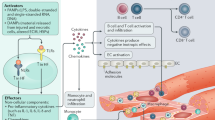
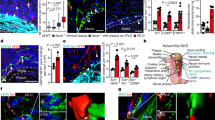
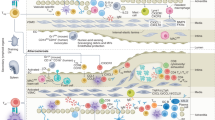
Abstract
Cardiovascular diseases (CVDs) make a substantial contribution to the global burden of disease. Prevention strategies have succeeded in reducing the effect of acute CVD events and deaths, but the long-term consequences of cardiovascular risk factors still represent the major cause of disability and chronic illness, suggesting that some pathophysiological mechanisms might not be adequately targeted by current therapies. Many of the underlying causes of CVD have now been recognized to have immune and inflammatory components. However, inflammation and immune activation were mostly regarded as a consequence of target-organ damage. Only more recent findings have indicated that immune dysregulation can be pathogenic for CVD, identifying a need for novel immunomodulatory therapeutic strategies. The nervous system, through an array of afferent and efferent arms of the autonomic nervous system, profoundly affects cardiovascular function. Interestingly, the autonomic nervous system also innervates immune organs, and neuroimmune interactions that are biologically relevant to CVD have been discovered, providing the foundation to target neural reflexes as an immunomodulatory therapeutic strategy. This Review summarizes how the neural regulation of immunity and inflammation participates in the onset and progression of CVD and explores promising opportunities for future therapeutic strategies.
Key points
-
The main routes of communication between the brain and the cardiovascular system are the brain circumventricular organs, the autonomic nervous system and the hypothalamus–pituitary–adrenal axis.
-
Immune organs are densely innervated by the peripheral nervous system.
-
The nervous system might contribute to cardiovascular disease either directly through innervation of the cardiovascular system or indirectly by influencing the immune responses involved in cardiovascular pathology.
-
Innovative approaches that are based on bioelectronic or ultrasound-driven modulation of immune organs are emerging options for the treatment of cardiovascular disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Joseph, P. et al. Reducing the global burden of cardiovascular disease, part 1: the epidemiology and risk factors. Circ. Res. 121, 677–694 (2017).
Leong, D. P. et al. Reducing the global burden of cardiovascular disease, part 2: prevention and treatment of cardiovascular disease. Circ. Res. 121, 695–710 (2017).
Tzoulaki, I., Elliott, P., Kontis, V. & Ezzati, M. Worldwide exposures to cardiovascular risk factors and associated health effects: current knowledge and data gaps. Circulation 133, 2314–2333 (2016).
Malpas, S. C. Sympathetic nervous system overactivity and its role in the development of cardiovascular disease. Physiol. Rev. 90, 513–557 (2010).
Elenkov, I. J., Wilder, R. L., Chrousos, G. P. & Vizi, E. S. The sympathetic nerve — an integrative interface between two supersystems: the brain and the immune system. Pharmacol. Rev. 52, 595–638 (2000).
Reardon, C., Murray, K. & Lomax, A. E. Neuroimmune communication in health and disease. Physiol. Rev. 98, 2287–2316 (2018).
Felten, D. L. et al. Noradrenergic sympathetic neural interactions with the immune system: structure and function. Immunol. Rev. 100, 225–260 (1987).
Bellinger, D. L., Lorton, D., Felten, S. Y. & Felten, D. L. Innervation of lymphoid organs and implications in development, aging, and autoimmunity. Int. J. Immunopharmacol. 14, 329–344 (1992).
Felten, D. L., Felten, S. Y., Carlson, S. L., Olschowka, J. A. & Livnat, S. Noradrenergic and peptidergic innervation of lymphoid tissue. J. Immunol. 135 (Suppl. 2), 755–765 (1985).
Felten, D. L. et al. Sympathetic innervation of lymph nodes in mice. Brain Res. Bull. 13, 693–699 (1984).
Tabarowski, Z., Gibson-Berry, K. & Felten, S. Y. Noradrenergic and peptidergic innervation of the mouse femur bone marrow. Acta Histochem. 98, 453–457 (1996).
Ballabh, P., Braun, A. & Nedergaard, M. The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol. Dis. 16, 1–13 (2004).
Banks, W. A. The blood-brain barrier in neuroimmunology: tales of separation and assimilation. Brain Behav. Immun. 44, 1–8 (2015).
Cancelliere, N. M., Black, E. A. & Ferguson, A. V. Neurohumoral integration of cardiovascular function by the lamina terminalis. Curr. Hypertens. Rep. 17, 93 (2015).
Johnson, A. K. & Gross, P. M. Sensory circumventricular organs and brain homeostatic pathways. FASEB J. 7, 678–686 (1993).
Siso, S., Jeffrey, M. & Gonzalez, L. Sensory circumventricular organs in health and disease. Acta Neuropathol. 120, 689–705 (2010).
