strategies for the design of enzymes that display catalytic promiscuity for new-to-nature reactions and highlight the successes of subsequent directed-evolution efforts to fine-tune these novel reactivities. For the former, we put a particular emphasis on the creation, stabilization and repurposing of reaction intermediates, which are key for unlocking new activities in an existing or designed active site. For the directed evolution of the resulting catalysts, we contrast approaches for enzyme design that make use of components found in nature and those that achieve new reactivities by incorporating synthetic components. Following the critical analysis of selected examples that are now available, we close this Review by providing a set of considerations and design principles for enzyme engineers, which will guide the future generation of efficient artificial enzymes for synthetically useful, abiotic transformations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Reetz, M. T. What are the limitations of enzymes in synthetic organic chemistry? Chem. Rec. 16, 2449–2459 (2016).
Khersonsky, O. & Tawfik, D. S. Enzyme promiscuity: a mechanistic and evolutionary perspective. Annu. Rev. Biochem. 79, 471–505 (2010).
Dietrich, J. A. et al. A novel semi-biosynthetic route for artemisinin production using engineered substrate-promiscuous P450BM3. ACS Chem. Biol. 4, 261–267 (2009).
Yoshikuni, Y., Ferrin, T. E. & Keasling, J. D. Designed divergent evolution of enzyme function. Nature 440, 1078–1082 (2006).
Jensen, R. A. Enzyme recruitment in evolution of new function. Annu. Rev. Microbiol. 30, 409–425 (1976).
Yčas, M. On earlier states of the biochemical system. J. Theor. Biol. 44, 145–160 (1974).
Miller, B. G. & Raines, R. T. Identifying latent enzyme activities: substrate ambiguity within modern bacterial sugar kinases. Biochemistry 43, 6387–6392 (2004).
Miller, B. G. & Raines, R. T. Reconstitution of a defunct glycolytic pathway via recruitment of ambiguous sugar kinases. Biochemistry 44, 10776–10783 (2005).
Seffernick, J. L. & Wackett, L. P. Rapid evolution of bacterial catabolic enzymes: a case study with atrazine chlorohydrolase. Biochemistry 40, 12747–12753 (2001).
Serdar, C. M., Gibson, D. T., Munnecke, D. M. & Lancaster, J. H. Plasmid involvement in parathion hydrolysis by Pseudomonas diminuta. Appl. Environ. Microbiol. 44, 246–249 (1982).
Scott, C. et al. Catalytic improvement and evolution of atrazine chlorohydrolase. Appl. Environ. Microbiol. 75, 2184–2191 (2009).
Noor, S. et al. Intramolecular epistasis and the evolution of a new enzymatic function. PLoS One 7, e39822 (2012).
Afriat-Jurnou, L., Jackson, C. J. & Tawfik, D. S. Reconstructing a missing link in the evolution of a recently diverged phosphotriesterase by active-site loop remodeling. Biochemistry 51, 6047–6055 (2012).
Meier, M. M. et al. Molecular engineering of organophosphate hydrolysis activity from a weak promiscuous lactonase template. J. Am. Chem. Soc. 135, 11670–11677 (2013).
Soskine, M. & Tawfik, D. S. Mutational effects and the evolution of new protein functions. Nat. Rev. Genet. 11, 572–582 (2010).
Aharoni, A. et al. The ‘evolvability’ of promiscuous protein functions. Nat. Genet. 37, 73–76 (2005).
Rothman, S. C. & Kirsch, J. F. How does an enzyme evolved in vitro compare to naturally occurring homologs possessing the targeted function? Tyrosine aminotransferase from aspartate aminotransferase. J. Mol. Biol. 327, 593–608 (2003).
Aharoni, A. et al. Directed evolution of mammalian paraoxonases PON1 and PON3 for bacterial expression and catalytic specialization. Proc. Natl Acad. Sci. USA 101, 482–487 (2004).
Gould, S. M. & Tawfik, D. S. Directed evolution of the promiscuous esterase activity of carbonic anhydrase II. Biochemistry 44, 5444–5452 (2005).
Larion, M., Moore, L. B., Thompson, S. M. & Miller, B. G. Divergent evolution of function in the ROK sugar kinase superfamily: role of enzyme loops in substrate specificity. Biochemistry 46, 13564–13572 (2007).
Bershtein, S., Goldin, K. & Tawfik, D. S. Intense neutral drifts yield robust and evolvable consensus proteins. J. Mol. Biol. 379, 1029–1044 (2008).
Whitman, C. P. The 4-oxalocrotonate tautomerase family of enzymes: how nature makes new enzymes using a β–α–β structural motif. Arch. Biochem. Biophys. 402, 1–13 (2002).
Zandvoort, E., Geertsema, E. M., Baas, B.-J., Quax, W. J. & Poelarends, G. J. Bridging between organocatalysis and biocatalysis: asymmetric addition of acetaldehyde to β-nitrostyrenes catalyzed by a promiscuous proline-based tautomerase. Angew. Chem. Int. Ed. 51, 1240–1243 (2012).
Geertsema, E. M. et al. Biocatalytic Michael-type additions of acetaldehyde to nitroolefins with the proline-based enzyme 4-oxalocrotonate tautomerase yielding enantioenriched γ-nitroaldehydes. Chem. Eur. J. 19, 14407–14410 (2013).
