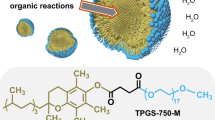
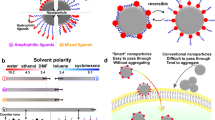
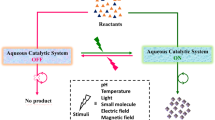
Abstract
Although aqueous solutions are considered to be sustainable, environmentally friendly reaction media, their use is often limited by poor reactant solubility. This limitation can be overcome by converting aqueous solutions into soft, dispersed interface-rich systems such as polyelectrolyte solutions, micellar solutions, oil-in-water microemulsions or vesicle dispersions. All consist of homogeneously distributed dynamic structures that, in a fashion reminiscent of enzymes, provide local environments that are different from the bulk solution. The presence of soft, dispersed interface-rich structures leads to not only selective reaction accelerations but also changes in reaction pathways, whereby chemical reactions are guided towards desired products. Once again, the analogy to enzyme-catalysed transformations is enticing. In this Review, we illustrate the general concepts applied in such systems and illustrate them with selected examples, ranging from enzyme mimics, the preparation of conductive polymers and transition-metal-catalysed organic syntheses on the industrial scale to the chemistry of prebiotic systems.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout












Similar content being viewed by others
References
Luisi, P. L. The Emergence of Life. From Chemical Origins of Synthetic Biology (Cambridge Univ. Press, Cambridge, 2006).
Deamer, D. First Life. Discovering the Connections between Stars, Cells, and How Life Began (University of California Press, Berkeley, 2011).
Ruiz-Mirazo, K., Briones, C. & de la Escosuras, A. Prebiotic systems chemistry: new perspectives for the origins of life. Chem. Rev. 114, 285–366 (2014).
Riuz-Mirazo, K., Peretó, J. & Moreno, A. A universal definition of life: autonomy and open-ended evolution. Origins Life Evol. Biospheres 34, 323–346 (2004).
Moreira, D. & López-García, P. Ten reasons to exclude viruses from the tree of life. Nat. Rev. Microbiol. 7, 306–311 (2009).
Alberts, B. et al. Molecular Biology of the Cell, 6th edn, (Garland Science, Taylor & Francis Group, New York, 2015).
Phillips, R., Kondev, J., Theriot, J. & Garcia, H. G. Physical Biology of the Cell 2nd edn, (Garland Science, Taylor & Francis Group, New York, 2013).
Ouzounis, C. A. & Karp, P. D. Global properties of the metabolic map of Escherichia coli. Genome Res. 10, 568–576 (2000).
Mouritsen, O. G. Life – as a Matter of Fat: The Emerging Science of Lipidomics. (Springer-Verlag Berlin Heidelberg, 2011).
Kohen, A. Role of dynamics in enzyme catalysis: substantial versus semantic controversies. Acc. Chem. Res. 48, 466–473 (2015).
Linsenmeier, A. M. & Braje, W. M. Efficient one-pot synthesis of dihydroquinolinones in water at room temperature. Tetrahedron 71, 6913–6919 (2015).
Gallou, F., Isley, N. A., Ganic, A., Onken, U. & Parmentier, M. Surfactant technology applied toward an active pharmaceutical ingredient: more than a simple green chemistry advance. Green Chem. 18, 14–19 (2016).
Lipshutz, B. L. When does organic chemistry follow nature’s lead and “make the switch”? J. Org. Chem. 82, 2806–2816 (2017).
Luisi, P. L. Why are enzymes macromolecules? Naturwissenschaften 66, 498–504 (1979).
Andreini, C., Cavallaro, G., Lorenzini, S. & Rosato, A. MetalPDB: a database of metal sites in biological macromolecular structures. Nucleic Acid Res. 41, D312–D319 (2013).
Sheldon, R. A. Enzyme immobilization: the quest for optimum performance. Adv. Synth. Catal. 349, 1289–1307 (2007).
Hanefeld, U., Gardossi, L. & Magner, E. Understanding enzyme immobilisation. Chem. Soc. Rev. 38, 453–468 (2009).
Küchler, A., Yoshimoto, M., Luginbühl, S., Mavelli, F. & Walde, P. Enzymatic reactions in confined environments. Nat. Nanotechnol. 11, 409–420 (2016).
Doherty, E. A. & Doudna, J. A. Ribozyme structures and mechanisms. Annu. Rev. Biophys. Biomol. Struct. 30, 457–475 (2001).
Silverman, S. K. Catalytic DNA: scope, applications, and biochemistry of deoxyribozymes. Trends Biochem. Sci. 41, 595–609 (2016).
Raynal, M., Ballester, P., Vidal-Ferran, A. & van Leeuwen, P. W. N. M. Supramolecular catalysis. Part 2: artificial enzyme mimics. Chem. Soc. Rev. 43, 1734–1787 (2014).
Kuah, E., Toh, S., Yee, J., Ma, Q. & Gao, Z. Enzyme mimics: advances and applications. Chem. Eur. J. 22, 8404–8430 (2016).
Fréchet, J. M. J. Dendrimers and supramolecular chemistry. Proc. Natl Acad. Sci. USA 99, 4782–4787 (2002).