Krause, E. G. et al. Angiotensin type 1 receptors in the subfornical organ mediate the drinking and hypothalamic-pituitary-adrenal response to systemic isoproterenol. Endocrinology 149, 6416–6424 (2008).
Lenkei, Z., Palkovits, M., Corvol, P. & Llorens-Cortes, C. Distribution of angiotensin type-1 receptor messenger RNA expression in the adult rat brain. Neuroscience 82, 827–841 (1998).
Frazier, C. J. et al. An angiotensin-responsive connection from the lamina terminalis to the paraventricular nucleus of the hypothalamus evokes vasopressin secretion to increase blood pressure in mice. J. Neurosci. 41, 1429–1442 (2021).
Johnson, A. K. & Xue, B. Central nervous system neuroplasticity and the sensitization of hypertension. Nat. Rev. Nephrol. 14, 750–766 (2018).
Li, Z. & Ferguson, A. V. Subfornical organ efferents to paraventricular nucleus utilize angiotensin as a neurotransmitter. Am. J. Physiol. 265, R302–R309 (1993).
Kawano, H. & Masuko, S. Region-specific projections from the subfornical organ to the paraventricular hypothalamic nucleus in the rat. Neuroscience 169, 1227–1234 (2010).
Stein, M. K. & Loewy, A. D. Area postrema projects to FoxP2 neurons of the pre-locus coeruleus and parabrachial nuclei: brainstem sites implicated in sodium appetite regulation. Brain Res. 1359, 116–127 (2010).
Guyenet, P. G. The sympathetic control of blood pressure. Nat. Rev. Neurosci. 7, 335–346 (2006).
Guyenet, P. G. et al. C1 neurons: the body’s EMTs. Am. J. Physiol. Regul. Integr. Comp. Physiol. 305, R187–R204 (2013).
D’Andrea, I. et al. Lack of kinase-independent activity of PI3Kγ in locus coeruleus induces ADHD symptoms through increased CREB signaling. EMBO Mol. Med. 7, 904–917 (2015).
Cook, A. D., Christensen, A. D., Tewari, D., McMahon, S. B. & Hamilton, J. A. Immune cytokines and their receptors in inflammatory pain. Trends Immunol. 39, 240–255 (2018).
Klose, C. S. N. & Veiga-Fernandes, H. Neuroimmune interactions in peripheral tissues. Eur. J. Immunol. 51, 1602–1614 (2021).
Huh, J. R. & Veiga-Fernandes, H. Neuroimmune circuits in inter-organ communication. Nat. Rev. Immunol. 20, 217–228 (2020).
Osborn, J. W. & Kuroki, M. T. Sympathetic signatures of cardiovascular disease: a blueprint for development of targeted sympathetic ablation therapies. Hypertension 59, 545–547 (2012).
Esler, M. The sympathetic nervous system through the ages: from Thomas Willis to resistant hypertension. Exp. Physiol. 96, 611–622 (2011).
Coffman, T. M. Under pressure: the search for the essential mechanisms of hypertension. Nat. Med. 17, 1402–1409 (2011).
Kingwell, B. A. et al. Heart rate spectral analysis, cardiac norepinephrine spillover, and muscle sympathetic nerve activity during human sympathetic nervous activation and failure. Circulation 90, 234–240 (1994).
Grassi, G., Colombo, M., Seravalle, G., Spaziani, D. & Mancia, G. Dissociation between muscle and skin sympathetic nerve activity in essential hypertension, obesity, and congestive heart failure. Hypertension 31, 64–67 (1998).
Esler, M. et al. Assessment of human sympathetic nervous system activity from measurements of norepinephrine turnover. Hypertension 11, 3–20 (1988).
Anderson, E. A., Sinkey, C. A., Lawton, W. J. & Mark, A. L. Elevated sympathetic nerve activity in borderline hypertensive humans. Evidence from direct intraneural recordings. Hypertension 14, 177–183 (1989).
Carnevale, D. et al. A cholinergic-sympathetic pathway primes immunity in hypertension and mediates brain-to-spleen communication. Nat. Commun. 7, 13035 (2016).
Carnevale, L. et al. Celiac vagus nerve stimulation recapitulates angiotensin II-induced splenic noradrenergic activation, driving egress of CD8 effector cells. Cell Rep. 33, 108494 (2020).
Kressel, A. M. et al. Identification of a brainstem locus that inhibits tumor necrosis factor. Proc. Natl Acad. Sci. USA 117, 29803–29810 (2020).
Butts, C. L. & Sternberg, E. M. Neuroendocrine factors alter host defense by modulating immune function. Cell Immunol. 252, 7–15 (2008).
Selye, H. Variations in organ size caused by chronic treatment with adrenal cortical compounds: an example of a dissociated adaptation to a hormone. J. Anat. 76, 94 (1941).
Marvar, P. J. & Harrison, D. G. Stress-dependent hypertension and the role of T lymphocytes. Exp. Physiol. 97, 1161–1167 (2012).