Miao, Y., Geertsema, E. M., Tepper, P. G., Zandvoort, E. & Poelarends, G. J. Promiscuous catalysis of asymmetric Michael-type additions of linear aldehydes to β-nitrostyrene by the proline-based enzyme 4-oxalocrotonate tautomerase. ChemBioChem 14, 191–194 (2013).
Poddar, H., Rahimi, M., Geertsema, E. M., Thunnissen, A.-M. W. H. & Poelarends, G. J. Evidence for the formation of an enamine species during aldol and Michael-type addition reactions promiscuously catalyzed by 4-oxalocrotonate tautomerase. ChemBioChem 16, 738–741 (2015).
van der Meer, J.-Y. et al. Using mutability landscapes of a promiscuous tautomerase to guide the engineering of enantioselective Michaelases. Nat. Commun. 7, 10911 (2016).
Branneby, C. et al. Carbon–carbon bonds by hydrolytic enzymes. J. Am. Chem. Soc. 125, 874–875 (2003).
Torre, O., Alfonso, I. & Gotor, V. Lipase catalysed Michael addition of secondary amines to acrylonitrile. Chem. Commun. 4, 1724–1725 (2004).
Svedendahl, M., Hult, K. & Berglund, P. Fast carbon–carbon bond formation by a promiscuous lipase. J. Am. Chem. Soc. 127, 17988–17989 (2005).
Carlqvist, P. et al. Exploring the active-site of a rationally redesigned lipase for catalysis of Michael-type additions. ChemBioChem 6, 331–336 (2005).
Svedendahl, M. et al. Direct epoxidation in Candida antarctica lipase B studied by experiment and theory. ChemBioChem 9, 2443–2451 (2008).
Albanese, D. C. M. & Gaggero, N. Albumin as a promiscuous biocatalyst in organic synthesis. RSC Adv. 5, 10588–10598 (2015).
Tokuriki, N. & Tawfik, D. S. Protein dynamism and evolvability. Science 324, 203–207 (2009).
Richter, F., Leaver-Fay, A., Khare, S. D., Bjelic, S. & Baker, D. De novo enzyme design using Rosetta3. PLoS One 6, e19230 (2011).
Bolon, D. N. & Mayo, S. L. Enzyme-like proteins by computational design. Proc. Natl Acad. Sci. USA 98, 14274–14279 (2001).
Althoff, E. A. et al. Robust design and optimization of retroaldol enzymes. Protein Sci. 21, 717–726 (2012).
Obexer, R. et al. Emergence of a catalytic tetrad during evolution of a highly active artificial aldolase. Nat. Chem. 9, 50–56 (2017).
Zeymer, C. & Hilvert, D. Directed evolution of protein catalysts. Annu. Rev. Biochem. 87, 131–157 (2018).
Blomberg, R. et al. Precision is essential for efficient catalysis in an evolved Kemp eliminase. Nature 503, 418–421 (2013).
Studer, S. et al. Evolution of a highly active and enantiospecific metalloenzyme from short peptides. Science 362, 1285–1288 (2018).
Giger, L. et al. Evolution of a designed retro-aldolase leads to complete active site remodeling. Nat. Chem. Biol. 9, 494–498 (2013).
Zanghellini, A. et al. New algorithms and an in silico benchmark for computational enzyme design. Protein Sci. 15, 2785–2794 (2006).
Hennig, M., Darimont, B., Jansonius, J. & Kirschner, K. The catalytic mechanism of indole-3-glycerol phosphate synthase: crystal structures of complexes of the enzyme from Sulfolobus solfataricus with substrate analogue, substrate, and product. J. Mol. Biol. 319, 757–766 (2002).
Kuhlman, B. et al. Design of a novel globular protein fold with atomic-level accuracy. Science 302, 1364–1368 (2003).
Schafer, J. W., Zoi, I., Antoniou, D. & Schwartz, S. D. Optimization of the turnover in artificial enzymes via directed evolution results in the coupling of protein dynamics to chemistry. J. Am. Chem. Soc. 141, 10431–10439 (2019).
Garrabou, X., Beck, T. & Hilvert, D. A promiscuous de novo retro-aldolase catalyzes asymmetric Michael additions via Schiff base intermediates. Angew. Chem. Int. Ed. 54, 5609–5612 (2015).
Garrabou, X., Macdonald, D. S., Wicky, B. I. M. & Hilvert, D. Stereodivergent evolution of artificial enzymes for the Michael reaction. Angew. Chem. Int. Ed. 57, 5288–5291 (2018).
Garrabou, X. et al. Fast Knoevenagel condensations catalyzed by an artificial Schiff-base-forming enzyme. J. Am. Chem. Soc. 138, 6972–6974 (2016).
Garrabou, X., Verez, R. & Hilvert, D. Enantiocomplementary synthesis of γ-nitroketones using designed and evolved carboligases. J. Am. Chem. Soc. 139, 103–106 (2017).
Garrabou, X., Macdonald, D. S. & Hilvert, D. Chemoselective Henry condensations catalyzed by artificial carboligases. Chem. Eur. J. 23, 6001–6003 (2017).
Fujimori, D. G. Radical SAM-mediated methylation reactions. Curr. Opin. Chem. Biol. 17, 597–604 (2013).
Mansoorabadi, S. O., Thibodeaux, C. J. & Liu, H. The diverse roles of flavin coenzymes — nature’s most versatile thespians. J. Org. Chem. 72, 6329–6342 (2007).