Astruc, D., Boisselier, E. & Ornelas, C. Dendrimers designed for functions: from physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, and nanomedicine. Chem. Rev. 110, 1857–1959 (2010).
Lehn, J. M. Supramolecular chemistry – scope and perspectives. Molecules, supermolecules, and molecular devices (nobel lecture). Angew. Chem. Int. Ed. Engl. 27, 89–112 (1988).
Moulin, E. & Giuseppone, N. in Supramolecular Chemistry: From Molecules to Nanomaterials (eds Gale, P. A., Steed, J. W.) 1543–1574 (John Wiley & Sons, Ltd., 2012).
Fersht, A. Structure and Mechanism in Protein Science (W. H. Freeman & Company, New York, 1999).
Silverman, R. B. The Organic Chemistry of Enzyme-Catalyzed Reactions (Academic Press, Amsterdam, 2002).
Berg, J. M., Tymoczko, J. L., Gatto, G. J. Jr. & Stryer, L. Biochemistry 8th edn, (W. H. Freeman & Company, New York, 2015).
Dodson, G. & Wlodawer, A. Catalytic triads and their relatives. Trends Pharm. Sci. 23, 347–352 (1998).
Nissen, P., Hansen, J., Ban, N., Moore, P. M. & Steitz, T. A. The structural basis of ribosome activity in peptide bond synthesis. Science 289, 920–930 (2000).
Sievers, A., Beringer, M., Rodnina, M. V. & Wolfenden, R. The ribosome as an entropy trap. Proc. Natl Acad. Sci. USA 101, 7897–7901 (2004); erratum 101, 12397–12398 (2004).
Bekturov, E. A. & Kudaibergenov, S. E. Catalysis by Polymers (WILEY-VCH Verlag GmbH & Co., KGaA, Weinheim, 2002).
Artar, M., Terashima, T., Sawamoto, M., Meijer, E. W. & Palmans, A. R. A. Understanding the catalytic activity of single-chain polymeric nanoparticles in water. J. Polym. Sci. A Polym. Chem. 52, 12–20 (2014).
Cole, J. P., Hanlon, A. M., Rodriguez, K. J. & Berda, E. B. Protein-like structure and activity in synthetic polymers. J. Polym. Sci. A Polym. Chem. 55, 191–206 (2017).
Artar, M., Souren, E. R. J., Terashima, T., Meijer, E. W. & Palmans, A. R. A. Single chain polymeric nanoparticles as selective hydrophobic reaction spaces in water. ACS Macro Lett. 4, 1099–1103 (2015).
Latorre-Sánchez, A. & Pomposo, J. A. Recent bioinspired applications of single-chain nanoparticles. Polym. Int. 65, 855–860 (2016).
Bungenberg de Jong, H. G. & Kruyt, H. R. Coacervation (partial miscibility in colloid systems). Proc. K. Ned. Akad. Wet. 32, 849–856 (1929).
Oparin, A. I. in In Prebiotic and Biochemical Evolution, (eds Kimball, A. P. & Oró, J.) 1–8 (North-Holland Publishing Company, Amsterdam, 1971).
Wang, Q. & Schlenoff, J. B. The polyelectrolyte complex/coacervate continuum. Macromolecules 47, 3108–3116 (2014).
Jho, Y. S., Yoo, H. Y., Lin, Y., Han, S. & Hwang, D. S. Molecular and structural basis of low interfacial energy of complex coacervates in water. Adv. Colloid Interface Sci. 239, 61–73 (2017).
Tomalia, D. A., Naylor, A. M. & Goddard III, W. A. Starburst dendrimers: molecular-level control of size, shape, surface chemistry, topology, and flexibility from atoms to macroscopic matter. Angew. Chem. Int. Ed. 29, 138–175 (1990).
Astruc, D. & Chardac, F. Dendritic catalysts and dendrimers in catalysis. Chem. Rev. 101, 2991–3023 (2001).
Kirkorian, K., Ellis, A. & Twyman, L. J. Catalytic hyperbranched polymers as enzyme mimics; exploiting the principles of encapsulation and supramolecular chemistry. Chem. Soc. Rev. 41, 6138–6159 (2012).
Plamper, F. A. & Richtering, W. Functional microgels and microgel systems. Acc. Chem. Res. 50, 131–140 (2017).
Fendler, E. J. & Fendler, J. H. Micellar catalysis in organic reactions: kinetic and mechanistic implications. Adv. Phys. Org. Chem. 8, 271–406 (1970).
Hiemenz, P. C. & Rajagopalan, R. Principles of Colloid and Surface Chemistry, 3rd edn, (Marcel Dekker, New York, 1997).
Israelachvili, J. N. Intermolecular and Surface Forces, 3rd edn, (Elsevier, Amsterdam, 2011).
Sjöblom, J., Lindberg, R. & Friberg, S. E. Microemulsions – phase equilibria characterization, structures, applications and chemical reactions. Adv. Colloid Interface Sci. 95, 125–287 (1996).
Wennerström, H., Söderman, O., Olsson, U. & Lindman, B. Macroemulsions versus microemulsions. Colloids Surf. A. 123–124, 13–26 (1997).
Holmberg, K. Organic reactions in microemulsions. Curr. Opin. Colloid Interf. Sci. 8, 187–196 (2003).
Murakami, Y., Kikuchi, J., Hisaeda, Y. & Hayashida, O. Artificial enzymes. Chem. Rev. 96, 721–758 (1996).
