Abstract
Antiretroviral therapy has largely transformed HIV infection into a chronic disease condition. As such, physicians and other providers caring for individuals living with HIV infection need to be aware of the potential cardiovascular complications of HIV infection and the nuances of how HIV infection increases the risk of cardiovascular diseases, including acute myocardial infarction, stroke, peripheral artery disease, heart failure and sudden cardiac death, as well as how to select available therapies to reduce this risk. In this Review, we discuss the epidemiology and clinical features of cardiovascular disease, with a focus on coronary heart disease, in the setting of HIV infection, which includes a substantially increased risk of myocardial infarction even when the HIV infection is well controlled. We also discuss the mechanisms underlying HIV-associated atherosclerotic cardiovascular disease, such as the high rates of traditional cardiovascular risk factors in patients with HIV infection and HIV-related factors, including the use of antiretroviral therapy and chronic inflammation in the setting of effectively treated HIV infection. Finally, we highlight available therapeutic strategies, as well as approaches under investigation, to reduce the risk of cardiovascular disease and lower inflammation in patients with HIV infection.
Key points
As improvements to antiretroviral therapies have led to better control of HIV infection (although not cured it), individuals with HIV infection are now ageing, and cardiovascular disease is an important health concern in this patient population.
Traditional risk factors including dyslipidaemia, hypertension, cigarette smoking, diabetes mellitus and metabolic syndrome are common among people with HIV infection and increase the risk of cardiovascular disease.
In addition to traditional risk factors, characteristics related to HIV infection, including low CD4+ T cell count, nadir CD4+ T cell count and viral detectability, and some antiretroviral therapies are independently associated with increased risk of cardiovascular disease.
In the setting of treated suppressed HIV replication, chronic inflammation and immune activation persist and are strongly predictive of mortality and cardiovascular events.
Potential strategies to reduce the risk of cardiovascular disease in patients with HIV infection include targeting traditional risk factors, initiation of antiretroviral therapy to reduce inflammation and other approaches to lower inflammation, including gut-related interventions, statin therapy and immune modulators.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Redfield, M. M. Heart failure with preserved ejection fraction. N. Engl. J. Med. 375, 1868–1877 (2016).
Smit, M. et al. Future challenges for clinical care of an ageing population infected with HIV: a modelling study. Lancet Infect. Dis. 15, 810–818 (2015).
Tseng, Z. H. et al. Sudden cardiac death in patients with human immunodeficiency virus infection. J. Am. Coll. Cardiol. 59, 1891–1896 (2012).
Freiberg, M. S. et al. HIV infection and the risk of acute myocardial infarction. JAMA Intern. Med. 173, 614–622 (2013).
Chow, F. C. et al. Comparison of ischemic stroke incidence in HIV-infected and non-HIV-infected patients in a US health care system. J. Acquir. Immune Defic. Syndr. 60, 351–358 (2012).
Freiberg, M. S. et al. Association between HIV infection and the risk of heart failure with reduced ejection fraction and preserved ejection fraction in the antiretroviral therapy era: results from the Veterans Aging Cohort Study. JAMA Cardiol. 2, 536–546 (2017).
Deeks, S. G., Lewin, S. R. & Havlir, D. V. The end of AIDS: HIV infection as a chronic disease. Lancet 382, 1525–1533 (2013).
Zanni, M. V. et al. Increased coronary atherosclerotic plaque vulnerability by coronary computed tomography angiography in HIV-infected men. AIDS 27, 1263–1272 (2013).
D’Ascenzo, F. et al. High prevalence at computed coronary tomography of non-calcified plaques in asymptomatic HIV patients treated with HAART: a meta-analysis. Atherosclerosis 240, 197–204 (2015).
Crane, H. M. et al. Types of myocardial infarction among human immunodeficiency virus-infected individuals in the United States. JAMA Cardiol. 2, 260–267 (2017).
Hsue, P. Y. et al. Clinical features of acute coronary syndromes in patients with human immunodeficiency virus infection. Circulation 109, 316–319 (2004).
Currie, P. F. et al. Heart muscle disease related to HIV infection: prognostic implications. BMJ 309, 1605–1607 (1994).
Hsue, P. Y. & Waters, D. D. Heart failure in persons living with HIV infection. Curr. Opin. HIV AIDS 12, 534–539 (2017).
Eisenberg, M. J., Gordon, A. S. & Schiller, N. B. HIV-associated pericardial effusions. Chest 102, 956–958 (1992).
Remick, J. et al. Heart failure in patients with human immunodeficiency virus infection: epidemiology, pathophysiology, treatment, and future research. Circulation 129, 1781–1789 (2014).
Tanuma, J. et al. Dilated cardiomyopathy in an adult human immunodeficiency virus type 1-positive patient treated with a zidovudine-containing antiretroviral regimen. Clin. Infect. Dis. 37, e109–e111 (2003).
Alvi, R. M. et al. Protease inhibitors and cardiovascular outcomes in patients with HIV and heart failure. J. Am. Coll. Cardiol. 72, 518–530 (2018).
Shah, A. S. V. et al. Global burden of atherosclerotic cardiovascular disease in people living with HIV. Circulation 138, 1100–1112 (2018).
Hsue, P. Y. & Waters, D. D. Time to recognize HIV infection as a major cardiovascular risk factor. Circulation 138, 1113–1115 (2018).
