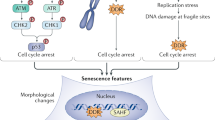
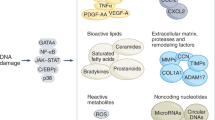
Abstract
Senescence is a state of permanent cell cycle arrest. Cellular senescence can promote tissue remodelling but can negatively affect regenerative capacities of tissues and contribute to inflammation and the progression of cancer and ageing-related diseases. This Review highlights the chemical characteristics of senescence and how we can target senescent cells with small molecules to induce senescence in hyperproliferative tissues. Alternatively, these small molecules can also be administered to inhibit senescence or eliminate senescent cells as the basis of a promising strategy to treat ageing-related diseases. We also describe advances in detecting senescence in in vitro and in vivo models, such that we can evaluate response to treatments intended to induce or eliminate senescent cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Hayflick, L. & Moorhead, P. S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 25, 585–621 (1961).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998).
Harley, C. B., Futcher, A. B. & Greider, C. W. Telomeres shorten during ageing of human fibroblasts. Nature 345, 458–460 (1990).
Olovnikov, A. M. Telomeres, telomerase, and aging: origin of the theory. Exp. Gerontol. 31, 443–448 (1996).
Yu, G.-L., Bradley, J. D., Attardi, L. D. & Blackburn, E. H. In vivo alteration of telomere sequences and senescence caused by mutated Tetrahymena telomerase RNAs. Nature 344, 126–132 (1990).
Bielak-Zmijewska, A., Mosieniak, G. & Sikora, E. Is DNA damage indispensable for stress-induced senescence? Mech. Ageing Dev. 170, 13–21 (2018).
Bloom, J. & Cross, F. R. Multiple levels of cyclin specificity in cell-cycle control. Nat. Rev. Mol. Cell. Biol. 8, 149–160 (2007).
Peeper, D. S. et al. A- and B-type cyclins differentially modulate substrate specificity of cyclin-cdk complexes. EMBO J. 12, 1947–1954 (1993).
Malumbres, M. & Barbacid, M. Cell cycle, CDKs and cancer: a changing paradigm. Nat. Rev. Cancer 9, 153–166 (2009).
Orzáez, M., Gortat, A., Mondragón, L., Bachs, O. & Pérez-Payá, E. ATP-noncompetitive inhibitors of CDK–cyclin complexes. ChemMedChem 4, 19–24 (2009).
Kong, X. et al. Interleukin-22 induces hepatic stellate cell senescence and restricts liver fibrosis in mice. Hepatology 56, 1150–1159 (2012).
Ritschka, B. et al. The senescence-associated secretory phenotype induces cellular plasticity and tissue regeneration. Genes Dev. 31, 172–183 (2017).
Mosteiro, L., Pantoja, C., de Martino, A. & Serrano, M. Senescence promotes in vivo reprogramming through p16INK4a and IL-6. Aging Cell 17, 12711–12722 (2018).
Hernandez-Segura, A., Nehme, J. & Demaria, M. Hallmarks of cellular senescence. Trends Cell Biol. 28, 436–453 (2018).
Sutherland, R. L. & Musgrove, E. A. CDK inhibitors as potential breast cancer therapeutics: new evidence for enhanced efficacy in ER+ disease. Breast Cancer Res. 11, 112–113 (2009).
Finn, R. S. et al. PD 0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially inhibits proliferation of luminal estrogen receptor-positive human breast cancer cell lines in vitro. Breast Cancer Res. 11, R77–R90 (2009).
Kirkland, J. L., Tchkonia, T., Zhu, Y., Niedernhofer, L. J. & Robbins, P. D. The clinical potential of senolytic drugs. J. Am. Geriatr. Soc. 65, 2297–2301 (2017).
Myrianthopoulos, V. The emerging field of senotherapeutic drugs. Future Med. Chem. 10, 2369–2372 (2018).
Rodier, F. et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat. Cell Biol. 11, 973–979 (2009).
Sagiv, A. et al. NKG2D ligands mediate immunosurveillance of senescent cells. Aging 8, 328–344 (2016).
Kang, T.-W. et al. Senescence surveillance of pre-malignant hepatocytes limits liver cancer development. Nature 479, 547–551 (2011).
Muñoz-Espín, D. & Serrano, M. Cellular senescence: from physiology to pathology. Nat. Rev. Mol. Cell. Biol. 15, 482–496 (2014).
Demaria, M. et al. An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev. Cell 31, 722–733 (2014).
Li, C. et al. Programmed cell senescence in skeleton during late puberty. Nat. Commun. 8, 1312 (2017).
Storer, M. et al. Senescence is a developmental mechanism that contributes to embryonic growth and patterning. Cell 155, 1119–1130 (2013).
