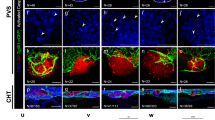
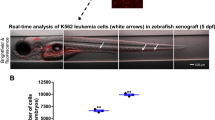
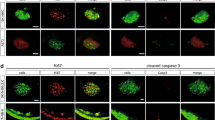
Abstract
In precision oncology, two major strategies are being pursued for predicting clinically relevant tumour behaviours, such as treatment response and emergence of drug resistance: inference based on genomic, transcriptomic, epigenomic and/or proteomic analysis of patient samples, and phenotypic assays in personalized cancer avatars. The latter approach has historically relied on in vivo mouse xenografts and in vitro organoids or 2D cell cultures. Recent progress in rapid combinatorial genetic modelling, the development of a genetically immunocompromised strain for xenotransplantation of human patient samples in adult zebrafish and the first clinical trial using xenotransplantation in zebrafish larvae for phenotypic testing of drug response bring this tiny vertebrate to the forefront of the precision medicine arena. In this Review, we discuss advances in transgenic and transplantation-based zebrafish cancer avatars, and how these models compare with and complement mouse xenografts and human organoids. We also outline the unique opportunities that these different models present for prediction studies and current challenges they face for future clinical deployment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
National Cancer Institute. Targeted cancer therapies. NCI https://www.cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet (2020).
Morrison, C. Fresh from the biotech pipeline — 2018. Nat. Biotechnol. 37, 118–123 (2019).
Friedman, A. A., Letai, A., Fisher, D. E. & Flaherty, K. T. Precision medicine for cancer with next-generation functional diagnostics. Nat. Rev. Cancer 15, 747–756 (2015).
Hyman, D. M., Taylor, B. S. & Baselga, J. Implementing genome-driven oncology. Cell 168, 584–599 (2017).
Clohessy, J. G. & Pandolfi, P. P. Mouse hospital and co-clinical trial project — from bench to bedside. Nat. Rev. Clin. Oncol. 12, 491 (2015). This review discusses mouse co-clinical trials for precision cancer therapy.
Drost, J. & Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 18, 407–418 (2018).
Tuveson, D. & Clevers, H. Cancer modeling meets human organoid technology. Science 364, 952–955 (2019).
Schwartzberg, L., Kim, E. S., Liu, D. & Schrag, D. Precision oncology: who, how, what, when, and when not? Am. Soc. Clin. Oncol. Educ. Book 37, 160–169 (2017).
Strauss, D. G. & Blinova, K. Clinical trials in a dish. Trends Pharmacol. Sci. 38, 4–7 (2017).
Aparicio, S., Hidalgo, M. & Kung, A. L. Examining the utility of patient-derived xenograft mouse models. Nat. Rev. Cancer 15, 311 (2015).
Tentler, J. J. et al. Patient-derived tumour xenografts as models for oncology drug development. Nat. Rev. Clin. Oncol. 9, 338 (2012).
Ablain, J. et al. Human tumor genomics and zebrafish modeling identify SPRED1 loss as a driver of mucosal melanoma. Science 362, 1055–1060 (2018). This article shows rapid generation of genetic avatars in zebrafish using combinatorial mosaic transgenesis.
Fior, R. et al. Single-cell functional and chemosensitive profiling of combinatorial colorectal therapy in zebrafish xenografts. Proc. Natl Acad. Sci. USA 114, E8234–E8243 (2017). This study conducts phenotypic drug response testing and migration scoring of PDXs in zebrafish larvae.
Yan, C. et al. Visualizing engrafted human cancer and therapy responses in immunodeficient zebrafish. Cell 177, 1903–1914 (2019). This article demonstrates establishment of PDXs in adult zebrafish.
White, R., Rose, K. & Zon, L. Zebrafish cancer: the state of the art and the path forward. Nat. Rev. Cancer 13, 624–636 (2013).
Pulak, R. Tools for automating the imaging of zebrafish larvae. Methods 96, 118–126 (2016).
Zhao, Y. et al. A review of automated microinjection of zebrafish embryos. Micromachines 10, 7 (2018).
Ablain, J., Durand, E. M., Yang, S., Zhou, Y. & Zon, L. I. A CRISPR/Cas9 vector system for tissue-specific gene disruption in zebrafish. Dev. Cell 32, 756–764 (2015).
Gengenbacher, N., Singhal, M. & Augustin, H. G. Preclinical mouse solid tumour models: status quo, challenges and perspectives. Nat. Rev. Cancer 17, 751 (2017).