Valdez, C. E., Smith, Q. A., Nechay, M. R. & Alexandrova, A. N. Mysteries of metals in metalloenzymes. Acc. Chem. Res. 47, 3110–3117 (2014).
Coelho, P. S., Brustad, E. M., Kannan, A. & Arnold, F. H. Olefin cyclopropanation via carbene transfer catalyzed by engineered cytochrome P450 enzymes. Science 339, 307–310 (2013).
Emmanuel, M. A., Greenberg, N. R., Oblinsky, D. G. & Hyster, T. K. Accessing non-natural reactivity by irradiating nicotinamide-dependent enzymes with light. Nature 540, 414–417 (2016).
Ponka, P. Cell biology of heme. Am. J. Med. Sci. 318, 241–256 (1999).
Poulos, T. L. & Kraut, J. A hypothetical model of the cytochrome c peroxidase.cytochrome c electron transfer complex. J. Biol. Chem. 255, 10322–10330 (1980).
Zhang, Z. et al. Electron transfer by domain movement in cytochrome bc 1. Nature 392, 677–684 (1998).
Ortiz de Montellano, P. R. Hydrocarbon hydroxylation by cytochrome P450 enzymes. Chem. Rev. 110, 932–948 (2010).
Omura, T. & Sato, R. A new cytochrome in liver microsomes. J. Biol. Chem. 237, PC1375–PC1376 (1962).
Rupasinghe, S. et al. The cytochrome P450 gene family CYP157 does not contain EXXR in the K-helix reducing the absolute conserved P450 residues to a single cysteine. FEBS Lett. 580, 6338–6342 (2006).
Meunier, B., de Visser, S. P. & Shaik, S. Mechanism of oxidation reactions catalyzed by cytochrome P450 enzymes. Chem. Rev. 104, 3947–3980 (2004).
Baek, H. K. & Van Wart, H. E. Elementary steps in the formation of horseradish peroxidase compound I: direct observation of compound 0, a new intermediate with a hyperporphyrin spectrum. Biochemistry 28, 5714–5719 (1989).
Rittle, J. & Green, M. T. Cytochrome P450 compound I: capture, characterization, and C-H bond activation kinetics. Science 933, 933–937 (2014).
Sono, M., Roach, M. P., Coulter, E. D. & Dawson, J. H. Heme-containing oxygenases. Chem. Rev. 96, 2841–2888 (1996).
Vaz, A. D. N., McGinnity, D. F. & Coon, M. J. Epoxidation of olefins by cytochrome P450: evidence from site-specific mutagenesis for hydroperoxo-iron as an electrophilic oxidant. Proc. Natl. Acad. Sci. USA 95, 3555–3560 (1998).
Groves, J. T., Nemo, T. E. & Myers, R. S. Hydroxylation and epoxidation catalyzed by iron-porphine complexes. Oxygen transfer from iodosylbenzene. J. Am. Chem. Soc. 101, 1032–1033 (1979).
Groves, J. T. & Nemo, T. E. Aliphatic hydroxylation catalyzed by iron porphyrin complexes. J. Am. Chem. Soc. 105, 6243–6248 (1983).
Groves, J. T. & Nemo, T. E. Epoxidation reactions catalyzed by iron porphyrins. Oxygen transfer from iodosylbenzene. J. Am. Chem. Soc. 105, 5786–5791 (1983).
Groves, J. T. & Quinn, R. Aerobic epoxidation of olefins with ruthenium porphyrin catalysts. J. Am. Chem. Soc. 107, 5790–5792 (1985).
Collman, J., Zhang, X., Lee, V., Uffelman, E. & Brauman, J. Regioselective and enantioselective epoxidation catalyzed by metalloporphyrins. Science 261, 1404–1411 (1993).
Grinstaff, M., Hill, M., Labinger, J. & Gray, H. Mechanism of catalytic oxygenation of alkanes by halogenated iron porphyrins. Science 264, 1311–1313 (1994).
Wolf, J. R., Hamaker, C. G., Djukic, J.-P., Kodadek, T. & Woo, L. K. Shape and stereoselective cyclopropanation of alkenes catalyzed by iron porphyrins. J. Am. Chem. Soc. 117, 9194–9199 (1995).
Morandi, B. & Carreira, E. M. Iron-catalyzed cyclopropanation in 6 M KOH with in situ generation of diazomethane. Science 335, 1471–1474 (2012).
Lewis, R. D. et al. Catalytic iron-carbene intermediate revealed in a cytochrome c carbene transferase. Proc. Natl Acad. Sci. USA 115, 7308–7313 (2018).
Breslow, R. & Gellman, S. H. Tosylamidation of cyclohexane by a cytochrome P-450 model. J. Chem. Soc. Chem. Commun. 24, 1400–1401 (1982).
Mahy, J. P., Bedi, G., Battioni, P. & Mansuy, D. Allylic amination of alkenes by tosyliminoiodobenzene: manganese porphyrins as suitable catalysts. Tetrahedron Lett. 29, 1927–1930 (1988).
Miura, Y. & Fulco, J. (ω–2) Hydroxylation of fatty acids by a soluble system from bacillus megaterium. J. Biol. Chem. 249, 1880–1888 (1974).
Kan, S. B. J., Lewis, R. D., Chen, K. & Arnold, F. H. Directed evolution of cytochrome c for carbon–silicon bond formation: bringing silicon to life. Science 354, 1048–1051 (2016).