Foster, K. E. et al. EphB-ephrin-B2 interactions are required for thymus migration during organogenesis. Proc. Natl Acad. Sci. USA 107, 13414–13419 (2010).
Nance, D. M., Hopkins, D. A. & Bieger, D. Re-investigation of the innervation of the thymus gland in mice and rats. Brain Behav. Immun. 1, 134–147 (1987).
Bulloch, K., Hausman, J., Radojcic, T. & Short, S. Calcitonin gene-related peptide in the developing and aging thymus. An immunocytochemical study. Ann. NY Acad. Sci. 621, 218–228 (1991).
Tollefson, L. & Bulloch, K. Dual-label retrograde transport: CNS innervation of the mouse thymus distinct from other mediastinum viscera. J. Neurosci. Res. 25, 20–28 (1990).
Vizi, E. S., Orso, E., Osipenko, O. N., Hasko, G. & Elenkov, I. J. Neurochemical, electrophysiological and immunocytochemical evidence for a noradrenergic link between the sympathetic nervous system and thymocytes. Neuroscience 68, 1263–1276 (1995).
Trotter, R. N., Stornetta, R. L., Guyenet, P. G. & Roberts, M. R. Transneuronal mapping of the CNS network controlling sympathetic outflow to the rat thymus. Auton. Neurosci. 131, 9–20 (2007).
de Leeuw, F. E. et al. The neural and neuro-endocrine component of the human thymus. I. Nerve-like structures. Brain Behav. Immun. 6, 234–248 (1992).
Pilipovic, I. et al. Catecholaminergic signalling through thymic nerve fibres, thymocytes and stromal cells is dependent on both circulating and locally synthesized glucocorticoids. Exp. Physiol. 97, 1211–1223 (2012).
Katayama, Y. et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell 124, 407–421 (2006).
Gao, X. et al. Nociceptive nerves regulate haematopoietic stem cell mobilization. Nature 589, 591–596 (2021).
Denes, A. et al. Central autonomic control of the bone marrow: multisynaptic tract tracing by recombinant pseudorabies virus. Neuroscience 134, 947–963 (2005).
Bajayo, A. et al. Skeletal parasympathetic innervation communicates central IL-1 signals regulating bone mass accrual. Proc. Natl Acad. Sci. USA 109, 15455–15460 (2012).
Jung, W. C., Levesque, J. P. & Ruitenberg, M. J. It takes nerve to fight back: the significance of neural innervation of the bone marrow and spleen for immune function. Semin. Cell Dev. Biol. 61, 60–70 (2017).
Maestroni, G. J. Catecholaminergic regulation of hematopoiesis in mice. Blood 92, 2971 (1998).
Mendez-Ferrer, S., Lucas, D., Battista, M. & Frenette, P. S. Haematopoietic stem cell release is regulated by circadian oscillations. Nature 452, 442–447 (2008).
Scheiermann, C. et al. Adrenergic nerves govern circadian leukocyte recruitment to tissues. Immunity 37, 290–301 (2012).
Fink, T. & Weihe, E. Multiple neuropeptides in nerves supplying mammalian lymph nodes: messenger candidates for sensory and autonomic neuroimmunomodulation? Neurosci. Lett. 90, 39–44 (1988).
Novotny, G. E. & Kliche, K. O. Innervation of lymph nodes: a combined silver impregnation and electron-microscopic study. Acta Anat. 127, 243–248 (1986).
Sloan, E. K. et al. Social stress enhances sympathetic innervation of primate lymph nodes: mechanisms and implications for viral pathogenesis. J. Neurosci. 27, 8857–8865 (2007).
Druzd, D. et al. Lymphocyte circadian clocks control lymph node trafficking and adaptive immune responses. Immunity 46, 120–132 (2017).
Suzuki, K., Hayano, Y., Nakai, A., Furuta, F. & Noda, M. Adrenergic control of the adaptive immune response by diurnal lymphocyte recirculation through lymph nodes. J. Exp. Med. 213, 2567–2574 (2016).
Nakai, A., Hayano, Y., Furuta, F., Noda, M. & Suzuki, K. Control of lymphocyte egress from lymph nodes through beta2-adrenergic receptors. J. Exp. Med. 211, 2583–2598 (2014).
Huang, S. et al. Lymph nodes are innervated by a unique population of sensory neurons with immunomodulatory potential. Cell 184, 441–459.e25 (2021).
Rosas-Ballina, M. et al. Acetylcholine-synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334, 98–101 (2011).
Vida, G., Pena, G., Deitch, E. A. & Ulloa, L. alpha7-cholinergic receptor mediates vagal induction of splenic norepinephrine. J. Immunol. 186, 4340–4346 (2011).
Carnevale, D. et al. The angiogenic factor PlGF mediates a neuroimmune interaction in the spleen to allow the onset of hypertension. Immunity 41, 737–752 (2014).