Howe, K. et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 496, 498–503 (2013).
Carneiro, M. C., de Castro, I. P. & Ferreira, M. G. Telomeres in aging and disease: lessons from zebrafish. Dis. Model. Mech. 9, 737–748 (2016).
Berghmans, S. et al. Making waves in cancer research: new models in the zebrafish. Biotechniques 39, 227–237 (2005).
Langenau, D. M. et al. Myc-induced T cell leukemia in transgenic zebrafish. Science 299, 887–890 (2003). This article reports the first transgenic cancer model in zebrafish.
Yang, H. W. et al. Targeted expression of human MYCN selectively causes pancreatic neuroendocrine tumors in transgenic zebrafish. Cancer Res. 64, 7256–7262 (2004).
Patton, E. E. et al. BRAF mutations are sufficient to promote nevi formation and cooperate with p53 in the genesis of melanoma. Curr. Biol. 15, 249–254 (2005).
Cagan, R. L., Zon, L. I. & White, R. M. Modeling cancer with flies and fish. Dev. Cell 49, 317–324 (2019).
Kirchberger, S., Sturtzel, C., Pascoal, S. & Distel, M. Quo natas, Danio? — Recent progress in modeling cancer in zebrafish. Front. Oncol. 7, 186 (2017).
Letrado, P., de Miguel, I., Lamberto, I., Diez-Martinez, R. & Oyarzabal, J. Zebrafish: speeding up the cancer drug discovery process. Cancer Res. 78, 6048–6058 (2018).
Vogelstein, B. et al. Cancer genome landscapes. Science 339, 1546–1558 (2013).
Vogelstein, B. & Kinzler, K. W. The path to cancer –three strikes and you’re out. N. Engl. J. Med. 373, 1895–1898 (2015).
McConnell, A. M. et al. Neural crest state activation in NRAS driven melanoma, but not in NRAS-driven melanocyte expansion. Dev. Biol. 449, 107–114 (2019).
Pea, A., Hruban, R. H. & Wood, L. D. Genetics of pancreatic neuroendocrine tumors: implications for the clinic. Expert Rev. Gastroenterol. Hepatol. 9, 1407–1419 (2015).
Berghmans, S. et al. tp53 mutant zebrafish develop malignant peripheral nerve sheath tumors. Proc. Natl Acad. Sci. USA 102, 407–412 (2005).
Ceol, C. J. et al. The histone methyltransferase SETDB1 is recurrently amplified in melanoma and accelerates its onset. Nature 471, 513–517 (2011).
Kendall, G. C. et al. PAX3-FOXO1 transgenic zebrafish models identify HES3 as a mediator of rhabdomyosarcoma tumorigenesis. eLife 7, e33800 (2018).
Langenau, D. M. et al. Effects of RAS on the genesis of embryonal rhabdomyosarcoma. Genes Dev. 21, 1382–1395 (2007).
Sanchez-Rivera, F. J. & Jacks, T. Applications of the CRISPR-Cas9 system in cancer biology. Nat. Rev. Cancer 15, 387–395 (2015).
Hwang, W. Y. et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat. Biotechnol. 31, 227–229 (2013). This study reports the first use of CRISPR in zebrafish.
Kawakami, K. et al. A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev. Cell 7, 133–144 (2004).
Heppt, M. V. et al. Prognostic significance of BRAF and NRAS mutations in melanoma: a German study from routine care. BMC Cancer 17, 536 (2017).
Shain, A. H. et al. The genetic evolution of melanoma from precursor lesions. N. Engl. J. Med. 373, 1926–1936 (2015).
Burns, M. A. et al. Hedgehog pathway mutations drive oncogenic transformation in high-risk T-cell acute lymphoblastic leukemia. Leukemia 32, 2126–2137 (2018).
Callahan, S. J. et al. Cancer modeling by transgene electroporation in adult zebrafish (TEAZ). Dis. Model. Mech. 11, (2018). This article shows mosaic transgenesis in adult zebrafish through electroporation of plasmid DNA.
White, R. M. et al. Transparent adult zebrafish as a tool for in vivo transplantation analysis. Cell Stem Cell 2, 183–189 (2008). This article reports development of the optically clear casper zebrafish strain often used for transplantation studies.
Weintraub, A. All eyes on zebrafish. Lab. Anim. 46, 323–326 (2017).