Bordeaux, M., Tyagi, V. & Fasan, R. Highly diastereoselective and enantioselective olefin cyclopropanation using engineered myoglobin-based catalysts. Angew. Chem. Int. Ed. 54, 1744–1748 (2015).
Chen, K., Huang, X., Kan, S. B. J., Zhang, R. K. & Arnold, F. H. Enzymatic construction of highly strained carbocycles. Science 360, 71–75 (2018).
Zhang, R. K. et al. Enzymatic assembly of carbon–carbon bonds via iron-catalysed sp 3 C–H functionalization. Nature 565, 67–72 (2019).
Wang, Z. J., Peck, N. E., Renata, H. & Arnold, F. H. Cytochrome P450-catalyzed insertion of carbenoids into N–H bonds. Chem. Sci. 5, 598–601 (2014).
Chen, K., Zhang, S. Q., Brandenberg, O. F., Hong, X. & Arnold, F. H. Alternate heme ligation steers activity and selectivity in engineered cytochrome P450-catalyzed carbene-transfer reactions. J. Am. Chem. Soc. 140, 16402–16407 (2018).
Kan, S. B. J., Huang, X., Gumulya, Y., Chen, K. & Arnold, F. H. Genetically programmed chiral organoborane synthesis. Nature 552, 132–136 (2017).
Svastits, E. W., Dawson, J. H., Breslow, R. & Gellman, S. H. Functionalized nitrogen atom transfer catalyzed by cytochrome P-450. J. Am. Chem. Soc. 107, 6427–6428 (1985).
Cho, I. et al. Enantioselective aminohydroxylation of styrenyl olefins catalyzed by an engineered hemoprotein. Angew. Chem. Int. Ed. 58, 3138–3142 (2019).
Prier, C. K., Zhang, R. K., Buller, A. R., Brinkmann-Chen, S. & Arnold, F. H. Enantioselective, intermolecular benzylic C–H amination catalysed by an engineered iron-haem enzyme. Nat. Chem. 9, 629–634 (2017).
Cho, I., Jia, Z.-J. & Arnold, F. H. Site-selective enzymatic C‒H amidation for synthesis of diverse lactams. Science 364, 575–578 (2019).
Coelho, P. S. et al. A serine-substituted P450 catalyzes highly efficient carbene transfer to olefins in vivo. Nat. Chem. Biol. 9, 485–487 (2013).
Wang, Z. J. et al. Improved cyclopropanation activity of histidine-ligated cytochrome P450 enables the enantioselective formal synthesis of levomilnacipran. Angew. Chem. Int. Ed. 53, 6810–6813 (2014).
Renata, H., Wang, Z. J., Kitto, R. Z. & Arnold, F. H. P450-catalyzed asymmetric cyclopropanation of electron-deficient olefins under aerobic conditions. Catal. Sci. Technol. 4, 3640–3643 (2014).
Stelter, M. et al. A novel type of monoheme cytochrome c: biochemical and structural characterization at 1.23 Å resolution of Rhodothermus marinus cytochrome c. Biochemistry 47, 11953–11963 (2008).
Kendrew, J. C. et al. Structure of myoglobin: a three-dimensional Fourier synthesis at 2 Å. resolution. Nature 185, 422–427 (1960).
Huang, X. et al. A biocatalytic platform for synthesis of chiral α-trifluoromethylated organoborons. ACS Cent. Sci. 5, 270–276 (2019).
Alonso-Cotchico, L. & Roelfes, G. A “broad spectrum” carbene transferase for synthesis of chiral α-trifluoromethylated organoborons. ACS Cent. Sci. 5, 206–208 (2019).
Nakano, Y., Biegasiewicz, K. F. & Hyster, T. K. Biocatalytic hydrogen atom transfer: an invigorating approach to free-radical reactions. Curr. Opin. Chem. Biol. 49, 16–24 (2019).
Fukuzumi, S., Hironaka, K. & Tanaka, T. Photoreduction of alkyl halides by an NADH model compound. An electron-transfer chain mechanism. J. Am. Chem. Soc. 105, 4722–4727 (1983).
Fukuzumi, S., Inada, O. & Suenobu, T. Mechanisms of electron-transfer oxidation of NADH analogues and chemiluminescence. Detection of the keto and enol radical cations. J. Am. Chem. Soc. 125, 4808–4816 (2003).
Brill, Z. G., Grover, H. K. & Maimone, T. J. Enantioselective synthesis of an ophiobolin sesterterpene via a programmed radical cascade. Science 352, 1078–1082 (2016).
Sibi, M. P., Manyem, S. & Zimmerman, J. Enantioselective radical processes. Chem. Rev. 103, 3263–3296 (2003).
Biegasiewicz, K. F., Cooper, S. J., Emmanuel, M. A., Miller, D. C. & Hyster, T. K. Catalytic promiscuity enabled by photoredox catalysis in nicotinamide-dependent oxidoreductases. Nat. Chem. 10, 770–775 (2018).
Sandoval, B. A., Meichan, A. J. & Hyster, T. K. Enantioselective hydrogen atom transfer: discovery of catalytic promiscuity in flavin-dependent ‘ene’-reductases. J. Am. Chem. Soc. 139, 11313–11316 (2017).
Sorigué, D. et al. An algal photoenzyme converts fatty acids to hydrocarbons. Science 357, 903–907 (2017).