O’Brien, W., Stenman, G. & Sager, R. Suppression of tumor growth by senescence in virally transformed human fibroblasts. Proc. Natl Acad. Sci. USA 83, 8659–8663 (1986).
Muñoz-Espín, D. et al. Programmed cell senescence during mammalian embryonic development. Cell 155, 1104–1118 (2013).
Sultana, Z., Maiti, K., Dedman, L. & Smith, R. Is there a role for placental senescence in the genesis of obstetric complications and fetal growth restriction? Am. J. Obstet. Gynecol. 218, S762–S773 (2018).
Schafer, M. J., Miller, J. D. & LeBrasseur, N. K. Cellular senescence: implications for metabolic disease. Mol. Cell. Endocrinol. 455, 93–102 (2017).
Khosla, S., Farr, J. N. & Kirkland, J. L. Inhibiting cellular senescence: a new therapeutic paradigm for age-related osteoporosis. J. Clin. Endocrinol. Metab. 103, 1282–1290 (2018).
Farr, J. N. et al. Targeting cellular senescence prevents age-related bone loss in mice. Nat. Med. 23, 1072–1079 (2017).
Schafer, M. J. et al. Cellular senescence mediates fibrotic pulmonary disease. Nat. Commun. 8, 14532 (2017).
Wandrer, F. et al. Senescence mirrors the extent of liver fibrosis in chronic hepatitis C virus infection. Aliment. Pharmacol. Ther. 48, 270–280 (2018).
Sun, R. et al. Senescence as a novel mechanism involved in β-adrenergic receptor mediated cardiac hypertrophy. PLoS One 12, e0182668 (2017).
Baker, D. J. & Petersen, R. C. Cellular senescence in brain aging and neurodegenerative diseases: evidence and perspectives. J. Clin. Invest. 128, 1208–1216 (2018).
Yang, H. & Fogo, A. B. Cell senescence in the aging kidney. J. Am. Soc. Nephrol. 21, 1436–1439 (2010).
Jimenez, R. et al. Replicative senescence in patients with chronic kidney failure. Kidney Int. 68, S11–S15 (2005).
He, S. & Sharpless, N. E. Senescence in health and disease. Cell 169, 1000–1011 (2017).
Krizhanovsky, V. et al. Senescence of activated stellate cells limits liver fibrosis. Cell 134, 657–667 (2008).
Ogrodnik, M. et al. Cellular senescence drives age-dependent hepatic steatosis. Nat. Commun. 8, 15691 (2017).
Tavana, O. & Zhu, C. Too many breaks (brakes): pancreatic β-cell senescence leads to diabetes. Cell Cycle 10, 2471–2484 (2011).
Krishnamurthy, J. et al. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature 443, 453–457 (2006).
Helman, A. et al. p16Ink4a-induced senescence of pancreatic beta cells enhances insulin secretion. Nat. Med. 22, 412–420 (2016).
Cindrova-Davies, T., Fogarty, N. M. E., Jones, C. J. P., Kingdom, J. & Burton, G. J. Evidence of oxidative stress-induced senescence in mature, post-mature and pathological human placentas. Placenta 68, 15–22 (2018).
Cox, L. S. & Redman, C. The role of cellular senescence in ageing of the placenta. Placenta 52, 139–145 (2017).
Collado, M. et al. Senescence in premalignant tumours. Nature 436, 642 (2005).
Chen, Z. et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436, 725–730 (2005).
Michaloglou, C. et al. BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436, 720–724 (2005).
Braig, M. et al. Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436, 660–665 (2005).
Schosserer, M., Grillari, J. & Breitenbach, M. The dual role of cellular senescence in developing tumors and their response to cancer therapy. Front. Oncol. 7, 2781–2793 (2017).
Wieland, E. et al. Endothelial Notch1 activity facilitates metastasis. Cancer Cell 31, 355–367 (2017).
Milanovic, M. et al. Senescence-associated reprogramming promotes cancer stemness. Nature 553, 96–100 (2017).
Parrinello, S., Coppe, J.-P., Krtolica, A. & Campisi, J. Stromal–epithelial interactions in aging and cancer: senescent fibroblasts alter epithelial cell differentiation. J. Cell Sci. 118, 485–496 (2005).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Baker, D. J. et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479, 232–236 (2011). In this work, the authors developed an ingenious genetic procedure to kill cells with high levels of p16. These cells overlap to a large extent with senescent cells. The authors observed that elimination of p16-positive cells improved the healthspan and extended the longevity of mice.
Baker, D. J. et al. Naturally occurring p16Ink4a-positive cells shorten healthy lifespan. Nature 530, 184–189 (2016).
Hernandez-Segura, A. et al. Unmasking transcriptional heterogeneity in senescent cells. Curr. Biol. 27, 2652–2660 (2017).