Paisible, A. L. et al. HIV infection, cardiovascular disease risk factor profile, and risk for acute myocardial infarction. J. Acquir. Immune Defic. Syndr. 68, 209–216 (2015).
Womack, J. A. et al. HIV infection and cardiovascular disease in women. J. Am. Heart Assoc. 3, e001035 (2014).
Benjamin, L. A. et al. HIV, antiretroviral treatment, hypertension, and stroke in Malawian adults: a case-control study. Neurology 86, 324–333 (2016).
Benjamin, L. A. et al. The role of human immunodeficiency virus-associated vasculopathy in the etiology of stroke. J. Infect. Dis. 216, 545–553 (2017).
Henry, K., Melroe, H., Huebesch, J., Hermundson, J. & Simpson, J. Atorvastatin and gemfibrozil for protease-inhibitor-related lipid abnormalities. Lancet 352, 1031–1032 (1998).
Friis-Moller, N. et al. Class of antiretroviral drugs and the risk of myocardial infarction. N. Engl. J. Med. 356, 1723–1735 (2007).
Monforte, A. et al. Atazanavir is not associated with an increased risk of cardio- or cerebrovascular disease events. AIDS 27, 407–415 (2013).
Ryom, L. et al. Cardiovascular disease and use of contemporary protease inhibitors: the D:A:D international prospective multicohort study. Lancet HIV 5, e291–e300 (2018).
Sabin, C. A. et al. Use of nucleoside reverse transcriptase inhibitors and risk of myocardial infarction in HIV-infected patients enrolled in the D:A:D study: a multi-cohort collaboration. Lancet 371, 1417–1426 (2008).
Sabin, C. A. et al. Is there continued evidence for an association between abacavir usage and myocardial infarction risk in individuals with HIV? A cohort collaboration. BMC Med. 14, 61 (2016).
Marcus, J. L. et al. Use of abacavir and risk of cardiovascular disease among HIV-infected individuals. J. Acquir. Immune Defic. Syndr. 71, 413–419 (2016).
Elion, R. A. et al. Recent abacavir use increases risk of type 1 and type 2 myocardial infarctions among adults with HIV. J. Acquir. Immune Defic. Syndr. 78, 62–72 (2018).
Alvarez, A. et al. Cardiovascular toxicity of abacavir: a clinical controversy in need of a pharmacological explanation. AIDS 31, 1781–1795 (2017).
Ding, X. et al. No association of abacavir use with myocardial infarction: findings of an FDA meta-analysis. J. Acquir. Immune Defic. Syndr. 61, 441–447 (2012).
Saag, M. S. et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA 320, 379–396 (2018).
Lundgren, J. D. et al. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N. Engl. J. Med. 373, 795–807 (2015).
Baker, J. V. et al. Early antiretroviral therapy at high CD4 counts does not improve arterial elasticity: a substudy of the strategic timing of antiretroviral treatment (START) trial. Open Forum Infect. Dis. 3, ofw213 (2016).
Baker, J. V. et al. Changes in cardiovascular disease risk factors with immediate versus deferred antiretroviral therapy initiation among HIV-positive participants in the START (Strategic Timing of Antiretroviral Treatment) trial. J. Am. Heart Assoc. 6, e004987 (2017).
Klein, D. B. et al. Declining relative risk for myocardial infarction among HIV-positive compared with HIV-negative individuals with access to care. Clin. Infect. Dis. 60, 1278–1280 (2015).
Delaney, J. A. et al. Cumulative human immunodeficiency viremia, antiretroviral therapy, and incident myocardial infarction. Epidemiology 30, 69–74 (2019).
Shahmanesh, M. et al. The cardiovascular risk management for people living with HIV in Europe: how well are we doing? AIDS 30, 2505–2518 (2016).
Rasmussen, L. D. et al. Myocardial infarction among Danish HIV-infected individuals: population-attributable fractions associated with smoking. Clin. Infect. Dis. 60, 1415–1423 (2015).
David, M. H., Hornung, R. & Fichtenbaum, C. J. Ischemic cardiovascular disease in persons with human immunodeficiency virus infection. Clin. Infect. Dis. 34, 98–102 (2002).
Matetzky, S. et al. Acute myocardial infarction in human immunodeficiency virus–infected patients. Arch. Intern. Med. 163, 457–460 (2003).
Escaut, L. et al. Coronary artery disease in HIV infected patients. Intensive Care Med. 29, 969–973 (2003).
Mehta, N. J. & Khan, I. A. HIV-associated coronary artery disease. Angiology 54, 269–275 (2003).
Ambrose, J. A. et al. Frequency of and outcome of acute coronary syndromes in patients with human immunodeficiency virus infection. Am. J. Cardiol. 92, 301–303 (2003).
Varriale, P., Saravi, G., Hernandez, E. & Carbon, F. Acute myocardial infarction in patients infected with human immunodeficiency virus. Am. Heart J. 147, 55–59 (2004).
Ren, X. et al. Comparison of outcomes using bare metal versus drug-eluting stents in coronary artery disease patients with and without human immunodeficiency virus infection. Am. J. Cardiol. 104, 216–222 (2009).
Schneider, S. et al. Association of increased CD8+ and persisting C-reactive protein levels with restenosis in HIV patients after coronary stenting. AIDS 30, 1413–1421 (2016).
