Abstract
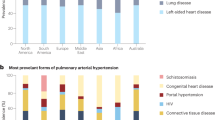
The epidemics of heart failure and, to a lesser extent, of pulmonary arterial hypertension continue unabated worldwide and are extremely costly in terms of loss of life and earnings, as well as the burden of health-care expenditure due to repeated hospitalization. The effectiveness of newly discovered therapies for the two conditions depends on their timely application. To date, symptoms have been used to guide the application and timing of therapy. Compelling evidence now exists that symptoms are preceded by several metabolic and haemodynamic changes, particularly a rise in intravascular pressures during exercise. These observations have stimulated the development of several implantable devices for the detection of impending unstable heart failure or pulmonary arterial hypertension, necessitating admission to hospital. In this Review, we summarize the rationale for monitoring patients with heart failure or pulmonary arterial hypertension, the transition from noninvasive to implantable devices and the current and anticipated clinical uses of these devices.
Key points
-
Long-term frequent or continuous haemodynamic monitoring is now feasible.
-
Measurements of pulmonary arterial pressure from a permanent implanted device have proven efficacy in reducing rehospitalization rates for patients with chronic heart failure.
-
Hypothetically, the integration of biomarker data, clinical signs and haemodynamic data will lead to improved care of patients with chronic cardiovascular conditions.
-
The emerging issue is the cost–benefit ratio for a variety of conditions and for different stages in progressive chronic diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ziaeian, B. & Fonarow, G. C. Epidemiology and aetiology of heart failure. Nat. Rev. Cardiol. 13, 368–378 (2016).
Savarese, G. & Lund, L. H. Global public health burden of heart failure. Card. Fail. Rev. 3, 7–11 (2017).
Ammar, K. A. et al. Prevalence and prognostic significance of heart failure stages: application of the American College of Cardiology/American Heart Association heart failure staging criteria in the community. Circulation 115, 1563–1570 (2007).
Keates, A. K. et al. Cardiovascular disease in Africa: epidemiological profile and challenges. Nat. Rev. Cardiol. 14, 273–293 (2017).
Lau, E. M. T., Giannoulatou, E., Celermajer, D. S. & Humbert, M. Epidemiology and treatment of pulmonary arterial hypertension. Nat. Rev. Cardiol. 14, 603–614 (2017).
Mocumbi, A. O., Thienemann, F. & Sliwa, K. A global perspective on the epidemiology of pulmonary hypertension. Can. J. Cardiol. 31, 375–381 (2015).
Heidenreich, P. A. et al. Forecasting the impact of heart failure in the United States a policy statement from the American Heart Association. Circ. Heart Fail. 6, 606–619 (2013).
Hoeper, M. M. et al. A global view of pulmonary hypertension. Lancet Respir. Med. 4, 306–322 (2016).
Gräf, S. et al. Identification of rare sequence variation underlying heritable pulmonary arterial hypertension. Nat. Commun. 9, 1416 (2018).
McGoon, M. D. & Miller, D. P. REVEAL: a contemporary US pulmonary arterial hypertension registry. Eur. Respir. Rev. 21, 8–18 (2012).
Peacock, A. Pulmonary hypertension. Eur. Respir. Rev. 22, 20–25 (2013).
Sikirica, M., Iorga, S. R., Bancroft, T. & Potash, J. The economic burden of pulmonary arterial hypertension (PAH) in the US on payers and patients. BMC Health Serv. Res. 14, 676 (2014).
McMurray, J. J. Neprilysin inhibition to treat heart failure: a tale of science, serendipity, and second chances. Eur. J. Heart Fail. 17, 242–247 (2015).
Hill, J. A. Braking bad hypertrophy. N. Engl. J. Med. 372, 2160–2162 (2015).
de Tombe, P. P. & Kohl, P. Which way to grow? Force over time may be the heart’s Dao de jing. Glob. Cardiol. Sci. Pract. 2016, e201621 (2016).
McMurray, J. J. et al. Angiotensin–neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 371, 993–1004 (2014).
Ghofrani, H. A. & Humbert, M. The role of combination therapy in managing pulmonary arterial hypertension. Eur. Respir. Rev. 23, 469–475 (2014).
Dorfs, S. et al. Pulmonary capillary wedge pressure during exercise and long-term mortality in patients with suspected heart failure with preserved ejection fraction. Eur. Heart J. 35, 3103–3112 (2014).
Nohria, A. et al. Clinical assessment identifies hemodynamic profiles that predict outcomes in patients admitted with heart failure. J. Am. Coll. Cardiol. 41, 1797–1804 (2003).
