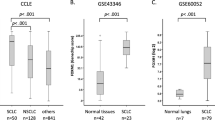
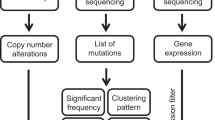
Abstract
Small cell lung cancer (SCLC) is an exceptionally lethal malignancy for which more effective therapies are urgently needed. Several lines of evidence, from SCLC primary human tumours, patient-derived xenografts, cancer cell lines and genetically engineered mouse models, appear to be converging on a new model of SCLC subtypes defined by differential expression of four key transcription regulators: achaete-scute homologue 1 (ASCL1; also known as ASH1), neurogenic differentiation factor 1 (NeuroD1), yes-associated protein 1 (YAP1) and POU class 2 homeobox 3 (POU2F3). In this Perspectives article, we review and synthesize these recent lines of evidence and propose a working nomenclature for SCLC subtypes defined by relative expression of these four factors. Defining the unique therapeutic vulnerabilities of these subtypes of SCLC should help to focus and accelerate therapeutic research, leading to rationally targeted approaches that may ultimately improve clinical outcomes for patients with this disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
07 June 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
27 June 2019
An author correction to Supplementary Table 1 was made, but the original file was not updated. We have now updated Supplementary Table 1 in the original article.
References
Gazdar, A. F., Bunn, P. A. & Minna, J. D. Small-cell lung cancer: what we know, what we need to know and the path forward. Nat. Rev. Cancer 17, 725–737 (2017).
Sabari, J. K. et al. Unravelling the biology of SCLC: implications for therapy. Nat. Rev. Clin. Oncol. 14, 549–561 (2017).
Hann, C. L. et al. in Cancer Principles & Practice of Oncology Ch. 49 (eds DeVita, V. T., Lawrence, T. S. & Rosenberg, S. A.) 671–700 (Wolters Kluwer, 2019).
Horn, L. et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N. Engl. J. Med. 379, 2220–2229 (2018).
von Pawel, J. et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J. Clin. Oncol. 17, 658–667 (1999).
Ready, N. et al. Third-line nivolumab monotherapy in recurrent SCLC: CheckMate 032. J. Thorac. Oncol. 14, 237–244 (2018).
Jordan, E. J. et al. Prospective comprehensive molecular characterization of lung adenocarcinomas for efficient patient matching to approved and emerging therapies. Cancer Discov. 7, 596–609 (2017).
Gazdar, A. F. et al. Establishment of continuous, clonable cultures of small-cell carcinoma of lung which have amine precursor uptake and decarboxylation cell properties. Cancer Res. 40, 3502–3507 (1980).
Carney, D. N. et al. Establishment and identification of small cell lung cancer cell lines having classic and variant features. Cancer Res. 45, 2913–2923 (1985).
Gazdar, A. F. et al. Characterization of variant subclasses of cell lines derived from small cell lung cancer having distinctive biochemical, morphological, and growth properties. Cancer Res. 45, 2924–2930 (1985).
Rudin, C. M. et al. Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat. Genet. 44, 1111–1116 (2012).
Peifer, M. et al. Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Nat. Genet. 44, 1104–1110 (2012).
George, J. et al. Comprehensive genomic profiles of small cell lung cancer. Nature 524, 47–53 (2015).
Poirier, J. T. et al. Selective tropism of Seneca Valley virus for variant subtype small cell lung cancer. J. Natl Cancer Inst. 105, 1059–1065 (2013).
Neptune, E. R. et al. Targeted disruption of NeuroD, a proneural basic helix-loop-helix factor, impairs distal lung formation and neuroendocrine morphology in the neonatal lung. J. Biol. Chem. 283, 21160–21169 (2008).
Borges, M. et al. An achaete-scute homologue essential for neuroendocrine differentiation in the lung. Nature 386, 852–855 (1997).
Ito, T. et al. Basic helix-loop-helix transcription factors regulate the neuroendocrine differentiation of fetal mouse pulmonary epithelium. Development 127, 3913–3921 (2000).
Poirier, J. T. et al. DNA methylation in small cell lung cancer defines distinct disease subtypes and correlates with high expression of EZH2. Oncogene 34, 5869–5878 (2015).
The Cancer Genome Atlas Research Network. Comprehensive genomic characterization of squamous cell lung cancers. Nature 489, 519–525 (2012).