Smith, A. C. et al. High-throughput cell transplantation establishes that tumor-initiating cells are abundant in zebrafish T-cell acute lymphoblastic leukemia. Blood 115, 3296–3303 (2010).
Blackburn, J. S. et al. Clonal evolution enhances leukemia-propagating cell frequency in T cell acute lymphoblastic leukemia through Akt/mTORC1 pathway activation. Cancer Cell 25, 366–378 (2014).
Tang, Q. et al. Optimized cell transplantation using adult rag2 mutant zebrafish. Nat. Methods 11, 821–824 (2014). This article reports allotransplantation of zebrafish tumours in immunocompromised recipients.
Tang, Q. et al. Imaging tumour cell heterogeneity following cell transplantation into optically clear immune-deficient zebrafish. Nat. Commun. 7, 10358 (2016).
Hayes, M. N. et al. Vangl2/RhoA signaling pathway regulates stem cell self-renewal programs and growth in rhabdomyosarcoma. Cell Stem Cell 22, 414–427.e6 (2018).
Ignatius, M. S. et al. The NOTCH1/SNAIL1/MEF2C pathway regulates growth and self-renewal in embryonal rhabdomyosarcoma. Cell Rep. 19, 2304–2318 (2017).
Moore, J. C. et al. Single-cell imaging of normal and malignant cell engraftment into optically clear prkdc-null SCID zebrafish. J. Exp. Med. 213, 2575–2589 (2016).
Tenente, I. M. et al. Myogenic regulatory transcription factors regulate growth in rhabdomyosarcoma. eLife 6, e19214 (2017).
Li, P. et al. Epoxyeicosatrienoic acids enhance embryonic haematopoiesis and adult marrow engraftment. Nature 523, 468–471 (2015).
Tamplin, O. J. et al. Hematopoietic stem cell arrival triggers dynamic remodeling of the perivascular niche. Cell 160, 241–252 (2015).
Heilmann, S. et al. A quantitative system for studying metastasis using transparent zebrafish. Cancer Res. 75, 4272–4282 (2015).
Zhang, M. et al. Adipocyte-derived lipids mediate melanoma progression via FATP proteins. Cancer Discov. 8, 1006–1025 (2018).
Nicoli, S., Ribatti, D., Cotelli, F. & Presta, M. Mammalian tumor xenografts induce neovascularization in zebrafish embryos. Cancer Res. 67, 2927–2931 (2007).
Haldi, M., Ton, C., Seng, W. L. & McGrath, P. Human melanoma cells transplanted into zebrafish proliferate, migrate, produce melanin, form masses and stimulate angiogenesis in zebrafish. Angiogenesis 9, 139–151 (2006).
Topczewska, J. M. et al. Embryonic and tumorigenic pathways converge via Nodal signaling: role in melanoma aggressiveness. Nat. Med. 12, 925–932 (2006).
Mercatali, L. et al. Development of a patient-derived xenograft (PDX) of breast cancer bone metastasis in a zebrafish model. Int. J. Mol. Sci. 17, 1375 (2016).
Bentley, V. L. et al. Focused chemical genomics using zebrafish xenotransplantation as a pre-clinical therapeutic platform for T-cell acute lymphoblastic leukemia. Haematologica 100, 70–76 (2015).
Lin, J. et al. A clinically relevant in vivo zebrafish model of human multiple myeloma to study preclinical therapeutic efficacy. Blood 128, 249–252 (2016).
Chapman, A. et al. Heterogeneous tumor subpopulations cooperate to drive invasion. Cell Rep. 8, 688–695 (2014).
Renshaw, S. A. & Trede, N. S. A model 450 million years in the making: zebrafish and vertebrate immunity. Dis. Model. Mech. 5, 38–47 (2012).
Langenau, D. M. et al. In vivo tracking of T cell development, ablation, and engraftment in transgenic zebrafish. Proc. Natl Acad. Sci. USA 101, 7369–7374 (2004).
Tang, Q. et al. Dissecting hematopoietic and renal cell heterogeneity in adult zebrafish at single-cell resolution using RNA sequencing. J. Exp. Med. 214, 2875–2887 (2017).
Trede, N. S., Langenau, D. M., Traver, D., Look, A. T. & Zon, L. I. The use of zebrafish to understand immunity. Immunity 20, 367–379 (2004).
Willett, C. E., Cortes, A., Zuasti, A. & Zapata, A. G. Early hematopoiesis and developing lymphoid organs in the zebrafish. Dev. Dyn. 214, 323–336 (1999).