Marcus, J. L. et al. Recurrence after hospitalization for acute coronary syndrome among HIV-infected and HIV-uninfected individuals. HIV Med. 20, 19–26 (2018).
O’Dwyer, E. J. et al. Lower coronary plaque burden in patients with HIV presenting with acute coronary syndrome. Open Heart 3, e000511 (2016).
Peyracchia, M. et al. Evaluation of coronary features of HIV patients presenting with ACS: the CUORE, a multicenter study. Atherosclerosis 274, 218–226 (2018).
Lo, J. et al. Effects of statin therapy on coronary artery plaque volume and high-risk plaque morphology in HIV-infected patients with subclinical atherosclerosis: a randomised, double-blind, placebo-controlled trial. Lancet HIV 2, e52–e63 (2015).
Thomas, G. P. et al. Associations between antiretroviral use and subclinical coronary atherosclerosis. AIDS 30, 2477–2486 (2016).
Post, W. S. et al. Associations between HIV infection and subclinical coronary atherosclerosis. Ann. Intern. Med. 160, 458–467 (2014).
Metkus, T. S. et al. HIV infection is associated with an increased prevalence of coronary noncalcified plaque among participants with a coronary artery calcium score of zero: multicenter AIDS Cohort Study (MACS). HIV Med. 16, 635–639 (2015).
Deeks, S. G. et al. International AIDS Society global scientific strategy: towards an HIV cure 2016. Nat. Med. 22, 839–850 (2016).
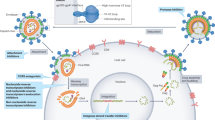
Krikke, M. et al. The role of T cells in the development of cardiovascular disease in HIV-infected patients. Atherosclerosis 237, 92–98 (2014).
Wang, T. et al. Increased cardiovascular disease risk in the HIV-positive population on ART: potential role of HIV-Nef and Tat. Cardiovasc. Pathol. 24, 279–282 (2015).
Ehrenreich, H. et al. Potent stimulation of monocytic endothelin-1 production by HIV-1 glycoprotein 120. J. Immunol. 150, 4601–4609 (1993).
Hsue, P. Y. et al. Progression of atherosclerosis as assessed by carotid intima-media thickness in patients with HIV infection. Circulation 109, 1603–1608 (2004).
Ho, J. E. et al. The association of CD4+ T-cell counts and cardiovascular risk in treated HIV disease. AIDS 26, 1115–1120 (2012).
Lichtenstein, K. A. et al. Low CD4+ T cell count is a risk factor for cardiovascular disease events in the HIV outpatient study. Clin. Infect. Dis. 51, 435–447 (2010).
Triant, V. A. et al. Association of immunologic and virologic factors with myocardial infarction rates in a U. S. Health Care System. J. Acquir. Immune Defic. Syndr. 55, 615–619 (2010).
Drozd, D. R. et al. Increased risk of myocardial infarction in HIV-infected individuals in North America compared with the general population. J. Acquir. Immune Defic. Syndr. 75, 568–576 (2017).
Serrano-Villar, S. et al. HIV-infected individuals with low CD4/CD8 ratio despite effective antiretroviral therapy exhibit altered T cell subsets, heightened CD8+T cell activation, and increased risk of non-AIDS morbidity and mortality. PLOS Pathog. 10, e1004078 (2014).
Serrano-Villar, S. et al. Increased risk of serious non-AIDS-related events in HIV-infected subjects on antiretroviral therapy associated with a low CD4/CD8 ratio. PLOS ONE 9, e85798 (2014).
Trickey, A. et al. CD4: CD8 ratio and CD8 count as prognostic markers for mortality in human immunodeficiency virus–infected patients on antiretroviral therapy: the Antiretroviral Therapy Cohort Collaboration (ART-CC). Clin. Infect. Dis. 65, 959–966 (2017).
Hanna, D. B. et al. Trends in cardiovascular disease mortality among persons with HIV in New York City, 2001–2012. Clin. Infect. Dis. 63, 1122–1129 (2016).
Hunt, P. W. et al. Relationship between T cell activation and CD4+ T cell count in HIV-seropositive individuals with undetectable plasma HIV RNA levels in the absence of therapy. J. Infect. Dis. 197, 126–133 (2008).
Naeger, D. M. et al. Cytomegalovirus-specific T cells persist at very high levels during long-term antiretroviral treatment of HIV disease. PLOS ONE 5, e8886 (2010).
Christensen-Quick, A., Vanpouille, C., Lisco, A. & Gianella, S. Cytomegalovirus and HIV persistence: pouring gas on the fire. AIDS Res. Hum. Retroviruses 33, S23–S30 (2017).
Hsue, P. Y. et al. Increased carotid intima-media thickness in HIV patients is associated with increased cytomegalovirus-specific T cell responses. AIDS 20, 2275–2283 (2006).
Masia, M. et al. Increased carotid intima-media thickness associated with antibody responses to varicella-zoster virus and cytomegalovirus in HIV-infected patients. PLOS ONE 8, e64327 (2013).
Knudsen, A. et al. Coronary artery calcium and intima-media thickness are associated with level of cytomegalovirus immunoglobulin G in HIV-infected patients. HIV Med. 20, 60–62 (2018).
