Abstract
Accumulating evidence points to the causal role of triglyceride-rich lipoproteins and their cholesterol-enriched remnants in atherogenesis. Genetic studies in particular have not only revealed a relationship between plasma triglyceride levels and the risk of atherosclerotic cardiovascular disease, but have also identified key proteins responsible for the regulation of triglyceride transport. Kinetic studies in humans using stable isotope tracers have been especially useful in delineating the function of these proteins and revealing the hitherto unappreciated complexity of triglyceride-rich lipoprotein metabolism. Given that triglyceride is an essential energy source for mammals, triglyceride transport is regulated by numerous mechanisms that balance availability with the energy demands of the body. Ongoing investigations are focused on determining the consequences of dysregulation as a result of either dietary imprudence or genetic variation that increases the risk of atherosclerosis and pancreatitis. The identification of molecular control mechanisms involved in triglyceride metabolism has laid the groundwork for a ‘precision-medicine’ approach to therapy. Novel pharmacological agents under development have specific molecular targets within a regulatory framework, and their deployment heralds a new era in lipid-lowering-mediated prevention of disease. In this Review, we outline what is known about the dysregulation of triglyceride transport in human hypertriglyceridaemia.
Key points
-
Triglyceride-rich lipoproteins (TRLs), in particular, their cholesterol-rich remnants, are now considered causal agents for atherogenesis and a suitable target for diet-based and drug-based interventions to prevent coronary heart disease.
-
The optimal level of plasma triglycerides is <1.2 mmol/l; plasma triglyceride levels >1.2 mmol/l are associated with increasing risk of atherosclerosis and pancreatitis (the latter in particular when plasma triglyceride levels are >10 mmol/l).
-
Highly regulated metabolic pathways control the release of TRLs in their tissues of origin (the intestine and liver), lipolysis and remodeling in the bloodstream, and clearance of remnant particles after the core triglycerides have been delivered to their destination is normally rapid and is facilitated by hepatic receptors.
-
Given that current treatments are inadequate in reducing triglycerides to optimal levels, novel treatments under development focus on increasing the efficiency of lipolysis and finding an approach to regulate plasma levels of TRLs and their remnants.
-
Large-scale outcome trials will be required to test the hypothesis that specifically lowering TRLs and remnants can reduce the risk of cardiovascular disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ginsberg, H. N. et al. Triglyceride-rich lipoproteins and their remnants: metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies-a consensus statement from the European Atherosclerosis Society. Eur. Heart J. 42, 4791–4806 (2021).
Emerging Risk Factors, C. et al. Major lipids, apolipoproteins, and risk of vascular disease. JAMA 302, 1993–2000 (2009).
Chapman, M. J. et al. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur. Heart J. 32, 1345–1361 (2011).
Austin, M. A. Plasma triglyceride and coronary heart disease. Arterioscler. Thromb. 11, 2–14 (1991).
Tall, A. R. HDL in morbidity and mortality: a 40+ year perspective. Clin. Chem. 67, 19–23 (2021).
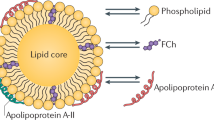
Olofsson, S. O. & Boren, J. Apolipoprotein B: a clinically important apolipoprotein which assembles atherogenic lipoproteins and promotes the development of atherosclerosis. J. Intern. Med. 258, 395–410 (2005).
Boren, J. et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 41, 2313–2330 (2020).
Davidson, N. O. & Shelness, G. S. APOLIPOPROTEIN B: mRNA editing, lipoprotein assembly, and presecretory degradation. Annu. Rev. Nutr. 20, 169–193 (2000).
Anant, S. & Davidson, N. O. Molecular mechanisms of apolipoprotein B mRNA editing. Curr. Opin. Lipidol. 12, 159–165 (2001).
Wang, A. B., Liu, D. P. & Liang, C. C. Regulation of human apolipoprotein B gene expression at multiple levels. Exp. Cell Res. 290, 1–12 (2003).
Chan, L. et al. From editosome to proteasome. Recent. Prog. Horm. Res. 55, 93–125 (2000). discussion 126.
Barrows, B. R. & Parks, E. J. Contributions of different fatty acid sources to very low-density lipoprotein-triacylglycerol in the fasted and fed states. J. Clin. Endocrinol. Metab. 91, 1446–1452 (2006).
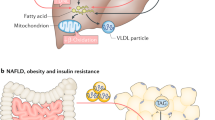
Donnelly, K. L. et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J. Clin. Invest. 115, 1343–1351 (2005).
Smith, G. I. et al. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J. Clin. Invest. 130, 1453–1460 (2020).
Browning, J. D. & Horton, J. D. Molecular mediators of hepatic steatosis and liver injury. J. Clin. Invest. 114, 147–152 (2004).
Koo, S. H., Dutcher, A. K. & Towle, H. C. Glucose and insulin function through two distinct transcription factors to stimulate expression of lipogenic enzyme genes in liver. J. Biol. Chem. 276, 9437–9445 (2001).
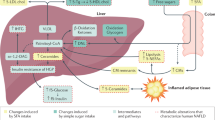
Deprince, A., Haas, J. T. & Staels, B. Dysregulated lipid metabolism links NAFLD to cardiovascular disease. Mol. Metab. 42, 101092 (2020).
Fisher, E. A. & Ginsberg, H. N. Complexity in the secretory pathway: the assembly and secretion of apolipoprotein B-containing lipoproteins. J. Biol. Chem. 277, 17377–17380 (2002).
Boren, J. et al. The assembly and secretion of ApoB 100-containing lipoproteins in Hep G2 cells. ApoB 100 is cotranslationally integrated into lipoproteins. J. Biol. Chem. 267, 9858–9867 (1992).
Gordon, D. A., Jamil, H., Gregg, R. E., Olofsson, S. O. & Boren, J. Inhibition of the microsomal triglyceride transfer protein blocks the first step of apolipoprotein B lipoprotein assembly but not the addition of bulk core lipids in the second step. J. Biol. Chem. 271, 33047–33053 (1996).
Olofsson, S. O. & Boren, J. Apolipoprotein B secretory regulation by degradation. Arterioscler. Thromb. Vasc. Biol. 32, 1334–1338 (2012).