Wiley, C. D. et al. Analysis of individual cells identifies cell-to-cell variability following induction of cellular senescence. Aging Cell 16, 1043–1050 (2017).
Petrova, N. V., Velichko, A. K., Razin, S. V. & Kantidze, O. L. Small molecule compounds that induce cellular senescence. Aging Cell 15, 999–1017 (2016).
Marusyk, A., Wheeler, L. J., Mathews, C. K. & DeGregori, J. p53 mediates senescence-like arrest induced by chronic replicational stress. Mol. Cell. Biol. 27, 5336–5351 (2007).
Yeo, E. J. et al. Senescence-like changes induced by hydroxyurea in human diploid fibroblasts. Exp. Gerontol. 35, 553–571 (2000).
Sumikawa, E., Matsumoto, Y., Sakemura, R., Fujii, M. & Ayusawa, D. Prolonged unbalanced growth induces cellular senescence markers linked with mechano transduction in normal and tumor cells. Biochem. Biophys. Res. Commun. 335, 558–565 (2005).
Michishita, E. et al. 5-Bromodeoxyuridine induces senescence-like phenomena in mammalian cells regardless of cell type or species. J. Biochem. 126, 1052–1059 (1999).
Modrak, D. E., Leon, E., Goldenberg, D. M. & Gold, D. V. Ceramide regulates gemcitabine-induced senescence and apoptosis in human pancreatic cancer cell lines. Mol. Cancer Res. 7, 890–896 (2009).
Huang, M., Whang, P., Lewicki, P. & Mitchell, B. S. Cyclopentenyl cytosine induces senescence in breast cancer cells through the nucleolar stress response and activation of p53. Mol. Pharmacol. 80, 40–48 (2011).
Chang, B. D. et al. A senescence-like phenotype distinguishes tumor cells that undergo terminal proliferation arrest after exposure to anticancer agents. Cancer Res. 59, 3761–3767 (1999).
te Poele, R. H., Okorokov, A. L., Jardine, L., Cummings, J. & Joel, S. P. DNA damage is able to induce senescence in tumor cells in vitro and in vivo. Cancer Res. 62, 1876–1883 (2002).
Mansilla, S., Piña, B. & Portugal, J. Daunorubicin-induced variations in gene transcription: commitment to proliferation arrest, senescence and apoptosis. Biochem. J. 372, 703–711 (2003).
Coppé, J.-P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 6, e301 (2008).
Han, Z. et al. Role of p21 in apoptosis and senescence of human colon cancer cells treated with camptothecin. J. Biol. Chem. 277, 17154–17160 (2002).
Wang, X. et al. Evidence of cisplatin-induced senescent-like growth arrest in nasopharyngeal carcinoma cells. Cancer Res. 58, 5019–5022 (1998).
Robles, S. J. & Adami, G. R. Agents that cause DNA double strand breaks lead to p16INK4a enrichment and the premature senescence of normal fibroblasts. Oncogene 16, 1113–1123 (1998).
Hirose, Y., Berger, M. S. & Pieper, R. O. p53 effects both the duration of G2/M arrest and the fate of temozolomide-treated human glioblastoma cells. Cancer Res. 61, 1957–1963 (2001).
Zhou, J.-M. et al. Senescence and telomere shortening induced by novel potent G-quadruplex interactive agents, quindoline derivatives, in human cancer cell lines. Oncogene 25, 503–511 (2006).
Huang, F.-C., Chang, C.-C., Wang, J.-M., Chang, T.-C. & Lin, J.-J. Induction of senescence in cancer cells by the G-quadruplex stabilizer, BMVC4, is independent of its telomerase inhibitory activity. Br. J. Pharmacol. 167, 393–406 (2012).
Müller, S. et al. Pyridostatin analogues promote telomere dysfunction and long-term growth inhibition in human cancer cells. Org. Biomol. Chem. 10, 6537–6546 (2012).
Riou, J. F. et al. Cell senescence and telomere shortening induced by a new series of specific G-quadruplex DNA ligands. Proc. Natl Acad. Sci. USA 99, 2672–2677 (2002).
Marconett, C. N. et al. Indole-3-carbinol downregulation of telomerase gene expression requires the inhibition of estrogen receptor-alpha and Sp1 transcription factor interactions within the hTERT promoter and mediates the G1 cell cycle arrest of human breast cancer cells. Carcinogenesis 32, 1315–1323 (2011).
Zhao, L. & Wink, M. The β-carboline alkaloid harmine inhibits telomerase activity of MCF-7 cells by down-regulating hTERT mRNA expression accompanied by an accelerated senescent phenotype. PeerJ 1, e174 (2013).
Vogt, M., Haggblom, C., Yeargin, J., Christiansen-Weber, T. & Haas, M. Independent induction of senescence by p16 INK4a and p21 CIP1 in spontaneously immortalized human fibroblasts. Cell Growth Differ. 9, 139–146 (1998).