Johansson, I. et al. Cytomegalovirus infection and disease reduce 10-year cardiac allograft vasculopathy-free survival in heart transplant recipients. BMC Infect. Dis. 15, 582 (2015).
Tincati, C., Douek, D. C. & Marchetti, G. Gut barrier structure, mucosal immunity and intestinal microbiota in the pathogenesis and treatment of HIV infection. AIDS Res. Ther. 13, 19 (2016).
Marchetti, G. et al. Microbial translocation predicts disease progression of HIV-infected antiretroviral-naive patients with high CD4+cell count. AIDS 25, 1385–1394 (2011).
Sandler, N. G. et al. Plasma levels of soluble CD14 independently predict mortality in HIV infection. J. Infect. Dis. 203, 780–790 (2011).
Reus Bañuls, S. et al. Association between inflammatory markers and microbial translocation in patients with human immunodeficiency virus infection taking antiretroviral treatment [Spanish]. Med. Clin. (Barc.) 142, 47–52 (2014).
Sandler, N. G. et al. Sevelamer does not decrease lipopolysaccharide or soluble CD14 levels but decreases soluble tissue factor, low-density lipoprotein (LDL) cholesterol, and oxidized LDL cholesterol levels in individuals with untreated HIV infection. J. Infect. Dis. 210, 1549–1554 (2014).
Ishizaki, A. et al. Effects of short-term probiotic ingestion on immune profiles and microbial translocation among HIV-1-infected Vietnamese children. Int. J. Mol. Sci. 18, 2185 (2017).
Somsouk, M. et al. The immunologic effects of mesalamine in treated HIV-infected individuals with incomplete CD4+ T cell recovery: a randomized crossover trial. PLOS ONE 9, e116306 (2014).
Kuller, L. H. et al. Inflammatory and coagulation biomarkers and mortality in patients with HIV infection. PLOS Med. 5, e203 (2008).
Borges, A. H. et al. Interleukin 6 is a stronger predictor of clinical events than high-sensitivity C-reactive protein or D-dimer during HIV infection. J. Infect. Dis. 214, 408–416 (2016).
Nordell, A. D. et al. Severity of cardiovascular disease outcomes among patients with HIV is related to markers of inflammation and coagulation. J. Am. Heart Assoc. 3, e000844 (2014).
Subramanian, S. et al. Arterial inflammation in patients with HIV. JAMA 308, 379–386 (2012).
Tawakol, A. et al. Association of arterial and lymph node inflammation with distinct inflammatory pathways in human immunodeficiency virus infection. JAMA Cardiol. 2, 163–171 (2017).
Ridker, P. M. Residual inflammatory risk: addressing the obverse side of the atherosclerosis prevention coin. Eur. Heart J. 37, 1720–1722 (2016).
Fichtenbaum, C. J., Yeh, T.-M., Evans, S. R. & Aberg, J. A. Treatment with pravastatin and fenofibrate improves atherogenic lipid profiles but not inflammatory markers in ACTG 5087. J. Clin. Lipidol. 4, 279–287 (2010).
Toribio, M. et al. Effects of pitavastatin and pravastatin on markers of immune activation and arterial inflammation in HIV. AIDS 31, 797–806 (2017).
Kelesidis, T. et al. Changes in inflammation and immune activation with atazanavir-, raltegravir-, darunavir-based initial antiviral therapy: ACTG 5260s. Clin. Infect. Dis. 61, 651–660 (2015).
Kim, C. J. et al. Impact of intensified antiretroviral therapy during early HIV infection on gut immunology and inflammatory blood biomarkers. AIDS 31, 1529–1534 (2017).
Hatano, H. et al. A randomized controlled trial assessing the effects of raltegravir intensification on endothelial function in treated HIV infection. J. Acquir. Immune Defic. Syndr. 61, 317–325 (2012).
O’Brien, M. P. et al. A randomized placebo controlled trial of aspirin effects on immune activation in chronically human immunodeficiency virus-infected adults on virologically suppressive antiretroviral therapy. Open Forum Infect. Dis. 4, ofw278 (2017).
Fahey, J. L. et al. The prognostic value of cellular and serologic markers in infection with human immunodeficiency virus type 1. N. Engl. J. Med. 322, 166–172 (1990).
Tenorio, A. R. et al. Soluble markers of inflammation and coagulation but not T cell activation predict non-AIDS-defining morbid events during suppressive antiretroviral treatment. J. Infect. Dis. 210, 1248–1259 (2014).
Longenecker, C. T. et al. Markers of inflammation and CD8 T cell activation, but not monocyte activation, are associated with subclinical carotid artery disease in HIV-infected individuals. HIV Med. 14, 385–390 (2013).
Kaplan, R. C. et al. T cell activation and senescence predict subclinical carotid artery disease in HIV-infected women. J. Infect. Dis. 203, 452–463 (2011).
Sinha, A. et al. Role of T-cell dysfunction, inflammation, and coagulation in microvascular disease in HIV. J. Am. Heart Assoc. 5, e004243 (2016).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Ridker, P. M. et al. Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet 391, 319–328 (2018).
Ridker, P. M. et al. Modulation of the interleukin-6 signalling pathway and incidence rates of atherosclerotic events and all-cause mortality: analyses from the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS). Eur. Heart J. 39, 3499–3507 (2018).
