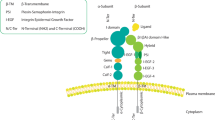
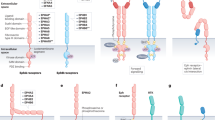
Abstract
Cell adhesion to the extracellular matrix is fundamental to tissue integrity and human health. Integrins are the main cellular adhesion receptors that through multifaceted roles as signalling molecules, mechanotransducers and key components of the cell migration machinery are implicated in nearly every step of cancer progression from primary tumour development to metastasis. Altered integrin expression is frequently detected in tumours, where integrins have roles in supporting oncogenic growth factor receptor (GFR) signalling and GFR-dependent cancer cell migration and invasion. In addition, integrins determine colonization of metastatic sites and facilitate anchorage-independent survival of circulating tumour cells. Investigations describing integrin engagement with a growing number of versatile cell surface molecules, including channels, receptors and secreted proteins, continue to lead to the identification of novel tumour-promoting pathways. Integrin-mediated sensing, stiffening and remodelling of the tumour stroma are key steps in cancer progression supporting invasion, acquisition of cancer stem cell characteristics and drug resistance. Given the complexity of integrins and their adaptable and sometimes antagonistic roles in cancer cells and the tumour microenvironment, therapeutic targeting of these receptors has been a challenge. However, novel approaches to target integrins and antagonism of specific integrin subunits in stringently stratified patient cohorts are emerging as potential ways forward.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
31 January 2019
In the originally published article, pertuzumab was incorrectly described as an anti-PI3K therapy in the section ‘Integrins in anticancer therapy’. The sentence should read ‘In mouse mammary tumour models, increased collagen levels and increased β1 integrin and SRC activity have been demonstrated to accompany, and promote, combined resistance to anti-human epidermal growth factor receptor 2 (HER2; also known as ERBB2) (trastuzumab and pertuzumab) and anti-PI3K (buparlisib) therapies164.’ This has now been corrected in all versions of the original article.
References
Humphries, J. D., Byron, A. & Humphries, M. J. Integrin ligands at a glance. J. Cell Sci. 119, 3901–3903 (2006).
Arruda Macêdo, J. K., Fox, J. W. & de Souza Castro, M. Disintegrins from snake venoms and their applications in cancer research and therapy. Curr. Protein Pept. Sci. 16, 532–548 (2015).
Hussein, H. A. M. et al. Beyond RGD: virus interactions with integrins. Arch. Virol. 160, 2669–2681 (2015).
De Franceschi, N., Hamidi, H., Alanko, J., Sahgal, P. & Ivaska, J. Integrin traffic — the update. J. Cell Sci. 128, 839–852 (2015).
Horton, E. R. et al. Definition of a consensus integrin adhesome and its dynamics during adhesion complex assembly and disassembly. Nat. Cell Biol. 17, 1577–1587 (2015).
Horton, E. R. et al. The integrin adhesome network at a glance. J. Cell Sci. 129, 4159–4163 (2016).
Zaidel-Bar, R., Itzkovitz, S., Ma’ayan, A., Iyengar, R. & Geiger, B. Functional atlas of the integrin adhesome. Nat. Cell Biol. 9, 858–867 (2007).
Winograd-Katz, S. E., Fässler, R., Geiger, B. & Legate, K. R. The integrin adhesome: from genes and proteins to human disease. Nat. Rev. Mol. Cell Biol. 15, 273–288 (2014).
Seguin, L., Desgrosellier, J. S., Weis, S. M. & Cheresh, D. A. Integrins and cancer: regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 25, 234–240 (2015).
Hamidi, H., Pietilä, M. & Ivaska, J. The complexity of integrins in cancer and new scopes for therapeutic targeting. Br. J. Cancer 115, 1017–1023 (2016).
Raab-Westphal, S., Marshall, J. F. & Goodman, S. L. Integrins as therapeutic targets: successes and cancers. Cancers 9, E110 (2017).
Parvani, J. G., Galliher-Beckley, A. J., Schiemann, B. J. & Schiemann, W. P. Targeted inactivation of β1 integrin induces β3 integrin switching, which drives breast cancer metastasis by TGF-β. Mol. Biol. Cell 24, 3449–3459 (2013).
van der Flier, A. et al. Endothelial alpha5 and alphav integrins cooperate in remodeling of the vasculature during development. Development 137, 2439–2449 (2010).
White, D. P., Caswell, P. T. & Norman, J. C. alpha v beta3 and alpha5beta1 integrin recycling pathways dictate downstream Rho kinase signaling to regulate persistent cell migration. J. Cell Biol. 177, 515–525 (2007).
Paul, N. R., Jacquemet, G. & Caswell, P. T. Endocytic trafficking of integrins in cell migration. Curr. Biol. 25, 1092 (2015).
Bouvard, D., Pouwels, J., De Franceschi, N. & Ivaska, J. Integrin inactivators: balancing cellular functions in vitro and in vivo. Nat. Rev. Mol. Cell Biol. 14, 430–442 (2013).
Sheldrake, H. M. & Patterson, L. H. Strategies to inhibit tumor associated integrin receptors: rationale for dual and multi-antagonists. J. Med. Chem. 57, 6301–6315 (2014).
Munshi, H. G. & Stack, M. S. Reciprocal interactions between adhesion receptor signaling and MMP regulation. Cancer Metastasis Rev. 25, 45–56 (2006).
Gaggioli, C. et al. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat. Cell Biol. 9, 1392–1400 (2007).
Attieh, Y. & Vignjevic, D. M. The hallmarks of CAFs in cancer invasion. Eur. J. Cell Biol. 95, 493–502 (2016).
