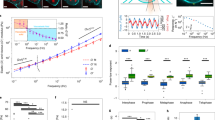
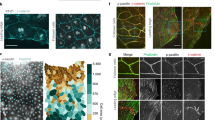
Abstract
During mitosis, or cell division, mammalian cells undergo extensive morphological changes, including elongation along the mitotic axis, which is perpendicular to the plane that bisects the two divided cells. Although much is known about the intracellular dynamics of mitosis, it is unclear how cells are able to divide in tissues, where the changes required for mitosis are mechanically constrained by surrounding cells and extracellular matrix. Here, by confining cells three dimensionally in hydrogels, we show that dividing cells generate substantial protrusive forces that deform their surroundings along the mitotic axis, clearing space for mitotic elongation. When forces are insufficient to create space for mitotic elongation, mitosis fails. We identify one source of protrusive force as the elongation of the interpolar spindle, an assembly of microtubules aligned with the mitotic axis. Another source of protrusive force is shown to be contraction of the cytokinetic ring, the polymeric structure that cleaves a dividing cell at its equator, which drives expansion along the mitotic axis. These findings reveal key functions for the interpolar spindle and cytokinetic ring in protrusive extracellular force generation, and explain how dividing cells overcome mechanical constraints in confining microenvironments, including some types of tumour.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Helmlinger, G., Netti, P. A., Lichtenbeld, H. C., Melder, R. J. & Jain, R. K. Solid stress inhibits the growth of multicellular tumor spheroids. Nat. Biotechnol. 15, 778–783 (1997).
Nia, H. T. et al. Solid stress and elastic energy as measures of tumour mechanopathology. Nat. Biomed. Eng. 1, 4 (2016).
Ramkumar, N. & Baum, B. Coupling changes in cell shape to chromosome segregation. Nat. Rev. Mol. Cell Biol. 17, 511–521 (2016).
Scholey, J. M., Brust-Mascher, I. & Mogilner, A. Cell division. Nature 422, 746–752 (2003).
Dumont, S. & Mitchison, T. J. Force and length in the mitotic spindle. Curr. Biol. 19, R749–R761 (2009).
Dumont, S., Salmon, E. D. & Mitchison, T. J. Deformations within moving kinetochores reveal different sites of active and passive force generation. Science 337, 355–358 (2012).
Shimamoto, Y., Maeda, Y. T., Ishiwata, S., Libchaber, A. J. & Kapoor, T. M. Insights into the micromechanical properties of the metaphase spindle. Cell 145, 1062–1074 (2011).
Stewart, M. P. et al. Hydrostatic pressure and the actomyosin cortex drive mitotic cell rounding. Nature 469, 226–230 (2011).
Ramanathan, S. P. et al. Cdk1-dependent mitotic enrichment of cortical myosin II promotes cell rounding against confinement. Nat. Cell Biol. 17, 148–159 (2015).
Fischer-Friedrich, E., Hyman, A. A., Jülicher, F., Müller, D. J. & Helenius, J. Quantification of surface tension and internal pressure generated by single mitotic cells. Sci. Rep. 4, 6213 (2015).
Cattin, C. J. et al. Mechanical control of mitotic progression in single animal cells. Proc. Natl Acad. Sci. USA 112, 11258–11263 (2015).
Sorce, B. et al. Mitotic cells contract actomyosin cortex and generate pressure to round against or escape epithelial confinement. Nat. Commun. 6, 8872 (2015).
Lancaster, O. et al. Mitotic rounding alters cell geometry to ensure efficient bipolar spindle formation. Dev. Cell 25, 270–283 (2013).
Lee, K. Y. & Mooney, D. J. Alginate: properties and biomedical applications. Prog. Polym. Sci. 37, 106–126 (2012).
Levental, I., Georges, P. C. & Janmey, P. A. Soft biological materials and their impact on cell function. Soft Matter 3, 299–306 (2007).
Chaudhuri, O. et al. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 15, 326–334 (2015).