Sandoval, B. A., Kurtoic, S. I., Chung, M. M., Biegasiewicz, K. F. & Hyster, T. K. Photoenzymatic catalysis enables radical-mediated ketone reduction in ene-reductases. Angew. Chem. Int. Ed. 58, 8714–8718 (2019).
Biegasiewicz, K. F. et al. Photoexcitation of flavoenzymes enables a stereoselective radical cyclization. Science 364, 1166–1169 (2019).
Schwizer, F. et al. Artificial metalloenzymes: reaction scope and optimization strategies. Chem. Rev. 118, 142–231 (2018).
Lu, Y., Yeung, N., Sieracki, N. & Marshall, N. M. Design of functional metalloproteins. Nature 460, 855–862 (2009).
Pochapsky, T. C. et al. in Nickel and Its Surprising Impact in Nature Vol. 2 (eds Sigel, A., Sigel, H. & Sigel, R. K. O.) 473–500 (Wiley, 2007).
Deshpande, A. R., Pochapsky, T. C. & Ringe, D. The metal drives the chemistry: dual functions of acireductone dioxygenase. Chem. Rev. 117, 10474–10501 (2017).
Wilkins, R. G. & Williams, K. R. Kinetics of formation and dissociation of manganese-bovine carbonic anhydrase B. J. Am. Chem. Soc. 96, 2241–2245 (1974).
Okrasa, K. & Kazlauskas, R. J. Manganese-substituted carbonic anhydrase as a new peroxidase. Chem. Eur. J. 12, 1587–1596 (2006).
Fernández-Gacio, A., Codina, A., Fastrez, J., Riant, O. & Soumillion, P. Transforming carbonic anhydrase into epoxide synthase by metal exchange. ChemBioChem 7, 1013–1016 (2006).
Jing, Q., Okrasa, K. & Kazlauskas, R. J. Stereoselective hydrogenation of olefins using rhodium-substituted carbonic anhydrase—a new reductase. Chem. Eur. J. 15, 1370–1376 (2009).
Jing, Q. & Kazlauskas, R. J. Regioselective hydroformylation of styrene using rhodium-substituted carbonic anhydrase. ChemCatChem 2, 953–957 (2010).
Key, H. M., Clark, D. S. & Hartwig, J. F. Generation, characterization, and tunable reactivity of organometallic fragments bound to a protein ligand. J. Am. Chem. Soc. 137, 8261–8268 (2015).
Oohora, K. & Hayashi, T. Reconstitution of heme enzymes with artificial metalloporphyrinoids. Methods Enzymol. 580, 439–454 (2016).
Teale, F. W. J. Cleavage of the haem-protein link by acid methylethylketone. Biochim. Biophys. Acta 35, 543 (1959).
Oohora, K., Kihira, Y., Mizohata, E., Inoue, T. & Hayashi, T. C(sp3)–H bond hydroxylation catalyzed by myoglobin reconstituted with manganese porphycene. J. Am. Chem. Soc. 135, 17282–17285 (2013).
Oohora, K. et al. Manganese(V) porphycene complex responsible for inert C–H bond hydroxylation in a myoglobin matrix. J. Am. Chem. Soc. 139, 18460–18463 (2017).
Oohora, K. et al. Catalytic cyclopropanation by myoglobin reconstituted with iron porphycene: acceleration of catalysis due to rapid formation of the carbene species. J. Am. Chem. Soc. 139, 17265–17268 (2017).
Sreenilayam, G., Moore, E. J., Steck, V. & Fasan, R. Metal substitution modulates the reactivity and extends the reaction scope of myoglobin carbene transfer catalysts. Adv. Synth. Catal. 359, 2076–2089 (2017).
Moore, E. J., Steck, V., Bajaj, P. & Fasan, R. Chemoselective cyclopropanation over carbene Y–H insertion catalyzed by an engineered carbene transferase. J. Org. Chem. 83, 7480–7490 (2018).
Key, H. M., Dydio, P., Clark, D. S. & Hartwig, J. F. Abiological catalysis by artificial haem proteins containing noble metals in place of iron. Nature 534, 534–537 (2016).
Dydio, P., Key, H. M., Hayashi, H., Clark, D. S. & Hartwig, J. F. Chemoselective, enzymatic C–H bond amination catalyzed by a cytochrome P450 containing an Ir(Me)-PIX cofactor. J. Am. Chem. Soc. 139, 1750–1753 (2017).
Key, H. M. et al. Beyond iron: iridium-containing P450 enzymes for selective cyclopropanations of structurally diverse alkenes. ACS Cent. Sci. 3, 302–308 (2017).
Dydio, P. et al. An artificial metalloenzyme with the kinetics of native enzymes. Science 354, 102–106 (2016).
Rabe, K. S., Kiko, K. & Niemeyer, C. M. Characterization of the peroxidase activity of CYP119, a thermostable P450 from Sulfolobus acidocaldarius. ChemBioChem 9, 420–425 (2008).
Knight, A. M. et al. Diverse engineered heme proteins enable stereodivergent cyclopropanation of unactivated alkenes. ACS Cent. Sci. 4, 372–377 (2018).
Brandenberg, O. F. et al. Stereoselective enzymatic synthesis of heteroatom-substituted cyclopropanes. ACS Catal. 8, 2629–2634 (2018).
Hernandez, K. E. et al. Highly stereoselective biocatalytic synthesis of key cyclopropane intermediate to ticagrelor. ACS Catal. 6, 7810–7813 (2016).