Attieh, Y. et al. Cancer-associated fibroblasts lead tumor invasion through integrin-β3-dependent fibronectin assembly. J. Cell Biol. 216, 3509–3520 (2017). This article demonstrates that cancer cell invasion requires not only CAF-mediated contractility of the matrix but also CAF-dependent remodelling of fibronectin by αvβ3 integrin.
Erdogan, B. et al. Cancer-associated fibroblasts promote directional cancer cell migration by aligning fibronectin. J. Cell Biol. 216, 3799–3816 (2017). This study describes a mechanism whereby CAFs align fibronectin fibres within the tumour ECM and, through application of traction forces mediated by α5β1 integrin, promote directional cancer cell migration.
Labernadie, A. et al. A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion. Nat. Cell Biol. 19, 224–237 (2017).
Lambert, A. W., Pattabiraman, D. R. & Weinberg, R. A. Emerging biological principles of metastasis. Cell 168, 670–691 (2017).
Frisch, S. M., Vuori, K., Ruoslahti, E. & Chan-Hui, P. Y. Control of adhesion-dependent cell survival by focal adhesion kinase. J. Cell Biol. 134, 793–799 (1996).
Strilic, B. & Offermanns, S. Intravascular survival and extravasation of tumor cells. Cancer Cell 32, 282–293 (2017).
Knowles, L. M. et al. Integrin αvβ3 and fibronectin upregulate slug in cancer cells to promote clot invasion and metastasis. Cancer Res. 73, 6175–6184 (2013). The authors in this study connect increased fibronectin and active αvβ3 integrin expression to upregulation of the transcription factor SLUG (also known as SNAI2), EMT and fibrin invasion as a mechanism selectively involved in lung metastasis.
Malik, G. et al. Plasma fibronectin promotes lung metastasis by contributions to fibrin clots and tumor cell invasion. Cancer Res. 70, 4327–4334 (2010).
Cagnet, S. et al. Signaling events mediated by α3β1 integrin are essential for mammary tumorigenesis. Oncogene 33, 4286–4295 (2014).
White, D. E. et al. Targeted disruption of beta1-integrin in a transgenic mouse model of human breast cancer reveals an essential role in mammary tumor induction. Cancer Cell 6, 159–170 (2004).
Ramirez, N. E. et al. The α2β1 integrin is a metastasis suppressor in mouse models and human cancer. J. Clin. Invest. 121, 226–237 (2011).
Ramovs, V., Te Molder, L. & Sonnenberg, A. The opposing roles of laminin-binding integrins in cancer. Matrix Biol. 57–58, 213–243 (2017).
Ludlow, A. et al. Characterization of integrin beta6 and thrombospondin-1 double-null mice. J. Cell. Mol. Med. 9, 421–437 (2005).
Moore, K. M. et al. Therapeutic targeting of integrin αvβ6 in breast cancer. J. Natl Cancer Inst. 106, dju169 (2014).
Truong, H. H. et al. β1 integrin inhibition elicits a prometastatic switch through the TGFβ-miR-200-ZEB network in E-cadherin-positive triple-negative breast cancer. Sci. Signal. 7, ra15 (2014).
Ahmed, N. et al. Overexpression of alpha(v)beta6 integrin in serous epithelial ovarian cancer regulates extracellular matrix degradation via the plasminogen activation cascade. Carcinogenesis 23, 237–244 (2002).
Baum, O. et al. Increased invasive potential and up-regulation of MMP-2 in MDA-MB-231 breast cancer cells expressing the beta3 integrin subunit. Int. J. Oncol. 30, 325–332 (2007).
Gu, X., Niu, J., Dorahy, D. J., Scott, R. & Agrez, M. V. Integrin alpha(v)beta6-associated ERK2 mediates MMP-9 secretion in colon cancer cells. Br. J. Cancer 87, 348–351 (2002).
Kesanakurti, D., Chetty, C., Dinh, D. H., Gujrati, M. & Rao, J. S. Role of MMP-2 in the regulation of IL-6/Stat3 survival signaling via interaction with α5β1 integrin in glioma. Oncogene 32, 327–340 (2013).
Yue, J., Zhang, K. & Chen, J. Role of integrins in regulating proteases to mediate extracellular matrix remodeling. Cancer Microenviron. 5, 275–283 (2012).
Roman, J., Ritzenthaler, J. D., Roser-Page, S., Sun, X. & Han, S. alpha5beta1-integrin expression is essential for tumor progression in experimental lung cancer. Am. J. Respir. Cell Mol. Biol. 43, 684–691 (2010).
Tuomi, S. et al. PKCepsilon regulation of an alpha5 integrin-ZO-1 complex controls lamellae formation in migrating cancer cells. Sci. Signal. 2, ra32 (2009).
González-Tarragó, V. et al. Binding of ZO-1 to α5β1 integrins regulates the mechanical properties of α5β1-fibronectin links. Mol. Biol. Cell 28, 1847–1852 (2017).
Barrow-McGee, R. et al. Beta 1-integrin-c-Met cooperation reveals an inside-in survival signalling on autophagy-related endomembranes. Nat. Commun. 7, 11942 (2016).
Alanko, J. et al. Integrin endosomal signalling suppresses anoikis. Nat. Cell Biol. 17, 1412–1421 (2015).
Guo, W. et al. Beta 4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis. Cell 126, 489–502 (2006).
Mai, A. et al. Distinct c-Met activation mechanisms induce cell rounding or invasion through pathways involving integrins, RhoA and HIP1. J. Cell Sci. 127, 1938–1952 (2014).
Novitskaya, V. et al. Integrin α3β1-CD151 complex regulates dimerization of ErbB2 via RhoA. Oncogene 33, 2779–2789 (2014).