Kastenhuber, E. R. & Lowe, S. W. Putting p53 in context. Cell 170, 1062–1078 (2017).
Efeyan, A. et al. Induction of p53-dependent senescence by the mdm2 antagonist nutlin-3a in mouse cells of fibroblast origin. Cancer Res. 67, 7350–7357 (2007).
Ling, X. et al. FL118 Induces p53-dependent senescence in colorectal cancer cells by promoting degradation of MdmX. Cancer Res. 74, 7487–7497 (2014).
Leontieva, O. V. & Blagosklonny, M. V. CDK4/6-inhibiting drug substitutes for p21 and p16 in senescence: duration of cell cycle arrest and MTOR activity determine geroconversion. Cell Cycle 12, 3063–3069 (2013).
Park, J.-Y., Park, S.-H. & Weiss, R. H. Disparate effects of roscovitine on renal tubular epithelial cell apoptosis and senescence: implications for autosomal dominant polycystic kidney disease. Am. J. Nephrol. 29, 509–515 (2009).
Ray, P. D., Huang, B.-W. & Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 24, 981–990 (2012).
Naylor, R. M., Baker, D. J. & van Deursen, J. M. Senescent cells: a novel therapeutic target for aging and age-related diseases. Clin. Pharmacol. Ther. 93, 105–116 (2013).
Sikora, E., Mosieniak, G. & Sliwinska, M. A. Morphological and functional characteristic of senescent cancer cells. Curr. Drug Targets 17, 377–387 (2016).
Funayama, R. & Ishikawa, F. Cellular senescence and chromatin structure. Chromosoma 116, 431–440 (2007).
Zhang, R. & Adams, P. D. Heterochromatin and its relationship to cell senescence and cancer therapy. Cell Cycle 6, 784–789 (2007).
Goldstein, S., Ballantyne, S. R., Robson, A. L. & Moerman, E. J. Energy metabolism in cultured human fibroblasts during aging in vitro. J. Cell. Physiol. 112, 419–424 (1982).
Bittles, A. H. & Harper, N. Increased glycolysis in ageing cultured human diploid fibroblasts. Biosci. Rep. 4, 751–756 (1984).
Muggleton-Harris, A. L. & Defuria, R. Age-dependent metabolic changes in cultured human fibroblasts. In Vitro Cell. Dev. Biol. 21, 271–276 (1985).
Childs, B. G., Baker, D. J., Kirkland, J. L., Campisi, J. & van Deursen, J. M. Senescence and apoptosis: dueling or complementary cell fates? EMBO Rep. 15, 1139–1153 (2014).
Muñoz-Espín, D. et al. A versatile drug delivery system targeting senescent cells. EMBO Mol. Med. 10, e9355 (2018). This work reports a silica-based nanoscaffold that releases its cargo preferentially in senescent cells. It demonstrates a principle that could be applied to other nanoformulations.
Takahashi, A., Ohtani, N. & Hara, E. Irreversibility of cellular senescence: dual roles of p16INK4a/Rb-pathway in cell cycle control. Cell Div. 2, 10–15 (2007).
Haaf, T. High-resolution analysis of DNA replication in released chromatin fibers containing 5-bromodeoxyuridine. Biotechniques 21, 1050–1054 (1996).
Gratzner, H. G. Monoclonal antibody to 5-bromo- and 5-iododeoxyuridine: a new reagent for detection of DNA replication. Science 218, 474–475 (1982).
Davalos, A. R. et al. p53-dependent release of alarmin HMGB1 is a central mediator of senescent phenotypes. J. Cell Biol. 201, 613–629 (2013).
Mather, K. A., Jorm, A. F., Parslow, R. A. & Christensen, H. Is telomere length a biomarker of aging? A review. J. Gerontol. A 66, 202–213 (2011).
Zietzer, A. & Hillmeister, P. Leucocyte telomere length as marker for cardiovascular ageing. Acta Physiol. 211, 251–256 (2014).
Bekaert, S., De Meyer, T. & Van Oostveldt, P. Telomere attrition as ageing biomarker. Anticancer Res. 25, 3011–3021 (2005).
Todaro, G. J. & Green, H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J. Cell Biol. 17, 299–313 (1963).
Blasco, M. A. et al. Telomere shortening and tumor formation by mouse cells lacking telomerase RNA. Cell 91, 25–34 (1997).
Blackburn, E. H. Structure and function of telomeres. Nature 350, 569–573 (1991).
Jaskelioff, M. et al. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature 469, 102–106 (2011).
Ourliac-Garnier, I. & Londoño-Vallejo, A. Telomere length analysis by quantitative fluorescent in situ hybridization (Q-FISH). Methods Mol. Biol. 735, 21–31 (2011).