Yu, Y., Hu, C., Xia, L. & Wang, J. Artificial metalloenzyme design with unnatural amino acids and non-native cofactors. ACS Catal. 8, 1851–1863 (2018).
Mirts, E. N., Petrik, I. D., Hosseinzadeh, P., Nilges, M. J. & Lu, Y. A designed heme-[4Fe-4S] metalloenzyme catalyzes sulfite reduction like the native enzyme. Science 361, 1098–1101 (2018).
Rosati, F. & Roelfes, G. Artificial metalloenzymes. ChemCatChem 2, 916–927 (2010).
Green, N. M. Avidin. Adv. Protein Chem. 29, 85–133 (1975).
Wilchek, M. & Bayer, E. A. Introduction to avidin-biotin technology. Methods Enzymol. 184, 5–13 (1990).
Wilson, M. E. & Whitesides, G. M. Conversion of a protein to a homogeneous asymmetric hydrogenation catalyst by site-specific modification with a diphosphinerhodium(I) moiety. J. Am. Chem. Soc. 100, 306–307 (1978).
Collot, J. et al. Artificial metalloenzymes for enantioselective catalysis based on biotin–avidin. J. Am. Chem. Soc. 125, 9030–9031 (2003).
Skander, M. et al. Artificial metalloenzymes: (strept)avidin as host for enantioselective hydrogenation by achiral biotinylated rhodium–diphosphine complexes. J. Am. Chem. Soc. 126, 14411–14418 (2004).
Pierron, J. et al. Artificial metalloenzymes for asymmetric allylic alkylation on the basis of the biotin–avidin technology. Angew. Chem. Int. Ed. 47, 701–705 (2008).
Chatterjee, A. et al. An enantioselective artificial Suzukiase based on the biotin–streptavidin technology. Chem. Sci. 7, 673–677 (2015).
Creus, M. et al. X-ray structure and designed evolution of an artificial transfer hydrogenase. Angew. Chem. Int. Ed. 47, 1400–1404 (2008).
Letondor, C. et al. Artificial transfer hydrogenases based on the biotin–(strept)avidin technology: fine tuning the selectivity by saturation mutagenesis of the host protein. J. Am. Chem. Soc. 128, 8320–8328 (2006).
Zimbron, J. M. et al. A dual anchoring strategy for the localization and activation of artificial metalloenzymes based on the biotin–streptavidin technology. J. Am. Chem. Soc. 135, 5384–5388 (2013).
Dürrenberger, M. et al. Artificial transfer hydrogenases for the enantioselective reduction of cyclic imines. Angew. Chem. Int. Ed. 50, 3026–3029 (2011).
Hyster, T. K. et al. Biotinylated Rh(III) complexes in engineered streptavidin for accelerated asymmetric C–H activation. Science 338, 500–503 (2012).
Ward, T. R. Artificial metalloenzymes based on the biotin–avidin technology: enantioselective catalysis and beyond. Acc. Chem. Res. 44, 47–57 (2011).
Heinisch, T. & Ward, T. R. Artificial metalloenzymes based on the biotin–streptavidin technology: challenges and opportunities. Acc. Chem. Res. 49, 1711–1721 (2016).
Hassan, I. S. et al. Asymmetric δ-lactam synthesis with a monomeric streptavidin artificial metalloenzyme. J. Am. Chem. Soc. 141, 4815–4819 (2019).
Nödling, A. R. et al. Reactivity and selectivity of iminium organocatalysis improved by a protein host. Angew. Chem. Int. Ed. 57, 12478–12482 (2018).
Pellizzoni, M. M. et al. Chimeric streptavidins as host proteins for artificial metalloenzymes. ACS Catal. 8, 1476–1484 (2018).
Mallin, H., Hestericová, M., Reuter, R. & Ward, T. R. Library design and screening protocol for artificial metalloenzymes based on the biotin-streptavidin technology. Nat. Protoc. 11, 835–852 (2016).
Wilson, Y. M., Duerrenberger, M., Nogueira, E. S. & Ward, T. R. Neutralizing the detrimental effect of glutathione on precious metal catalysts. J. Am. Chem. Soc. 136, 8928–8932 (2014).
Jeschek, M. et al. Directed evolution of artificial metalloenzymes for in vivo metathesis. Nature 537, 661–665 (2016).
Heinisch, T. et al. E. coli surface display of streptavidin for directed evolution of an allylic deallylase. Chem. Sci. 9, 5383–5388 (2018).
Rebelein, J. G., Cotelle, Y., Garabedian, B. & Ward, T. R. Chemical optimization of whole-cell transfer hydrogenation using carbonic anhydrase as host protein. ACS Catal. 9, 4173–4178 (2019).
Monnard, F. W., Nogueira, E. S., Heinisch, T., Schirmer, T. & Ward, T. R. Human carbonic anhydrase II as host protein for the creation of artificial metalloenzymes: the asymmetric transfer hydrogenation of imines. Chem. Sci. 4, 3269 (2013).
Heinisch, T. et al. Improving the catalytic performance of an artificial metalloenzyme by computational design. J. Am. Chem. Soc. 137, 10414–10419 (2015).
Bersellini, M. & Roelfes, G. Multidrug resistance regulators (MDRs) as scaffolds for the design of artificial metalloenzymes. Org. Biomol. Chem. 15, 3069–3073 (2017).
Grkovic, S., Brown, M. H. & Skurray, R. A. Regulation of bacterial drug export system. Microbiol. Mol. Biol. Rev. 66, 671–701 (2002).