Trusolino, L., Bertotti, A. & Comoglio, P. M. A signaling adapter function for alpha6beta4 integrin in the control of HGF-dependent invasive growth. Cell 107, 643–654 (2001).
Desgrosellier, J. S. et al. Integrin αvβ3 drives slug activation and stemness in the pregnant and neoplastic mammary gland. Dev. Cell 30, 295–308 (2014).
Desgrosellier, J. S. et al. An integrin alpha(v)beta(3)-c-Src oncogenic unit promotes anchorage-independence and tumor progression. Nat. Med. 15, 1163–1169 (2009).
Sun, Q. et al. Proapoptotic PUMA targets stem-like breast cancer cells to suppress metastasis. J. Clin. Invest. 128, 531–544 (2018).
Mitra, A. K. et al. Ligand-independent activation of c-Met by fibronectin and α(5)β(1)-integrin regulates ovarian cancer invasion and metastasis. Oncogene 30, 1566–1576 (2011).
Mayorca-Guiliani, A. E. et al. ISDoT: in situ decellularization of tissues for high-resolution imaging and proteomic analysis of native extracellular matrix. Nat. Med. 23, 890–898 (2017). This article describes a whole-organ decellularization technique to study native ECM architecture and components, and to monitor cancer cell-induced changes in ECM during disease progression.
Naba, A. et al. Characterization of the extracellular matrix of normal and diseased tissues using proteomics. J. Proteome Res. 16, 3083–3091 (2017).
Handorf, A. M., Zhou, Y., Halanski, M. A. & Li, W. Tissue stiffness dictates development, homeostasis, and disease progression. Organogenesis 11, 1–15 (2015).
Matsui, H., Harada, I. & Sawada, Y. Src, p130Cas, and mechanotransduction in cancer cells. Genes Cancer 3, 394–401 (2012).
Sun, Z., Guo, S. S. & Fässler, R. Integrin-mediated mechanotransduction. J. Cell Biol. 215, 445–456 (2016).
Kaushik, S., Pickup, M. W. & Weaver, V. M. From transformation to metastasis: deconstructing the extracellular matrix in breast cancer. Cancer Metastasis Rev. 35, 655–667 (2016).
Acerbi, I. et al. Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration. Integr. Biol. 7, 1120–1134 (2015).
Paszek, M. J. et al. Tensional homeostasis and the malignant phenotype. Cancer Cell 8, 241–254 (2005).
Rubashkin, M. G. et al. Force engages vinculin and promotes tumor progression by enhancing PI3K activation of phosphatidylinositol (3,4,5)-triphosphate. Cancer Res. 74, 4597–4611 (2014).
Aragona, M. et al. A mechanical checkpoint controls multicellular growth through YAP/TAZ regulation by actin-processing factors. Cell 154, 1047–1059 (2013).
Dupont, S. et al. Role of YAP/TAZ in mechanotransduction. Nature 474, 179–183 (2011).
Kaukonen, R. et al. Normal stroma suppresses cancer cell proliferation via mechanosensitive regulation of JMJD1a-mediated transcription. Nat. Commun. 7, 12237 (2016).
Elosegui-Artola, A. et al. Mechanical regulation of a molecular clutch defines force transmission and transduction in response to matrix rigidity. Nat. Cell Biol. 18, 540–548 (2016).
Das, A., Fischer, R. S., Pan, D. & Waterman, C. M. YAP nuclear localization in the absence of cell-cell contact is mediated by a filamentous actin-dependent, myosin II- and phospho-YAP-independent pathway during extracellular matrix mechanosensing. J. Biol. Chem. 291, 6096–6110 (2016).
Kim, N. & Gumbiner, B. M. Adhesion to fibronectin regulates Hippo signaling via the FAK-Src-PI3K pathway. J. Cell Biol. 210, 503–515 (2015).
Foster, C. T., Gualdrini, F. & Treisman, R. Mutual dependence of the MRTF-SRF and YAP-TEAD pathways in cancer-associated fibroblasts is indirect and mediated by cytoskeletal dynamics. Genes Dev. 31, 2361–2375 (2017).
Si, Y. et al. Src inhibits the hippo tumor suppressor pathway through tyrosine phosphorylation of Lats1. Cancer Res. 77, 4868–4880 (2017).
Nardone, G. et al. YAP regulates cell mechanics by controlling focal adhesion assembly. Nat. Commun. 8, 15321 (2017).
Cox, T. R. et al. LOX-mediated collagen crosslinking is responsible for fibrosis-enhanced metastasis. Cancer Res. 73, 1721–1732 (2013).
Khan, Z. & Marshall, J. F. The role of integrins in TGFβ activation in the tumour stroma. Cell Tissue Res. 365, 657–673 (2016).
Voloshenyuk, T. G., Landesman, E. S., Khoutorova, E., Hart, A. D. & Gardner, J. D. Induction of cardiac fibroblast lysyl oxidase by TGF-β1 requires PI3K/Akt, Smad3, and MAPK signaling. Cytokine 55, 90–97 (2011).
Gao, A. E., Sullivan, K. E. & Black, L. D. Lysyl oxidase expression in cardiac fibroblasts is regulated by α2β1 integrin interactions with the cellular microenvironment. Biochem. Biophys. Res. Commun. 475, 70–75 (2016).
Franco-Barraza, J. et al. Matrix-regulated integrin αvβ5 maintains α5β1-dependent desmoplastic traits prognostic of neoplastic recurrence. eLife 6, e20600 (2017). This study compares normal fibroblasts and CAFs from patients with pancreatic cancer to reveal alterations in the subcellular localization of α5β1 integrin as a potential prognostic marker for disease recurrence.
Maritzen, T., Schachtner, H. & Legler, D. F. On the move: endocytic trafficking in cell migration. Cell. Mol. Life Sci. 72, 2119–2134 (2015).
