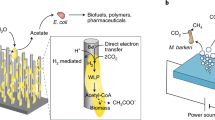
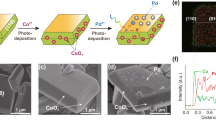
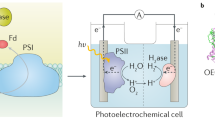
Abstract
Sunlight is our most abundant, clean and inexhaustible energy source. However, its diffuse and intermittent nature makes it difficult to use directly, suggesting that we should instead store this energy. One of the most attractive avenues for this involves using solar energy to split H2O and afford H2 through artificial photosynthesis, the practical realization of which requires low-cost, robust photocatalysts. Colloidal quantum dots (QDs) of IIB–VIA semiconductors appear to be an ideal material from which to construct highly efficient photocatalysts for H2 photogeneration. In this Review, we highlight recent developments in QD-based artificial photosynthetic systems for H2 evolution using sacrificial reagents. These case studies allow us to introduce strategies — including size optimization, structural modification and surface design — to increase the H2 evolution activities of QD-based artificial photosystems. Finally, we describe photocatalytic biomass reforming and unassisted photoelectrochemical H2O splitting — two new pathways that could make QD-based solar-to-fuel conversion practically viable and cost-effective in the near future.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Armaroli, N. & Balzani, V. The future of energy supply: challenges and opportunities. Angew. Chem. Int. Ed. 46, 52–66 (2007).
Lewis, N. S. & Nocera, D. G. Powering the planet: chemical challenges in solar energy utilization. Proc. Natl Acad. Sci. USA 103, 15729–15735 (2006).
Gray, H. B. Powering the planet with solar fuel. Nat. Chem. 1, 7 (2009).
Nocera, D. G. Solar fuels and solar chemicals industry. Acc. Chem. Res. 50, 616–619 (2017).
Lubitz, W. & Tumas, W. Hydrogen: an overview. Chem. Rev. 107, 3900–3903 (2007).
Armaroli, N. & Balzani, V. The hydrogen issue. ChemSusChem 4, 21–36 (2011).
Kim, D., Sakamoto, K. K., Hong, D. & Yang, P. Artificial photosynthesis for sustainable fuel and chemical production. Angew. Chem. Int. Ed. 54, 3259–3266 (2015).
Frischmann, P. D., Mahata, K. & Würthner, F. Powering the future of molecular artificial photosynthesis with light-harvesting metallosupramolecular dye assemblies. Chem. Soc. Rev. 42, 1847–1870 (2013).
Barber, J. Photosynthetic energy conversion: natural and artificial. Chem. Soc. Rev. 38, 185–196 (2009).
Chen, X., Shen, S., Guo, L. & Mao, S. S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 110, 6503–6570 (2010).
Wu, L.-Z., Chen, B., Li, Z.-J. & Tung, C.-H. Enhancement of the efficiency of photocatalytic reduction of protons to hydrogen via molecular assembly. Acc. Chem. Res. 47, 2177–2185 (2014).
Blankenship, R. E. et al. Comparing photosynthetic and photovoltaic efficiencies and recognizing the potential for improvement. Science 332, 805–809 (2011).
Armaroli, N. & Balzani, V. Solar electricity and solar fuels: status and perspectives in the context of the energy transition. Chem. Eur. J. 22, 32–57 (2016).
Wang, Q. et al. Scalable water splitting on particulate photocatalyst sheets with a solar-to-hydrogen energy conversion efficiency exceeding 1%. Nat. Mater. 15, 611 (2016).
Kudo, A. & Miseki, Y. Heterogeneous photocatalyst materials for water splitting. Chem. Soc. Rev. 38, 253–278 (2009).
Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
Brugger, P. A., Cuendet, P. & Graetzel, M. Ultrafine and specific catalysts affording efficient hydrogen evolution from water under visible light illumination. J. Am. Chem. Soc. 103, 2923–2927 (1981).