Baerlocher, G. M., Vulto, I., de Jong, G. & Lansdorp, P. M. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nat. Protoc. 1, 2365–2376 (2006).
Martens, U. M. et al. Measurement of telomere length in haematopoietic cells using in situ hybridization techniques. Biochem. Soc. Trans. 28, 245–250 (2000).
Kelesidis, T. & Schmid, I. Assessment of telomere length, phenotype, and DNA content. Curr. Protoc. Cytom. 79, 1–23 (2017).
Brugat, T., Nguyen-Khac, F., Grelier, A., Merle-Béral, H. & Delic, J. Telomere dysfunction-induced foci arise with the onset of telomeric deletions and complex chromosomal aberrations in resistant chronic lymphocytic leukemia cells. Blood 116, 239–249 (2010).
Krtolica, A., Parrinello, S., Lockett, S., Desprez, P. Y. & Campisi, J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc. Natl Acad. Sci. USA 98, 12072–12077 (2001). This paper was the first to report that senescent cells secrete soluble factors with biological activity on non-senescent neighbour cells. In this particular case, the authors found that the secretome of senescent cells stimulates the tumorigenic properties of cancer cells.
Kuilman, T. et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 133, 1019–1031 (2008).
Acosta, J. C. et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133, 1006–1018 (2008).
Campisi, J., Andersen, J. K., Kapahi, P. & Melov, S. Cellular senescence: a link between cancer and age-related degenerative disease? Semin. Cancer Biol. 21, 354–359 (2011).
Acosta, J. C. et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol. 15, 978–990 (2013).
Jung, T., Bader, N. & Grune, T. Lipofuscin: formation, distribution, and metabolic consequences. Ann. NY Acad. Sci. 1119, 97–111 (2007).
Katz, M. L., Robinson, W. G., Herrmann, R. K., Groome, A. B. & Bieri, J. G. Lipofuscin accumulation resulting from senescence and vitamin E deficiency: spectral properties and tissue distribution. Mech. Ageing Dev. 25, 149–159 (1984).
Georgakopoulou, E. et al. Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging 5, 37–50 (2012).
Gatenby, J. B. & Moussa, T. A. The sudan black B technique in cytology. J. Microsc. Soc. 69, 72–75 (1949).
Rasmussen, G. L. A method of staining the statoacoustic nerve in bulk with Sudan black B. Anat. Rec. 139, 465–469 (1961).
Jung, T., Höhn, A. & Grune, T. Lipofuscin: detection and quantification by microscope techniques. Methods Mol. Biol. 594, 173–193 (2010).
Evangelou, K. et al. Robust, universal biomarker assay to detect senescent cells in biological specimens. Aging Cell 16, 192–197 (2017).
Dimri, G. P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995). This paper was the first to report a useful marker of senescence, namely, senescence-associated β-galactosidase (SA-β-Gal). Many current methods to detect senescence are based on their elevated SA-β-Gal.
Biran, A. et al. Quantitative identification of senescent cells in aging and disease. Aging Cell 16, 661–671 (2017).
Rotman, B. Measurement of activity of single molecules of β-d-galactosidase. Proc. Natl Acad. Sci. USA 47, 1981–1991 (1961).
Rotman, B., Zderic, J. A. & Edelstein, M. Fluorogenic substrates for β-d-galactosidases and phosphatases derived from flurescein (3,6-dihydroxyfluoran) and its monomethylether. Proc. Natl Acad. Sci. USA 50, 1–6 (1963).
Strachan, R., Wood, J. & Hirschmann, R. Synthesis and properties of 4-methyl-2-oxo-1,2-benzopyran-7-yl β-d-galactoside (galactoside of 4-methylumbelliferone). J. Org. Chem. 27, 1074–1075 (1962).
Zhang, J. et al. A novel near-infrared fluorescent probe for sensitive detection of β-galactosidase in living cells. Anal. Chim. Acta 968, 97–104 (2017).
Aizawa, K. Studien über Carbohydrasen, I. I. Die fermentative Hydrolyse des p-nitrophenol-β-galactoside. Enzymologia 6, 321–324 (1939).
Horwitz, J. P. et al. Substrates for cytochemical demonstration of enzyme activity. I. Some substituted 3-indolyl-β-d-glycopyranosides. J. Med. Chem. 7, 574–575 (1964).
Yang, N.-C. & Hu, M.-L. The limitations and validities of senescence-associated β-galactosidase activity as an aging marker for human foreskin fibroblast Hs68 cells. Exp. Gerontol. 40, 813–819 (2005).
Calado, R. T. & Young, N. S. Telomere diseases. N. Engl. J. Med. 361, 2353–2365 (2009).
Chatterjee, S. K., Bhattacharya, M. & Barlow, J. J. Glycosyltransferase and glycosidase activities in ovarian cancer patients. Cancer Res. 39, 1943–1951 (1979).