Waclaw, B. et al. A spatial model predicts that dispersal and cell turnover limit intratumour heterogeneity. Nature 525, 261–264 (2015). This study presents a model that provides insights into tumour growth and evolution on the basis of short-range cell movement and cell turnover within the tumour.
Chan, C. E. & Odde, D. J. Traction dynamics of filopodia on compliant substrates. Science 322, 1687–1691 (2008).
Gardel, M. L. et al. Traction stress in focal adhesions correlates biphasically with actin retrograde flow speed. J. Cell Biol. 183, 999–1005 (2008).
Mitchison, T. & Kirschner, M. Cytoskeletal dynamics and nerve growth. Neuron 1, 761–772 (1988).
Provenzano, P. P. et al. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 4, 38 (2006).
Barcus, C. E. et al. Elevated collagen-I augments tumor progressive signals, intravasation and metastasis of prolactin-induced estrogen receptor alpha positive mammary tumor cells. Breast Cancer Res 19, 9 (2017).
Drifka, C. R. et al. Human pancreatic stellate cells modulate 3D collagen alignment to promote the migration of pancreatic ductal adenocarcinoma cells. Biomed. Microdevices 18, 105 (2016).
Drifka, C. R. et al. Highly aligned stromal collagen is a negative prognostic factor following pancreatic ductal adenocarcinoma resection. Oncotarget 7, 76197–76213 (2016).
Laklai, H. et al. Genotype tunes pancreatic ductal adenocarcinoma tissue tension to induce matricellular fibrosis and tumor progression. Nat. Med. 22, 497–505 (2016).
Hanley, C. J. et al. A subset of myofibroblastic cancer-associated fibroblasts regulate collagen fiber elongation, which is prognostic in multiple cancers. Oncotarget 7, 6159–6174 (2016).
Pointer, K. B. et al. Association of collagen architecture with glioblastoma patient survival. J. Neurosurg. 126, 1812–1821 (2017).
Gupton, S. L. et al. Mena binds α5 integrin directly and modulates α5β1 function. J. Cell Biol. 198, 657–676 (2012).
Oudin, M. J. et al. Tumor cell-driven extracellular matrix remodeling drives haptotaxis during metastatic progression. Cancer Discov. 6, 516–531 (2016). This study describes how a direct interaction between the pro-metastatic, actin-regulatory protein MENA(INV) and α5β1 integrin increases tumour cell haptotaxis and contributes to the formation of perpendicular collagen capsules at the tumour edge.
Georgiadou, M. & Ivaska, J. Tensins: bridging AMP-activated protein kinase with integrin activation. Trends Cell Biol. 27, 703–711 (2017).
Georgiadou, M. et al. AMPK negatively regulates tensin-dependent integrin activity. J. Cell Biol. 216, 1107–1121 (2017).
Duan, W. et al. Desmoplasia suppression by metformin-mediated AMPK activation inhibits pancreatic cancer progression. Cancer Lett. 385, 225–233 (2017).
Te Boekhorst, V., Preziosi, L. & Friedl, P. Plasticity of cell migration in vivo and in silico. Annu. Rev. Cell Dev. Biol. 32, 491–526 (2016).
Te Boekhorst, V. & Friedl, P. Plasticity of cancer cell invasion-mechanisms and implications for therapy. Adv. Cancer Res. 132, 209–264 (2016).
Yilmaz, M. & Christofori, G. EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev. 28, 15–33 (2009).
Macpherson, I. R. et al. p120-catenin is required for the collective invasion of squamous cell carcinoma cells via a phosphorylation-independent mechanism. Oncogene 26, 5214–5228 (2007).
Canel, M. et al. Quantitative in vivo imaging of the effects of inhibiting integrin signaling via Src and FAK on cancer cell movement: effects on E-cadherin dynamics. Cancer Res. 70, 9413–9422 (2010). This study points to a novel role for integrin signalling in regulating E-cadherin internalization and therefore cell–cell adhesion strength and collective cancer cell movement.
Canel, M., Serrels, A., Frame, M. C. & Brunton, V. G. E-cadherin-integrin crosstalk in cancer invasion and metastasis. J. Cell Sci. 126, 393–401 (2013).
Borghi, N., Lowndes, M., Maruthamuthu, V., Gardel, M. L. & Nelson, W. J. Regulation of cell motile behavior by crosstalk between cadherin- and integrin-mediated adhesions. Proc. Natl Acad. Sci. USA 107, 13324–13329 (2010).
Gimond, C. et al. Induction of cell scattering by expression of beta1 integrins in beta1-deficient epithelial cells requires activation of members of the rho family of GTPases and downregulation of cadherin and catenin function. J. Cell Biol. 147, 1325–1340 (1999).
Martinez-Rico, C., Pincet, F., Thiery, J. & Dufour, S. Integrins stimulate E-cadherin-mediated intercellular adhesion by regulating Src-kinase activation and actomyosin contractility. J. Cell Sci. 123, 712–722 (2010). This study builds on the reported crosstalk between integrins and cadherins to show that E-cadherin-dependent adhesion strength can be stimulated in the presence of fibronectin.
Hegerfeldt, Y., Tusch, M., Bröcker, E. & Friedl, P. Collective cell movement in primary melanoma explants: plasticity of cell-cell interaction, beta1-integrin function, and migration strategies. Cancer Res. 62, 2125–2130 (2002).
Friedl, P., Sahai, E., Weiss, S. & Yamada, K. M. New dimensions in cell migration. Nat. Rev. Mol. Cell Biol. 13, 743–747 (2012).
Ridley, A. J. Rho GTPase signalling in cell migration. Curr. Opin. Cell Biol. 36, 103–112 (2015).