Umakoshi, H., Morimoto, K., Yasuda, N., Shimanouchi, T. & Kuboi, R. Development of liposome-based mimics of superoxide dismutase and peroxidase based on the “LIPOzyme” concept. J. Biotechnol. 147, 59–63 (2010).
Mancin, F. et al. Hydrolytic metallo-nanozymes: from micelles and vesicles to gold nanoparticles. Molecules 21, 1014 (2016).
Barriga, H. M. G., Holme, M. N. & Stevens, M. M. Cubosomes; the next generation of smart lipid nanoparticles? Angew. Chem. Int. Ed. https://doi.org/10.1002/anie.201804067 (2018).
Torchilin, V. P. & Weissig, V. (eds) Liposomes – A Practical Approach 2nd edn (Oxford Univ. Press, 2003).
Walde, P. in Encyclopedia of Nanoscience and Nanotechnology (ed. Nalwa, H. S.) 43–79 (APS, Valencia, CA, 2004).
Korlach, J., Schwille, P., Webb, W. W. & Feigenson, G. W. Characterization of lipid bilayer phases by confocal microscopy and fluorescence correlation spectroscopy. Proc. Natl Acad. Sci. USA 96, 8461–8466 (1999).
Veatch, S. L. & Keller, S. L. Separation of liquid phases in giant vesicles of ternary mixtures of phospholipids and cholesterol. Biophys. J. 85, 3074–3083 (2003).
Suga, K. & Umakoshi, H. Detection of nanosized ordered domains in DOPC/DPPC and DOPC/Ch binary lipid mixture systems of large unilamellar vesicles using a TEMPO quenching method. Langmuir 29, 4830–4838 (2013).
Huerta, E., Stals, P. J. M., Meijer, E. W. & Palmans, A. R. A. Consequences of folding a water-soluble polymer around an organocatalyst. Angew. Chem. Int. Ed. 52, 2906–2910 (2013).
Hanlon, A. M., Lyon, C. K. & Berda, E. B. What is next in single-chain nanoparticles? Macromolecules 49, 2–14 (2016).
Klotz, I. M., Royer, G. P. & Scarpa, I. S. Synthetic derivatives of polyethyleneimine with enzyme-like catalytic activity (synzymes). Proc. Natl Acad. Sci. USA 68, 263–264 (1971).
Kiefer, H. C., Congdon, W. I., Scarpa, I. S. & Klotz, I. M. Catalytic accelerations of 1012-fold by an enzyme-like synthetic polymer. Proc. Natl Acad. Sci. USA 69, 2155–2159 (1972).
Avenier, F., Domingos, J. B., Van Vliet, L. D. & Hollfelder, F. Poyethylene imine derivatives (“synzymes”) accelerate phosphate transfer in the absence of metal. J. Am. Chem. Soc. 129, 7611–7619 (2007).
Suh, J., Scarpa, I. S. & Klotz, I. M. Catalysis of decarboxylation of nitrobenzisoxazolecarboxylic acid and of cyanophenylacetic acid by modified polyethyleneimines. J. Am. Chem. Soc. 98, 7060–7064 (1976).
Klotz, I. M. & Suh, J. in Artificial Enzymes (ed. Breslow, R.) 63–88 (WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 2005).
Chi, Y., Scroggins, S. T., Boz, E. & Fréchet, J. M. J. Control of aldol reaction pathways of enolizable aldehydes in an aqueous environment with a hyperbranched polymeric catalyst. J. Am. Chem. Soc. 130, 17287–17289 (2008).
Chevalier, Y. & Bolzinger, A.-M. Emulsions stabilized with solid nanoparticles: pickering emulsions. Colloids Surf. A 439, 23–34 (2013).
Yang, Y. et al. An overview of pickering emulsions: solid-particle materials, classification, morphology, and applications. Front. Pharmacol. 8, 287 (2017).
Zhang, H., Zhang, Q., Hong, C. & Zou, G. Asymmetric Michael addition in an aqueous environment with the assistance of optically active hyperbranched polymers. Polym. Chem. 8, 1771–1777 (2017).
Tan, Y. Y. & Challa, G. in Encyclopedia of Polymer Science and Engineering 554–569. (John Wiley & Sons, Inc., New York, 1989.
Połowinski, S. Template polymerisation and co-polymerisation. Prog. Polym. Sci. 27, 537–577 (2002).
Ferguson, J. & Shah, A. O. Further studies on polymerization in interacting polymer systems. Eur. Polym. J. 4, 611–619 (1968).
Liu, J. M., Sun, L., Hwang, J.-H. & Yang, S. C. Novel template guided synthesis of polyaniline. Mat. Res. Soc. Symp. Proc. 247, 601–606 (1992).
Samuelson, L. A., Anagnostopoulos, A., Alva, K. S., Kumar, J. & Tripathy, S. K. Biologically derived conducting and water soluble polyaniline. Macromolecules 31, 4376–4378 (1998).
Liu, W., Kumar, J., Tripathy, S., Senecal, K. J. & Samuelson, L. Enzymatically synthesized conducting polyaniline. J. Am. Chem. Soc. 121, 71–78 (1999).