Maeda, K. et al. GaN:ZnO solid solution as a photocatalyst for visible-light-driven overall water splitting. J. Am. Chem. Soc. 127, 8286–8287 (2005).
Nocera, D. G. The artificial leaf. Acc. Chem. Res. 45, 767–776 (2012).
Qu, Y. & Duan, X. Progress, challenge and perspective of heterogeneous photocatalysts. Chem. Soc. Rev. 42, 2568–2580 (2013).
Romain, S., Vigara, L. & Llobet, A. Oxygen−oxygen bond formation pathways promoted by ruthenium complexes. Acc. Chem. Res. 42, 1944–1953 (2009).
Concepcion, J. J. et al. Making oxygen with ruthenium complexes. Acc. Chem. Res. 42, 1954–1965 (2009).
Min, S. & Lu, G. Enhanced electron transfer from the excited eosin Y to mpg-C3N4 for highly efficient hydrogen evolution under 550 nm irradiation. J. Phys. Chem. C 116, 19644–19652 (2012).
Yamada, Y., Miyahigashi, T., Kotani, H., Ohkubo, K. & Fukuzumi, S. Photocatalytic hydrogen evolution under highly basic conditions by using Ru nanoparticles and 2-phenyl-4-(1-naphthyl)quinolinium ion. J. Am. Chem. Soc. 133, 16136–16145 (2011).
Wang, C., Cao, S. & Fu, W.-F. A stable dual-functional system of visible-light-driven Ni(ii) reduction to a nickel nanoparticle catalyst and robust in situ hydrogen production. Chem. Commun. 49, 11251–11253 (2013).
Willkomm, J., Muresan, N. M. & Reisner, E. Enhancing H2 evolution performance of an immobilised cobalt catalyst by rational ligand design. Chem. Sci. 6, 2727–2736 (2015).
Cai, J.-G., Yu, Z.-T., Yuan, Y.-J., Li, F. & Zou, Z.-G. Dinuclear iridium(iii) complexes containing bibenzimidazole and their application to water photoreduction. ACS Catal. 4, 1953–1963 (2014).
Cline, E. D., Adamson, S. E. & Bernhard, S. Homogeneous catalytic system for photoinduced hydrogen production utilizing iridium and rhodium complexes. Inorg. Chem. 47, 10378–10388 (2008).
Lakshminarasimhan, N., Bokare, A. D. & Choi, W. Effect of agglomerated state in mesoporous TiO2 on the morphology of photodeposited Pt and photocatalytic activity. J. Phys. Chem. C 116, 17531–17539 (2012).
Wang, X. et al. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 8, 76–80 (2008).
Smith, A. M. & Nie, S. Semiconductor nanocrystals: structure, properties, and band gap engineering. Acc. Chem. Res. 43, 190–200 (2009).
Talapin, D. V., Lee, J.-S., Kovalenko, M. V. & Shevchenko, E. V. Prospects of colloidal nanocrystals for electronic and optoelectronic applications. Chem. Rev. 110, 389–458 (2009).
Zrazhevskiy, P., Sena, M. & Gao, X. Designing multifunctional quantum dots for bioimaging, detection, and drug delivery. Chem. Soc. Rev. 39, 4326–4354 (2010).
Kalyanasundaram, K., Borgarello, E., Duonghong, D. & Grätzel, M. Cleavage of water by visible-light irradiation of colloidal CdS solutions; inhibition of photocorrosion by RuO2. Angew. Chem. Int. Ed. 20, 987–988 (1981).
Wu, K. & Lian, T. Quantum confined colloidal nanorod heterostructures for solar-to-fuel conversion. Chem. Soc. Rev. 45, 3781–3810 (2016).
Artero, V. & Fontecave, M. Solar fuels generation and molecular systems: is it homogeneous or heterogeneous catalysis? Chem. Soc. Rev. 42, 2338–2356 (2013).