Jacquemet, G. et al. RCP-driven α5β1 recycling suppresses Rac and promotes RhoA activity via the RacGAP1-IQGAP1 complex. J. Cell Biol. 202, 917–935 (2013).
Shafaq-Zadah, M. et al. Persistent cell migration and adhesion rely on retrograde transport of β(1) integrin. Nat. Cell Biol. 18, 54–64 (2016).
De Franceschi, N. et al. Selective integrin endocytosis is driven by interactions between the integrin α-chain and AP2. Nat. Struct. Mol. Biol. 23, 172–179 (2016).
Adorno, M. et al. A mutant-p53/Smad complex opposes p63 to empower TGFbeta-induced metastasis. Cell 137, 87–98 (2009).
Morton, J. P. et al. Mutant p53 drives metastasis and overcomes growth arrest/senescence in pancreatic cancer. Proc. Natl Acad. Sci. USA 107, 246–251 (2010).
Muller, P. A. J. et al. Mutant p53 drives invasion by promoting integrin recycling. Cell 139, 1327–1341 (2009).
Paul, N. R. et al. α5β1 integrin recycling promotes Arp2/3-independent cancer cell invasion via the formin FHOD3. J. Cell Biol. 210, 1013–1031 (2015).
Arjonen, A. et al. Mutant p53-associated myosin-X upregulation promotes breast cancer invasion and metastasis. J. Clin. Invest. 124, 1069–1082 (2014).
Zhang, H. et al. Myosin-X provides a motor-based link between integrins and the cytoskeleton. Nat. Cell Biol. 6, 523–531 (2004).
Jacquemet, G., Hamidi, H. & Ivaska, J. Filopodia in cell adhesion, 3D migration and cancer cell invasion. Curr. Opin. Cell Biol. 36, 23–31 (2015).
Jacquemet, G. et al. FiloQuant reveals increased filopodia density during breast cancer progression. J. Cell Biol. 216, 3387–3403 (2017).
Jacquemet, G. et al. L-type calcium channels regulate filopodia stability and cancer cell invasion downstream of integrin signalling. Nat. Commun. 7, 13297 (2016).
Beaty, B. T. & Condeelis, J. Digging a little deeper: the stages of invadopodium formation and maturation. Eur. J. Cell Biol. 93, 438–444 (2014).
Destaing, O., Block, M. R., Planus, E. & Albiges-Rizo, C. Invadosome regulation by adhesion signaling. Curr. Opin. Cell Biol. 23, 597–606 (2011).
Paterson, E. K. & Courtneidge, S. A. Invadosomes are coming: new insights into function and disease relevance. FEBS J. 285, 8–27 (2017).
Caswell, P. T., Vadrevu, S. & Norman, J. C. Integrins: masters and slaves of endocytic transport. Nat. Rev. Mol. Cell Biol. 10, 843–853 (2009).
Pellinen, T. et al. Small GTPase Rab21 regulates cell adhesion and controls endosomal traffic of beta1-integrins. J. Cell Biol. 173, 767–780 (2006).
Nader, G. P. F., Ezratty, E. J. & Gundersen, G. G. FAK, talin and PIPKIγ regulate endocytosed integrin activation to polarize focal adhesion assembly. Nat. Cell Biol. 18, 491–503 (2016).
Jahangiri, A. et al. Cross-activating c-Met/β1 integrin complex drives metastasis and invasive resistance in cancer. Proc. Natl Acad. Sci. USA 114, E8694 (2017).
Yoshioka, T. et al. β4 Integrin signaling induces expansion of prostate tumor progenitors. J. Clin. Invest. 123, 682–699 (2013).
Gao, J. et al. Mechanistic insights into EGFR membrane clustering revealed by super-resolution imaging. Nanoscale 7, 2511–2519 (2015).
Lau, L. F. Cell surface receptors for CCN proteins. J. Cell Commun. Signal. 10, 121–127 (2016).
Xie, D., Nakachi, K., Wang, H., Elashoff, R. & Koeffler, H. P. Elevated levels of connective tissue growth factor, WISP-1, and CYR61 in primary breast cancers associated with more advanced features. Cancer Res. 61, 8917–8923 (2001).
Huang, Y., Lan, Q., Lorusso, G., Duffey, N. & Rüegg, C. The matricellular protein CYR61 promotes breast cancer lung metastasis by facilitating tumor cell extravasation and suppressing anoikis. Oncotarget 8, 9200–9215 (2017).
Lakshminarayan, R. et al. Galectin-3 drives glycosphingolipid-dependent biogenesis of clathrin-independent carriers. Nat. Cell Biol. 16, 595–606 (2014).
Commisso, C. et al. Macropinocytosis of protein is an amino acid supply route in Ras-transformed cells. Nature 497, 633–637 (2013).
Seguin, L. et al. An integrin β3-KRAS-RalB complex drives tumour stemness and resistance to EGFR inhibition. Nat. Cell Biol. 16, 457–468 (2014).
Seguin, L. et al. Galectin-3, a druggable vulnerability for KRAS-addicted cancers. Cancer Discov. 7, 1464–1479 (2017). This study identifies an exciting therapeutic opportunity to target KRAS-addicted tumours by dampening the activity of galectin, a cell surface carbohydrate that binds to, and clusters, αvβ3 integrin to activate KRAS signalling.
Zhang, K. et al. Oncogenic K-Ras upregulates ITGA6 expression via FOSL1 to induce anoikis resistance and synergizes with αV-Class integrins to promote EMT. Oncogene 36, 5681–5694 (2017).
Colombo, M., Raposo, G. & Théry, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 30, 255–289 (2014).
E. L. Andaloussi, S., Mäger, I., Breakefield, X. O. & Wood, M. J. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat. Rev. Drug Discov. 12, 347–357 (2013).