Christensen, C. L. et al. Targeting transcriptional addictions in small cell lung cancer with a covalent CDK7 inhibitor. Cancer Cell 26, 909–922 (2014).
Lan, M. S. & Breslin, M. B. Structure, expression, and biological function of INSM1 transcription factor in neuroendocrine differentiation. FASEB J. 23, 2024–2033 (2009).
Fujino, K. et al. Insulinoma-associated protein 1 is a crucial regulator of neuroendocrine differentiation in lung cancer. Am. J. Pathol. 185, 3164–3177 (2015).
Gierl, M. S. et al. The zinc-finger factor Insm1 (IA-1) is essential for the development of pancreatic beta cells and intestinal endocrine cells. Genes Dev. 20, 2465–2478 (2006).
Wildner, H. et al. Insm1 (IA-1) is a crucial component of the transcriptional network that controls differentiation of the sympatho-adrenal lineage. Development 135, 473–481 (2008).
Borromeo, M. D. et al. ASCL1 and NEUROD1 reveal heterogeneity in pulmonary neuroendocrine tumors and regulate distinct genetic programs. Cell Rep. 16, 1259–1272 (2016).
Gratton, M. O. et al. Hes6 promotes cortical neurogenesis and inhibits Hes1 transcription repression activity by multiple mechanisms. Mol. Cell. Biol. 23, 6922–6935 (2003).
Wooten, D. et al. Small cell lung cancer subtypes identified by systems-level modeling of transcription factor networks. Preprint at bioRxiv https://www.biorxiv.org/content/10.1101/506402v2 (2018).
Meuwissen, R. et al. Induction of small cell lung cancer by somatic inactivation of both Trp53 and Rb1 in a conditional mouse model. Cancer Cell 4, 181–189 (2003).
Sutherland, K. D. et al. Cell of origin of small cell lung cancer: inactivation of Trp53 and Rb1 in distinct cell types of adult mouse lung. Cancer Cell 19, 754–764 (2011).
Yang, D. et al. Intertumoral heterogeneity in SCLC is influenced by the cell type of origin. Cancer Discov. 8, 1316–1331 (2018).
Schaffer, B. E. et al. Loss of p130 accelerates tumor development in a mouse model for human small-cell lung carcinoma. Cancer Res. 70, 3877–3883 (2010).
Mollaoglu, G. et al. MYC drives progression of small cell lung cancer to a variant neuroendocrine subtype with vulnerability to aurora kinase inhibition. Cancer Cell 31, 270–285 (2017).
Little, C. D. et al. Amplification and expression of the c-myc oncogene in human lung cancer cell lines. Nature 306, 194–196 (1983).
McColl, K. et al. Reciprocal expression of INSM1 and YAP1 defines subgroups in small cell lung cancer. Oncotarget 8, 73745–73756 (2017).
Huang, Y. H. et al. POU2F3 is a master regulator of a tuft cell-like variant of small cell lung cancer. Genes Dev. 32, 915–928 (2018).
Yamashita, J. et al. Skn-1a/Pou2f3 functions as a master regulator to generate Trpm5-expressing chemosensory cells in mice. PLOS ONE 12, e0189340 (2017).
Zhang, W. et al. Small cell lung cancer tumors and preclinical models display heterogeneity of neuroendocrine phenotypes. Transl Lung Cancer Res. 7, 32–49 (2018).
The Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 511, 543–550 (2014).
Lim, J. S. et al. Intratumoural heterogeneity generated by Notch signalling promotes small-cell lung cancer. Nature 545, 360–364 (2017).
Hodgkinson, C. L. et al. Tumorigenicity and genetic profiling of circulating tumor cells in small-cell lung cancer. Nat. Med. 20, 897–903 (2014).
Drapkin, B. J. et al. Genomic and functional fidelity of small cell lung cancer patient-derived xenografts. Cancer Discov. 8, 600–615 (2018).
Calbo, J. et al. A functional role for tumor cell heterogeneity in a mouse model of small cell lung cancer. Cancer Cell 19, 244–256 (2011).
Kwon, M. C. et al. Paracrine signaling between tumor subclones of mouse SCLC: a critical role of ETS transcription factor Pea3 in facilitating metastasis. Genes Dev. 29, 1587–1592 (2015).