Ginsberg, H. N. & Fisher, E. A. The ever-expanding role of degradation in the regulation of apolipoprotein B metabolism. J. Lipid Res. 50, S162–S166 (2009).
Stillemark, P. et al. The assembly and secretion of apolipoprotein B-48-containing very low density lipoproteins in McA-RH7777 cells. J. Biol. Chem. 275, 10506–10513 (2000).
Wang, X. et al. Receptor-mediated ER export of lipoproteins controls lipid homeostasis in mice and humans. Cell Metab. 33, 350–-366 e357 (2021).
Rustaeus, S., Stillemark, P., Lindberg, K., Gordon, D. & Olofsson, S. O. The microsomal triglyceride transfer protein catalyzes the post-translational assembly of apolipoprotein B-100 very low density lipoprotein in McA-RH7777 cells. J. Biol. Chem. 273, 5196–5203 (1998).
Adiels, M. et al. Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia 49, 755–765 (2006).
Boren, J. et al. Influence of triacylglycerol biosynthesis rate on the assembly of apoB-100-containing lipoproteins in Hep G2 cells. Arterioscler. Thromb. 13, 1743–1754 (1993).
Packard, C. J., Boren, J. & Taskinen, M. R. Causes and consequences of hypertriglyceridemia. Front. Endocrinol. 11, 252 (2020).
Malmstrom, R. et al. Metabolic basis of hypotriglyceridemic effects of insulin in normal men. Arterioscler. Thromb. Vasc. Biol. 17, 1454–1464 (1997).
Lewis, G. F. et al. VLDL production is decreased to a similar extent by acute portal vs. peripheral venous insulin. Am. J. Physiol. 267, E566–E572 (1994).
Cummings, M. H. et al. Acute hyperinsulinemia decreases the hepatic secretion of very-low-density lipoprotein apolipoprotein B-100 in NIDDM. Diabetes 44, 1059–1065 (1995).
Malmstrom, R. et al. Effects of insulin and acipimox on VLDL1 and VLDL2 apolipoprotein B production in normal subjects. Diabetes 47, 779–787 (1998).
Lewis, G. F., Uffelman, K. D., Szeto, L. W., Weller, B. & Steiner, G. Interaction between free fatty acids and insulin in the acute control of very low density lipoprotein production in humans. J. Clin. Invest. 95, 158–166 (1995).
Adiels, M. et al. Acute suppression of VLDL1 secretion rate by insulin is associated with hepatic fat content and insulin resistance. Diabetologia 50, 2356–2365 (2007).
Pavlic, M., Xiao, C., Szeto, L., Patterson, B. W. & Lewis, G. F. Insulin acutely inhibits intestinal lipoprotein secretion in humans in part by suppressing plasma free fatty acids. Diabetes 59, 580–587 (2010).
Loomba, R., Friedman, S. L. & Shulman, G. I. Mechanisms and disease consequences of nonalcoholic fatty liver disease. Cell 184, 2537–2564 (2021).
Xiao, C., Pavlic, M., Szeto, L., Patterson, B. W. & Lewis, G. F. Effects of acute hyperglucagonemia on hepatic and intestinal lipoprotein production and clearance in healthy humans. Diabetes 60, 383–390 (2011).
Nielsen, S. & Karpe, F. Determinants of VLDL-triglycerides production. Curr. Opin. Lipidol. 23, 321–326 (2012).
Palmisano, B. T., Zhu, L. & Stafford, J. M. Role of estrogens in the regulation of liver lipid metabolism. Adv. Exp. Med. Biol. 1043, 227–256 (2017).
Fielding, B. A. et al. Ethanol with a mixed meal increases postprandial triacylglycerol but decreases postprandial non-esterified fatty acid concentrations. Br. J. Nutr. 83, 597–604 (2000).
Brinton, E. A. Effects of ethanol intake on lipoproteins and atherosclerosis. Curr. Opin. Lipidol. 21, 346–351 (2010).
Taskinen, M. R. et al. Alcohol-induced changes in serum lipoproteins and in their metabolism. Am. Heart J. 113, 458–464 (1987).
Prinsen, B. H. et al. Endogenous cholesterol synthesis is associated with VLDL-2 apoB-100 production in healthy humans. J. Lipid Res. 44, 1341–1348 (2003).
Gaw, A. et al. Overproduction of small very low density lipoproteins (Sf 20-60) in moderate hypercholesterolemia: relationships between apolipoprotein B kinetics and plasma lipoproteins. J. Lipid Res. 36, 158–171 (1995).
James, R. W. et al. Apolipoprotein B metabolism in homozygous familial hypercholesterolemia. J. Lipid Res. 30, 159–169 (1989).
Xiao, C., Stahel, P. & Lewis, G. F. Regulation of chylomicron secretion: focus on post-assembly mechanisms. Cell Mol. Gastroenterol. Hepatol. 7, 487–501 (2019).
Hussain, M. M., Fatma, S., Pan, X. & Iqbal, J. Intestinal lipoprotein assembly. Curr. Opin. Lipidol. 16, 281–285 (2005).
Levy, E., Beaulieu, J. F. & Spahis, S. From congenital disorders of fat malabsorption to understanding intra-enterocyte mechanisms behind chylomicron assembly and secretion. Front. Physiol. 12, 629222 (2021).
Stahel, P., Xiao, C., Nahmias, A. & Lewis, G. F. Role of the gut in diabetic dyslipidemia. Front. Endocrinol. 11, 116 (2020).
Giammanco, A., Cefalu, A. B., Noto, D. & Averna, M. R. The pathophysiology of intestinal lipoprotein production. Front. Physiol. 6, 61 (2015).
Cartwright, I. J., Plonne, D. & Higgins, J. A. Intracellular events in the assembly of chylomicrons in rabbit enterocytes. J. Lipid Res. 41, 1728–1739 (2000).
Xiao, C., Stahel, P., Nahmias, A. & Lewis, G. F. Emerging role of lymphatics in the regulation of intestinal lipid mobilization. Front. Physiol. 10, 1604 (2019).
Lambert, J. E. & Parks, E. J. Postprandial metabolism of meal triglyceride in humans. Biochim. Biophys. Acta 1821, 721–726 (2012).
Mattes, R. D. Oral fat exposure increases the first phase triacylglycerol concentration due to release of stored lipid in humans. J. Nutr. 132, 3656–3662 (2002).