Konantz, M. et al. Zebrafish xenografts as a tool for in vivo studies on human cancer. Ann. NY Acad. Sci. 1266, 124–137 (2012).
He, S. et al. Neutrophil-mediated experimental metastasis is enhanced by VEGFR inhibition in a zebrafish xenograft model. J. Pathol. 227, 431–445 (2012).
Visvader, J. E. & Lindeman, G. J. Cancer stem cells: current status and evolving complexities. Cell Stem Cell 10, 717–728 (2012).
Stoletov, K., Montel, V., Lester, R. D., Gonias, S. L. & Klemke, R. High-resolution imaging of the dynamic tumor cell vascular interface in transparent zebrafish. Proc. Natl Acad. Sci. USA 104, 17406–17411 (2007).
Traver, D. et al. Transplantation and in vivo imaging of multilineage engraftment in zebrafish bloodless mutants. Nat. Immunol. 4, 1238–1246 (2003).
Traver, D. et al. Effects of lethal irradiation in zebrafish and rescue by hematopoietic cell transplantation. Blood 104, 1298–1305 (2004).
Larsen, E. C. et al. Dexamethasone and high-dose methotrexate improve outcome for children and young adults with high-risk B-acute lymphoblastic leukemia: a report from Children’s Oncology Group Study AALL0232. J. Clin. Oncol. 34, 2380–2388 (2016).
Matsuda, M. et al. Human NK cell development in hIL-7 and hIL-15 knockin NOD/SCID/IL2rgKO mice. Life Sci. Alliance 2, 201800195 (2019).
Herndler-Brandstetter, D. et al. Humanized mouse model supports development, function, and tissue residency of human natural killer cells. Proc. Natl Acad. Sci. USA 114, E9626–E9634 (2017). This article reports engineering of NSG mice towards humanized models and more robust PDX support.
Dang, M., Henderson, R. E., Garraway, L. A. & Zon, L. I. Long-term drug administration in the adult zebrafish using oral gavage for cancer preclinical studies. Dis. Model. Mech. 9, 811–820 (2016). This study administers a drug in adult zebrafish through intraperitoneal injection and oral gavage.
Samaee, S. M., Seyedin, S. & Varga, Z. M. An affordable intraperitoneal injection setup for juvenile and adult zebrafish. Zebrafish 14, 77–79 (2017).
Usai, A. et al. A model of zebrafish avatar for co-clinical trials. Cancers 12, 677 (2019). This article reports preliminary results of zebrafish co-clinical trial NCT03668418.
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Goessling, W., North, T. E. & Zon, L. I. Ultrasound biomicroscopy permits in vivo characterization of zebrafish liver tumors. Nat. Methods 4, 551–553 (2007).
Jin, Y. et al. Comparison of efficacy and toxicity of bevacizumab, endostar and apatinib in transgenic and human lung cancer xenograft zebrafish model. Sci. Rep. 8, 15837 (2018).
Chen, C. et al. A multiplex preclinical model for adenoid cystic carcinoma of the salivary gland identifies regorafenib as a potential therapeutic drug. Sci. Rep. 7, 11410 (2017).
Wu, J. Q. et al. Patient-derived xenograft in zebrafish embryos: a new platform for translational research in gastric cancer. J. Exp. Clin. Cancer Res. 36, 160 (2017).
Wang, L. et al. Patient-derived heterogeneous xenograft model of pancreatic cancer using zebrafish larvae as hosts for comparative drug assessment. J. Vis. Exp. 146, e59507 (2019). This is a method guide for the drug testing of zPDXs in larvae.
Ikonomopoulou, M. P. et al. Gomesin inhibits melanoma growth by manipulating key signaling cascades that control cell death and proliferation. Sci. Rep. 8, 11519 (2018).
Wang, G. et al. The novel autophagy inhibitor elaiophylin exerts antitumor activity against multiple myeloma with mutant TP53 in part through endoplasmic reticulum stress-induced apoptosis. Cancer Biol. Ther. 18, 584–595 (2017).
von Massenhausen, A. et al. Targeting DDR2 in head and neck squamous cell carcinoma with dasatinib. Int. J. Cancer 139, 2359–2369 (2016).
Ochoa-Alvarez, J. A. et al. Antibody and lectin target podoplanin to inhibit oral squamous carcinoma cell migration and viability by distinct mechanisms. Oncotarget 6, 9045–9060 (2015).