Hsue, P. Y. et al. IL-1beta inhibition reduces atherosclerotic inflammation in HIV infection. J. Am. Coll. Cardiol. 72, 2809–2811 (2018).
Hsue, P. Y. et al. Safety and impact of low-dose methotrexate on endothelial function and inflammation in individuals with treated human immunodeficiency virus: AIDS Clinical Trials Group Study A5314. Clin. Infect. Dis. https://doi.org/10.1093/cid/ciy781 (2018).
Stein, J. H. et al. Brachial artery echogenicity and grayscale texture changes in HIV-infected individuals receiving low-dose methotrexate. Arterioscler. Thromb. Vasc. Biol. 38, 2870–2878 (2018).
Grund, B. et al. Relevance of interleukin-6 and D-dimer for serious non-AIDS morbidity and death among HIV-positive adults on suppressive antiretroviral therapy. PLOS ONE 11, e0155100 (2016).
Swirski, F. K. & Nahrendorf, M. Cardioimmunology: the immune system in cardiac homeostasis and disease. Nat. Rev. Immunol. 18, 733–744 (2018).
Deeks, S. G., Overbaugh, J., Phillips, A. & Buchbinder, S. HIV infection. Nat. Rev. Dis. Primers 1, 15035 (2015).
Campbell, J. H., Hearps, A. C., Martin, G. E., Williams, K. C. & Crowe, S. M. The importance of monocytes and macrophages in HIV pathogenesis, treatment, and cure. AIDS 28, 2175–2187 (2014).
Wacleche, V. S., Tremblay, C. L., Routy, J. P. & Ancuta, P. The biology of monocytes and dendritic cells: contribution to HIV pathogenesis. Viruses 10, E65 (2018).
Walker, J. A. et al. Elevated numbers of CD163+ macrophages in hearts of simian immunodeficiency virus-infected monkeys correlate with cardiac pathology and fibrosis. AIDS Res. Hum. Retroviruses 30, 685–694 (2014).
Walker, J. A. et al. Anti-alpha4 integrin antibody blocks monocyte/macrophage traffic to the heart and decreases cardiac pathology in a SIV infection model of AIDS. J. Am. Heart Assoc. 4, e001932 (2015).
Scherzer, R. et al. Association of biomarker clusters with cardiac phenotypes and mortality in patients with HIV infection. Circ. Heart Fail. 11, e004312 (2018).
Leyes, P. et al. Increased cholesterol absorption rather than synthesis is involved in boosted protease inhibitor-associated hypercholesterolaemia. AIDS 32, 1309–1316 (2018).
Fontas, E. et al. Lipid profiles in HIV-infected patients receiving combination antiretroviral therapy: are different antiretroviral drugs associated with different lipid profiles? J. Infect. Dis. 189, 1056–1074 (2004).
Worm, S. W. et al. Risk of myocardial infarction in patients with HIV infection exposed to specific individual antiretroviral drugs from the 3 major drug classes: the data collection on adverse events of anti-HIV drugs (D:A:D) study. J. Infect. Dis. 201, 318–330 (2010).
Nix, L. M. & Tien, P. C. Metabolic syndrome, diabetes, and cardiovascular risk in HIV. Curr. HIV/AIDS Rep. 11, 271–278 (2014).
Hsue, P. Y. et al. Association of abacavir and impaired endothelial function in treated and suppressed HIV-infected patients. AIDS 23, 2021–2027 (2009).
Marconi, V. C. et al. Bilirubin is inversely associated with cardiovascular disease among HIV-positive and HIV-negative individuals in VACS (Veterans Aging Cohort Study). J. Am. Heart Assoc. 7, e007792 (2018).
Maseri, A. Inflammation, atherosclerosis, and ischemic events — exploring the hidden side of the moon. N. Engl. J. Med. 336, 1014–1016 (1997).
Hansson, G. K. Inflammation and atherosclerosis: the end of a controversy. Circulation 136, 1875–1877 (2017).
Triant, V. A. et al. Cardiovascular risk prediction functions underestimate risk in HIV infection. Circulation 137, 2203–2214 (2018).
Ganz, P. et al. Development and validation of a protein-based risk score for cardiovascular outcomes among patients with stable coronary heart disease. JAMA 315, 2532–2541 (2016).
Riddler, S. A. et al. Impact of HIV infection and HAART on serum lipids in men. JAMA 289, 2978–2982 (2003).
Fourie, C. M., Van Rooyen, J. M., Kruger, A. & Schutte, A. E. Lipid abnormalities in a never-treated HIV-1 subtype C-infected African population. Lipids 45, 73–80 (2010).
Periard, D. et al. Atherogenic dyslipidemia in HIV-infected individuals treated with protease inhibitors. The Swiss HIV Cohort Study. Circulation 100, 700–705 (1999).
Ucciferri, C. et al. Improved metabolic profile after switch to darunavir/ritonavir in HIV positive patients previously on protease inhibitor therapy. J. Med. Virol. 85, 755–759 (2013).
Daar, E. S. et al. Atazanavir plus ritonavir or efavirenz as part of a 3-drug regimen for initial treatment of HIV-1. Ann. Intern. Med. 154, 445–456 (2011).