Hoshino, A. et al. Tumour exosome integrins determine organotropic metastasis. Nature 527, 329–335 (2015).
Holash, J. et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science 284, 1994–1998 (1999).
Parikh, S. M. et al. Excess circulating angiopoietin-2 may contribute to pulmonary vascular leak in sepsis in humans. PLOS Med. 3, e46 (2006).
Hakanpaa, L. et al. Endothelial destabilization by angiopoietin-2 via integrin β1 activation. Nat. Commun. 6, 5962 (2015).
Cao, Y. et al. Neuropilin-2 promotes extravasation and metastasis by interacting with endothelial α5 integrin. Cancer Res. 73, 4579–4590 (2013).
Handa, A. et al. Neuropilin-2 expression affects the increased vascularization and is a prognostic factor in osteosarcoma. Int. J. Oncol. 17, 291–295 (2000).
Yasuoka, H. et al. Neuropilin-2 expression in breast cancer: correlation with lymph node metastasis, poor prognosis, and regulation of CXCR4 expression. BMC Cancer 9, 220 (2009).
Wang, H. et al. Tumor cell alpha3beta1 integrin and vascular laminin-5 mediate pulmonary arrest and metastasis. J. Cell Biol. 164, 935–941 (2004).
Barbazán, J. et al. Liver metastasis is facilitated by the adherence of circulating tumor cells to vascular fibronectin deposits. Cancer Res. 77, 3431–3441 (2017).
Bos, P. D. et al. Genes that mediate breast cancer metastasis to the brain. Nature 459, 1005–1009 (2009).
Barney, L. E. et al. A cell-ECM screening method to predict breast cancer metastasis. Integr. Biol. 7, 198–212 (2015).
Huck, L., Pontier, S. M., Zuo, D. M. & Muller, W. J. Beta1-integrin is dispensable for the induction of ErbB2 mammary tumors but plays a critical role in the metastatic phase of tumor progression. Proc. Natl Acad. Sci. USA 107, 15559–15564 (2010).
Grzesiak, J. J. et al. Knockdown of the β(1) integrin subunit reduces primary tumor growth and inhibits pancreatic cancer metastasis. Int. J. Cancer 129, 2905–2915 (2011).
Stoletov, K. et al. Visualizing extravasation dynamics of metastatic tumor cells. J. Cell Sci. 123, 2332–2341 (2010).
Reymond, N. et al. Cdc42 promotes transendothelial migration of cancer cells through β1 integrin. J. Cell Biol. 199, 653–668 (2012).
Chen, M. B., Lamar, J. M., Li, R., Hynes, R. O. & Kamm, R. D. Elucidation of the roles of tumor integrin β1 in the extravasation stage of the metastasis cascade. Cancer Res. 76, 2513–2524 (2016). The authors of this study use a 3D microfluidic model of the human microvasculature to reveal β1 integrin adhesion to subendothelial laminin as a critical prerequisite for successful transmigration.
Kato, H. et al. The primacy of β1 integrin activation in the metastatic cascade. PLOS ONE 7, e46576 (2012).
Barbazán, J. et al. Molecular characterization of circulating tumor cells in human metastatic colorectal cancer. PLOS ONE 7, e40476 (2012).
Lai, M. et al. Talin-1 overexpression defines high risk for aggressive oral squamous cell carcinoma and promotes cancer metastasis. J. Pathol. 224, 367–376 (2011).
Sakamoto, S., McCann, R. O., Dhir, R. & Kyprianou, N. Talin1 promotes tumor invasion and metastasis via focal adhesion signaling and anoikis resistance. Cancer Res. 70, 1885–1895 (2010).
Sin, S. et al. Role of the focal adhesion protein kindlin-1 in breast cancer growth and lung metastasis. J. Natl Cancer Inst. 103, 1323–1337 (2011).
Sarvi, S. et al. Kindlin-1 promotes pulmonary breast cancer metastasis. Cancer Res. 78, 1484–1496 (2018).
Barkan, D. et al. Inhibition of metastatic outgrowth from single dormant tumor cells by targeting the cytoskeleton. Cancer Res. 68, 6241–6250 (2008).
Shibue, T. & Weinberg, R. A. Integrin beta1-focal adhesion kinase signaling directs the proliferation of metastatic cancer cells disseminated in the lungs. Proc. Natl Acad. Sci. USA 106, 10290–10295 (2009).
Shibue, T., Brooks, M. W. & Weinberg, R. A. An integrin-linked machinery of cytoskeletal regulation that enables experimental tumor initiation and metastatic colonization. Cancer Cell 24, 481–498 (2013).
Mellor, H. The role of formins in filopodia formation. Biochim. Biophys. Acta 1803, 191–200 (2010).
Shibue, T., Brooks, M. W., Inan, M. F., Reinhardt, F. & Weinberg, R. A. The outgrowth of micrometastases is enabled by the formation of filopodium-like protrusions. Cancer Discov. 2, 706–721 (2012).
Hanker, A. B. et al. Extracellular matrix/integrin signaling promotes resistance to combined inhibition of HER2 and PI3K in HER2+ breast cancer. Cancer Res. 77, 3280–3292 (2017).
Huang, C. et al. β1 integrin mediates an alternative survival pathway in breast cancer cells resistant to lapatinib. Breast Cancer Res. 13, R84 (2011).
Sethi, T. et al. Extracellular matrix proteins protect small cell lung cancer cells against apoptosis: a mechanism for small cell lung cancer growth and drug resistance in vivo. Nat. Med. 5, 662–668 (1999).
Kanda, R. et al. Erlotinib resistance in lung cancer cells mediated by integrin β1/Src/Akt-driven bypass signaling. Cancer Res. 73, 6243–6253 (2013).