Ghotra, V. P. et al. SYK is a candidate kinase target for the treatment of advanced prostate cancer. Cancer Res. 75, 230–240 (2015).
van der Ent, W. et al. Ewing sarcoma inhibition by disruption of EWSR1-FLI1 transcriptional activity and reactivation of p53. J. Pathol. 233, 415–424 (2014).
Tan, D. S. et al. Bosutinib inhibits migration and invasion via ACK1 in KRAS mutant non-small cell lung cancer. Mol. Cancer 13, 13 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01858168 (2013).
Liu, P. H. et al. An IRAK1-PIN1 signalling axis drives intrinsic tumour resistance to radiation therapy. Nat. Cell Biol. 21, 203–213 (2019).
Smith, M. P. et al. Effect of SMURF2 targeting on susceptibility to MEK inhibitors in melanoma. J. Natl Cancer Inst. 105, 33–46 (2013).
Yoganantharjah, P. & Gibert, Y. The use of the zebrafish model to aid in drug discovery and target validation. Curr. Top. Med. Chem. 17, 2041–2055 (2017).
Sarmah, S. & Marrs, J. A. Zebrafish as a vertebrate model system to evaluate effects of environmental toxicants on cardiac development and function. Int. J. Mol. Sci. 17, 2123 (2016).
Chakraborty, C., Sharma, A. R., Sharma, G. & Lee, S. S. Zebrafish: a complete animal model to enumerate the nanoparticle toxicity. J. Nanobiotech. 14, 65 (2016).
Kanungo, J., Cuevas, E., Ali, S. F. & Paule, M. G. Zebrafish model in drug safety assessment. Curr. Pharm. Des. 20, 5416–5429 (2014).
Chakravarthy, S., Sadagopan, S., Nair, A. & Sukumaran, S. K. Zebrafish as an in vivo high-throughput model for genotoxicity. Zebrafish 11, 154–166 (2014).
Peterson, R. T. & Macrae, C. A. Systematic approaches to toxicology in the zebrafish. Annu. Rev. Pharmacol. Toxicol. 52, 433–453 (2012).
Boumahdi, S. & de Sauvage, F. J. The great escape: tumour cell plasticity in resistance to targeted therapy. Nat. Rev. Drug Discov. 19, 39–56 (2020).
Yao, Y. et al. Patient-derived organoids predict chemoradiation responses of locally advanced rectal cancer. Cell Stem Cell 26, 17–26.e6 (2020). This article reports the use of organoids to assess drug response in the neoadjuvant setting.
Fazio, M. & Zon, L. I. Fishing for answers in precision cancer medicine. Proc. Natl Acad. Sci. USA 114, 10306–10308 (2017).
Baeten, J. T. & de Jong, J. L. O. Genetic models of leukemia in zebrafish. Front. Cell Dev. Biol. 6, 115 (2018).
Kurata, M. et al. Highly multiplexed genome engineering using CRISPR/Cas9 gRNA arrays. PLoS one 13, e0198714 (2018).
Zhang, Y. et al. A gRNA-tRNA array for CRISPR-Cas9 based rapid multiplexed genome editing in Saccharomyces cerevisiae. Nat. Commun. 10, 1053 (2019).
Kuzu, O. F., Nguyen, F. D., Noory, M. A. & Sharma, A. Current state of animal (mouse) modeling in melanoma research. Cancer Growth Metastasis 8 (Suppl. 1), 81–94 (2015).
Xue, W. et al. CRISPR-mediated direct mutation of cancer genes in the mouse liver. Nature 514, 380–384 (2014).
Sanchez-Rivera, F. J. et al. Rapid modelling of cooperating genetic events in cancer through somatic genome editing. Nature 516, 428–431 (2014). This article reports mosaic transgenesis in mouse cancer models.
National Research Council. Guide for the Care and Use of Laboratory Animals 8th edn (National Academies Press, 2011)
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT03668418 (2018).
Vlachogiannis, G. et al. Patient-derived organoids model treatment response of metastatic gastrointestinal cancers. Science 359, 920–926 (2018).
Tiriac, H. et al. Organoid profiling identifies common responders to chemotherapy in pancreatic cancer. Cancer Discov. 8, 1112–1129 (2018).
Ganesh, K. et al. A rectal cancer organoid platform to study individual responses to chemoradiation. Nat. Med. 25, 1607–1614 (2019).
Ben-David, U., Beroukhim, R. & Golub, T. R. Genomic evolution of cancer models: perils and opportunities. Nat. Rev. Cancer 19, 97–109 (2019).