Chavez-Jauregui, R. N., Mattes, R. D. & Parks, E. J. Dynamics of fat absorption and effect of sham feeding on postprandial lipema. Gastroenterology 139, 1538–1548 (2010).
Khan, N. A. & Besnard, P. Oro-sensory perception of dietary lipids: new insights into the fat taste transduction. Biochim. Biophys. Acta 1791, 149–155 (2009).
Mattes, R. D. Brief oral stimulation, but especially oral fat exposure, elevates serum triglycerides in humans. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G365–G371 (2009).
Robertson, M. D. et al. Mobilisation of enterocyte fat stores by oral glucose in humans. Gut 52, 834–839 (2003).
Xiao, C. et al. Oral glucose mobilizes triglyceride stores from the human intestine. Cell Mol. Gastroenterol. Hepatol. 7, 313–337 (2019).
Bjornson, E. et al. Investigation of human apoB48 metabolism using a new, integrated non-steady-state model of apoB48 and apoB100 kinetics. J. Intern. Med. 285, 562–577 (2019).
Bjornson, E. et al. Apolipoprotein B48 metabolism in chylomicrons and very low-density lipoproteins and its role in triglyceride transport in normo- and hypertriglyceridemic human subjects. J. Intern. Med. 288, 422–438 (2020).
Xiao, C., Stahel, P., Carreiro, A. L., Buhman, K. K. & Lewis, G. F. Recent advances in triacylglycerol mobilization by the gut. Trends Endocrinol. Metab. 29, 151–163 (2018).
Nahmias, A., Stahel, P., Tian, L., Xiao, C. & Lewis, G. F. GLP-1 (glucagon-like peptide-1) is physiologically relevant for chylomicron secretion beyond its known pharmacological role. Arterioscler. Thromb. Vasc. Biol. 41, 1893–1900 (2021).
Stahel, P., Xiao, C., Nahmias, A., Tian, L. & Lewis, G. F. Multi-organ coordination of lipoprotein secretion by hormones, nutrients and neural networks. Endocr. Rev. 42, 815–838 (2021).
Taskinen, M. R. et al. Effects of liraglutide on the metabolism of triglyceride-rich lipoproteins in type 2 diabetes. Diabetes Obes. Metab. 23, 1191–1201 (2021).
Stemmer, K., Finan, B., DiMarchi, R. D., Tschop, M. H. & Muller, T. D. Insights into incretin-based therapies for treatment of diabetic dyslipidemia. Adv. Drug. Deliv. Rev. 159, 34–53 (2020).
Hein, G. J., Baker, C., Hsieh, J., Farr, S. & Adeli, K. GLP-1 and GLP-2 as yin and yang of intestinal lipoprotein production: evidence for predominance of GLP-2-stimulated postprandial lipemia in normal and insulin-resistant states. Diabetes 62, 373–381 (2013).
Verges, B. et al. Liraglutide reduces postprandial hyperlipidemia by increasing ApoB48 (apolipoprotein B48) catabolism and by reducing apob48 production in patients with type 2 diabetes mellitus. Arterioscler. Thromb. Vasc. Biol. 38, 2198–2206 (2018).
Berberich, A. J. & Hegele, R. A. Lipid effects of glucagon-like peptide 1 receptor analogs. Curr. Opin. Lipidol. 32, 191–199 (2021).
Julve, J., Martin-Campos, J. M., Escola-Gil, J. C. & Blanco-Vaca, F. Chylomicrons: advances in biology, pathology, laboratory testing, and therapeutics. Clin. Chim. Acta 455, 134–148 (2016).
Lee, J. & Hegele, R. A. Abetalipoproteinemia and homozygous hypobetalipoproteinemia: a framework for diagnosis and management. J. Inherit. Metab. Dis. 37, 333–339 (2014).
Paquette, M., Dufour, R., Hegele, R. A. & Baass, A. A tale of 2 cousins: an atypical and a typical case of abetalipoproteinemia. J. Clin. Lipidol. 10, 1030–1034 (2016).
Ginsberg, H. N. ApoB SURFs a ride from the ER to the Golgi. Cell Metab. 33, 231–233 (2021).
Strong, A. & Rader, D. J. Sortilin as a regulator of lipoprotein metabolism. Curr. Atheroscler. Rep. 14, 211–218 (2012).
Wilson, D. E. et al. Phenotypic expression of heterozygous lipoprotein lipase deficiency in the extended pedigree of a proband homozygous for a missense mutation. J. Clin. Invest. 86, 735–750 (1990).
Gilham, D. et al. Inhibitors of hepatic microsomal triacylglycerol hydrolase decrease very low density lipoprotein secretion. Faseb J. 17, 1685–1687 (2003).
Dolinsky, V. W., Gilham, D., Alam, M., Vance, D. E. & Lehner, R. Triacylglycerol hydrolase: role in intracellular lipid metabolism. Cell Mol. Life Sci. 61, 1633–1651 (2004).
Boren, J. et al. Effects of TM6SF2 E167K on hepatic lipid and very low-density lipoprotein metabolism in humans. JCI Insight 5, e144079 (2020).
Boren, J. et al. Effects of PNPLA3 I148M on hepatic lipid and very low-density lipoprotein metabolism in humans. J. Intern. Med. 291, 218–223 (2022).
Havel, R. J. & Gordon, R. S. Jr. Idiopathic hyperlipemia: metabolic studies in an affected family. J. Clin. Invest. 39, 1777–1790 (1960).
Kersten, S. Physiological regulation of lipoprotein lipase. Biochim. Biophys. Acta 1841, 919–933 (2014).
Semenkovich, C. F., Wims, M., Noe, L., Etienne, J. & Chan, L. Insulin regulation of lipoprotein lipase activity in 3T3-L1 adipocytes is mediated at posttranscriptional and posttranslational levels. J. Biol. Chem. 264, 9030–9038 (1989).
Taskinen, M. R. & Nikkila, E. A. Lipoprotein lipase activity of adipose tissue and skeletal muscle in insulin-deficient human diabetes. Relation to high-density and very-low-density lipoproteins and response to treatment. Diabetologia 17, 351–356 (1979).