Eke, I. et al. Simultaneous β1 integrin-EGFR targeting and radiosensitization of human head and neck cancer. J. Natl Cancer Inst. 107, dju419 (2015).
Hirata, E. et al. Intravital imaging reveals how BRAF inhibition generates drug-tolerant microenvironments with high integrin β1/FAK signaling. Cancer Cell 27, 574–588 (2015). This article describes paradoxical activation of melanoma-associated fibroblasts by a BRAF inhibitor, which leads to increased stromal matrix stiffening and elevated β1 integrin–SRC–FAK signalling and drug resistance in melanoma cells.
Hu, T., Zhou, R., Zhao, Y. & Wu, G. Integrin α6/Akt/Erk signaling is essential for human breast cancer resistance to radiotherapy. Sci. Rep. 6, 33376 (2016).
Park, C. C., Zhang, H. J., Yao, E. S., Park, C. J. & Bissell, M. J. Beta1 integrin inhibition dramatically enhances radiotherapy efficacy in human breast cancer xenografts. Cancer Res. 68, 4398–4405 (2008).
Lomonaco, S. L. et al. Cilengitide induces autophagy-mediated cell death in glioma cells. Neuro Oncol. 13, 857–865 (2011).
Mikkelsen, T. et al. Radiation sensitization of glioblastoma by cilengitide has unanticipated schedule-dependency. Int. J. Cancer 124, 2719–2727 (2009).
Mason, W. P. End of the road: confounding results of the CORE trial terminate the arduous journey of cilengitide for glioblastoma. Neuro Oncol. 17, 634–635 (2015).
Nabors, L. B. et al. Two cilengitide regimens in combination with standard treatment for patients with newly diagnosed glioblastoma and unmethylated MGMT gene promoter: results of the open-label, controlled, randomized phase II CORE study. Neuro Oncol. 17, 708–717 (2015).
Stupp, R. et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071–22072 study): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 15, 1100–1108 (2014).
Élez, E. et al. Abituzumab combined with cetuximab plus irinotecan versus cetuximab plus irinotecan alone for patients with KRAS wild-type metastatic colorectal cancer: the randomised phase I/II POSEIDON trial. Ann. Oncol. 26, 132–140 (2015).
Aslan, B. et al. The ZNF304-integrin axis protects against anoikis in cancer. Nat. Commun. 6, 7351 (2015).
Lee, B. Y., Timpson, P., Horvath, L. G. & Daly, R. J. FAK signaling in human cancer as a target for therapeutics. Pharmacol. Ther. 146, 132–149 (2015).
Leng, C. et al. An integrin beta4-EGFR unit promotes hepatocellular carcinoma lung metastases by enhancing anchorage independence through activation of FAK-AKT pathway. Cancer Lett. 376, 188–196 (2016).
Jiang, H. et al. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nat. Med. 22, 851–860 (2016).
Serrels, A. et al. Nuclear FAK controls chemokine transcription, Tregs, and evasion of anti-tumor immunity. Cell 163, 160–173 (2015).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02428270 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02546531 (2018).
Canel, M. et al. Nuclear FAK and Runx1 cooperate to regulate IGFBP3, cell-cycle progression, and tumor growth. Cancer Res. 77, 5301–5312 (2017).
Johnston, K. A. & Lopez, K. M. Lysyl oxidase in cancer inhibition and metastasis. Cancer Lett. 417, 174–181 (2018).
Miller, B. W. et al. Targeting the LOX/hypoxia axis reverses many of the features that make pancreatic cancer deadly: inhibition of LOX abrogates metastasis and enhances drug efficacy. EMBO Mol. Med. 7, 1063–1076 (2015).
Pickup, M. W. et al. Stromally derived lysyl oxidase promotes metastasis of transforming growth factor-β-deficient mouse mammary carcinomas. Cancer Res. 73, 5336–5346 (2013).
Wei, Y. et al. Fibroblast-specific inhibition of TGF-β1 signaling attenuates lung and tumor fibrosis. J. Clin. Invest. 127, 3675–3688 (2017).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139, 891–906 (2009).
Baker, A. et al. The role of lysyl oxidase in SRC-dependent proliferation and metastasis of colorectal cancer. J. Natl Cancer Inst. 103, 407–424 (2011).
Rao, C. V., Janakiram, N. B. & Mohammed, A. Molecular pathways: mucins and drug delivery in cancer. Clin. Cancer Res. 23, 1373–1378 (2017).
Freeman, S. A. et al. Integrins form an expanding diffusional barrier that coordinates phagocytosis. Cell 164, 128–140 (2016).
Woods, E. C. et al. A bulky glycocalyx fosters metastasis formation by promoting G1 cell cycle progression. eLife 6, e25752 (2017).
Raina, D. et al. Direct targeting of the mucin 1 oncoprotein blocks survival and tumorigenicity of human breast carcinoma cells. Cancer Res. 69, 5133–5141 (2009).
Joshi, M. D. et al. MUC1 oncoprotein is a druggable target in human prostate cancer cells. Mol. Cancer Ther. 8, 3056–3065 (2009).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01279603 (2014).
Rao, C. V. et al. Small-molecule inhibition of GCNT3 disrupts mucin biosynthesis and malignant cellular behaviors in pancreatic cancer. Cancer Res. 76, 1965–1974 (2016).
Notni, J. et al. Variation of specific activities of 68Ga-aquibeprin and 68Ga-avebetrin enables selective PET imaging of different expression levels of integrins α5β1 and αvβ3. J. Nucl. Med. 57, 1618–1624 (2016).
Notni, J. et al. Complementary, selective PET imaging of integrin subtypes α5β1 and αvβ3 using 68Ga-aquibeprin and 68Ga-avebetrin. J. Nucl. Med. 57, 460–466 (2016).