Piers, W. E. Future trends in organometallic chemistry: organometallic approaches to water splitting. Organometallics 30, 13–16 (2011).
Han, Z. & Eisenberg, R. Fuel from water: the photochemical generation of hydrogen from water. Acc. Chem. Res. 47, 2537–2544 (2014).
Holmes, M. A., Townsend, T. K. & Osterloh, F. E. Quantum confinement controlled photocatalytic water splitting by suspended CdSe nanocrystals. Chem. Commun. 48, 371–373 (2012).
Han, Z., Qiu, F., Eisenberg, R., Holland, P. L. & Krauss, T. D. Robust photogeneration of H2 in water using semiconductor nanocrystals and a nickel catalyst. Science 338, 1321–1324 (2012).
Jaramillo, T. F. et al. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts. Science 317, 100–102 (2007).
Weingarten, A. S. et al. Self-assembling hydrogel scaffolds for photocatalytic hydrogen production. Nat. Chem. 6, 964–970 (2014).
Tard, C. & Pickett, C. J. Structural and functional analogues of the active sites of the [Fe]-, [NiFe]-, and [FeFe]-hydrogenases. Chem. Rev. 109, 2245–2274 (2009).
Schilter, D., Camara, J. M., Huynh, M. T., Hammes-Schiffer, S. & Rauchfuss, T. B. Hydrogenase enzymes and their synthetic models: the role of metal hydrides. Chem. Rev. 116, 8693–8749 (2016).
Wang, F. et al. Artificial photosynthetic systems based on [FeFe]-hydrogenase mimics: the road to high efficiency for light-driven hydrogen evolution. ACS Catal. 2, 407–416 (2012).
Brown, K. A., Dayal, S., Ai, X., Rumbles, G. & King, P. W. Controlled assembly of hydrogenase–CdTe nanocrystal hybrids for solar hydrogen production. J. Am. Chem. Soc. 132, 9672–9680 (2010).
Wang, F. et al. A highly efficient photocatalytic system for hydrogen production by a robust hydrogenase mimic in an aqueous solution. Angew. Chem. Int. Ed. 50, 3193–3197 (2011).
Wu, H. B., Xia, B. Y., Yu, L., Yu, X.-Y. & Lou, X. W. Porous molybdenum carbide nano-octahedrons synthesized via confined carburization in metal–organic frameworks for efficient hydrogen production. Nat. Commun. 6, 6512 (2015).
Liu, C., Qiu, F., Peterson, J. J. & Krauss, T. D. Aqueous photogeneration of H2 with CdSe nanocrystals and nickel catalysts: electron transfer dynamics. J. Phys. Chem. B 119, 7349–7357 (2015).
Li, Z.-J. et al. A robust “artificial catalyst” in situ formed from CdTe QDs and inorganic cobalt salts for photocatalytic hydrogen evolution. Energy Environ. Sci 6, 465–469 (2013).
Li, Z.-J. et al. Direct synthesis of all-inorganic heterostructured CdSe/CdS QDs in aqueous solution for improved photocatalytic hydrogen generation. J. Mater. Chem. A 5, 10365–10373 (2017).
Li, Z.-J. et al. Visible light catalysis-assisted assembly of Nih-QD hollow nanospheres in situ via hydrogen bubbles. J. Am. Chem. Soc. 136, 8261–8268 (2014).
Li, Z.-J. et al. An exceptional artificial photocatalyst, Nih-CdSe/CdS core/shell hybrid, made in situ from CdSe quantum dots and nickel salts for efficient hydrogen evolution. Adv. Mater. 25, 6613–6618 (2013).
Alivisatos, A. P. Semiconductor clusters, nanocrystals, and quantum dots. Science 271, 933–937 (1996).
Alivisatos, A. P. Perspectives on the physical chemistry of semiconductor nanocrystals. J. Phys. Chem. 100, 13226–13239 (1996).