Grkovic, S., Hardie, K. M., Brown, M. H. & Skurray, R. A. Interactions of the QacR multidrug-binding protein with structurally diverse ligands: implications for the evolution of the binding pocket. Biochemistry 42, 15226–15236 (2003).
Agustiandari, H. et al. LmrR is a transcriptional repressor of expression of the multidrug ABC transporter LmrCD in Lactococcus lactis. J. Bacteriol. 190, 759–763 (2008).
Roelfes, G. LmrR: a privileged scaffold for artificial metalloenzymes. Acc. Chem. Res. 52, 545–556 (2019).
Madoori, P. K., Agustiandari, H., Driessen, A. J. M. & Thunnissen, A.-M. W. H. Structure of the transcriptional regulator LmrR and its mechanism of multidrug recognition. EMBO J. 28, 156–166 (2009).
Bos, J., Fusetti, F., Driessen, A. J. M. & Roelfes, G. Enantioselective artificial metalloenzymes by creation of a novel active site at the protein dimer interface. Angew. Chem. Int. Ed. 51, (7472–7475 (2012).
Bos, J., García-Herraiz, A. & Roelfes, G. An enantioselective artificial metallo-hydratase. Chem. Sci. 4, 3578–3582 (2013).
Yang, H. et al. Evolving artificial metalloenzymes via random mutagenesis. Nat. Chem. 10, 318–324 (2018).
Liu, C. C. & Schultz, P. G. Adding new chemistries to the genetic code. Annu. Rev. Biochem. 79, 413–444 (2010).
Drienovská, I., Rioz-Martínez, A., Draksharapu, A. & Roelfes, G. Novel artificial metalloenzymes by in vivo incorporation of metal-binding unnatural amino acids. Chem. Sci. 6, 770–776 (2014).
Drienovská, I. et al. Design of an enantioselective artificial metallo-hydratase enzyme containing an unnatural metal-binding amino acid. Chem. Sci. 8, 7228–7235 (2017).
Schumacher, M. A. et al. Structural mechanisms of QacR induction and multidrug recognition. Science 294, 2158–2163 (2001).
Itou, H. et al. The CGL2612 protein from Corynebacterium glutamicum is a drug resistance-related transcriptional repressor: structural and functional analysis of a newly identified transcription factor from genomic DNA analysis. J. Biol. Chem. 280, 38711–38719 (2005).
Yamasaki, S. et al. The crystal structure of multidrug-resistance regulator RamR with multiple drugs. Nat. Commun. 4, 2078 (2013).
Bos, J., Browne, W. R., Driessen, A. J. M. & Roelfes, G. Supramolecular assembly of artificial metalloenzymes based on the dimeric protein LmrR as promiscuous scaffold. J. Am. Chem. Soc. 137, 9796–9799 (2015).
Villarino, L. et al. An artificial heme enzyme for cyclopropanation reactions. Angew. Chem. Int. Ed. 57, 7785–7789 (2018).
Drienovská, I., Mayer, C., Dulson, C. & Roelfes, G. A designer enzyme for hydrazone and oxime formation featuring an unnatural catalytic aniline residue. Nat. Chem. 10, 946–952 (2018).
Chin, J. W. et al. Addition of p-Azido-L-phenylalanine to the genetic code of Escherichia coli. J. Am. Chem. Soc. 124, 9026–9027 (2002).
Mehl, R. A. et al. Generation of a bacterium with a 21 amino acid genetic code. J. Am. Chem. Soc. 125, 935–939 (2003).
Mayer, C., Dulson, C., Reddem, E., Thunnissen, A.-M. W. H. & Roelfes, G. Directed evolution of a designer enzyme featuring an unnatural catalytic amino acid. Angew. Chem. Int. Ed. 58, 2083–2087 (2019).
Erkkilä, A., Majander, I. & Pihko, P. M. Iminium catalysis. Chem. Rev. 107, 5416–5470 (2007).
Lewis, J. C. Beyond the second coordination sphere: engineering dirhodium artificial metalloenzymes to enable protein control of transition metal catalysis. Acc. Chem. Res. 52, 576–584 (2019).
Rea, D. & Fülöp, V. Structure-function properties of prolyl oligopeptidase family enzymes. Cell Biochem. Biophys. 44, 349–365 (2006).
Harwood, V. J., Denson, J. D., Robinson-Bidle, K. A. & Schreier, H. J. Overexpression and characterization of a prolyl endopeptidase from the hyperthermophilic archaeon Pyrococcus furiosus. J. Bacteriol. 179, 3613–3618 (1997).
Nicolas, I., Le Maux, P. & Simonneaux, G. Asymmetric catalytic cyclopropanation reactions in water. Coord. Chem. Rev. 252, 727–735 (2008).
Harris, M. N., Madura, J. D., Ming, L.-J. & Harwood, V. J. Kinetic and mechanistic studies of prolyl oligopeptidase from the hyperthermophile Pyrococcus furiosus. J. Biol. Chem. 276, 19310–19317 (2001).
Reetz, M. T. Directed evolution of artificial metalloenzymes: a universal means to tune the selectivity of transition metal catalysts? Acc. Chem. Res. 52, 336–344 (2019).
Burke, A. J. et al. Design and evolution of an enzyme with a non-canonical organocatalytic mechanism. Nature 570, 219–223 (2019).