Hochman, J. S. & Katz, S. Back to the future in cardiogenic shock — initial PCI of the culprit lesion only. N. Engl. J. Med. 377, 2486–2488 (2017).
ElGuindy, A. & Yacoub, M. H. Heart failure with preserved ejection fraction. Glob. Cardiol. Sci. Pract. 2012, 10 (2012).
Dunlay, S. M., Roger, V. L. & Redfield, M. M. Epidemiology of heart failure with preserved ejection fraction. Nat. Rev. Cardiol. 14, 591–602 (2017).
American Heart Association. Classes of heart failure. AHA http://www.heart.org/HEARTORG/Conditions/HeartFailure/AboutHeartFailure/Classes-of-Heart-Failure_UCM_306328_Article.jsp#.Wss15IhubDc (2017).
Gheorghiade, M. & Pang, P. S. Acute heart failure syndromes. J. Am. Coll. Cardiol. 53, 557–573 (2009).
Gheorghiade, M., Vaduganathan, M., Fonarow, G. C. & Bonow, R. O. Rehospitalization for heart failure: problems and perspectives. J. Am. Coll. Cardiol. 61, 391–403 (2013).
Gheorghiade, M. et al. Acute heart failure syndromes: current state and framework for future research. Circulation 112, 3958–3968 (2005).
Albaghdadi, M., Gheorghiade, M. & Pitt, B. Mineralocorticoid receptor antagonism: therapeutic potential in acute heart failure syndromes. Eur. Heart J. 32, 2626–2633 (2011).
Damasceno, A. et al. The causes, treatment, and outcome of acute heart failure in 1006 Africans from 9 countries: results of the sub-Saharan Africa survey of heart failure. Arch. Intern. Med. 172, 1386–1394 (2012).
Sulaiman, K. et al. Clinical characteristics, management, and outcomes of acute heart failure patients: observations from the Gulf acute heart failure registry (Gulf CARE). Eur. J. Heart Fail. 17, 374–384 (2015).
Follath, F. et al. Clinical presentation, management and outcomes in the Acute Heart Failure Global Survey of Standard Treatment (ALARM-HF). Intensive Care Med. 37, 619–626 (2011).
Hassan, M., Latif, N. & Yacoub, M. Adipose tissue: friend or foe? Nat. Rev. Cardiol. 9, 689–702 (2012).
Austin, E. D. & Loyd, J. E. The genetics of pulmonary arterial hypertension. Circ. Res. 115, 189–200 (2014).
Newman, J. H. et al. Genetic basis of pulmonary arterial hypertension: current understanding and future directions. J. Am. Coll. Cardiol. 43 (Suppl.), 33–39 (2004).
Schermuly, R. T., Ghofrani, H. A., Wilkins, M. R. & Grimminger, F. Mechanisms of disease: pulmonary arterial hypertension. Nat. Rev. Cardiol. 8, 443–455 (2011).
Rabinovitch, M. Molecular pathogenesis of pulmonary arterial hypertension. J. Clin. Invest. 122, 4306–4313 (2012).
Ryan, J. J. & Archer, S. L. Emerging concepts in the molecular basis of pulmonary arterial hypertension. Part I: metabolic plasticity and mitochondrial dynamics in the pulmonary circulation and right ventricle in pulmonary arterial hypertension. Circulation 131, 1691–1702 (2015).
Haddad, R. N. & Mielniczuk, L. M. An evidence-based approach to screening and diagnosis of pulmonary hypertension. Can. J. Cardiol. 31, 382–390 (2015).
Inglis, S. C. et al. Structured telephone support or non-invasive telemonitoring for patients with heart failure. Cochrane Database Syst. Rev. 10, CD007228 (2015).
Koehler, F. et al. Impact of remote telemedical management on mortality and hospitalizations in ambulatory patients with chronic heart failure: the Telemedical Interventional Monitoring in Heart Failure study. Circulation 123, 1873–1880 (2011).
Chaudhry, S. I. et al. Telemonitoring in patients with heart failure. N. Engl. J. Med. 363, 2301–2309 (2010).
Seto, E. et al. Mobile phone-based telemonitoring for heart failure management: a randomized controlled trial. J. Med. Internet Res. 14, e31 (2012).
Hindricks, G. et al. Implant-based multiparameter telemonitoring of patients with heart failure (IN-TIME): a randomised controlled trial. Lancet 384, 583–590 (2014).