Williamson, S. C. et al. Vasculogenic mimicry in small cell lung cancer. Nat. Commun. 7, 13322 (2016).
Saunders, L. R. et al. A DLL3-targeted antibody-drug conjugate eradicates high-grade pulmonary neuroendocrine tumor-initiating cells in vivo. Sci. Transl Med. 7, 302ra136 (2015).
Cardnell, R. J. et al. Protein expression of TTF1 and cMYC define distinct molecular subgroups of small cell lung cancer with unique vulnerabilities to aurora kinase inhibition, DLL3 targeting, and other targeted therapies. Oncotarget 8, 73419–73432 (2017).
Jia, D. et al. Crebbp loss drives small cell lung cancer and increases sensitivity to HDAC inhibition. Cancer Discov. 8, 1422–1437 (2018).
Takagi, S. et al. LSD1 inhibitor T-3775440 inhibits SCLC cell proliferation by disrupting LSD1 interactions with SNAG domain proteins INSM1 and GFI1B. Cancer Res. 77, 4652–4662 (2017).
Augert, A. et al. Targeting NOTCH activation in small cell lung cancer through LSD1 inhibition. Sci. Signal. 12, eaau2922 (2019).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02034123 (2019).
Rudin, C. M. et al. Phase I clinical study of Seneca Valley Virus (SVV-001), a replication-competent picornavirus, in advanced solid tumors with neuroendocrine features. Clin. Cancer Res. 17, 888–895 (2011).
Belani, C. P. et al. Vismodegib or cixutumumab in combination with standard chemotherapy for patients with extensive-stage small cell lung cancer: a trial of the ECOG-ACRIN Cancer Research Group (E1508). Cancer 122, 2371–2378 (2016).
Sen, T., Gay, C. M. & Byers, L. A. Targeting DNA damage repair in small cell lung cancer and the biomarker landscape. Transl Lung Cancer Res. 7, 50–68 (2018).
Sen, T. et al. CHK1 inhibition in small-cell lung cancer produces single-agent activity in biomarker-defined disease subsets and combination activity with cisplatin or olaparib. Cancer Res. 77, 3870–3884 (2017).
Udyavar, A. R. et al. Novel hybrid phenotype revealed in small cell lung cancer by a transcription factor network model that can explain tumor heterogeneity. Cancer Res. 77, 1063–1074 (2017).
Hellmann, M. D. et al. Tumor mutational burden and efficacy of nivolumab monotherapy and in combination with ipilimumab in small-cell lung cancer. Cancer Cell 33, 853–861 (2018).
Ott, P. A. et al. Pembrolizumab in patients with extensive-stage small-cell lung cancer: results from the phase Ib KEYNOTE-028 Study. J. Clin. Oncol. 35, 3823–3829 (2017).
Sen, T. et al. Targeting DNA damage response promotes anti-tumor immunity through STING-mediated T cell activation in small cell lung cancer. Cancer Discov. https://doi.org/10.1158/2159-8290.CD-18-1020 (2019).
Skoulidis, F. et al. STK11/LKB1 mutations and PD-1 inhibitor resistance in KRAS-mutant lung adenocarcinoma. Cancer Discov. 8, 822–835 (2018).
Sequist, L. V. et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci. Transl Med. 3, 75ra26 (2011).
Niederst, M. J. et al. RB loss in resistant EGFR mutant lung adenocarcinomas that transform to small-cell lung cancer. Nat. Commun. 6, 6377 (2015).
Mu, P. et al. SOX2 promotes lineage plasticity and antiandrogen resistance in TP53- and RB1-deficient prostate cancer. Science 355, 84–88 (2017).
Ku, S. Y. et al. Rb1 and Trp53 cooperate to suppress prostate cancer lineage plasticity, metastasis, and antiandrogen resistance. Science 355, 78–83 (2017).
Rekhtman, N. et al. Next-generation sequencing of pulmonary large cell neuroendocrine carcinoma reveals small cell carcinoma-like and non-small cell carcinoma-like subsets. Clin. Cancer Res. 22, 3618–3629 (2016).
George, J. et al. Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. Nat. Commun. 9, 1048 (2018).