Notni, J. et al. In vivo PET imaging of the cancer integrin αvβ6 using (68)Ga-labeled cyclic RGD nonapeptides. J. Nucl. Med. 58, 671–677 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT00565721 (2014).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02683824 (2018).
Gundem, G. et al. The evolutionary history of lethal metastatic prostate cancer. Nature 520, 353–357 (2015).
Shattil, S. J., Kim, C. & Ginsberg, M. H. The final steps of integrin activation: the end game. Nat. Rev. Mol. Cell Biol. 11, 288–300 (2010).
Lilja, J. et al. SHANK proteins limit integrin activation by directly interacting with Rap1 and R-Ras. Nat. Cell Biol. 19, 292–305 (2017).
Avraamides, C. J., Garmy-Susini, B. & Varner, J. A. Integrins in angiogenesis and lymphangiogenesis. Nat. Rev. Cancer 8, 604–617 (2008).
Topalian, S. L., Drake, C. G. & Pardoll, D. M. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 27, 450–461 (2015).
Kwan, B. H. et al. Integrin-targeted cancer immunotherapy elicits protective adaptive immune responses. J. Exp. Med. 214, 1679–1690 (2017).
Yu, X. et al. Inhibiting metastasis and preventing tumor relapse by triggering host immunity with tumor-targeted photodynamic therapy using photosensitizer-loaded functional nanographenes. ACS Nano 11, 10147–10158 (2017).
Liu, X. et al. Tumor-penetrating peptide enhances transcytosis of silicasome-based chemotherapy for pancreatic cancer. J. Clin. Invest. 127, 2007–2018 (2017).
Man, Y. K. S. et al. The novel oncolytic adenoviral mutant Ad5-3Δ-A20T retargeted to αvβ6 integrins efficiently eliminates pancreatic cancer cells. Mol. Cancer Ther. 17, 575–587 (2018).
Acknowledgements
The authors apologize to all colleagues whose work was not mentioned here owing to space limitations. The authors thank the Ivaska laboratory members for their constructive criticisms of this review and J. Marshall for insightful discussion and contributions to Supplementary Table 1. Work in the authors’ laboratory was supported by funding from the Academy of Finland, a European Research Council (ERC) Consolidator Grant, the Sigrid Juselius Foundation and the Cancer Society of Finland.
Reviewer information
Nature Reviews Cancer thanks D. Calderwood, D. Cheresh and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
J.I. researched the data for the article and wrote the body of the manuscript before submission. H.H. assembled the table, drafted the display items and contributed to writing of the manuscript before submission. J.I. and H.H. equally contributed to revising and editing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Glossary
- Angiogenesis
-
A physiological process characterized by the formation of new blood vessels from pre-existing vasculature, which can be deregulated during disease to promote the spread of cancer cells.
- Intravasation
-
The invasion of cancer cells through a basement membrane to enter blood or lymphatic vessels.
- Extravasation
-
The movement of cells out of a blood vessel, which involves traversing an endothelial cell layer and basement membrane, into the surrounding tissue.
- Anoikis
-
A specialized form of programmed cell death, which occurs upon loss of integrin-mediated adhesion to the extracellular matrix.
- Thrombus
-
Also known as a blood clot, a structure that is the final result of blood coagulation. A thrombus consists of aggregated platelets and red blood cells and a mesh of crosslinked fibrin.
- Desmoplasia
-
The growth of fibrous or connective tissue wherein resident cells produce excess fibrous matrix components such as collagen.
- Mechanoreceptors
-
A term used to define receptors capable of receiving and translating mechanical cues from the environment.
- Focal adhesion
-
Integrin-mediated cell–extracellular matrix contact that is connected to the actin cytoskeleton and acts as both a physical anchor and a signalling hub to regulate the cell’s response to extracellular cues.
- Lysyl oxidase (LOX) enzymes
-
Extracellular copper-dependent enzymes that act on lysine residues in collagen and elastin to promote crosslinking of these matrix molecules.
- Haptotaxis
-
Directional cell migration on an extracellular matrix gradient towards higher matrix concentrations.
- Filopodia
-
Actin-rich, finger-like membrane protrusions that extend out of the cell to probe the extracellular matrix.
- Invadopodia
-
Actin-based membrane protrusions found in invasive carcinoma cells and associated with sites of extracellular matrix degradation.
- Glycocalyx
-
Also known as the pericellular matrix, an external cell layer that contains a fibrous meshwork of carbohydrates (oligosaccharides, glycoproteins and mucins). This layer projects from the cell surface to cover the cell membrane in many animal cells and bacteria.
- Photodynamic therapy
-
A medical treatment that uses a photosensitizing molecule (frequently a drug that becomes activated by light exposure) and a light source to activate the administered drug.
Rights and permissions
About this article
Cite this article
Hamidi, H., Ivaska, J. Every step of the way: integrins in cancer progression and metastasis. Nat Rev Cancer 18, 533–548 (2018). https://doi.org/10.1038/s41568-018-0038-z
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41568-018-0038-z
This article is cited by
-
The interaction of platelet-related factors with tumor cells promotes tumor metastasis
Journal of Translational Medicine (2024)
-
Matrix stiffness affects tumor-associated macrophage functional polarization and its potential in tumor therapy
Journal of Translational Medicine (2024)
-
Tumor-derived small extracellular vesicles facilitate omental metastasis of ovarian cancer by triggering activation of mesenchymal stem cells
Cell Communication and Signaling (2024)
-
Integrinβ-1 in disorders and cancers: molecular mechanisms and therapeutic targets
Cell Communication and Signaling (2024)
-
Unveiling the mechanisms and challenges of cancer drug resistance
Cell Communication and Signaling (2024)