Li, X.-B. et al. Mechanistic insights into the interface-directed transformation of thiols into disulfides and molecular hydrogen by visible-light irradiation of quantum dots. Angew. Chem. Int. Ed. 53, 2085–2089 (2014).
Huang, J., Stockwell, D., Huang, Z., Mohler, D. L. & Lian, T. Photoinduced ultrafast electron transfer from CdSe quantum dots to Re–bipyridyl complexes. J. Am. Chem. Soc. 130, 5632–5633 (2008).
Zhao, J., Holmes, M. A. & Osterloh, F. E. Quantum confinement controls photocatalysis: a free energy analysis for photocatalytic proton reduction at CdSe nanocrystals. ACS Nano 7, 4316–4325 (2013).
Norris, D. J. & Bawendi, M. G. Measurement and assignment of the size-dependent optical spectrum in CdSe quantum dots. Phys. Rev. B 53, 16338–16346 (1996).
Sapra, S. & Sarma, D. D. Evolution of the electronic structure with size in II-VI semiconductor nanocrystals. Phys. Rev. B 69, 125304 (2004).
Hutchison, G. R., Ratner, M. A. & Marks, T. J. Intermolecular charge transfer between heterocyclic oligomers. Effects of heteroatom and molecular packing on hopping transport in organic semiconductors. J. Am. Chem. Soc. 127, 16866–16881 (2005).
Jian, J.-X. et al. Comparison of H2 photogeneration by [FeFe]-hydrogenase mimics with CdSe QDs and Ru(bpy)3Cl2 in aqueous solution. Energy Environ. Sci. 9, 2083–2089 (2016).
Peng, X. et al. Shape control of CdSe nanocrystals. Nature 404, 59–61 (2000).
Baranov, A. V. et al. Effect of ZnS shell thickness on the phonon spectra in CdSe quantum dots. Phys. Rev. B 68, 165306 (2003).
Wu, X.-J. et al. Controlled growth of high-density CdS and CdSe nanorod arrays on selective facets of two-dimensional semiconductor nanoplates. Nat. Chem. 8, 470–475 (2016).
Fisher, A. A. E. & Osborne, M. A. Sizing up excitons in core–shell quantum dots via shell-dependent photoluminescence blinking. ACS Nano 11, 7829–7840 (2017).
Zhu, H., Song, N. & Lian, T. Controlling charge separation and recombination rates in CdSe/ZnS type I core–shell quantum dots by shell thicknesses. J. Am. Chem. Soc. 132, 15038–15045 (2010).
Huang, J., Mulfort, K. L., Du, P. & Chen, L. X. Photodriven charge separation dynamics in CdSe/ZnS core/shell quantum dot/cobaloxime hybrid for efficient hydrogen production. J. Am. Chem. Soc. 134, 16472–16475 (2012).
Zhu, H., Song, N. & Lian, T. Wave function engineering for ultrafast charge separation and slow charge recombination in type II core/shell quantum dots. J. Am. Chem. Soc. 133, 8762–8771 (2011).
Nan, W. et al. Crystal structure control of zinc-blende CdSe/CdS core/shell nanocrystals: synthesis and structure-dependent optical properties. J. Am. Chem. Soc. 134, 19685–19693 (2012).
Thibert, A. et al. Sequestering high-energy electrons to facilitate photocatalytic hydrogen generation in CdSe/CdS nanocrystals. J. Phys. Chem. Lett. 2, 2688–2694 (2011).
Wang, P. et al. Shell thickness engineering significantly boosts the photocatalytic H2 evolution efficiency of CdS/CdSe core/shell quantum dots. ACS Appl. Mater. Interfaces 9, 35712–35720 (2017).
Kilina, S., Ivanov, S. & Tretiak, S. Effect of surface ligands on optical and electronic spectra of semiconductor nanoclusters. J. Am. Chem. Soc. 131, 7717–7726 (2009).