Bellinger, D. L., Felten, S. Y., Lorton, D. & Felten, D. L. Origin of noradrenergic innervation of the spleen in rats. Brain Behav. Immun. 3, 291–311 (1989).
Ackerman, K., Felten, S., Bellinger, D. & Felten, D. Noradrenergic sympathetic innervation of the spleen: III. Development of innervation in the rat spleen. J. Neurosci. Res. 18, 49–54 (1987).
Bellinger, D. L., Felten, S. Y., Collier, T. J. & Felten, D. L. Noradrenergic sympathetic innervation of the spleen: IV. Morphometric analysis in adult and aged F344 rats. J. Neurosci. Res. 18, 55–63 (1987).
Felten, S. Y. & Olschowka, J. Noradrenergic sympathetic innervation of the spleen: II. Tyrosine hydroxylase (TH)-positive nerve terminals form synapticlike contacts on lymphocytes in the splenic white pulp. J. Neurosci. Res. 18, 37–48 (1987).
Williams, J. M. & Felten, D. L. Sympathetic innervation of murine thymus and spleen: a comparative histofluorescence study. Anat. Rec. 199, 531–542 (1981).
Nance, D. M. & Sanders, V. M. Autonomic innervation and regulation of the immune system (1987-2007). Brain Behav. Immun. 21, 736–745 (2007).
Straub, R. H. Complexity of the bi-directional neuroimmune junction in the spleen. Trends Pharmacol. Sci. 25, 640–646 (2004).
Wang, H. et al. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature 421, 384–388 (2003).
Ding, X. et al. Panicle-shaped sympathetic architecture in the spleen parenchyma modulates antibacterial innate immunity. Cell Rep. 27, 3799–3807.e3 (2019).
Lucas, T. A., Zhu, L. & Buckwalter, M. S. Spleen glia are a transcriptionally unique glial subtype interposed between immune cells and sympathetic axons. Glia 69, 1799–1815 (2021).
Griffin, J. W. & Thompson, W. J. Biology and pathology of nonmyelinating Schwann cells. Glia 56, 1518–1531 (2008).
Cano, G., Sved, A. F., Rinaman, L., Rabin, B. S. & Card, J. P. Characterization of the central nervous system innervation of the rat spleen using viral transneuronal tracing. J. Comp. Neurol. 439, 1–18 (2001).
Randall, T. D. & Mebius, R. E. The development and function of mucosal lymphoid tissues: a balancing act with micro-organisms. Mucosal Immunol. 7, 455–466 (2014).
Carragher, D. M., Rangel-Moreno, J. & Randall, T. D. Ectopic lymphoid tissues and local immunity. Semin. Immunol. 20, 26–42 (2008).
Bienenstock, J., Croitoru, K., Ernst, P. B. & Stanisz, A. M. Nerves and neuropeptides in the regulation of mucosal immunity. Adv. Exp. Med. Biol. 257, 19–26 (1989).
Basbaum, A. I., Bautista, D. M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Blackshaw, L. A. & Gebhart, G. F. The pharmacology of gastrointestinal nociceptive pathways. Curr. Opin. Pharmacol. 2, 642–649 (2002).
Lai, N. Y. et al. Gut-innervating nociceptor neurons regulate peyer’s patch microfold cells and SFB levels to mediate salmonella host defense. Cell 180, 33–49.e22 (2020).
Kratz, A., Campos-Neto, A., Hanson, M. S. & Ruddle, N. H. Chronic inflammation caused by lymphotoxin is lymphoid neogenesis. J. Exp. Med. 183, 1461–1472 (1996).
Ghosh, S., Steere, A. C., Stollar, B. D. & Huber, B. T. In situ diversification of the antibody repertoire in chronic Lyme arthritis synovium. J. Immunol. 174, 2860–2869 (2005).
Rupprecht, T. A. et al. The chemokine CXCL13 (BLC): a putative diagnostic marker for neuroborreliosis. Neurology 65, 448–450 (2005).
Mazzucchelli, L. et al. BCA-1 is highly expressed in Helicobacter pylori-induced mucosa-associated lymphoid tissue and gastric lymphoma. J. Clin. Invest. 104, R49–R54 (1999).
Baddoura, F. K. et al. Lymphoid neogenesis in murine cardiac allografts undergoing chronic rejection. Am. J. Transpl. 5, 510–516 (2005).
Thaunat, O. et al. Lymphoid neogenesis in chronic rejection: evidence for a local humoral alloimmune response. Proc. Natl Acad. Sci. USA 102, 14723–14728 (2005).
Andersson, A. et al. Spatial deconvolution of HER2-positive breast cancer delineates tumor-associated cell type interactions. Nat. Commun. 12, 6012 (2021).
Rodriguez, A. B. et al. Immune mechanisms orchestrate tertiary lymphoid structures in tumors via cancer-associated fibroblasts. Cell Rep. 36, 109422 (2021).