Wang, H. & Eckel, R. H. Lipoprotein lipase: from gene to obesity. Am. J. Physiol. Endocrinol. Metab. 297, E271–E288 (2009).
Kirchgessner, T. G. et al. Genetic and developmental regulation of the lipoprotein lipase gene: loci both distal and proximal to the lipoprotein lipase structural gene control enzyme expression. J. Biol. Chem. 264, 1473–1482 (1989).
Wu, S. A., Kersten, S. & Qi, L. Lipoprotein lipase and its regulators: an unfolding story. Trends Endocrinol. Metab. 32, 48–61 (2021).
Young, S. G. et al. GPIHBP1 and lipoprotein lipase, partners in plasma triglyceride metabolism. Cell Metab. 30, 51–65 (2019).
Ruppert, P. M. M. & Kersten, S. A lipase fusion feasts on fat. J. Biol. Chem. 295, 2913–2914 (2020).
Beigneux, A. P. et al. Glycosylphosphatidylinositol-anchored high-density lipoprotein-binding protein 1 plays a critical role in the lipolytic processing of chylomicrons. Cell Metab. 5, 279–291 (2007).
Goldberg, I. J. et al. Lipolytic enzymes and free fatty acids at the endothelial interface. Atherosclerosis 329, 1–8 (2021).
Rabacchi, C. et al. Clinical and genetic features of 3 patients with familial chylomicronemia due to mutations in GPIHBP1 gene. J. Clin. Lipidol. 10, 915–921.e914 (2016).
Paquette, M., Hegele, R. A., Pare, G. & Baass, A. A novel mutation in GPIHBP1 causes familial chylomicronemia syndrome. J. Clin. Lipidol. 12, 506–510 (2018).
Dron, J. S. & Hegele, R. A. Genetics of hypertriglyceridemia. Front. Endocrinol. 11, 455 (2020).
Wolska, A., Reimund, M. & Remaley, A. T. Apolipoprotein C-II: the re-emergence of a forgotten factor. Curr. Opin. Lipidol. 31, 147–153 (2020).
Dai, W., Zhang, Z., Yao, C. & Zhao, S. Emerging evidences for the opposite role of apolipoprotein C3 and apolipoprotein A5 in lipid metabolism and coronary artery disease. Lipids Health Dis. 18, 220 (2019).
Kersten, S. Long-lost friend is back in the game. J. Lipid Res. 62, 100072 (2021).
Olivecrona, G. & Beisiegel, U. Lipid binding of apolipoprotein CII is required for stimulation of lipoprotein lipase activity against apolipoprotein CII-deficient chylomicrons. Arterioscler. Thromb. Vasc. Biol. 17, 1545–1549 (1997).
Jong, M. C., Hofker, M. H. & Havekes, L. M. Role of ApoCs in lipoprotein metabolism: functional differences between ApoC1, ApoC2, and ApoC3. Arterioscler. Thromb. Vasc. Biol. 19, 472–484 (1999).
Wolska, A. et al. Apolipoprotein C-II: new findings related to genetics, biochemistry, and role in triglyceride metabolism. Atherosclerosis 267, 49–60 (2017).
Reimund, M. et al. Apolipoprotein C-II mimetic peptide is an efficient activator of lipoprotein lipase in human plasma as studied by a calorimetric approach. Biochem. Biophys. Res. Commun. 519, 67–72 (2019).
Wolska, A. et al. A dual apolipoprotein C-II mimetic-apolipoprotein C-III antagonist peptide lowers plasma triglycerides. Sci. Transl. Med. 12, eaaw7905 (2020).
Chen, Y. Q. et al. ApoA5 lowers triglyceride levels via suppression of ANGPTL3/8-mediated LPL inhibition. J. Lipid Res. 62, 100068 (2021).
Reeskamp, L. F., Tromp, T. R. & Stroes, E. S. G. The next generation of triglyceride-lowering drugs: will reducing apolipoprotein C-III or angiopoietin like protein 3 reduce cardiovascular disease? Curr. Opin. Lipidol. 31, 140–146 (2020).
Boren, J., Packard, C. J. & Taskinen, M. R. The roles of ApoC-III on the metabolism of triglyceride-rich lipoproteins in humans. Front. Endocrinol. 11, 474 (2020).
Jorgensen, A. B., Frikke-Schmidt, R., Nordestgaard, B. G. & Tybjaerg-Hansen, A. Loss-of-function mutations in APOC3 and risk of ischemic vascular disease. N. Engl. J. Med. 371, 32–41 (2014).
Tg et al. Loss-of-function mutations in APOC3, triglycerides, and coronary disease. N. Engl. J. Med. 371, 22–31 (2014).
Gaudet, D. et al. Targeting APOC3 in the familial chylomicronemia syndrome. N. Engl. J. Med. 371, 2200–2206 (2014).
Liu, H. et al. Characterization of recombinant wild type and site-directed mutations of apolipoprotein C-III: lipid binding, displacement of ApoE, and inhibition of lipoprotein lipase. Biochemistry 39, 9201–9212 (2000).
Sparks, J. D. & Dong, H. H. FoxO1 and hepatic lipid metabolism. Curr. Opin. Lipidol. 20, 217–226 (2009).
Taskinen, M. R. & Boren, J. New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis 239, 483–495 (2015).
Jackson, K. G., Wolstencroft, E. J., Bateman, P. A., Yaqoob, P. & Williams, C. M. Greater enrichment of triacylglycerol-rich lipoproteins with apolipoproteins E and C-III after meals rich in saturated fatty acids than after meals rich in unsaturated fatty acids. Am. J. Clin. Nutr. 81, 25–34 (2005).
Faghihnia, N., Mangravite, L. M., Chiu, S., Bergeron, N. & Krauss, R. M. Effects of dietary saturated fat on LDL subclasses and apolipoprotein CIII in men. Eur. J. Clin. Nutr. 66, 1229–1233 (2012).
Pieke, B. et al. Treatment of hypertriglyceridemia by two diets rich either in unsaturated fatty acids or in carbohydrates: effects on lipoprotein subclasses, lipolytic enzymes, lipid transfer proteins, insulin and leptin. Int. J. Obes. Relat. Metab. Disord. 24, 1286–1296 (2000).
Hiukka, A. et al. Alterations of lipids and apolipoprotein CIII in very low density lipoprotein subspecies in type 2 diabetes. Diabetologia 48, 1207–1215 (2005).