Zhang, Y., Douglas, J. F., Ermi, B. D. & Amis, E. J. Influence of counterion valency on the scattering properties of highly charged polyelectrolyte solutions. J. Chem. Phys. 114, 3299–3313 (2001).
Kim, Y.-J., Uyama, H. & Kobayashi, S. Regioselective synthesis of poly(phenylene) as a complex with poly(ethylene glycol) by template polymerization of phenol in water. Macromolecules 36, 5058–5060 (2003).
Romsted, L. in Supramolecular Chemistry: From Molecules to Nanomaterials (eds Gale, P. A. & Steed J. W.)181–203 (John Wiley & Sons, Ltd., 2012).
Smart, T. et al. Block copolymer nanostructures. Nano Today 3, 38–46 (2008).
Heald, C. R. et al. Poly(lactic acid)-poly(ethylene oxide) (PLA-PEG) nanoparticles: NMR studies of the central solidlike PLA core and the liquid PEG corona. Langmuir 18, 3669–3675 (2002).
Lund, R. et al. Equilibrium chain exchange kinetics of diblock copolymer micelles: effect of morphology. Macromolecules 44, 6145–6154 (2011).
Bernheim-Groswasser, A., Zana, R. & Talmon, Y. Sphere-to-cylinder transition in aqueous micellar solution of a dimeric (gemini) surfactant. J. Phys. Chem. B. 104, 4005–4009 (2000).
Almgren, M. Mixed micelles and other structures in the solubilization of bilayer lipid membranes by surfactants. Biochim. Biophys. Acta 1508, 146–163 (2000).
Menger, F. M. & Portnoy, C. E. On the chemistry of reactions proceeding inside molecular aggregates. J. Am. Chem. Soc. 89, 4698–4703 (1967).
Namani, T. & Walde, P. From decanoate micelles to decanoic acid/ dodecylbenzenesulfonate vesicles. Langmuir 21, 6210–6219 (2005).
Bunton, C. A., Minch, M. J., Hidalgo, J. & Sepulveda, L. Electrolyte effects on the cationic micelle-catalyzed decarboxylation of 6-nitrobenzisoxazole-3-carboxylate anion. J. Am. Chem. Soc. 95, 3262–3272 (1973).
Tascioglu, S. Micellar solutions as reaction media. Tetrahedron 52, 11113–11152 (1996).
Dwars, T., Paetzold, E. & Oehme, G. Reactions in micellar systems. Angew. Chem. Int. Ed. 44, 7174–7199 (2005).
La Sorella, G., Strukul, G. & Scarso, A. Recent advances in catalysis in micellar media. Green Chem. 17, 644–683 (2015).
Bunton, C. A. The dependence of micellar rate effects upon reaction mechanism. Adv. Colloid Interf. Sci. 123–126, 333–343 (2006).
Bunton, C. A., Romsted, L. S. & Savelli, G. Tests of the pseudophase model of micellar catalysis: its partial failure. J. Am. Chem. Soc. 101, 1253–1259 (1979).
Shinoda, K. & Hutchinson, E. Pseudo-phase separation model for thermodynamic calculations on micellar solutions. J. Phys. Chem. 66, 577–582 (1962).
Romsted, L. S., Bunton, C. A. & Yao, J. Micellar catalysis, a useful misnomer. Curr. Opin. Colloid Interface Sci. 2, 622–628 (1997).
Peng, Y.-Y., Ding, Q.-P., Li, Z., Wang, P. G. & Cheng, J.-P. Proline catalyzed aldol reactions in aqueous micelles: an environmentally friendly reaction system. Tetrahedron Lett. 44, 3871–3875 (2003).
Scrimin, P. in Supramolecular Control of Structure and Reactivity (ed. Hamilton, A. D.) 101–153 (John Wiley & Sons, Chichester, 1996).
Ravichandran, S. et al. Micellar nanoreactors for hematin catalyzed synthesis of electrically conducting polypyrrole. Langmuir 28, 13380–13386 (2012).
Cardellicchio, C., Capozzi, A. M. M. & Naso, F. The Betti base: the awakening of a sleeping beauty. Tetrahedron: Asymm. 21, 507–517 (2010).
Kumar, A., Gupta, M. K. & Kumar, M. Non-ionic surfactant catalyzed synthesis of Betti base in water. Tetrahedron Lett. 51, 1582–1584 (2010).
Kumar, A., Gupta, M. K., Kumar, M. & Saxena, D. Micelle promoted multicomponent synthesis of 3-amino alkylated indoles via a Mannich-type reaction in water. RSC Adv. 3, 1673–1678 (2013).
Gabriel, C. M. et al. Effects of co-solvents on reactions run under micellar catalysis conditions. Org. Lett. 19, 194–197 (2017).
Menger, F. M. & Elrington, A. R. Organic reactivity in microemulsion systems. J. Am. Chem. Soc. 113, 9621–9624 (1991).
Menger, F. M. & Elrington, A. R. Rapid deactivation of mustard via microemulsion technology. J. Am. Chem. Soc. 112, 8201–8203 (1990).
Chhatre, A. S., Joshi, R. A. & Kulkarni, B. D. Microemuslions as media for organic synthesis: selective nitration of phenol to ortho-nitrophenol using dilute nitric acid. J. Colloid Interface Sci. 158, 183–187 (1993).
Strey, R. Microemulsion microstructure and interfacial curvature. Colloid Polym. Sci. 272, 1005–1019 (1994).