He, C., Weinberg, D. J., Nepomnyashchii, A. B., Lian, S. & Weiss, E. A. Control of the redox activity of PbS quantum dots by tuning electrostatic interactions at the quantum dot/solvent interface. J. Am. Chem. Soc. 138, 8847–8854 (2016).
Boles, M. A., Ling, D., Hyeon, T. & Talapin, D. V. The surface science of nanocrystals. Nat. Mater. 15, 141–153 (2016).
Hines, D. A. & Kamat, P. V. Recent advances in quantum dot surface chemistry. ACS Appl. Mater. Interfaces 6, 3041–3057 (2014).
Weiss, E. A. Designing the surfaces of semiconductor quantum dots for colloidal photocatalysis. ACS Energy Lett. 2, 1005–1013 (2017).
Peterson, M. D. et al. The role of ligands in determining the exciton relaxation dynamics in semiconductor quantum dots. Annu. Rev. Phys. Chem. 65, 317–339 (2014).
Weller, H. Colloidal semiconductor Q-particles: chemistry in the transition region between solid state and molecules. Angew. Chem. Int. Ed. 32, 41–53 (1993).
Das, A., Han, Z., Haghighi, M. G. & Eisenberg, R. Photogeneration of hydrogen from water using CdSe nanocrystals demonstrating the importance of surface exchange. Proc. Natl Acad. Sci. USA 110, 16716–16723 (2013).
Wang, P., Zhang, J., He, H., Xu, X. & Jin, Y. The important role of surface ligand on CdSe/CdS core/shell nanocrystals in affecting the efficiency of H2 photogeneration from water. Nanoscale 7, 5767–5775 (2015).
Nag, A. et al. Metal-free inorganic ligands for colloidal nanocrystals: S2−, HS−, Se2−, HSe−, Te2−, HTe−, TeS3 2−, OH−, and NH2 − as surface ligands. J. Am. Chem. Soc. 133, 10612–10620 (2011).
Kovalenko, M. V., Scheele, M. & Talapin, D. V. Colloidal nanocrystals with molecular metal chalcogenide surface ligands. Science 324, 1417–1420 (2009).
Kuehnel, M. F., Wakerley, D. W., Orchard, K. L. & Reisner, E. Photocatalytic formic acid conversion on CdS nanocrystals with controllable selectivity for H2 or CO. Angew. Chem. Int. Ed. 54, 9627–9631 (2015).
Chang, C. M., Orchard, K. L., Martindale, B. C. M. & Reisner, E. Ligand removal from CdS quantum dots for enhanced photocatalytic H2 generation in pH neutral water. J. Mater. Chem. A 4, 2856–2862 (2016).
Wen, M. et al. Secondary coordination sphere accelerates hole transfer for enhanced hydrogen photogeneration from [FeFe]-hydrogenase mimic and CdSe QDs in water. Sci. Rep. 6, 29851 (2016).
Li, X.-B. et al. Self-assembled framework enhances electronic communication of ultrasmall-sized nanoparticles for exceptional solar hydrogen evolution. J. Am. Chem. Soc. 139, 4789–4796 (2017).
Kuehnel, M. F. & Reisner, E. Solar hydrogen generation from lignocellulose. Angew. Chem. Int. Ed. 57, 3290–3296 (2018).
You, B., Liu, X., Jiang, N. & Sun, Y. A general strategy for decoupled hydrogen production from water splitting by integrating oxidative biomass valorization. J. Am. Chem. Soc. 138, 13639–13646 (2016).
Wang, J.-J. et al. Photocatalytic hydrogen evolution from glycerol and water over nickel-hybrid cadmium sulfide quantum dots under visible-light irradiation. ChemSusChem 7, 1468–1475 (2014).
Wakerley, D. W. et al. Solar-driven reforming of lignocellulose to H2 with a CdS/CdOx photocatalyst. Nat. Energy 2, 17021 (2017).
Sahai, S. et al. Quantum dots sensitization for photoelectrochemical generation of hydrogen: a review. Renew. Sust. Energ. Rev. 68, 19–27 (2017).