Molina, J. M. et al. Rilpivirine versus efavirenz with tenofovir and emtricitabine in treatment-naive adults infected with HIV-1 (ECHO): a phase 3 randomised double-blind active-controlled trial. Lancet 378, 238–246 (2011).
Sax, P. E. et al. Tenofovir alafenamide versus tenofovir disoproxil fumarate in single tablet regimens for initial HIV-1 therapy: a randomized phase 2 study. J. Acquir. Immune Defic. Syndr. 67, 52–58 (2014).
Srinivasa, S. & Grinspoon, S. K. Metabolic and body composition effects of newer antiretrovirals in HIV-infected patients. Eur. J. Endocrinol. 170, R185–R102 (2014).
Raffi, F. et al. Once-daily dolutegravir versus raltegravir in antiretroviral-naive adults with HIV-1 infection: 48 week results from the randomised, double-blind, non-inferiority SPRING-2 study. Lancet 381, 735–743 (2013).
Libby, P., Bonow, R. O., Mann, D. L. & Zipes, D. P. Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine, 2-Volume Set (Elsevier Health Sciences, 2007).
Ekoru, K. et al. HIV treatment is associated with a two-fold higher probability of raised triglycerides: pooled analyses in 21 023 individuals in sub-Saharan Africa. Glob. Health Epidemiol. Genom. 3, e7 (2018).
Joy, T. et al. Dietary fat intake and relationship to serum lipid levels in HIV-infected patients with metabolic abnormalities in the HAART era. AIDS 21, 1591–1600 (2007).
Landmesser, U. et al. 2017 update of ESC/EAS Task Force on practical clinical guidance for proprotein convertase subtilisin/kexin type 9 inhibition in patients with atherosclerotic cardiovascular disease or in familial hypercholesterolaemia. Eur. Heart J. 39, 1131–1143 (2017).
Jacobson, T. A. NLA task force on statin safety-2014 update. J. Clin. Lipidol. 8, S1–S4 (2014).
Grundy, S. M. et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J. Am. Coll. Cardiol. https://doi.org/10.1016/j.jacc.2018.11.002 (2018).
Gilbert, J. M., Fitch, K. V. & Grinspoon, S. K. HIV-related cardiovascular disease, statins, and the REPRIEVE trial. Top. Antivir. Med. 23, 146–149 (2015).
Feinstein, M. J., Achenbach, C. J., Stone, N. J. & Lloyd-Jones, D. M. A. Systematic review of the usefulness of statin therapy in HIV-infected patients. Am. J. Cardiol. 115, 1760–1766 (2015).
Dube, M. P. et al. Guidelines for the evaluation and management of dyslipidemia in human immunodeficiency virus (HIV)-infected adults receiving antiretroviral therapy: recommendations of the HIV Medical Association of the Infectious Disease Society of America and the Adult AIDS Clinical Trials Group. Clin. Infect. Dis. 37, 613–627 (2003).
Busti, A. J. et al. Effects of atazanavir/ritonavir or fosamprenavir/ritonavir on the pharmacokinetics of rosuvastatin. J. Cardiovasc. Pharmacol. 51, 605–610 (2008).
Waters, D. D. & Boekholdt, S. M. An evidence-based guide to cholesterol-lowering guidelines. Can. J. Cardiol. 33, 343–349 (2017).
Mosepele, M., Molefe-Baikai, O. J., Grinspoon, S. K. & Triant, V. A. Benefits and risks of statin therapy in the HIV-infected population. Curr. Infect. Dis. Rep. 20, 20 (2018).
Aberg, J. A. et al. Pitavastatin versus pravastatin in adults with HIV-1 infection and dyslipidaemia (INTREPID): 12 week and 52 week results of a phase 4, multicentre, randomised, double-blind, superiority trial. Lancet HIV 4, e284–e294 (2017).
Levy, M. E., Greenberg, A. E., Magnus, M., Younes, N. & Castel, A. Evaluation of statin eligibility, prescribing practices, and therapeutic responses using ATP III, ACC/AHA, and NLA dyslipidemia treatment guidelines in a large urban cohort of HIV-infected outpatients. AIDS Patient Care STDS 32, 58–69 (2018).
Burkholder, G. A. et al. Low-density lipoprotein cholesterol response after statin initiation among persons living with human immunodeficiency virus. J. Clin. Lipidol 12, 988–998 (2018).
Rosenson, R. S., Colantonio, L. D., Burkholder, G. A., Chen, L. & Muntner, P. Trends in utilization of statin therapy and contraindicated statin use in HIV—infected adults treated with antiretroviral therapy from 2007 through 2015. J. Am. Heart Assoc. 7, e010345 (2018).
Wohl, D. A. et al. Ezetimibe alone reduces low-density lipoprotein cholesterol in HIV-infected patients receiving combination antiretroviral therapy. Clin. Infect. Dis. 47, 1105–1108 (2008).
[No authors listed]. Corrigendum to: ‘2016 ESC/EAS Guidelines for the Management of Dyslipidaemias’. Eur. Heart J. 39, 1254 (2018).
Eron, J. J. et al. Switch to a raltegravir-based regimen versus continuation of a lopinavir-ritonavir-based regimen in stable HIV-infected patients with suppressed viraemia (SWITCHMRK 1 and 2): two multicentre, double-blind, randomised controlled trials. Lancet 375, 396–407 (2010).