Kanter, J. E. et al. Increased apolipoprotein C3 drives cardiovascular risk in type 1 diabetes. J. Clin. Invest. 129, 4165–4179 (2019).
Basu, A. et al. Apolipoprotein-defined lipoprotein subclasses, serum apolipoproteins, and carotid intima-media thickness in T1D. J. Lipid Res. 59, 872–883 (2018).
Valladolid-Acebes, I., Berggren, P. O. & Juntti-Berggren, L. Apolipoprotein CIII Is an important piece in the type-1 diabetes jigsaw puzzle. Int. J. Mol. Sci. 22, 932 (2021).
Adiels, M. et al. Role of apolipoprotein C-III overproduction in diabetic dyslipidaemia. Diabetes Obes. Metab. 21, 1861–1870 (2019).
Jansson Sigfrids, F. et al. Remnant cholesterol predicts progression of diabetic nephropathy and retinopathy in type 1 diabetes. J. Intern. Med. 290, 632–645 (2021).
Ginsberg, H. N. & Reyes-Soffer, G. Is APOC3 the driver of cardiovascular disease in people with type I diabetes mellitus? J. Clin. Invest. 129, 4074–4076 (2019).
Kersten, S. New insights into angiopoietin-like proteins in lipid metabolism and cardiovascular disease risk. Curr. Opin. Lipidol. 30, 205–211 (2019).
Koishi, R. et al. Angptl3 regulates lipid metabolism in mice. Nat. Genet. 30, 151–157 (2002).
Kersten, S. Angiopoietin-like 3 in lipoprotein metabolism. Nat. Rev. Endocrinol. 13, 731–739 (2017).
Nidhina Haridas, P. A. et al. Regulation of angiopoietin-like proteins (ANGPTLs) 3 and 8 by insulin. J. Clin. Endocrinol. Metab. 100, E1299–E1307 (2015).
Shimamura, M. et al. Angiopoietin-like protein3 regulates plasma HDL cholesterol through suppression of endothelial lipase. Arterioscler. Thromb. Vasc. Biol. 27, 366–372 (2007).
Musunuru, K. et al. Exome sequencing, ANGPTL3 mutations, and familial combined hypolipidemia. N. Engl. J. Med. 363, 2220–2227 (2010).
Romeo, S. et al. Rare loss-of-function mutations in ANGPTL family members contribute to plasma triglyceride levels in humans. J. Clin. Invest. 119, 70–79 (2009).
Willer, C. J. et al. Newly identified loci that influence lipid concentrations and risk of coronary artery disease. Nat. Genet. 40, 161–169 (2008).
Arca, M., Minicocci, I. & Maranghi, M. The angiopoietin-like protein 3: a hepatokine with expanding role in metabolism. Curr. Opin. Lipidol. 24, 313–320 (2013).
Stitziel, N. O. et al. ANGPTL3 deficiency and protection against coronary artery disease. J. Am. Coll. Cardiol. 69, 2054–2063 (2017).
Dewey, F. E. et al. Genetic and pharmacologic inactivation of ANGPTL3 and cardiovascular disease. N. Engl. J. Med. 377, 211–221 (2017).
Gaudet, D. et al. Vupanorsen, an N-acetyl galactosamine-conjugated antisense drug to ANGPTL3 mRNA, lowers triglycerides and atherogenic lipoproteins in patients with diabetes, hepatic steatosis, and hypertriglyceridaemia. Eur. Heart J. 41, 3936–3945 (2020).
Romeo, S. et al. Population-based resequencing of ANGPTL4 uncovers variations that reduce triglycerides and increase HDL. Nat. Genet. 39, 513–516 (2007).
Ruppert, P. M. M. et al. Fasting induces ANGPTL4 and reduces LPL activity in human adipose tissue. Mol. Metab. 40, 101033 (2020).
Bini, S. et al. The interplay between angiopoietin-like proteins and adipose tissue: another piece of the relationship between adiposopathy and cardiometabolic diseases? Int. J. Mol. Sci. 22, 742 (2021).
Dewey, F. E. et al. Inactivating variants in ANGPTL4 and risk of coronary artery disease. N. Engl. J. Med. 374, 1123–1133 (2016).
Liu, D. J. et al. Exome-wide association study of plasma lipids in >300,000 individuals. Nat. Genet. 49, 1758–1766 (2017).
Cooper, A. D. Hepatic uptake of chylomicron remnants. J. Lipid Res. 38, 2173–2192 (1997).
Boren, J. et al. Identification of the low density lipoprotein receptor-binding site in apolipoprotein B100 and the modulation of its binding activity by the carboxyl terminus in familial defective apo-B100. J. Clin. Invest. 101, 1084–1093 (1998).
Goldstein, J. L. & Brown, M. S. The LDL receptor. Arterioscler. Thromb. Vasc. Biol. 29, 431–438 (2009).
Lagace, T. A. PCSK9 and LDLR degradation: regulatory mechanisms in circulation and in cells. Curr. Opin. Lipidol. 25, 387–393 (2014).
Chan, D. C. et al. Comparative effects of PCSK9 (proprotein convertase subtilisin/kexin type 9) inhibition and statins on postprandial triglyceride-rich lipoprotein metabolism. Arterioscler. Thromb. Vasc. Biol. 38, 1644–1655 (2018).
Vallejo-Vaz, A. J., Corral, P., Schreier, L. & Ray, K. K. Triglycerides and residual risk. Curr. Opin. Endocrinol. Diabetes Obes. 27, 95–103 (2020).
Taskinen, M. R. et al. Effects of evolocumab on the postprandial kinetics of apo (apolipoprotein) B100- and B48-containing lipoproteins in subjects with type 2 diabetes. Arterioscler. Thromb. Vasc. Biol. 41, 962–975 (2021).
Klop, B. et al. Daytime triglyceride variability in men and women with different levels of triglyceridemia. Clin. Chim. Acta 412, 2183–2189 (2011).
Ceriello, A. et al. Evidence for an independent and cumulative effect of postprandial hypertriglyceridemia and hyperglycemia on endothelial dysfunction and oxidative stress generation: effects of short- and long-term simvastatin treatment. Circulation 106, 1211–1218 (2002).