Knas, M. et al. The profile of lysosomal exoglycosidases in replicative and stress-induced senescence in early passage human fibroblasts. Folia Histochem. Cytobiol. 50, 220–227 (2012).
Hildebrand, D. et al. α-Fucosidase as a novel convenient biomarker for cellular senescence. Cell Cycle 12, 1922–1927 (2013).
Esterly, J. R., Standen, A. C. & Pearson, B. The histochemical demonstration of intestinal β-d-fucosidase with 5-bromo-4-chloroindole-3-yl-β-d-fucopyranoside. J. Histochem. Cytochem. 15, 470–474 (1967).
Rushton, A. R. & Dawson, G. Glycosphingolipid β-galactosidases of cultured mammalian cells: characterization of the enzymes from mouse cell line LMTK− and human lesch-nyhan fibroblasts. Biochim. Biophys. Acta 388, 92–105 (1975).
Gey, C. & Seeger, K. Metabolic changes during cellular senescence investigated by proton NMR-spectroscopy. Mech. Ageing Dev. 134, 130–138 (2013).
Campisi, J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120, 513–522 (2005).
Campisi, J. & d’Adda di Fagagna, F. Cellular senescence: when bad things happen to good cells. Nat. Rev. Mol. Cell. Biol. 8, 729–740 (2007).
Krishnamurthy, J. et al. Ink4a/Arf expression is a biomarker of aging. J. Clin. Invest. 114, 1299–1307 (2004).
Vandenberk, B., Brouwers, B., Hatse, S. & Wildiers, H. p16INK4a: a central player in cellular senescence and a promising aging biomarker in elderly cancer patients. J. Geriatr. Oncol. 2, 259–269 (2011).
Serrano, M., Hannon, G. J. & Beach, D. A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature 366, 704–707 (1993).
Serrano, M., Lin, A. W., McCurrach, M. E., Beach, D. & Lowe, S. W. Oncogenic ras provokes premature cell senescence associated with accumulation of p53 and p16INK4a. Cell 88, 593–602 (1997).
Beauséjour, C. M. et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 22, 4212–4222 (2003).
Rheinwald, J. G. et al. A two-stage, p16INK4A- and p53-dependent keratinocyte senescence mechanism that limits replicative potential independent of telomere status. Mol. Cell. Biol. 22, 5157–5172 (2002).
Herbig, U., Jobling, W. A., Chen, B. P. C., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21CIP1, but not p16INK4a. Mol. Cell 14, 501–513 (2004).
Hall, B. M. et al. Aging of mice is associated with p16(Ink4a)- and β-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells. Aging 8, 1294–1315 (2016).
Yamakoshi, K. et al. Real-time in vivo imaging of p16Ink4a reveals cross talk with p53. J. Cell Biol. 186, 393–407 (2009).
Lozano-Torres, B. et al. An OFF–ON two-photon fluorescent probe for tracking cell senescence in vivo. J. Am. Chem. Soc. 139, 8808–8811 (2017).
Bonten, E. J., Annunziata, I. & d’Azzo, A. Lysosomal multienzyme complex: pros and cons of working together. Cell. Mol. Life Sci. 71, 2017–2032 (2014).
Soto-Gamez, A. & Demaria, M. Therapeutic interventions for aging: the case of cellular senescence. Drug Discov. Today 22, 786–795 (2017).
Jeon, O. H. et al. Local clearance of senescent cells attenuates the development of post-traumatic osteoarthritis and creates a pro-regenerative environment. Nat. Med. 23, 775–781 (2017).
Baker, D. J. et al. Opposing roles for p16Ink4a and p19Arf in senescence and ageing caused by BubR1 insufficiency. Nat. Cell Biol. 10, 825–836 (2008).
Childs, B. G. et al. Senescent intimal foam cells are deleterious at all stages of atherosclerosis. Science 354, 472–477 (2016).
Justice, J. N. et al. Senolytics in idiopathic pulmonary fibrosis: results from a first-in-human, open-label, pilot study. EBioMedicine 40, 554–563 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03513016 (2019).
Zhu, Y. et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell 14, 644–658 (2015). This was the first demonstration that senescent cells are hypersensitive to certain drugs, now known as senolytic drugs. In this pioneering work, the authors discovered that the combination of dasatinib and quercetin is senolytic.
Fuhrmann-Stroissnigg, H. et al. Identification of HSP90 inhibitors as a novel class of senolytics. Nat. Commun. 8, 422 (2017).
Kim, H.-N. et al. DNA damage and senescence in osteoprogenitors expressing Osx1 may cause their decrease with age. Aging Cell 16, 693–703 (2017).
Chang, J. et al. Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat. Med. 22, 78–83 (2016).
Demaria, M. et al. Cellular senescence promotes adverse effects of chemotherapy and cancer relapse. Cancer Discov. 7, 165–176 (2017).