Houtkamp, M. A., de Boer, O. J., van der Loos, C. M., van der Wal, A. C. & Becker, A. E. Adventitial infiltrates associated with advanced atherosclerotic plaques: structural organization suggests generation of local humoral immune responses. J. Pathol. 193, 263–269 (2001).
Lotzer, K. et al. Mouse aorta smooth muscle cells differentiate into lymphoid tissue organizer-like cells on combined tumor necrosis factor receptor-1/lymphotoxin beta-receptor NF-kappaB signaling. Arterioscler. Thromb. Vasc. Biol. 30, 395–402 (2010).
Mohanta, S. K. et al. Artery tertiary lymphoid organs contribute to innate and adaptive immune responses in advanced mouse atherosclerosis. Circ. Res. 114, 1772–1787 (2014).
Hu, D. et al. Artery tertiary lymphoid organs control aorta immunity and protect against atherosclerosis via vascular smooth muscle cell lymphotoxin beta receptors. Immunity 42, 1100–1115 (2015).
Drayton, D. L., Liao, S., Mounzer, R. H. & Ruddle, N. H. Lymphoid organ development: from ontogeny to neogenesis. Nat. Immunol. 7, 344–353 (2006).
Olivier, B. J. et al. Vagal innervation is required for the formation of tertiary lymphoid tissue in colitis. Eur. J. Immunol. 46, 2467–2480 (2016).
Borovikova, L. V. et al. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 405, 458–462 (2000).
Chavan, S. S., Pavlov, V. A. & Tracey, K. J. Mechanisms and therapeutic relevance of neuro-immune communication. Immunity 46, 927–942 (2017).
Ebringer, A. & Doyle, A. E. Raised serum IgG levels in hypertension. Br. Med. J. 2, 146–148 (1970).
Hilme, E., Herlitz, H., Soderstrom, T. & Hansson, L. Increased secretion of immunoglobulins in malignant hypertension. J. Hypertens. 7, 91–95 (1989).
Suryaprabha, P., Padma, T. & Rao, U. B. Increased serum IgG levels in essential hypertension. Immunol. Lett. 8, 143–145 (1984).
Guzik, T. J. et al. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J. Exp. Med. 204, 2449–2460 (2007).
Seniuk, A. et al. B6.Rag1 knockout mice generated at the Jackson laboratory in 2009 show a robust wild-type hypertensive phenotype in response to Ang II (Angiotensin II). Hypertension 75, 1110–1116 (2020).
Ji, H. et al. Loss of resistance to angiotensin II-induced hypertension in the jackson laboratory recombination-activating gene null mouse on the C57BL/6J background. Hypertension 69, 1121–1127 (2017).
Drummond, G. R., Vinh, A., Guzik, T. J. & Sobey, C. G. Immune mechanisms of hypertension. Nat. Rev. Immunol. 19, 517–532 (2019).
Trott, D. W. et al. Oligoclonal CD8+ T cells play a critical role in the development of hypertension. Hypertension 64, 1108–1115 (2014).
Abboud, F. M. The Walter B. Cannon Memorial Award Lecture, 2009. Physiology in perspective: The wisdom of the body. In search of autonomic balance: the good, the bad, and the ugly. Am. J. Physiol. Regul. Integr. Comp. Physiol. 298, R1449–R1467 (2010).
Abboud, F. M., Harwani, S. C. & Chapleau, M. W. Autonomic neural regulation of the immune system: implications for hypertension and cardiovascular disease. Hypertension 59, 755–762 (2012).
Ganta, C. K. et al. Central angiotensin II-enhanced splenic cytokine gene expression is mediated by the sympathetic nervous system. Am. J. Physiol. Heart Circ. Physiol. 289, H1683–H1691 (2005).
Marvar, P. J. et al. Central and peripheral mechanisms of T-lymphocyte activation and vascular inflammation produced by angiotensin II-induced hypertension. Circ. Res. 107, 263–270 (2010).
Perrotta, M. et al. Deoxycorticosterone acetate-salt hypertension activates placental growth factor in the spleen to couple sympathetic drive and immune system activation. Cardiovasc. Res. 114, 456–467 (2018).
Ahmari, N. et al. Elevated bone marrow sympathetic drive precedes systemic inflammation in angiotensin II hypertension. Am. J. Physiol. Heart Circ. Physiol. 317, H279–H289 (2019).
Wilck, N. et al. Salt-responsive gut commensal modulates TH17 axis and disease. Nature 551, 585–589 (2017).
Faraco, G. et al. Dietary salt promotes neurovascular and cognitive dysfunction through a gut-initiated TH17 response. Nat. Neurosci. 21, 240–249 (2018).
van Gils, J. M. et al. The neuroimmune guidance cue netrin-1 promotes atherosclerosis by inhibiting the emigration of macrophages from plaques. Nat. Immunol. 13, 136–143 (2012).