Dey, J., Saha, M., Pal, A. K. & Ismail, K. Regioselective nitration of aromatic compounds in an aqueous sodium dodecylsulfate and nitric acid medium. RSC Adv. 3, 18609–18613 (2013).
Chern, C. S. Emulsion polymerization mechanisms and kinetics. Prog. Polym. Sci. 31, 443–486 (2006).
Asua, J. M. Miniemulsion polymerization. Prog. Polym. Sci. 27, 1283–1346 (2002).
Landfester, K. Polyreactions in miniemulsions. Macromol. Rapid Commun. 22, 896–936 (2001).
Handa, S., Fennewald, J. C. & Lipshutz, B. H. Aerobic oxidation in nanomicelles of aryl alkynes, in water at room temperature. Angew. Chem. Int. Ed. 53, 3432–3435 (2014).
Lipshutz, B. H. et al. TPGS-750M: a second-generation amphiphile for metal-catalyzed cross-couplings in water at room temperature. J. Org. Chem. 76, 4379–4391 (2011).
Lipshutz, B. H., Ghorai, S. & Cortes-Clerget, M. The hydrophobic effect applied to organic synthesis: recent synthetic chemistry “in water”. Chem. Eur. J. 24, 6672–6695 (2018).
Klumphu, P. & Lipshutz, B. H. “Nok”: a phytosterol-based amphiphile enabling transition-metal-catalyzed couplings in water at room temperature. J. Org. Chem. 79, 888–900 (2014).
Lee, N. R., Gallou, F. & Lipshutz, B. H. SNAr reactions in aqueous nanomicelles: from milligrams to grams with no dipolar aprotic solvents needed. Org. Process Res. Dev. 21, 218–221 (2017).
Lipshutz, B., Aguinaldo, G. T., Ghorai, S. & Voigtritter, K. Olefin cross-metathesis reactions at room temperature using the nonionic amphiphile “PTS”: just add water. Org. Lett. 10, 1325–1328 (2008).
Gallou, F., Guo, P., Parmentier, M. & Zhou, J. A. General and practical alternative to polar aprotic solvents exemplified on an amide bond formation. Org. Proc. Res. Dev. 20, 1388–1391 (2016).
Andersson, M. P., Gallou, F., Klumphu, P., Takale, B. & Lipshutz, B. H. Structure of nanoparticles derived from designer surfactant TPGS-750-M in water, as used in organic synthesis. Chem. Eur. J. 24, 6778–6786 (2018).
Gelbart, W. M. & Ben-Shaul, A. The “New” Science of “Complex Fluids”. J. Phys. Chem. 100, 13169–13189 (1996).
Walde, P., Umakoshi, H., Stano, P. & Mavelli, F. Emergent properties arising from the assembly of amphiphiles. Artificial vesicle membranes as reaction promoters and regulators. Chem. Commun. 50, 10177–10197 (2014).
Fernandez-Trillo, F., Grover, L. M., Stephenson-Brown, A., Harrison, P. & Mendes, P. M. Vesicles in nature and the laboratory: elucidation of their biological properties and synthesis of increasingly complex synthetic vesicles. Angew. Chem. Int. Ed. 56, 3142–3160 (2017).
Bagatolli, L. A. & Mouritsen, O. G. Is the fluid mosaic (and the accompanying raft hypothesis) a suitable model to describe fundamental features of biological membranes? What may be missing? Front. Plant Sci. 4, 457 (2013).
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Brown, D. A. & London, E. Structure and origin of ordered lipid domains in biological membranes. J. Membr. Biol. 164, 103–114 (1998).
Carquin, M., D’Auria, L., Pollet, H., Bongarzone, E. R. & Tyteca, D. Recent progress on lipid lateral heterogeneity in plasma membranes: from rafts to submicrometric domains. Prog. Lipid Res. 62, 1–24 (2016).
Sezgin, E., Lebental, I., Mayor, S. & Eggeling, C. The mystery of membrane organization: composition, regulation and roles of lipid rafts. Nat. Rev. Mol. Cell. Biol. 18, 361–374 (2017).
Pintre, I. C. & Webb, S. J. Binding and reactivity at bilayer membranes. Adv. Phys. Org. Chem. 47, 129–183 (2013).
Fendler, J. H. & Hinze, W. L. Reactivity control in micelles and surfactant vesicles. Kinetics and mechanism of base-catalyzed hydrolysis of 5,5′-Dithiobis(2-nitrobenzoic acid) in water, hexadecyltrimethylammonium bromide micelles, and dioctadecyldimethylammonium chloride surfactant vesicles. J. Am. Chem. Soc. 103, 5439–5447 (1981).
Riddles, P. W., Blakeley, R. P. & Zerner, B. Reassessment of ellman’s reagent. Methods Enzymol. 91, 49–60 (1983).
Iwasaki, F., Suga, K. & Umakoshi, H. Pseudo-interphase of liposome promotes 1,3-dipolar cycloaddition reaction of benzonitrile oxide and N-ethylmaleimide in aqueous solution. J. Phys. Chem. B 119, 9772–9779 (2015).
Iwasaki, F., Suga, K., Okamoto, Y. & Umakoshi, H. Enantioselective C–C bond formation enhanced by self-assembly of achiral surfactants. ACS Omega 2, 1447–1453 (2017).