Bussian, T. J. et al. Clearance of senescent glial cells prevents tau-dependent pathology and cognitive decline. Nature 562, 578–582 (2018).
Yosef, R. et al. Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat. Commun. 7, 11190–11201 (2016).
Zhu, Y. et al. New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging 9, 1–9 (2017).
Baar, M. P. et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169, 132–147 (2017).
Roos, C. M. et al. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell 15, 973–977 (2016).
Xu, M. et al. Senolytics improve physical function and increase lifespan in old age. Nat. Med. 24, 1246–1256 (2018).
Wang, Y. et al. Discovery of piperlongumine as a potential novel lead for the development of senolytic agents. Aging 8, 2915–2926 (2016).
Zhang, X. X. et al. Oxidation resistance 1 is a novel senolytic target. Aging Cell 17, 12780 (2018).
Boskovic, Z. V., Hussain, M. M., Adams, D. J., Dai, M. & Schreiber, S. L. Synthesis of piperlogs and analysis of their effects on cells. Tetrahedron 69, 7559–7567 (2013).
Liu, X. et al. Senolytic activity of piperlongumine analogues: synthesis and biological evaluation. Bioorg. Med. Chem. 26, 3925–3938 (2018).
Adams, D. J. et al. Synthesis, cellular evaluation, and mechanism of action of piperlongumine analogs. Proc. Natl Acad. Sci. USA 109, 15115–15120 (2012).
He, Y., Li, W., Hu, G., Sun, H. & Kong, Q. Bioactivities of EF24, a novel curcumin analog: a review. Front. Oncol. 8, 614–622 (2018).
Li, W., He, Y., Zhang, R., Zheng, G. & Zhou, D. The curcumin analog EF24 is a novel senolytic agent. Aging 11, 771–782 (2019).
Samaraweera, L., Adomako, A., Rodriguez-Gabin, A. & McDaid, H. M. A novel indication for panobinostat as a senolytic drug in NSCLC and HNSCC. Sci. Rep. 7, 1900 (2017).
Ozsvari, B., Nuttall, J. R., Sotgia, F. & Lisanti, M. P. Azithromycin and roxithromycin define a new family of “senolytic” drugs that target senescent human fibroblasts. Aging 10, 3294–3307 (2018).
Bernardos, A. et al. Enzyme-responsive controlled release using mesoporous silica supports capped with lactose. Angew. Chem. Int. Ed. 48, 5884–5887 (2009).
Agostini, A. et al. Targeted cargo delivery in senescent cells using capped mesoporous silica nanoparticles. Angew. Chem. Int. Ed. 51, 10556–10560 (2012).
Niedernhofer, L. J. & Robbins, P. D. Senotherapeutics for healthy ageing. Nat. Rev. Drug Discov. 17, 377 (2018).
Harrison, D. E. et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 460, 392–395 (2009).
Herranz, N. et al. mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype. Nat. Cell Biol. 17, 1205–1217 (2015).
Laberge, R.-M. et al. MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat. Cell Biol. 17, 1049–1061 (2015).
Thapa, R. K. et al. Progressive slowdown/prevention of cellular senescence by CD9-targeted delivery of rapamycin using lactose-wrapped calcium carbonate nanoparticles. Sci. Rep. 7, 43299 (2017).
Laberge, R.-M. et al. Glucocorticoids suppress selected components of the senescence-associated secretory phenotype. Aging Cell 11, 569–578 (2012).
Kang, H. T. et al. Chemical screening identifies ATM as a target for alleviating senescence. Nat. Chem. Biol. 13, 616–623 (2017).
Harrison, D. E. et al. Acarbose, 17-α-estradiol, and nordihydroguaiaretic acid extend mouse lifespan preferentially in males. Aging Cell 13, 273–282 (2014).
Liu, S. et al. Simvastatin suppresses breast cancer cell proliferation induced by senescent cells. Sci. Rep. 5, 17895 (2016).
Freund, A., Patil, C. K. & Campisi, J. p38MAPK is a novel DNA damage response-independent regulator of the senescence-associated secretory phenotype. EMBO J. 30, 1536–1548 (2011).
Xu, M. et al. JAK inhibition alleviates the cellular senescence-associated secretory phenotype and frailty in old age. Proc. Natl Acad. Sci. USA 112, 6301–6310 (2015).
Katz, M. L. & Robison, W. G. What is lipofuscin? Defining characteristics and differentiation from other autofluorescent lysosomal storage bodies. Arch. Gerontol. Geriatr. 34, 169–184 (2002).
Burd, C. E. et al. Monitoring tumorigenesis and senescence in vivo with a p16 INK4a-luciferase model. Cell 152, 340–351 (2013).