Johansson, M. E. et al. α7 Nicotinic acetylcholine receptor is expressed in human atherosclerosis and inhibits disease in mice — brief report. Arterioscler. Thromb. Vasc. Biol. 34, 2632–2636 (2014).
Christoffersson, G., Ratliff, S. S. & von Herrath, M. G. Interference with pancreatic sympathetic signaling halts the onset of diabetes in mice. Sci. Adv. 6, eabb2878 (2020).
Ramkhelawon, B. et al. Netrin-1 promotes adipose tissue macrophage retention and insulin resistance in obesity. Nat. Med. 20, 377–384 (2014).
Carnevale, D. & Lembo, G. Neuroimmune interactions in cardiovascular diseases. Cardiovasc. Res. 117, 402–410 (2021).
Vasamsetti, S. B. et al. Sympathetic neuronal activation triggers myeloid progenitor proliferation and differentiation. Immunity 49, 93–106.e7 (2018).
Cardoso, F. et al. Neuro-mesenchymal units control ILC2 and obesity via a brain-adipose circuit. Nature 597, 410–414 (2021).
Lambert, E. et al. Association between the sympathetic firing pattern and anxiety level in patients with the metabolic syndrome and elevated blood pressure. J. Hypertens. 28, 543–550 (2010).
Lambert, E. A. & Lambert, G. W. Stress and its role in sympathetic nervous system activation in hypertension and the metabolic syndrome. Curr. Hypertens. Rep. 13, 244–248 (2011).
Marvar, P. J. et al. T lymphocytes and vascular inflammation contribute to stress-dependent hypertension. Biol. Psychiatry 71, 774–782 (2012).
Heidt, T. et al. Chronic variable stress activates hematopoietic stem cells. Nat. Med. 20, 754–758 (2014).
McAlpine, C. S. et al. Sleep modulates haematopoiesis and protects against atherosclerosis. Nature 566, 383–387 (2019).
Arsenault, B. J. & Despres, J. P. Cardiovascular disease prevention: lifestyle attenuation of genetic risk. Nat. Rev. Cardiol. 14, 187–188 (2017).
Yusuf, S. et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet 364, 937–952 (2004).
Fiuza-Luces, C. et al. Exercise benefits in cardiovascular disease: beyond attenuation of traditional risk factors. Nat. Rev. Cardiol. 15, 731–743 (2018).
Frodermann, V. et al. Exercise reduces inflammatory cell production and cardiovascular inflammation via instruction of hematopoietic progenitor cells. Nat. Med. 25, 1761–1771 (2019).
Lymperopoulos, A., Rengo, G. & Koch, W. J. Adrenergic nervous system in heart failure: pathophysiology and therapy. Circ. Res. 113, 739–753 (2013).
Triposkiadis, F. et al. The sympathetic nervous system in heart failure physiology, pathophysiology, and clinical implications. J. Am. Coll. Cardiol. 54, 1747–1762 (2009).
Cohn, J. N. et al. Plasma norepinephrine as a guide to prognosis in patients with chronic congestive heart failure. N. Engl. J. Med. 311, 819–823 (1984).
Nahrendorf, M. Myeloid cell contributions to cardiovascular health and disease. Nat. Med. 24, 711–720 (2018).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Ismahil, M. A. et al. Remodeling of the mononuclear phagocyte network underlies chronic inflammation and disease progression in heart failure: critical importance of the cardiosplenic axis. Circ. Res. 114, 266–282 (2014).
Hulsmans, M. et al. Cardiac macrophages promote diastolic dysfunction. J. Exp. Med. 215, 423–440 (2018).
Fujiu, K. et al. A heart-brain-kidney network controls adaptation to cardiac stress through tissue macrophage activation. Nat. Med. 23, 611–622 (2017).
Epelman, S. et al. Embryonic and adult-derived resident cardiac macrophages are maintained through distinct mechanisms at steady state and during inflammation. Immunity 40, 91–104 (2014).
Zaman, R. et al. Selective loss of resident macrophage-derived insulin-like growth factor-1 abolishes adaptive cardiac growth to stress. Immunity 54, 2057–2071.e6 (2021).
Wong, N. R. et al. Resident cardiac macrophages mediate adaptive myocardial remodeling. Immunity 54, 2072–2088.e7 (2021).
Coote, J. H. & Chauhan, R. A. The sympathetic innervation of the heart: important new insights. Auton. Neurosci. 199, 17–23 (2016).
Ziegler, K. A. et al. Local sympathetic denervation attenuates myocardial inflammation and improves cardiac function after myocardial infarction in mice. Cardiovasc. Res. 114, 291–299 (2018).
DiBona, G. F. & Kopp, U. C. Neural control of renal function. Physiol. Rev. 77, 75–197 (1997).
Gupta, J. et al. Association between albuminuria, kidney function, and inflammatory biomarker profile in CKD in CRIC. Clin. J. Am. Soc. Nephrol. 7, 1938–1946 (2012).