Varma, N., Michalski, J., Epstein, A. E. & Schweikert, R. Automatic remote monitoring of implantable cardioverter-defibrillator lead and generator performance the lumos-T safely reduces routine office device follow-up (TRUST) trial. Circ. Arrhythm. Electrophysiol. 3, 428–436 (2010).
Lara, B. A. et al. Accurate monitoring of intravascular fluid volume: a novel application of intrathoracic impedance measures for the guidance of volume reduction therapy. Int. J. Cardiol. Heart Vasc. 8, 47–51 (2015).
Van Veldhuisen, D. J. Intrathoracic impedance monitoring, audible patient alerts, and outcome in patients with heart failure. Circulation 124, 1719–1726 (2011).
Boehmer, J. P. et al. A multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. JACC Heart Fail. 5, 216–225 (2017).
Soga, Y. et al. Efficacy of fluid assessment based on intrathoracic impedance monitoring in patients with systolic heart failure. Circ. J. 75, 129–134 (2011).
Rickards, A. F., Bombardini, T., Corbucci, G. & Plicchi, G. An implantable intracardiac accelerometer for monitoring myocardial contractility: the multicenter PEA study group. Pacing Clin. Electrophysiol. 19, 2066–2071 (1996).
Mansourati, J., Heurteau, M. & Abalea, J. Heart failure monitoring with a cardiac resynchronization therapy device-based cardiac contractility sensor: a case series. J. Med. Case Rep. 8, 27 (2014).
Morgan, J. M. et al. Remote management of heart failure using implantable electronic devices. Eur. Heart J. 38, 2352–2360 (2017).
Cournand, A. Cardiac catheterization; development of the technique, its contributions to experimental medicine, and its initial applications in man. Acta Med. Scand. Suppl. 579, 3–32 (1975).
Gibbs, J. S. et al. Diurnal variation of pulmonary artery pressure in chronic heart failure. Br. Heart J. 62, 30–35 (1989).
Gibbs, J. S., Keegan, J., Wright, C., Fox, K. M. & Poole-Wilson, P. A. Pulmonary artery pressure changes during exercise and daily activities in chronic heart failure. J. Am. Coll. Cardiol. 15, 52–61 (1990).
Bourge, R. C. et al. Randomized controlled trial of an implantable continuous hemodynamic monitor in patients with advanced heart failure: the COMPASS-HF study. J. Am. Coll. Cardiol. 51, 1073–1079 (2008).
Ritzema, J. et al. Direct left atrial pressure monitoring in ambulatory heart failure patients: initial experience with a new permanent implantable device. Circulation 116, 2952–2959 (2007).
Adamson, P. B. et al. Continuous hemodynamic monitoring in patients with mild to moderate heart failure: results of The Reducing Decompensation Events Utilizing Intracardiac Pressures in Patients With Chronic Heart Failure (REDUCEhf) trial. Congest. Heart Fail. 17, 248–254 (2011).
Zile, M. R. et al. Intracardiac pressures measured using an implantable hemodynamic monitor: relationship to mortality in patients with chronic heart failure. Circ. Heart Fail. 10, e003594 (2017).
Ritzema, J. et al. Physician-directed patient self-management of left atrial pressure in advanced chronic heart failure. Circulation 121, 1086–1095 (2010).
Abraham, W. T. et al. Hemodynamic monitoring in advanced heart failure: results from the LAPTOP-HF trial [abstract]. J. Card. Fail. 22, 940 (2016).
Abraham, W. T. et al. Wireless pulmonary artery haemodynamic monitoring in chronic heart failure: a randomised controlled trial. Lancet 377, 658–666 (2011).
Hubbert, L., Baranowski, J., Delshad, B. & Ahn, H. Left atrial pressure monitoring with an implantable wireless pressure sensor after implantation of a left ventricular assist device. ASAIO J. 63, e60–e65 (2017).
Petersan, P. J. & Anlage, S. M. Measurement of resonant frequency and quality factor of microwave resonators: comparison of methods. J. Appl. Phys. 84, 3392–3402 (1998).
Murphy, O. H. et al. Continuous in vivo blood pressure measurements using a fully implantable wireless SAW sensor. Biomed. Microdevices 15, 737–749 (2013).
Zile, M. R. et al. Transition from chronic compensated to acute decompensated heart failure: pathophysiological insights obtained from continuous monitoring of intracardiac pressures. Circulation 118, 1433–1441 (2008).
Braunwald, E. Biomarkers in heart failure. N. Engl. J. Med. 358, 2148–2159 (2008).
Chow, S. L. et al. Role of biomarkers for the prevention, assessment, and management of heart failure: a scientific statement from the American Heart Association. Circulation 135, e1054–e1091 (2017).