Gatell, J. M. et al. Switching from a ritonavir-boosted protease inhibitor to a dolutegravir-based regimen for maintenance of HIV viral suppression in patients with high cardiovascular risk. AIDS 31, 2503–2514 (2017).
Lee, F. J. et al. Rosuvastatin versus protease inhibitor switching for hypercholesterolaemia: a randomized trial. HIV Med. 17, 605–614 (2016).
Rasmussen, L. D. et al. Risk of diabetes mellitus in persons with and without HIV: a Danish nationwide population-based cohort study. PLOS ONE 7, e44575 (2012).
Mathabire Rücker, S. C. et al. High rates of hypertension, diabetes, elevated low-density lipoprotein cholesterol, and cardiovascular disease risk factors in HIV-infected patients in Malawi. AIDS 32, 253–260 (2018).
Lake, J. E. & Currier, J. S. Metabolic disease in HIV infection. Lancet Infect. Dis. 13, 964–975 (2013).
Mirza, F. S., Luthra, P. & Chirch, L. Endocrinological aspects of HIV infection. J. Endocrinol. Invest. 41, 881–899 (2018).
Grunfeld, C. et al. Association of upper trunk and visceral adipose tissue volume with insulin resistance in control and HIV-infected subjects in the FRAM study. J. Acquir. Immune Defic. Syndr. 46, 283–290 (2007).
Martinez, E. et al. Risk of lipodystrophy in HIV-1-infected patients treated with protease inhibitors: a prospective cohort study. Lancet 357, 592–598 (2001).
Rakotoambinina, B. et al. Lipodystrophic syndromes and hyperlipidemia in a cohort of HIV-1-infected patients receiving triple combination antiretroviral therapy with a protease inhibitor. J. Acquir. Immune Defic. Syndr. 27, 443–449 (2001).
Kurella, M., Lo, J. C. & Chertow, G. M. Metabolic syndrome and the risk for chronic kidney disease among nondiabetic adults. J. Am. Soc. Nephrol. 16, 2134–2140 (2005).
Grundy, S. M. et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112, 2735–2752 (2005).
Mocroft, A. et al. Cumulative and current exposure to potentially nephrotoxic antiretrovirals and development of chronic kidney disease in HIV-positive individuals with a normal baseline estimated glomerular filtration rate: a prospective international cohort study. Lancet HIV 3, e23–e32 (2016).
Armah, K. A. et al. Prehypertension, hypertension, and the risk of acute myocardial infarction in HIV-infected and -uninfected veterans. Clin. Infect. Dis. 58, 121–129 (2014).
Choi, A. I. et al. Association between kidney function and albuminuria with cardiovascular events in HIV-infected persons. Circulation 121, 651–658 (2010).
Ryom, L. et al. Renal impairment and cardiovascular disease in HIV-positive individuals: the D:A:D study. J. Infect. Dis. 214, 1212–1220 (2016).
Helleberg, M. et al. Mortality attributable to smoking among HIV-1-infected individuals: a nationwide, population-based cohort study. Clin. Infect. Dis. 56, 727–734 (2013).
Keith, A., Dong, Y., Shuter, J. & Himelhoch, S. Behavioral interventions for tobacco use in HIV-infected smokers: a meta-analysis. J. Acquir. Immune Defic. Syndr. 72, 527–533 (2016).
Pool, E. R., Dogar, O., Lindsay, R. P., Weatherburn, P. & Siddiqi, K. Interventions for tobacco use cessation in people living with HIV and AIDS. Cochrane Database Syst. Rev. 6, CD011120 (2016).
Huber, M. et al. Outcome of smoking cessation counselling of HIV-positive persons by HIV care physicians. HIV Med. 13, 387–397 (2012).
Cui, Q. et al. Safety and tolerability of varenicline tartrate (Champix(®)/Chantix(®)) for smoking cessation in HIV-infected subjects: a pilot open-label study. AIDS Patient Care STDS 26, 12–19 (2012).
Balfour, L. et al. An HIV-tailored quit-smoking counselling pilot intervention targeting depressive symptoms plus nicotine replacement therapy. AIDS Care 29, 24–31 (2017).
Petoumenos, K. et al. Rates of cardiovascular disease following smoking cessation in patients with HIV infection: results from the D:A:D study(*). HIV Med. 12, 412–421 (2011).
Behrens, G., Schmidt, H., Meyer, D., Stoll, M. & Schmidt, R. E. Vascular complications associated with use of HIV protease inhibitors. Lancet 351, 1958 (1998).
Himelman, R. B., Chung, W. S., Chernoff, D. N., Schiller, N. B. & Hollander, H. Cardiac manifestations of human immunodeficiency virus infection: a two-dimensional echocardiographic study. J. Am. Coll. Cardiol. 13, 1030–1036 (1989).
Vittecoq, D., Escaut, L. & Monsuez, J. J. Vascular complications associated with use of HIV protease inhibitors. Lancet 351, 1959 (1998).
Holmberg, S. D. et al. Protease inhibitors and cardiovascular outcomes in patients with HIV-1. Lancet 360, 1747–1748 (2002).
Fletcher, C. V. Overview of antiretroviral agents used to treat HIV. UpToDate https://www.uptodate.com/contents/overview-of-antiretroviral-agents-used-to-treat-hiv (2018).