Ronco, C., Bellomo, R. & Kellum, J. A. Acute kidney injury. Lancet 394, 1949–1964 (2019).
Griffin, K. A. Hypertensive kidney injury and the progression of chronic kidney disease. Hypertension 70, 687–694 (2017).
Go, A. S., Chertow, G. M., Fan, D., McCulloch, C. E. & Hsu, C. Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 351, 1296–1305 (2004).
Schlaich, M. P. Sympathetic activation in chronic kidney disease: out of the shadow. Hypertension 57, 683–685 (2011).
Yeboah, M. M. et al. Cholinergic agonists attenuate renal ischemia-reperfusion injury in rats. Kidney Int. 74, 62–69 (2008).
Chatterjee, P. K. et al. Nicotinic acetylcholine receptor agonists attenuate septic acute kidney injury in mice by suppressing inflammation and proteasome activity. PLoS ONE 7, e35361 (2012).
Gigliotti, J. C. et al. Ultrasound prevents renal ischemia-reperfusion injury by stimulating the splenic cholinergic anti-inflammatory pathway. J. Am. Soc. Nephrol. 24, 1451–1460 (2013).
Tanaka, S. et al. Vagus nerve stimulation activates two distinct neuroimmune circuits converging in the spleen to protect mice from kidney injury. Proc. Natl Acad. Sci. USA 118, e2021758118 (2021).
Xiao, L. et al. Renal denervation prevents immune cell activation and renal inflammation in angiotensin II-induced hypertension. Circ. Res. 117, 547–557 (2015).
Banek, C. T. et al. Resting afferent renal nerve discharge and renal inflammation: elucidating the role of afferent and efferent renal nerves in deoxycorticosterone acetate salt hypertension. Hypertension 68, 1415–1423 (2016).
Banek, C. T. et al. Targeted afferent renal denervation reduces arterial pressure but not renal inflammation in established DOCA-salt hypertension in the rat. Am. J. Physiol. Regul. Integr. Comp. Physiol. 314, R883–R891 (2018).
Banek, C. T., Gauthier, M. M., Van Helden, D. A., Fink, G. D. & Osborn, J. W. Renal inflammation in DOCA-salt hypertension. Hypertension 73, 1079–1086 (2019).
Veelken, R. et al. Autonomic renal denervation ameliorates experimental glomerulonephritis. J. Am. Soc. Nephrol. 19, 1371–1378 (2008).
Kim, J. & Padanilam, B. J. Renal nerves drive interstitial fibrogenesis in obstructive nephropathy. J. Am. Soc. Nephrol. 24, 229–242 (2013).
Iadecola, C. The pathobiology of vascular dementia. Neuron 80, 844–866 (2013).
Nation, D. A. et al. Blood-brain barrier breakdown is an early biomarker of human cognitive dysfunction. Nat. Med. 25, 270–276 (2019).
Carnevale, L. et al. Brain MRI fiber-tracking reveals white matter alterations in hypertensive patients without damage at conventional neuroimaging. Cardiovasc. Res. 114, 1536–1546 (2018).
Carnevale, D. et al. Hypertension induces brain beta-amyloid accumulation, cognitive impairment, and memory deterioration through activation of receptor for advanced glycation end products in brain vasculature. Hypertension 60, 188–197 (2012).
Toth, P. et al. Age-related autoregulatory dysfunction and cerebromicrovascular injury in mice with angiotensin II-induced hypertension. J. Cereb. Blood Flow Metab. 33, 1732–1742 (2013).
Faraco, G. et al. Perivascular macrophages mediate the neurovascular and cognitive dysfunction associated with hypertension. J. Clin. Invest. 126, 4674–4689 (2016).
Santisteban, M. M. et al. Endothelium-macrophage crosstalk mediates blood-brain barrier dysfunction in hypertension. Hypertension 76, 795–807 (2020).
Faraco, G. et al. Dietary salt promotes cognitive impairment through tau phosphorylation. Nature 574, 686–690 (2019).
Carnevale, L., Perrotta, M. & Lembo, G. A focused review of neural recording and stimulation techniques with immune-modulatory targets. Front. Immunol. 12, 689344 (2021).
Pavlov, V. A., Chavan, S. S. & Tracey, K. J. Bioelectronic medicine: from preclinical studies on the inflammatory reflex to new approaches in disease diagnosis and treatment. Cold Spring Harb. Perspect. Med. 10, a034140 (2020).
Martelli, D., Farmer, D. G. S., McKinley, M. J., Yao, S. T. & McAllen, R. M. Anti-inflammatory reflex action of splanchnic sympathetic nerves is distributed across abdominal organs. Am. J. Physiol. Regul. Integr. Comp. Physiol. 316, R235–R242 (2019).