Fard, A. et al. Acute elevations of plasma asymmetric dimethylarginine and impaired endothelial function in response to a high-fat meal in patients with type 2 diabetes. Arterioscler. Thromb. Vasc. Biol. 20, 2039–2044 (2000).
Taskinen, M. R. Diabetic dyslipidaemia: from basic research to clinical practice. Diabetologia 46, 733–749 (2003).
Esan, O. & Wierzbicki, A. S. Triglycerides and cardiovascular disease. Curr. Opin. Cardiol. 36, 469–477 (2021).
Cohn, J. S. et al. Contribution of apoB-48 and apoB-100 triglyceride-rich lipoproteins (TRL) to postprandial increases in the plasma concentration of TRL triglycerides and retinyl esters. J. Lipid Res. 34, 2033–2040 (1993).
Karpe, F., Bell, M., Bjorkegren, J. & Hamsten, A. Quantification of postprandial triglyceride-rich lipoproteins in healthy men by retinyl ester labeling and simultaneous measurement of apolipoproteins B-48 and B-100. Arterioscler. Thromb. Vasc. Biol. 15, 199–207 (1995).
Schneeman, B. O., Kotite, L., Todd, K. M. & Havel, R. J. Relationships between the responses of triglyceride-rich lipoproteins in blood plasma containing apolipoproteins B-48 and B-100 to a fat-containing meal in normolipidemic humans. Proc. Natl Acad. Sci. USA 90, 2069–2073 (1993).
Hiukka, A. et al. Long-term effects of fenofibrate on VLDL and HDL subspecies in participants with type 2 diabetes mellitus. Diabetologia 50, 2067–2075 (2007).
Adiels, M. et al. Postprandial accumulation of chylomicrons and chylomicron remnants is determined by the clearance capacity. Atherosclerosis 222, 222–228 (2012).
Peterson, J. et al. Fatty acid control of lipoprotein lipase: a link between energy metabolism and lipid transport. Proc. Natl Acad. Sci. USA 87, 909–913 (1990).
Miles, J. M. et al. Systemic and forearm triglyceride metabolism: fate of lipoprotein lipase-generated glycerol and free fatty acids. Diabetes 53, 521–527 (2004).
Vallejo-Vaz, A. J. et al. Triglyceride-rich lipoprotein cholesterol and risk of cardiovascular events among patients receiving statin therapy in the TNT trial. Circulation 138, 770–781 (2018).
Boren, J., Matikainen, N., Adiels, M. & Taskinen, M. R. Postprandial hypertriglyceridemia as a coronary risk factor. Clin. Chim. Acta 431, 131–142 (2014).
Skalen, K. et al. Subendothelial retention of atherogenic lipoproteins in early atherosclerosis. Nature 417, 750–754 (2002).
Boren, J. et al. Identification of the principal proteoglycan-binding site in LDL. A single-point mutation in apo-B100 severely affects proteoglycan interaction without affecting LDL receptor binding. J. Clin. Invest. 101, 2658–2664 (1998).
Flood, C. et al. Identification of the proteoglycan binding site in apolipoprotein B48. J. Biol. Chem. 277, 32228–32233 (2002).
Salinas, C. A. A. & Chapman, M. J. Remnant lipoproteins: are they equal to or more atherogenic than LDL? Curr. Opin. Lipidol. 31, 132–139 (2020).
Schwartz, E. A. & Reaven, P. D. Lipolysis of triglyceride-rich lipoproteins, vascular inflammation, and atherosclerosis. Biochim. Biophys. Acta 1821, 858–866 (2012).
Cabodevilla, A. G. et al. Eruptive xanthoma model reveals endothelial cells internalize and metabolize chylomicrons, leading to extravascular triglyceride accumulation. J. Clin. Invest. 131, e145800 (2021).
Fischer, A. W. et al. Lysosomal lipoprotein processing in endothelial cells stimulates adipose tissue thermogenic adaptation. Cell Metab. 33, 547–564.e7 (2021).
Moreton, J. R. Physical state of lipids and foreign substances producing atherosclerosis. Science 107, 371–373 (1948).
Zilversmit, D. B. Atherogenesis: a postprandial phenomenon. Circulation 60, 473–485 (1979).
Baass, A., Paquette, M., Bernard, S. & Hegele, R. A. Familial chylomicronemia syndrome: an under-recognized cause of severe hypertriglyceridaemia. J. Intern. Med. 287, 340–348 (2020).
Goldberg, R. B. & Chait, A. A comprehensive update on the chylomicronemia syndrome. Front. Endocrinol. 11, 593931 (2020).
D’Erasmo, L. et al. Spectrum of mutations and long-term clinical outcomes in genetic chylomicronemia syndromes. Arterioscler. Thromb. Vasc. Biol. 39, 2531–2541 (2019).
Hegele, R. A. et al. Clinical and biochemical features of different molecular etiologies of familial chylomicronemia. J. Clin. Lipidol. 12, 920–927.e4 (2018).
Dron, J. S. et al. Severe hypertriglyceridemia is primarily polygenic. J. Clin. Lipidol. 13, 80–88 (2019).
Moulin, P. et al. Identification and diagnosis of patients with familial chylomicronaemia syndrome (FCS): Expert panel recommendations and proposal of an “FCS score”. Atherosclerosis 275, 265–272 (2018).
Gill, P. K., Dron, J. S. & Hegele, R. A. Genetics of hypertriglyceridemia and atherosclerosis. Curr. Opin. Cardiol. 36, 264–271 (2021).
Pedersen, S. B., Langsted, A. & Nordestgaard, B. G. Nonfasting mild-to-moderate hypertriglyceridemia and risk of acute pancreatitis. JAMA Intern. Med. 176, 1834–1842 (2016).
Hansen, S. E. J., Madsen, C. M., Varbo, A., Tybjaerg-Hansen, A. & Nordestgaard, B. G. Genetic variants associated with increased plasma levels of triglycerides, via effects on the lipoprotein lipase pathway, increase risk of acute pancreatitis. Clin. Gastroenterol. Hepatol. 19, 1652–1660.e6 (2021).
Hansen, S. E. J., Madsen, C. M., Varbo, A. & Nordestgaard, B. G. Body mass index, triglycerides, and risk of acute pancreatitis: a population-based study of 118 000 individuals. J. Clin. Endocrinol. Metab. 105, dgz059 (2020).