Macdonald, T. J. & Nann, T. Quantum dot sensitized photoelectrodes. Nanomaterials 1, 79 (2011).
Nann, T. et al. Water splitting by visible light: a nanophotocathode for hydrogen production. Angew. Chem. Int. Ed. 49, 1574–1577 (2010).
Ruberu, T. P. A., Dong, Y., Das, A. & Eisenberg, R. Photoelectrochemical generation of hydrogen from water using a CdSe quantum dot-sensitized photocathode. ACS Catal. 5, 2255–2259 (2015).
Liu, B. et al. A solution-processed, mercaptoacetic acid-engineered CdSe quantum dot photocathode for efficient hydrogen production under visible light irradiation. Energy Environ. Sci. 8, 1443–1449 (2015).
Li, X.-B. et al. Hole-accepting-ligand-modified CdSe QDs for dramatic enhancement of photocatalytic and photoelectrochemical hydrogen evolution by solar energy. Adv. Sci. 3, 1500282 (2016).
Wen, M. et al. Hole-transfer-layer modification of quantum dot-sensitized photocathodes for dramatically enhanced hydrogen evolution. Part. Part. Syst. Charact. 35, 1700278 (2017).
Li, J. et al. Graphdiyne: a metal-free material as hole transfer layer to fabricate quantum dot-sensitized photocathodes for hydrogen production. J. Am. Chem. Soc. 138, 3954–3957 (2016).
Lv, H. et al. Semiconductor quantum dot-sensitized rainbow photocathode for effective photoelectrochemical hydrogen generation. Proc. Natl Acad. Sci. USA 114, 11297–11302 (2017).
Dong, Y. et al. Efficient photoelectrochemical hydrogen generation from water using a robust photocathode formed by CdTe QDs and nickel ion. ACS Sustainable Chem. Eng. 3, 2429–2434 (2015).
Macdonald, T. J. et al. SWCNT photocathodes sensitised with InP/ZnS core–shell nanocrystals. J. Mater. Chem. C 4, 3379–3384 (2016).
Dong, Y. et al. Efficient and stable MoS2/CdSe/NiO photocathode for photoelectrochemical hydrogen generation from water. Chem. Asian J. 10, 1660–1667 (2015).
Meng, P., Wang, M., Yang, Y., Zhang, S. & Sun, L. CdSe quantum dots/molecular cobalt catalyst co-grafted open porous NiO film as a photocathode for visible light driven H2 evolution from neutral water. J. Mater. Chem. A 3, 18852–18859 (2015).
Yang, H. B. et al. Stable quantum dot photoelectrolysis cell for unassisted visible light solar water splitting. ACS Nano 8, 10403–10413 (2014).
Jing, L. et al. Aqueous based semiconductor nanocrystals. Chem. Rev. 116, 10623–10730 (2016).
Kuehnel, M. F. et al. ZnSe quantum dots modified with a Ni(cyclam) catalyst for efficient visible-light driven CO2 reduction in water. Chem. Sci. 9, 2501–2509 (2018).
Jang, Y. J. et al. Unbiased sunlight-driven artificial photosynthesis of carbon monoxide from CO2 using a ZnTe-based photocathode and a perovskite solar cell in tandem. ACS Nano 10, 6980–6987 (2016).
Jang, Y. J., Lee, J., Lee, J. & Lee, J. S. Solar hydrogen production from zinc telluride photocathode modified with carbon and molybdenum sulfide. ACS Appl. Mater. Interfaces 8, 7748–7755 (2016).
Fang, Z. et al. Defect engineering and phase junction architecture of wide-bandgap ZnS for conflicting visible light activity in photocatalytic H2 evolution. ACS Appl. Mater. Interfaces 7, 13915–13924 (2015).
Lian, Z. et al. Durian-shaped CdS@ZnSe core@mesoporous-shell nanoparticles for enhanced and sustainable photocatalytic hydrogen evolution. J. Phys. Chem. Lett. 9, 2212–2217 (2018).