Paprocki, D., Koszelewski, D., Walde, P. & Ostaszewski, R. Efficient Passerini reactions in an aqueous vesicle system. RSC Adv. 5, 102828–102835 (2015).
Grochmal, A., Prout, L., Makin-Taylor, R., Prohens, R. & Tomas, S. Modulation of reactivity in the cavity of liposomes promotes the formation of peptide bonds. J. Am. Chem. Soc. 137, 12269–12275 (2015).
Luginbühl, S., Bertschi, L., Willeke, M., Schuler, L. D. & Walde, P. How anionic vesicles steer the oligomerization of enzymatically oxidized p-aminodiphenylamine (PADPA) toward a polyaniline emeraldine salt (PANI-ES)-type product. Langmuir 32, 9765–9779 (2016).
Janosevic Ležaic, A. et al. Insight into the template effect of vesicles on the laccase-catalyzed oligomerization of N-phenyl-1,4-phenylenediamine from Raman spectroscopy and cyclic voltammetry measurements. Sci. Rep. 6, 30724 (2016).
Duss, M. et al. Lipidic mesophases as novel nanoreactor scaffolds for organocatalysts: heterogeneously catalyzed asymmetric aldol reactions in confined water. ACS Appl. Mater. Interfaces 10, 5114–5124 (2018).
Zhou, T. et al. Efficient asymmetric synthesis of carbohydrates by aldolase nano- confined in lipidic cubic mesophases. ACS Catal. 8, 5810–5815 (2018).
Hutchison, C. A. III et al. Design and synthesis of a minimal bacterial genome. Science 35, aad6253 (2016).
Keller, M. A., Turchyn, A. V. & Ralser, M. Non-enzymatic glycolysis and pentose phosphate pathway-like reactions in a plausible Archean ocean. Mol. Syst. Biol. 10, 725 (2014).
Keller, M. A. et al. Conditional iron and pH-dependent activity of a non-enzymatic glycolysis and pentose phosphate pathway. Sci. Adv. 2, e1501235 (2016).
Muchowska, K. et al. Metals promote sequences of the reverse Krebs cycle. Nat. Ecol. Evol. 1, 1716–1721 (2017).
Orgel, L. E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 39, 99–123 (2004).
Joyce, G. F. & Orgel, L. E. in The RNA World (eds Gesteland, R. F., Cech, T. R., & Atkins, J. F.) 23–56 (Cold Spring Harbor Laboratory Press, New York, 2006).
Powner, M. W., Gerland, B. & Sutherland, J. D. Synthesis of activated pyrimidine nucleotides in prebiotically plausible conditions. Nature 459, 239–242 (2009).
Patel, B. H., Percivalle, C., Ritson, D. J., Duffy, C. D. & Sutherland, J. D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 7, 301–307 (2015).
Saladino, R., Crestini, C., Pino, S., Costanzo, G. & Di Mauro, E. Formamide and the origin of life. Phys. Life Rev. 9, 84–104 (2012).
Saladino, R., Šponer, J. E., Šponer, J. & Di Mauro, E. Rewarming the primordial soup: revisitations and rediscoveries in prebiotic chemistry. ChemBioChem 19, 22–25 (2018).
Lincoln, T. A. & Joyce, G. F. Self-sustained replication of an RNA enzyme. Science 323, 1229–1232 (2009).
Vaidya, N. et al. Spontaneous network formation among cooperative RNA replicators. Nature 491, 72–77 (2012).
Vasas, V., Fernando, C. T., Santos, M., Kauffman, S. A. & Szathmáry, E. Evolution before genes. Biol. Direct 7, 1 (2012).
de la Escosura, A., Briones, C. & Ruiz-Mirazo, K. The systems perspective at the crossroads between chemistry and biology. J. Theor. Biol. 381, 11–22 (2015).
Sutherland, J. D. Studies on the origin of life - the end of the beginning. Nat. Rev. Chem. 1, 1–7 (2017).
Gibard, C. et al. Phosphorylation, oligomerization and self-assembly in water under potential prebiotic conditions. Nat. Chem. 10, 212–217 (2018).
Maurer, S. E., Deamer, D. W., Boncella, J. M. & Monnard, P.-A. Chemical evolution of amphiphiles: glycerol monoacyl derivatives stabilize plausible prebiotic membranes. Astrobiology 9, 979–987 (2009).
Szostak, J. W. An optimal degree of physical and chemical heterogeneity for the origin of life? Phil. Trans. Roy. Soc. B 366, 2894–2901 (2011).
Ruiz-Mirazo, K., Briones, C. & de la Escosura, A. Chemical roots of biological evolution: the origins of life as a process of development of autonomous functional systems. Open Biol. 7, 170050 (2017).
Zhou, H. X., Rivas, G. & Minton, A. P. Macromolecular crowding and confinement: biochemical, biophysical, and potential physiological consequences. Annu. Rev. Biophys. 37, 375–397 (2008).
Minton, A. P. How can biochemical reactions within cells differ from those in test tubes? J. Cell Sci. 119, 2863–2869 (2006).
Spitzer, J. & Poolman, B. The role of biomacromolecular crowding, ionic strength and physicochemical gradients in the complexities of life’s emergence. Microbiol. Mol. Biol. Rev. 73, 371–388 (2009).