Wang, K. et al. Patient-derived xenotransplants can recapitulate the genetic driver landscape of acute leukemias. Leukemia 31, 151–158 (2017).
Ben-David, U. et al. Patient-derived xenografts undergo mouse-specific tumor evolution. Nat. Genet. 49, 1567–1575 (2017). This article shows the evolution of mouse PDXs compared with donor patients.
Ooft, S. N. et al. Patient-derived organoids can predict response to chemotherapy in metastatic colorectal cancer patients. Sci. Transl Med. 11, eaay2574 (2019).
Hirata, E. & Sahai, E. Tumor microenvironment and differential responses to therapy. Cold Spring Harb. Perspect. Med. https://doi.org/10.1101/cshperspect.a026781 (2017).
Binnewies, M. et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 24, 541–550 (2018).
Vargas, R. et al. Case study: patient-derived clear cell adenocarcinoma xenograft model longitudinally predicts treatment response. NPJ Precis. Oncol. 2, 14 (2018).
Nardella, C., Lunardi, A., Patnaik, A., Cantley, L. C. & Pandolfi, P. P. The APL paradigm and the ‘co-clinical trial’ project. Cancer Discov. 1, 108–116 (2011).
Acknowledgements
L.I.Z. is supported by the Melanoma Research Alliance and US National Institutes of Health (grants P01CA163222 and R01 CA103846). M.F. was supported by Boehringer Ingelheim Fonds. D.M.L. is funded by the US National Institutes of Health (grants R01CA154923, R01CA211734, R01CA215118, R01CA226926 and R24OD016761) and the MGH Research Scholars Program. Y.C. is supported by the Alex Lemonade Stand Foundation.
Author information
Authors and Affiliations
Contributions
M.F. and J.A. researched data for the article, substantially contributed to discussion of the content, wrote the manuscript, and reviewed and edited the manuscript before submission. Y.C. researched data for the article and wrote the manuscript. D.L. substantially contributed to discussion of the content, and reviewed and edited the manuscript before submission. L.I.Z. substantially contributed to discussion of the content, wrote the manuscript, and reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
L.I.Z. is a founder and stockholder of Fate Therapeutics Inc., Scholar Rock and Camp4 Therapeutics Inc., and is a scientific adviser for Stemgent. The other authors declare no competing interests.
Additional information
Peer review information
Nature Reviews Cancer thanks M. G. Ferreira, Z. Gong, R. Stewart and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- One-cell stage
-
The zebrafish embryonic stage where the egg has been fertilized by sperm, but before the first cell division.
- Nevi
-
Benign skin lesions of melanocytes, also known as moles, which are thought to be senescent.
- Tol2 transposon
-
Tol2 is an active transposon derived from the medaka fish and is used as a gene delivery method.
- U6 promoter
-
An RNA polymerase III type 3 promoter commonly used to drive short hairpin RNAs or guide RNAs.
- Casper zebrafish strain
-
An optically clear strain of zebrafish obtained by crossing the roy (mpv17−/−) and nacre (mitfa−/−) mutants, which results in the lack of iridophores and melanocytes, respectively.
- NSG mice
-
Genetically engineered immunocompromised strain of mice widely used as recipients for engraftment of human primary cells or tumours (for example, patient-derived xenografts).
- RECIST criteria
-
A set of published rules that define when patients with cancer improve (‘respond’), stay the same (‘stable’) or worsen (‘progression’) during treatments.
- Hydrodynamic injection
-
Rapid injection of a relatively large volume of DNA solution, which is used for gene delivery in mouse livers.
Rights and permissions
About this article
Cite this article
Fazio, M., Ablain, J., Chuan, Y. et al. Zebrafish patient avatars in cancer biology and precision cancer therapy. Nat Rev Cancer 20, 263–273 (2020). https://doi.org/10.1038/s41568-020-0252-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-020-0252-3
This article is cited by
-
Zebrafish tumour xenograft models: a prognostic approach to epithelial ovarian cancer
npj Precision Oncology (2024)
-
Zebrafish live imaging: a strong weapon in anticancer drug discovery and development
Clinical and Translational Oncology (2024)
-
Applications of lung cancer organoids in precision medicine: from bench to bedside
Cell Communication and Signaling (2023)
-
The tumor ecosystem in head and neck squamous cell carcinoma and advances in ecotherapy
Molecular Cancer (2023)
-
Alternative lengthening of telomeres (ALT) cells viability is dependent on C-rich telomeric RNAs
Nature Communications (2023)