Zhou, Y., Hu, W., Ludwig, J. & Huang, J. Exceptionally robust CuInS2/ZnS nanoparticles as single component photocatalysts for H2 evolution. J. Phys. Chem. C 121, 19031–19035 (2017).
Gao, Y.-J. et al. Self-assembled inorganic clusters of semiconducting quantum dots for effective solar hydrogen evolution. Chem. Commun. 54, 4858–4861 (2018).
Peng, Z. A. & Peng, X. Formation of high-quality CdTe, CdSe, and CdS nanocrystals using CdO as precursor. J. Am. Chem. Soc. 123, 183–184 (2001).
Qiu, F. et al. Photocatalytic hydrogen generation by CdSe/CdS nanoparticles. Nano Lett. 16, 5347–5352 (2016).
Gimbert-Suriñach, C. et al. Efficient and limiting reactions in aqueous light-induced hydrogen evolution systems using molecular catalysts and quantum dots. J. Am. Chem. Soc. 136, 7655–7661 (2014).
Han, K. et al. Photochemical hydrogen production from water catalyzed by CdTe quantum dots/molecular cobalt catalyst hybrid systems. Chem. Commun. 51, 7008–7011 (2015).
Li, C.-B. et al. Interface-directed assembly of a simple precursor of [FeFe]-H2ase mimics on CdSe QDs for photosynthetic hydrogen evolution in water. Energy Environ. Sci. 6, 2597–2602 (2013).
Wang, F. et al. Exceptional poly(acrylic acid)-based artificial [FeFe]-hydrogenases for photocatalytic H2 production in water. Angew. Chem. Int. Ed. 52, 8134–8138 (2013).
Jian, J.-X. et al. Chitosan confinement enhances hydrogen photogeneration from a mimic of the diiron subsite of [FeFe]-hydrogenase. Nat. Commun. 4, 2695 (2013).
Lai, L.-H. et al. Organic–inorganic hybrid solution-processed H2-evolving photocathodes. ACS Appl. Mater. Interfaces 7, 19083–19090 (2015).
Wen, M. et al. Integrating CdSe quantum dots with a [FeFe]-hydrogenase mimic into a photocathode for hydrogen evolution at a low bias voltage. ChemPhotoChem 1, 260–264 (2017).
Acknowledgements
The authors are grateful for financial support from the Ministry of Science and Technology of China (2014CB239402 and 2017YFA0206903), the National Science Foundation of China (21390404, 21861132004 and 21603248), the Strategic Priority Research Program of the Chinese Academy of Science (XDB17000000), Key Research Program of Frontier Science of the Chinese Academy of Sciences (QYZDY-SSW-JSCO29) and the Youth Innovation Promotion Association of Chinese Academy of Sciences (2018031).
Author information
Authors and Affiliations
Contributions
All authors contributed to researching the article, discussing the content and writing and editing of the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, XB., Tung, CH. & Wu, LZ. Semiconducting quantum dots for artificial photosynthesis. Nat Rev Chem 2, 160–173 (2018). https://doi.org/10.1038/s41570-018-0024-8
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41570-018-0024-8
This article is cited by
-
Silver nanoparticle enhanced metal-organic matrix with interface-engineering for efficient photocatalytic hydrogen evolution
Nature Communications (2023)
-
Counterbalancing of electron and hole transfer in quantum dots for enhanced photocatalytic H2 evolution
Nano Research (2023)
-
Metal-Free 2D/2D van der Waals Heterojunction Based on Covalent Organic Frameworks for Highly Efficient Solar Energy Catalysis
Nano-Micro Letters (2023)
-
Recent Advancements in Photoelectrochemical Water Splitting for Hydrogen Production
Electrochemical Energy Reviews (2023)
-
Unraveling of cocatalysts photodeposited selectively on facets of BiVO4 to boost solar water splitting
Nature Communications (2022)