Spitzer, J., Pielak, G. & Poolman, B. Emergence of life: physical chemistry changes the paradigm. Biol. Direct 10, 33 (2015).
Oparin, A. I., Serebroskaya, K. B., Pantskhava, S. N. & Vasil’yeva, N. V. Enzymatic synthesis of polyadenylic acid in coacervate drops. Biokhimiya 28, 671–673 (1963).
Monnard, P.-A. & Walde, P. Current ideas about prebiological compartmentalization. Life 5, 1239–1263 (2015).
Ferris, J. P. Montmorillonite-catalysed formation of RNA oligomers: the possible role of catalysis in the origins of life. Phil. Trans. R. Soc., B 361, 1777–1786 (2006).
Lambert, J. F. Adsorption and polymerization of amino acids on mineral surfaces: a review. Origins Life Evol. Biospheres 38, 211–242 (2008).
Rimola, A., Costa, D., Sodupe, M., Lambert, J. F. & Ugliengo, P. Silica surface features and their role in the adsorption of biomolecules: computational modeling and experiments. Chem. Rev. 113, 4216–4313 (2013).
Erastova, V., Degiacomi, M. T., Fraser, D. G. & Greenwell, H. C. Mineral surface chemistry control for origin of prebiotic peptides. Nat. Commun. 8, 2033 (2017).
Sokolova, E. et al. Enhanced transcription rates in membrane-free protocells formed by coacervation of cell lysate. Proc. Natl Acad. Sci. USA 110, 11692–11697 (2013).
Tang, T.-Y. D. et al. Fatty acid membrane assembly on coacervate microdroplets as a step towards a hybrid protocell model. Nat. Chem. 6, 527–533 (2014).
Douliez, J.-P. et al. Catanionic coacervate droplets as a surfactant-based membrane-free protocell model. Angew. Chem. Int. Ed. 56, 13689–13693 (2017).
Bachmann, P. A., Luisi, P. L. & Lang, J. Autocatalytic self-replicating micelles as models for prebiotic structures. Nature 357, 57–59 (1992).
Walde, P., Goto, A., Monnard, P. A., Wessicken, M. & Luisi, P. L. Oparin’s reactions revisited: enzymatic synthesis of poly(adenylic acid) in micelles and self- reproducing vesicles. J. Am. Chem. Soc. 116, 7541–7547 (1994).
Morowitz, H. J., Heinz, B. & Deamer, D. W. The chemical logic of a minimum protocell. Origins Life Evol. Biospheres 18, 281–287 (1988).
Blocher, M., Liu, D., Walde, P. & Luisi, P. L. Liposome-assisted selective polycondensation of α-amino acids and peptides. Macromolecules 32, 7332–7334 (1999).
Hitz, T., Blocher, M., Walde, P. & Luisi, P. L. Stereoselectivity aspects in the condensation of racemic NCA-amino acids in the presence and absence of liposomes. Macromolecules 34, 2443–2449 (2001).
Zepik, H. H., Rajamani, S., Maurel, M.-C. & Deamer, D. Oligomerization of thioglutamic acid: encapsulated reactions and lipid catalysis. Origins Life Evol. Biospheres 37, 495–505 (2007).
Murillo-Sánchez, S., Beaufils, D., González Mañas, J. M., Pascal, R. & Ruiz-Mirazo, K. Fatty acids’ double role in the prebiotic formation of a hydrophobic dipeptide. Chem. Sci. 7, 3406–3414 (2016).
Vlassov, A., Khvorova, A. & Yarus, M. Binding and disruption of phospholipid bilayers by supramolecular RNA complexes. Proc. Natl Acad. Sci. USA 98, 7706–7711 (2001).
Janas, T., Janas, T. & Yarus, M. Specific RNA binding to ordered phospholipid bilayers. Nucleic Acids Res. 34, 2128–2136 (2006).
Kamat, N. P., Tobé, S., Hill, I. T. & Szostak, J. W. Electrostatic localization of RNA to protocell membranes by cationic hydrophobic peptides. Angew. Chem. Int. Ed. 54, 11735–11739 (2015).
Budin, I. & Szostak, J. Physical effects underlying the transition from primitive to modern cell membranes. Proc. Natl Acad. Sci. USA 108, 5249–5254 (2011).
Budin, I., Prywes, N., Zhang, N. & Szostak, J. W. Chain-length heterogeneity allows for the assembly of fatty acid vesicles in dilute solutions. Biophys. J. 107, 1582–1590 (2014).
Mansy, S. S. et al. Template-directed synthesis of a genetic polymer in a model protocell. Nature 454, 122–125 (2008).
Sacerdote, M. G. & Szostak, J. W. Semipermeable lipid bilayers exhibit diastereoselectivity favoring ribose. Proc. Natl Acad. Sci. USA 102, 6004–6008 (2005).
Hanczyc, M. M., Fujikawa, S. M. & Szostak, J. W. Experimental models of primitive cellular compartments: encapsulation, growth, and division. Science 302, 618–622 (2003).
Chen, I. A., Roberts, R. W. & Szostak, J. W. The emergence of competition between model protocells. Science 305, 1474–1476 (2004).
Zhu, T. F. & Szostak, J. W. Coupled growth and division of model protocell membranes. J. Am. Chem. Soc. 131, 5705–5713 (2009).