Travis, W. D. et al. The 2015 World Health Organization Classification of Lung Tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J. Thorac Oncol. 10, 1243–1260 (2015).
Guinee, D. G. et al. The spectrum of immunohistochemical staining of small-cell lung carcinoma in specimens from transbronchial and open-lung biopsies. Am. J. Clin. Pathol. 102, 406–414 (1994).
Junker, K., Wiethege, T. & Muller, K. M. Pathology of small-cell lung cancer. J. Cancer Res. Clin. Oncol. 126, 361–368 (2000).
Kim, Y. H. et al. Combined microarray analysis of small cell lung cancer reveals altered apoptotic balance and distinct expression signatures of MYC family gene amplification. Oncogene 25, 130–138 (2006).
Barretina, J. et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 483, 603–607 (2012).
Acknowledgements
The authors thank N. Rekhtman for insightful comment regarding pathological criteria for small cell lung cancer diagnosis. This work was supported by grants from the US National Institutes of Health, including U24CA213274 (C.M.R., J.T.P., A.D., J.D.M. and A.F.G.), R01CA197936 (C.M.R., J.T.P. and C.D.), R01CA207295 (L.A.B.), U01CA213273 (L.A.B., J.V.H. and J.S.), P50CA70907 and U01CA213338 (J.E.J., J.D.M. and A.F.G.), U54CA217450 (J.M.L. and V.Q.), UG1CA233259 (J.M.L.) and R21CA216504 (T.G.O.); by Veterans Affairs Merit Review I01CX001425 (P.P.M.); by a LUNGevity Foundation Career Development Award (J.M.L.); and by Cancer Research UK A27412, A25254 and A20465 (C.D.).
Reviewer information
Nature Reviews Cancer thanks T. Bivona, E. Brambilla and other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.M.R. and J.T.P. researched data for the article, made substantial contributions to the discussion of content, wrote the article and reviewed or edited the article before submission. The other authors all made substantial contributions to the discussion of content and reviewed or edited the article before submission.
Corresponding authors
Ethics declarations
Competing interests
C.M.R. has consulted for AbbVie, Amgen, Ascentage, AstraZeneca, BMS, Celgene, Daiichi Sankyo, Genentech/Roche, Ipsen, Loxo and PharmaMar; is on the scientific advisory board for Elucida and Harpoon; and receives research funding from Daiichi Sankyo. L.A.B. has consulted for AbbVie, AstraZeneca, BerGenBio, Genmab and PharmaMar and receives research support from AbbVie, AstraZeneca, Genmab and Tolero. C.D. has consulted for AstraZeneca and Merck and receives research funding from AstraZeneca, Epigene, Amgen, FLX Bio, Menarini and Angel. J.V.H. has consulted for AstraZeneca, Boehringer Ingelheim, Exelixis, Genentech, GlaxoSmithKline, Guardant, Hengrui, Lilly, Novartis, Spectrum, EMD Serono and Synta; has received research support from AstraZeneca, Bayer, GlaxoSmithKline and Spectrum; and receives royalties and licensing fees from Spectrum. J.M.L. receives research funding from Ipsen and AbbVie. D.M. received research funding from Janssen and Roche. J.D.M. and A.F.G. receive licensing royalties for cell lines from the US National Institutes of Health and the University of Texas Southwestern Medical Center. J.S. receives research funding from AbbVie. C.R.V. is an adviser to KSQ Therapeutics. All other authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Rudin, C.M., Poirier, J.T., Byers, L.A. et al. Molecular subtypes of small cell lung cancer: a synthesis of human and mouse model data. Nat Rev Cancer 19, 289–297 (2019). https://doi.org/10.1038/s41568-019-0133-9
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-019-0133-9
This article is cited by
-
C-Myc protein expression indicates unfavorable clinical outcome in surgically resected small cell lung cancer
World Journal of Surgical Oncology (2024)
-
Small cells – big issues: biological implications and preclinical advancements in small cell lung cancer
Molecular Cancer (2024)
-
Molecular profile of bladder cancer progression to clinically aggressive subtypes
Nature Reviews Urology (2024)
-
Genetically-engineered mouse models of small cell lung cancer: the next generation
Oncogene (2024)
-
Dynamic phenotypic reprogramming and chemoresistance induced by lung fibroblasts in small cell lung cancer
Scientific Reports (2024)