Martelli, D., McKinley, M. J. & McAllen, R. M. The cholinergic anti-inflammatory pathway: a critical review. Auton. Neurosci. 182, 65–69 (2014).
Martelli, D., Yao, S. T., McKinley, M. J. & McAllen, R. M. Reflex control of inflammation by sympathetic nerves, not the vagus. J. Physiol. 592, 1677–1686 (2014).
Donega, M. et al. Human-relevant near-organ neuromodulation of the immune system via the splenic nerve. Proc. Natl Acad. Sci. USA 118, e2025428118 (2021).
Cotero, V. et al. Noninvasive sub-organ ultrasound stimulation for targeted neuromodulation. Nat. Commun. 10, 952 (2019).
Zachs, D. P. et al. Noninvasive ultrasound stimulation of the spleen to treat inflammatory arthritis. Nat. Commun. 10, 951 (2019).
Ulloa, L., Quiroz-Gonzalez, S. & Torres-Rosas, R. Nerve stimulation: immunomodulation and control of inflammation. Trends Mol. Med. 23, 1103–1120 (2017).
Liu, S. et al. A neuroanatomical basis for electroacupuncture to drive the vagal-adrenal axis. Nature 598, 641–645 (2021).
Yeboah, M. et al. Cholinergic agonists attenuate renal ischemia–reperfusion injury in rats. Kidney Int. 74, 62–69 (2008).
Gigliotti, J. C. et al. Ultrasound modulates the splenic neuroimmune axis in attenuating AKI. J. Am. Soc. Nephrol. 26, 2470–2481 (2015).
Schmued, L. C. & Fallon, J. H. Fluoro-Gold: a new fluorescent retrograde axonal tracer with numerous unique properties. Brain Res. 377, 147–154 (1986).
Glover, J. C., Petursdottir, G. & Jansen, J. K. Fluorescent dextran-amines used as axonal tracers in the nervous system of the chicken embryo. J. Neurosci. Methods 18, 243–254 (1986).
Lanciego, J. L. et al. Complex brain circuits studied via simultaneous and permanent detection of three transported neuroanatomical tracers in the same histological section. J. Neurosci. Methods 103, 127–135 (2000).
Aston-Jones, G. & Card, J. P. Use of pseudorabies virus to delineate multisynaptic circuits in brain: opportunities and limitations. J. Neurosci. Methods 103, 51–61 (2000).
Loewy, A. D. Viruses as transneuronal tracers for defining neural circuits. Neurosci. Biobehav. Rev. 22, 679–684 (1998).
Jansen, A. S. & Loewy, A. D. Viral tracing of innervation. Science 265, 121–122 (1994).
Strack, A. M. & Loewy, A. D. Pseudorabies virus: a highly specific transneuronal cell body marker in the sympathetic nervous system. J. Neurosci. 10, 2139–2147 (1990).
Acknowledgements
Work in the author’s laboratory is supported by the European Research Council (ERC-StG-GA759921), the European ERA-CVD Joint Transnational Call 2018 (NEMO-IMMUNEagainstHF) and 2019 (Gut-Brain-Immune-HHD), the Italian Ministry of University and Research (FARE-MIUR-R18RLRPS2R), and the Italian Ministry of Health (Ricerca Corrente). The author thanks G. Lembo, M. Perrotta and L. Carnevale from the laboratory for helpful discussion and inspiration for preparing the figures before submission.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks John Osborn, Janine van Gils and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Angiotensin II
-
A biologically generated peptide hormone of the renin–angiotensin–aldosterone system, with potent vasoconstrictor effects and signalling functions, also used to induce hypertension in experimental models.
- Optogenetics
-
A technology that uses light to control and sense neurons that have been genetically modified to express channelrhodopsin 2, a light-gated cation channel.
- Deoxycorticosterone acetate
-
(DOCA). A mineralocorticoid that is used together with salt in drinking water in a well-established preclinical model to recapitulate moderate-to-severe, salt-sensitive hypertension.
- 6-Hydroxydopamine
-
A hydroxylated analogue of dopamine used as a neurotoxic synthetic organic compound that can selectively destroy dopaminergic and noradrenergic neurons in the brain.
Rights and permissions
About this article
Cite this article
Carnevale, D. Neuroimmune axis of cardiovascular control: mechanisms and therapeutic implications. Nat Rev Cardiol 19, 379–394 (2022). https://doi.org/10.1038/s41569-022-00678-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-022-00678-w
This article is cited by
-
Immune and inflammatory mechanisms in hypertension
Nature Reviews Cardiology (2024)
-
Neuroimmunology of Cardiovascular Disease
Current Hypertension Reports (2024)
-
Heart disease causes sleep disturbances via neuroimmune mechanisms
Nature Reviews Cardiology (2023)
-
Role of Inflammatory Processes in the Brain-Body Relationship Underlying Hypertension
Current Hypertension Reports (2023)
-
Targeted therapies for cardiac diseases
Nature Reviews Cardiology (2022)