Brandenberg, O. F., Fasan, R. & Arnold, F. H. Exploiting and engineering hemoproteins for abiological carbene and nitrene transfer reactions. Curr. Opin. Biotechnol. 47, 102–111 (2017).
Ward, T. R. Artificial metalloenzymes based on the biotin–avidin technology: enantioselective catalysis and beyond. Acc. Chem. Res. 44, 47–57 (2011).
Tawfik, D. S. Loop grafting and the origins of enzyme species. Science 311, 475–476 (2006).
Alonso-Cotchico, L., Rodríguez-Guerra Pedregal, J., Lledós, A. & Maréchal, J.-D. The effect of cofactor binding on the conformational plasticity of the biological receptors in artificial metalloenzymes: the case study of LmrR. Front. Chem. 7, 211 (2019).
Takeuchi, K., Tokunaga, Y., Imai, M., Takahashi, H. & Shimada, I. Dynamic multidrug recognition by multidrug transcriptional repressor LmrR. Sci. Rep. 4, 6922 (2015).
Watkins, D. W. et al. Construction and in vivo assembly of a catalytically proficient and hyperthermostable de novo enzyme. Nat. Commun. 8, 358 (2017).
Burton, A. J., Thomson, A. R., Dawson, W. M., Brady, R. L. & Woolfson, D. N. Installing hydrolytic activity into a completely de novo protein framework. Nat. Chem. 8, 837–844 (2016).
Song, W. J., Yu, J. & Tezcan, F. A. Importance of scaffold flexibility/rigidity in the design and directed evolution of artificial metallo-β-lactamases. J. Am. Chem. Soc. 139, 16772–16779 (2017).
Song, W. J. & Tezcan, F. A. A designed supramolecular protein assembly with in vivo enzymatic activity. Science 346, 1525–1528 (2014).
Tantillo, D. J., Jiangang, C. & Houk, K. N. Theozymes and compuzymes: theoretical models for biological catalysis. Curr. Opin. Chem. Biol. 2, 743–750 (1998).
Kiss, G., Çelebi-Ölçüm, N., Moretti, R., Baker, D. & Houk, K. N. Computational enzyme design. Angew. Chem. Int. Ed. 52, 5700–5725 (2013).
Röthlisberger, D. et al. Kemp elimination catalysts by computational enzyme design. Nature 453, 190–195 (2008).
Jiang, L. et al. De novo computational design of retro-aldol enzymes. Science 319, 1387–1391 (2008).
Siegel, J. B. et al. Computational design of an enzyme catalyst for a stereoselective bimolecular Diels–Alder reaction. Science 329, 309–313 (2010).
Wang, L., Brock, A., Herberich, B. & Schultz, P. G. Expanding the genetic code of Escherichia coli. Science 292, 498–500 (2001).
Mirts, E. N., Bhagi-Damodaran, A. & Lu, Y. Understanding and modulating metalloenzymes with unnatural amino acids, non-native metal ions, and non-native metallocofactors. Acc. Chem. Res. 52, 935–944 (2019).
Acknowledgements
The authors gratefully acknowledge financial support from the Netherlands Organisation for Scientific Research (NWO, Vici grant 724.013.003 and Veni grant 722.017.007). G.R. acknowledges support from the Ministry of Education, Culture and Science (Gravitation programme no. 024.001.035).
Author information
Authors and Affiliations
Contributions
R.B.L.-G researched data for the article, wrote the manuscript and prepared the figures, with contributions from C.M. All authors contributed to the discussion, reviewing and editing of the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Chemistry thanks T. Ward and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Theozyme
-
A theoretical structure produced during computational enzyme design, where stabilizing amino acid residues are positioned around a computed transition state (complex).
- Whole-cell catalysis
-
Catalytic (enzymatic) conversions which employ intact cells, exploiting either the native metabolism of the organism or by using the organism to (over)produce a particular enzyme.
- Site-directed mutagenesis
-
Altering (a) particular amino acid(s) in a protein through mutation of the gene that encodes it.
- Apo-form
-
In cofactor-dependent enzymes, the form of the enzyme without its cofactor.
- Holoenzyme
-
In cofactor-dependent enzymes, the form of the enzyme complete with its cofactor.
- Periplasm
-
Principally in Gram-negative bacteria, the region of the cell between the outer and inner cytoplasmic membrane.
Rights and permissions
About this article
Cite this article
Leveson-Gower, R.B., Mayer, C. & Roelfes, G. The importance of catalytic promiscuity for enzyme design and evolution. Nat Rev Chem 3, 687–705 (2019). https://doi.org/10.1038/s41570-019-0143-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41570-019-0143-x
This article is cited by
-
Functional analysis of single enzymes combining programmable molecular circuits with droplet-based microfluidics
Nature Nanotechnology (2024)
-
Characterization of a novel amidohydrolase with promiscuous esterase activity from a soil metagenomic library and its application in degradation of amide herbicides
Environmental Science and Pollution Research (2024)
-
Tartrate Dehydrogenase in Bacillus Species: Deciphering Unique Catalytic Diversity Through Kinetic, Structural and Molecular Docking Analysis
The Protein Journal (2024)
-
Direct visible-light-excited flavoproteins for redox-neutral asymmetric radical hydroarylation
Nature Catalysis (2023)
-
Catalytic site flexibility facilitates the substrate and catalytic promiscuity of Vibrio dual lipase/transferase
Nature Communications (2023)