Chen, F., Yang, J., Li, Y. & Wang, H. Circulating microRNAs as novel biomarkers for heart failure. Hellenic J. Cardiol. https://doi.org/10.1016/j.hjc.2017.10.002 (2017).
Angermann, C. E. et al. Safety and feasibility of pulmonary artery pressure-guided heart failure therapy: rationale and design of the prospective CardioMEMS Monitoring Study for Heart Failure (MEMS-HF). Clin. Res. Cardiol. https://doi.org/10.1007/s00392-018-1281-8 (2018).
Kastrup, M. Current practice of hemodynamic monitoring and vasopressor and inotropic therapy in post-operative cardiac surgery patients in Germany: results from a postal survey. Acta Anaesthesiol. Scand. 51, 347–358 (2007).
Terracciano, C. M., Miller, L. W. & Yacoub, M. Contemporary use of ventricular assist devices. Ann. Rev. Med. 61, 255–270 (2010).
Kim, G. H., Uriel, N. & Burkhoff, D. Reverse remodelling and myocardial recovery in heart failure. Nat. Rev. Cardiol. 15, 83–96 (2018).
Birks, E. J. et al. Left ventricular assist device and drug therapy for the reversal of heart failure. N. Engl. J. Med. 355, 1873–1884 (2006).
Yacoub, M. H. & Terracciano, C. M. Bridge to recovery and the search for decision nodes. Circ. Heart Fail. 4, 393–395 (2011).
Morrell, N. W. et al. Cellular and molecular basis of pulmonary arterial hypertension. J. Am. Coll. Cardiol. 54, S20–S31 (2009).
Rich, S. Future of clinical trials for pulmonary hypertension. Circulation 123, 2919–2921 (2011).
Frantz, R. P. Hemodynamic monitoring in pulmonary arterial hypertension. Expert Rev. Resp. Med. 5, 173–178 (2011).
Raina, A., Abraham, W. T., Adamson, P. B., Bauman, J. & Benza, R. L. Limitations of right heart catheterization in the diagnosis and risk stratification of patients with pulmonary hypertension related to left heart disease: insights from a wireless pulmonary artery pressure monitoring system. J. Heart Lung Transplant. 34, 438–447 (2015).
Ghio, S., Schirinzi, S. & Pica, S. Pulmonary arterial compliance: how and why should we measure it? Glob. Cardiol. Sci. Pract. 2015, 58 (2015).
Klersy, C. et al. A meta-analysis of remote monitoring of heart failure patients. J. Am. Coll. Cardiol. 54, 1683–1694 (2009).
Kong, X. et al. Brown adipose tissue controls skeletal muscle function via the secretion of myostatin. Cell Metab. 28, 631–643.e3 (2018).
Binkley, P. F. et al. Feasibility of using multivector impedance to monitor pulmonary congestion in heart failure patients. J. Interv. Card. Electriphysiol. 35, 197–206 (2012).
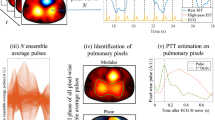
Lee, S. L. et al. Spatial orientation and morphology of the pulmonary artery: relevance to optimising design and positioning of a continuous pressure monitoring device. J. Cardiovasc. Transl Res. 9, 239–248 (2016).
Acknowledgements
The authors acknowledge the Wellcome Trust and the UK Department of Health for their support during the development of the SAW sensor system.
Author information
Authors and Affiliations
Contributions
Both authors researched data for the article, discussed its contents, wrote the manuscript and reviewed and edited it before submission.
Corresponding author
Ethics declarations
Competing interests
The authors are co-founders of Cardian Limited, a recent spinout from Imperial College London, UK, for the further development of the SAW sensor system.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yacoub, M.H., McLeod, C. The expanding role of implantable devices to monitor heart failure and pulmonary hypertension. Nat Rev Cardiol 15, 770–779 (2018). https://doi.org/10.1038/s41569-018-0103-z
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41569-018-0103-z
This article is cited by
-
Implantable haemodynamic sensor to remotely MONITOR HF
Nature Reviews Cardiology (2023)
-
Hydrogel interfaces for merging humans and machines
Nature Reviews Materials (2022)
-
Graphene electronic tattoos 2.0 with enhanced performance, breathability and robustness
npj 2D Materials and Applications (2022)
-
CD146-HIF-1α hypoxic reprogramming drives vascular remodeling and pulmonary arterial hypertension
Nature Communications (2019)
-
Looking back and thinking forwards — 15 years of cardiology and cardiovascular research
Nature Reviews Cardiology (2019)