Lim, H., Park, H. & Kim, H. P. Effects of flavonoids on senescence-associated secretory phenotype formation from bleomycin-induced senescence in BJ fibroblasts. Biochem. Pharmacol. 96, 337–348 (2015).
Acknowledgements
R.M. laboratory members thank the financial support from the Spanish Government (projects MAT2015-64139-C4-1-R, RTI2018-100910-B-C41 and AGL2015-70235-C2-2-R) and the Generalitat Valenciana (project PROMETEO 2018/024). B.L-T. and A.E. are grateful to the Spanish Ministry of Economy for their PhD grants (FPU15/02707 and FPU17/05454). M.R. was holder of a “la Caixa”-Severo Ochoa PhD scholarship. Work in the laboratory of M.S. was funded by IRB and by grants from the Spanish Ministry of Economy (MINECO, SAF), the European Research Council (ERC Advanced Grant) and by “la Caixa” Foundation. M.O. thanks the financial support from SAF2017-84689-R project and MINECO/AEI/FEDER, UE. The IRB Barcelona is the recipient of a Severo Ochoa Award of Excellence from the MINECO. The funders had no role in the design, data collection, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to the preparation of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The laboratory of R.M. and the laboratory of M.S. have had a research contract with Pfizer, Inc. on research not mentioned in this work. R.M. and M.S. are founders and shareholders of Senolytic Therapeutics, Inc. (USA) and Senolytic Therapeutics, S.L. (Spain). The remaining authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Telomeres
-
Regions at the ends of chromosomes that comprise repetitive DNA sequences. These regions protect the chromosome from deterioration and from the undesirable fusion with other chromosomes.
- Oncogenes
-
Proto-oncogenes are genes that have various functions in cellular proliferation and differentiation. Activating mutations in these genes or an increased copy number give rise to what is known as oncogenes that stimulate uncontrolled cell proliferation contributing to cancer progression.
- Tumour suppressor genes
-
Genes that inhibit uncontrolled cell division in normal cells. Mutations that inactivate or reduce the function of tumour suppressor genes increase the risk of cancer.
- Spindle stress
-
The spindle apparatus is a cytoskeletal structure that separates the chromosomes into daughter cells during cell division. Oxidative stress, monopolar spindle 1 kinase inhibition or heat shock induce the deformation of spindles and trigger mitotic abnormalities.
- Nucleolar stress
-
The nucleolus is a subnuclear compartment responsible for ribosome biogenesis. The nucleolus senses different stress signals and coordinates the activation of p53, a protein involved in cell cycle arrest, DNA repair and apoptosis
- Natural killer cells
-
(NK cells). NK cells are lymphocytes of the innate immune system that kill cancer cells and cells infected by viruses.
- Macrophages
-
These cells form part of the innate immune system. They are phagocytes that can detect, engulf and eliminate pathogens and apoptotic cells.
- Neutrophils
-
These are the first white blood cells recruited to sites of inflammation. They are responsible for the phagocytosis of pathogens and the release of antimicrobial factors stored in their cytoplasmic granules.
- CD4+ T lymphocytes
-
These cells function in the immune response by stimulating other immune cells to fight against infection.
- CD8+ T lymphocytes
-
These cells (also known as cytotoxic T lymphocytes, CTLs) neutralize tumour and infected cells by injecting them with toxic enzymes, such as perforin and granzyme. They are commonly called CD8+, due to the presence of the CD8 membrane receptor which recognizes peptides of major histocompatibility complex (MHC) class I protein on infected cells.
- B lymphocytes
-
White blood cells of the adaptive immune system responsible for the secretion of antibodies that recognize antigenic molecules of pathogens. They are also responsible for the presentation of antigens to T lymphocytes, the negative regulation of inflammatory responses and the regulation of responses to autoantigens.
- Immune clearance
-
Elimination of microorganisms or various types of cells, such as senescent, infected or tumour cells, by the immune system.
- Steatosis
-
The accumulation of extra fat in the liver, producing the disease that is also known as ‘fatty liver’.
Rights and permissions
About this article
Cite this article
Lozano-Torres, B., Estepa-Fernández, A., Rovira, M. et al. The chemistry of senescence. Nat Rev Chem 3, 426–441 (2019). https://doi.org/10.1038/s41570-019-0108-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41570-019-0108-0
This article is cited by
-
A renal clearable fluorogenic probe for in vivo β-galactosidase activity detection during aging and senolysis
Nature Communications (2024)
-
Targeting NEDD8-activating enzyme for cancer therapy: developments, clinical trials, challenges and future research directions
Journal of Hematology & Oncology (2023)
-
Discovering small-molecule senolytics with deep neural networks
Nature Aging (2023)
-
Local H2 release remodels senescence microenvironment for improved repair of injured bone
Nature Communications (2023)
-
Hautalterung – zelluläre Seneszenz
Die Dermatologie (2023)