Adamala, K. & Szostak, J. W. Competition between model protocells driven by an encapsulated catalyst. Nat. Chem. 5, 495–501 (2013).
Hardy, M. D. et al. Self-reproducing catalyst drives repeated phospholipid synthesis and membrane growth. Proc. Natl Acad. Sci. USA 112, 8187–8192 (2015).
O’Flaherty, D. K. et al. Copying of mixed-sequence RNA templates inside model protocells. J. Am. Chem. Soc. 140, 5171–5178 (2018).
Albanese, D. C. M. & Gaggero, N. Albumin as a promiscuous biocatalyst in organic synthesis. RSC Adv. 5, 10588–10598 (2015).
Hollfelder, F., Kirby, A. J., Tawfik, D. S., Kikuchi, K. & Hilvert, D. Characterization of proton-transfer catalysis by serum albumins. J. Am. Chem. Soc. 122, 1022–1029 (2000).
Lockridge, O. et al. Pseudo-esterase activity of human albumin. J. Biol. Chem. 283, 22582–22590 (2008).
Saunders, B. R. & Vincent, B. Microgel particles as model colloids: theory, properties and applications. Adv. Colloid Interf. Sci. 80, 1–25 (1999).
Israelachvili, J. N., Mitchell, D. J. & Ninham, B. W. Theory of self-assembly of lipid bilayers and vesicles. Biochim. Biophys. Acta 470, 185–201 (1977).
Roussel, G., Michaux, C. & Perpète, E. A. Multiscale molecular dynamics simulations of sodium dodecyl sulfate micelles: from coarse-grained to all-atom resolution. J. Mol. Model. 20, 2469 (2014).
Hagan, S. A. et al. Polylactide-poly(ethylene glycol) copolymers as drug delivery systems. 1. Characterization of water dispersible micelle-forming systems. Langmuir 12, 2153–2161 (1996).
Wu, D. et al. Effect of molecular parameters on the architecture and membrane properties of 3D assemblies of amphiphilic copolymers. Macromolecules 47, 5060–5069 (2014).
Ma, J.-G., Boyd, B. J. & Drummond, C. J. Positional isomers of linear sodium dodecyl benzene sulfonate: solubility, self-assembly, and air/water interfacial activity. Langmuir 22, 8646–8654 (2006).
Guo, Z. et al. Vesicles as soft templates for the enzymatic polymerization of aniline. Langmuir 25, 11390–11404 (2009).
Acknowledgements
Dedicated to P. L. Luisi and F. M. Menger. The financial support from the European Cooperation in Science and Technology (COST) action CM1304 for the stimulating meetings on the ‘Emergence and Evolution of Complex Chemical Systems’ is highly appreciated, as well as the financial support from the Swiss National Science Foundation (project No. 200020_150254), the Polish National Science Center through HARMONIA project No. 2014/14/M/ST5/00030 and the Basque Government (project IT 590–13).
Reviewer information
Nature Reviews Chemistry thanks B. Lipshutz and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
All authors contributed to all aspects of manuscript preparation, revision and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Dispersed
-
To be more or less evenly distributed throughout the entire volume considered.
- Interface
-
The surface area of a dispersed entity that is in direct contact with the solution in which the entity is dispersed.
- Green industrial processes
-
Industrial processes that address and follow the guidance of the principles of green chemistry and engineering.
- In vivo catalytic activity
-
The ability to catalyse a particular chemical reaction at the place where the enzyme is localized within a cell (in the case of intracellular enzymes) or in the environment into which it is secreted from cells (the case of extracellular enzymes).
- Catalytic triad
-
A set of three specifically arranged amino acids involved in the catalysis at the active site of certain enzymes.
- Soft interface
-
Fluid in the sense that there is no fixed relationship between nearest-neighbour molecules or between different parts of the molecules constituting the interface.
- Dendrimers
-
Radially symmetric molecules with a well-defined monodisperse structure of tree-like branches.
- Amphiphiles
-
Chemical compounds consisting of ‘water-loving’ (hydrophilic) and ‘fat-loving’ (lipophilic), for example, ‘water-hating’ (hydrophobic), parts.
- Pickering emulsions
-
Emulsions that are stabilized by solid colloidal particles instead of conventional amphiphiles.
- Amphiphilic block copolymers
-
Polymer molecules consisting of adjacent blocks that differ in their constitution and water solubility such that they behave as macromolecular amphiphiles.
Rights and permissions
About this article
Cite this article
Serrano-Luginbühl, S., Ruiz-Mirazo, K., Ostaszewski, R. et al. Soft and dispersed interface-rich aqueous systems that promote and guide chemical reactions. Nat Rev Chem 2, 306–327 (2018). https://doi.org/10.1038/s41570-018-0042-6
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41570-018-0042-6
This article is cited by
-
Synthesising a minimal cell with artificial metabolic pathways
Communications Chemistry (2023)
-
Non-enzymatic oligonucleotide ligation in coacervate protocells sustains compartment-content coupling
Nature Communications (2023)
-
Probing solvation electrostatics at the air–water interface
Theoretical Chemistry Accounts (2023)
-
Switchable aqueous catalytic systems for organic transformations
Communications Chemistry (2022)
-
Self-reproducing catalytic micelles as nanoscopic protocell precursors
Nature Reviews Chemistry (2021)