Mach, F. et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur. Heart J. 41, 111–188 (2020).
Kotseva, K. et al. Lifestyle and impact on cardiovascular risk factor control in coronary patients across 27 countries: results from the european society of cardiology ESC-EORP EUROASPIRE V registry. Eur. J. Prev. Cardiol. 26, 824–835 (2019).
Arnett, D. K. et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 74, 1376–1414 (2019).
Blaak, E. E., Riccardi, G. & Cho, L. Carbohydrates: separating fact from fiction. Atherosclerosis 328, 114–123 (2021).
Martinez-Gonzalez, M. A. et al. Carbohydrate quality changes and concurrent changes in cardiovascular risk factors: a longitudinal analysis in the PREDIMED-Plus randomized trial. Am. J. Clin. Nutr. 111, 291–306 (2020).
Sacks, F. M. et al. Dietary fats and cardiovascular disease: a presidential advisory from the American Heart Association. Circulation 136, e1–e23 (2017).
Ruscica, M., Ferri, N., Santos, R. D., Sirtori, C. R. & Corsini, A. Lipid lowering drugs: present status and future developments. Curr. Atheroscler. Rep. 23, 17 (2021).
Watts, G. F., Raal, F. J. & Chan, D. C. Transcriptomic therapy for dyslipidemias utilizing nucleic acids targeted at ANGPTL3. Future Cardiol. 18, 143–153 (2021).
Xu, J. & Ashjian, E. Treatment of hypertriglyceridemia: a review of therapies in the pipeline. J. Pharm. Pract. https://doi.org/10.1177/08971900211053489 (2021).
Parhofer, K. G. & Laufs, U. The diagnosis and treatment of hypertriglyceridemia. Dtsch. Arztebl Int. 116, 825–832 (2019).
Laufs, U., Parhofer, K. G., Ginsberg, H. N. & Hegele, R. A. Clinical review on triglycerides. Eur. Heart J. 41, 99–109c (2020).
Keech, A. et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366, 1849–1861 (2005).
Scott, R. et al. Effects of fenofibrate treatment on cardiovascular disease risk in 9,795 individuals with type 2 diabetes and various components of the metabolic syndrome: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetes Care 32, 493–498 (2009).
ACCORD Study Group. Effects of combination lipid therapy in type 2 diabetes mellitus. N. Engl. J. Med. 362, 1563–1574 (2010).
Bruckert, E., Labreuche, J., Deplanque, D., Touboul, P. J. & Amarenco, P. Fibrates effect on cardiovascular risk is greater in patients with high triglyceride levels or atherogenic dyslipidemia profile: a systematic review and meta-analysis. J. Cardiovasc. Pharmacol. 57, 267–272 (2011).
Lee, M., Saver, J. L., Towfighi, A., Chow, J. & Ovbiagele, B. Efficacy of fibrates for cardiovascular risk reduction in persons with atherogenic dyslipidemia: a meta-analysis. Atherosclerosis 217, 492–498 (2011).
Araki, E. et al. Effects of pemafibrate, a novel selective PPARalpha modulator, on lipid and glucose metabolism in patients with type 2 diabetes and hypertriglyceridemia: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care 41, 538–546 (2018).
Pradhan, A. D. et al. Rationale and design of the pemafibrate to reduce cardiovascular outcomes by reducing triglycerides in patients with diabetes (PROMINENT) study. Am. Heart J. 206, 80–93 (2018).
Mangili, O. C. et al. Favorable effects of ezetimibe alone or in association with simvastatin on the removal from plasma of chylomicrons in coronary heart disease subjects. Atherosclerosis 233, 319–325 (2014).
Toth, P. P. et al. Comparing remnant lipoprotein cholesterol measurement methods to evaluate efficacy of ezetimibe/statin vs statin therapy. J. Clin. Lipidol. 13, 997–1007.e8 (2019).
Sakamoto, K. et al. Effect of ezetimibe on LDL-C lowering and atherogenic lipoprotein profiles in type 2 diabetic patients poorly controlled by statins. PLoS ONE 10, e0138332 (2015).
Ahmed, O. et al. Ezetimibe in combination with simvastatin reduces remnant cholesterol without affecting biliary lipid concentrations in gallstone patients. J. Am. Heart Assoc. 7, e009876 (2018).
Morrone, D. et al. Lipid-altering efficacy of ezetimibe plus statin and statin monotherapy and identification of factors associated with treatment response: a pooled analysis of over 21,000 subjects from 27 clinical trials. Atherosclerosis 223, 251–261 (2012).
Tremblay, A. J., Lamarche, B., Cohn, J. S., Hogue, J. C. & Couture, P. Effect of ezetimibe on the in vivo kinetics of apoB-48 and apoB-100 in men with primary hypercholesterolemia. Arterioscler. Thromb. Vasc. Biol. 26, 1101–1106 (2006).
Arsenault, B. J., Perrot, N. & Puri, R. Therapeutic agents targeting cardiometabolic risk for preventing and treating atherosclerotic cardiovascular diseases. Clin. Pharmacol. Ther. 104, 257–268 (2018).
Blom, D. J., Raal, F. J., Santos, R. D. & Marais, A. D. Lomitapide and mipomersen-inhibiting microsomal triglyceride transfer protein (MTP) and apoB100 synthesis. Curr. Atheroscler. Rep. 21, 48 (2019).
Gallo, A., Beliard, S., D’Erasmo, L. & Bruckert, E. Familial chylomicronemia syndrome (FCS): recent data on diagnosis and treatment. Curr. Atheroscler. Rep. 22, 63 (2020).
Casula, M. et al. Omega-3 polyunsaturated fatty acids supplementation and cardiovascular outcomes: do formulation, dosage, and baseline cardiovascular risk matter? An updated meta-analysis of randomized controlled trials. Pharmacol. Res. 160, 105060 (2020).
Marston, N. A. et al. Association between triglyceride lowering and reduction of cardiovascular risk across multiple lipid-lowering therapeutic classes: a systematic review and meta-regression analysis of randomized controlled trials. Circulation 140, 1308–1317 (2019).
Nicholls, S. J. et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: the strength randomized clinical trial. JAMA 324, 2268–2280 (2020).