Bozzette, S. A., Ake, C. F., Tam, H. K., Chang, S. W. & Louis, T. A. Cardiovascular and cerebrovascular events in patients treated for human immunodeficiency virus infection. N. Engl. J. Med. 348, 702–710 (2003).
Friis-Moller, N. et al. Combination antiretroviral therapy and the risk of myocardial infarction. N. Engl. J. Med. 349, 1993–2003 (2003).
El-Sadr, W. M. et al. CD4+ count-guided interruption of antiretroviral treatment. N. Engl. J. Med. 355, 2283–2296 (2006).
Stein, J. H. et al. A prospective, randomized clinical trial of antiretroviral therapies on carotid wall thickness: AIDS Clinical Trial Group Study A5260s. AIDS 29, 1775–1783 (2015).
INSIGHT START Study Group. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N. Engl. J. Med. 373, 795–807 (2015).
Duprez, D. A. et al. Inflammation, coagulation and cardiovascular disease in HIV-infected individuals. PLOS ONE 7, e44454 (2012).
Tien, P. C. et al. Inflammation and mortality in HIV-infected adults: analysis of the FRAM study cohort. J. Acquir. Immune Defic. Syndr. 55, 316–322 (2010).
So-Armah, K. A. et al. Do biomarkers of inflammation, monocyte activation, and altered coagulation explain excess mortality between HIV infected and uninfected people? J. Acquir. Immune Defic. Syndr. 72, 206–213 (2016).
Wada, N. I. et al. Inflammatory biomarkers and mortality risk among HIV-suppressed men: a multisite prospective cohort study. Clin. Infect. Dis. 63, 984–990 (2016).
Baker, J. V. et al. Systemic inflammation, coagulation, and clinical risk in the START trial. Open Forum Infect. Dis. 4, ofx262 (2017).
U.S. Department of Health and Human Services. FDA-approved HIV medicines. AIDSinfo https://aidsinfo.nih.gov/understanding-hiv-aids/fact-sheets/21/58/fda-approved-hiv-medicines (updated 12 Apr 2019).
Ofotokun, I. et al. Comparison of the metabolic effects of ritonavir-boosted darunavir or atazanavir versus raltegravir, and the impact of ritonavir plasma exposure: ACTG 5257. Clin. Infect. Dis. 60, 1842–1851 (2015).
Bergersen, B. M. Cardiovascular risk in patients with HIV Infection: impact of antiretroviral therapy. Drugs 66, 1971–1987 (2006).
Tebas, P. et al. Lipid levels and changes in body fat distribution in treatment-naive, HIV-1-infected adults treated with rilpivirine or efavirenz for 96 weeks in the ECHO and THRIVE trials. Clin. Infect. Dis. 59, 425–434 (2014).
Crane, H. M. et al. Impact of NRTIs on lipid levels among a large HIV-infected cohort initiating antiretroviral therapy in clinical care. AIDS 25, 185–195 (2011).
Maggi, P. et al. Cardiovascular risk factors in patients on long-term treatment with nevirapine- or efavirenz-based regimens. J. Antimicrob. Chemother. 66, 896–900 (2011).
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed its content, wrote the manuscript and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- CD4+ T cell counts
-
T cell subset that has a role in the immune system response against pathogens, infections and illnesses. A normal CD4+ T cell count is 500–1,500 cells per µl of plasma. CD4+ T cells are the main target cell of HIV, and the CD4+ T cell count is used to monitor the status of the HIV infection and the efficacy of the antiretroviral therapy.
- Nadir CD4+ T cell count
-
The lowest CD4+ T cell count an individual has had, which serves as a marker for immunodeficiency.
- Immunosenescence
-
Changes to the immune system that can be associated with age.
- Viraemia
-
Presence of viral particles in the blood.
- Latent HIV infection
-
A dormant or non-replicative HIV infection within a cell; in this state, the virus is not actively infecting other cells and individuals do not usually have noticeable symptoms.
- Virological failure
-
Refers to the failure of the HIV treatment to supress the virus completely; the virus is detectable in the blood (>200 copies per ml). This failure can occur as a result of drug resistance, drug toxicity or noncompliance with antiretroviral therapy.
Rights and permissions
About this article
Cite this article
Hsue, P.Y., Waters, D.D. HIV infection and coronary heart disease: mechanisms and management. Nat Rev Cardiol 16, 745–759 (2019). https://doi.org/10.1038/s41569-019-0219-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-019-0219-9
This article is cited by
-
Prevalence and risk factors of cardiovascular disease among people living with HIV in the Asia-Pacific region: a systematic review
BMC Public Health (2023)
-
Incidence and risk factors of hypertriglyceridemia in males with human immunodeficiency virus who are treated with combination antiretroviral therapy: a retrospective cohort study
Lipids in Health and Disease (2023)
-
The Gut Microbiome, Microbial Metabolites, and Cardiovascular Disease in People Living with HIV
Current HIV/AIDS Reports (2023)
-
Lipid metabolism patterns and relevant clinical and molecular features of coronary artery disease patients: an integrated bioinformatic analysis
Lipids in Health and Disease (2022)
-
Cardiovascular disease (CVD) risk assessment of HIV medication regimens using hematopoietic CD34+ progenitor cells
Stem Cell Research & Therapy (2022)