Nicholls, S. J. et al. Assessment of omega-3 carboxylic acids in statin-treated patients with high levels of triglycerides and low levels of high-density lipoprotein cholesterol: rationale and design of the STRENGTH trial. Clin. Cardiol. 41, 1281–1288 (2018).
Bhatt, D. L. et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N. Engl. J. Med. 380, 11–22 (2019).
Doi, T., Langsted, A. & Nordestgaard, B. G. A possible explanation for the contrasting results of REDUCE-IT vs. STRENGTH: cohort study mimicking trial designs. Eur. Heart J. 42, 4807–4817 (2021).
Gencer, B. et al. Effect of long-term marine omega-3 fatty acids supplementation on the risk of atrial fibrillation in randomized controlled trials of cardiovascular outcomes: a systematic review and meta-analysis. Circulation 144, 1981–1990 (2021).
Macchi, C. et al. A new dawn for managing dyslipidemias: the era of RNA-based therapies. Pharmacol. Res. 150, 104413 (2019).
Tsimikas, S. RNA-targeted therapeutics for lipid disorders. Curr. Opin. Lipidol. 29, 459–466 (2018).
Raal, F. J. et al. Evinacumab for homozygous familial hypercholesterolemia. N. Engl. J. Med. 383, 711–720 (2020).
Hsieh, J. & Adeli, K. Regulation of intestinal chylomicron production by glucagon-like peptides. Cardiovasc. Hematol. Disord. Drug Targets 12, 92–97 (2012).
Packard, C. J., Munro, A., Lorimer, A. R., Gotto, A. M. & Shepherd, J. Metabolism of apolipoprotein B in large triglyceride-rich very low density lipoproteins of normal and hypertriglyceridemic subjects. J. Clin. Invest. 74, 2178–2192 (1984).
Boren, J. et al. The assembly and secretion of apoB 100 containing lipoproteins in Hep G2 cells. Evidence for different sites for protein synthesis and lipoprotein assembly. J. Biol. Chem. 265, 10556–10564 (1990).
Ginsberg, H. N. Role of lipid synthesis, chaperone proteins and proteasomes in the assembly and secretion of apoprotein B-containing lipoproteins from cultured liver cells. Clin. Exp. Pharmacol. Physiol. 24, A29–A32 (1997).
Zhang, X. & Fernandez-Hernando, C. Transport of LDLs into the arterial wall: impact in atherosclerosis. Curr. Opin. Lipidol. 31, 279–285 (2020).
Huang, L. et al. SR-B1 drives endothelial cell LDL transcytosis via DOCK4 to promote atherosclerosis. Nature 569, 565–569 (2019).
Tabas, I., Williams, K. J. & Boren, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation 116, 1832–1844 (2007).
Camejo, G. The interaction of lipids and lipoproteins with the intercellular matrix of arterial tissue: its possible role in atherogenesis. Adv. Lipid Res. 19, 1–53 (1982).
Proctor, S. D. & Mamo, J. C. Intimal retention of cholesterol derived from apolipoprotein B100- and apolipoprotein B48-containing lipoproteins in carotid arteries of Watanabe heritable hyperlipidemic rabbits. Arterioscler. Thromb. Vasc. Biol. 23, 1595–1600 (2003).
Flood, C. et al. Molecular mechanism for changes in proteoglycan binding on compositional changes of the core and the surface of low-density lipoprotein-containing human apolipoprotein B100. Arterioscler. Thromb. Vasc. Biol. 24, 564–570 (2004).
Hiukka, A. et al. ApoCIII-enriched LDL in type 2 diabetes displays altered lipid composition, increased susceptibility for sphingomyelinase, and increased binding to biglycan. Diabetes 58, 2018–2026 (2009).
Johannesen, C. D. L., Mortensen, M. B., Langsted, A. & Nordestgaard, B. G. Apolipoprotein B and non-HDL cholesterol better reflect residual risk than LDL cholesterol in statin-treated patients. J. Am. Coll. Cardiol. 77, 1439–1450 (2021).
Balling, M. et al. VLDL cholesterol accounts for one-half of the risk of myocardial infarction associated with apob-containing lipoproteins. J. Am. Coll. Cardiol. 76, 2725–2735 (2020).
Nordestgaard, B. G. Triglyceride-rich lipoproteins and atherosclerotic cardiovascular disease: new insights from epidemiology, genetics, and biology. Circ. Res. 118, 547–563 (2016).
Nakajima, K. et al. Postprandial lipoprotein metabolism: VLDL vs chylomicrons. Clin. Chim. Acta 412, 1306–1318 (2011).
Acknowledgements
The authors acknowledge funding from the Swedish Research Council, the Swedish Heart–Lung Foundation, the Swedish state under the agreement between the Swedish government and the county councils (ALFGBG-965404; J.B.), and the Finnish Foundation of Cardiovascular Research (M.-R.T.). The authors thank R. Perkins (University of Gothenburg, Sweden) for help with editing this manuscript.
Author information
Authors and Affiliations
Contributions
J.B., M.-R.T. and C.J.P. researched data for this manuscript. All the authors contributed to the discussion of its content and wrote, reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer-review information
Nature Reviews Cardiology thanks David Cohen, who co-reviewed with Michele Alves-Bezerra; Daniel Rader; and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Borén, J., Taskinen, MR., Björnson, E. et al. Metabolism of triglyceride-rich lipoproteins in health and dyslipidaemia. Nat Rev Cardiol 19, 577–592 (2022). https://doi.org/10.1038/s41569-022-00676-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-022-00676-y
This article is cited by
-
Correlations between genetically predicted lipid-lowering drug targets and inflammatory bowel disease
Lipids in Health and Disease (2024)
-
Prevalence of dyslipidaemia within Polish nurses. Cross-sectional study - single and multiple linear regression models and ROC analysis
BMC Public Health (2024)
-
Remnant cholesterol trajectory and subclinical arteriosclerosis: a 10-year longitudinal study of Chinese adults
Scientific Reports (2024)
-
Elevated blood remnant cholesterol and triglycerides are causally related to the risks of cardiometabolic multimorbidity
Nature Communications (2024)
-
Hyperlipidaemia in diabetes: are there particular considerations for next-generation therapies?
Diabetologia (2024)