Vig, D. K., Hamby, A. E. & Wolgemuth, C. W. On the quantification of cellular velocity fields. Biophys. J. 110, 1469–1475 (2016).
Legant, W. R. et al. Measurement of mechanical tractions exerted by cells in three-dimensional matrices. Nat. Methods 7, 969–971 (2010).
Franck, C., Maskarinec, S. A., Tirrell, D. A. & Ravichandran, G. Three-dimensional traction force microscopy: A new tool for quantifying cell-matrix interactions. PLoS One 6, 17833 (2011).
Son, S. et al. Resonant microchannel volume and mass measurements show that suspended cells swell during mitosis. J. Cell Biol. 211, 757–763 (2015).
Musacchio, A. & Salmon, E. D. The spindle-assembly checkpoint in space and time. Nat. Rev. Mol. Cell Biol. 8, 379–393 (2007).
Kastl, J. et al. Mad2 inhibitor-1 (M2I-1): a small molecule protein–protein interaction inhibitor targeting the mitotic spindle assembly checkpoint. ACS Chem. Biol. 10, 1661–1666 (2015).
Zlotek-Zlotkiewicz, E., Monnier, S., Cappello, G., Le Berre, M. & Piel, M. Optical volume and mass measurements show that mammalian cells swell during mitosis. J. Cell Biol. 211, 765–774 (2015).
Pollard, T. D. Mechanics of cytokinesis in eukaryotes. Curr. Opin. Cell Biol. 22, 50–56 (2010).
Jaramillo, C. et al. ESCRT-III assembly and cytokinetic abscission are induced by tension release in the intercellular bridge. Science 339, 882–886 (2010).
Fededa, J. P. & Gerlich, D. W. Molecular control of animal cell cytokinesis. Nat. Cell Biol. 14, 440–447 (2012).
Ward, J. J., Roque, H., Antony, C. & Nédélec, F. Mechanical design principles of a mitotic spindle. eLife 3, e03398 (2014).
Heald, R. & Khodjakov, A. Thirty years of search and capture: The complex simplicity of mitotic spindle assembly. J. Cell Biol. 211, 1103–1111 (2015).
Forth, S. & Kapoor, T. M. The mechanics of microtubule networks in cell division. J. Cell Biol. 216, 1525–1531 (2017).
Nicklas, R. B. Measurements of the force produced by the mitotic spindle in anaphase. J. Cell Biol. 97, 542–548 (1983).
Guild, J., Ginzberg, M. B., Hueschen, C. L., Mitchison, T. J. & Dumont, S. Increased lateral microtubule contact at the cell cortex is sufficient to drive mammalian spindle elongation. Mol. Biol. Cell 28, 1975–1983 (2017).
Yang, C.-F. et al. Kinesin-5 contributes to spindle-length scaling in the evolution of cancer toward metastasis. Sci. Rep. 6, 35767 (2016).
Tanenbaum, M. E. et al. Kif15 cooperates with Eg5 to promote bipolar spindle assembly. Curr. Biol. 19, 1703–1711 (2009).
Straight, A. F. et al. Dissecting temporal and spatial control of cytokinesis with a myosin II inhibitor. Science 299, 1743–1747 (2003).
Mitsushima, M. et al. Revolving movement of a dynamic cluster of actin filaments during mitosis. J. Cell Biol. 191, 453–462 (2010).
He, L. et al. Local 3D matrix confinement determines division axis through cell shape. Oncotarget 7, 6994–7011 (2015).
Matzke, R., Jacobson, K. & Radmacher, M. Direct, high-resolution measurement of furrow stiffening during division of adherent cells. Nat. Cell Biol. 3, 607–610 (2001).
Mogessie, B. & Schuh, M. Actin protects mammalian eggs against chromosome segregation errors. Science 357, eaal1647 (2017).
Thielicke, W. & Stamhuis, E. J. PIVlab – Towards user-friendly, affordable and accurate digital particle image velocimetry in MATLAB. J. Open Res. Softw. 2, e30 (2014).
Skoufias, D. A. et al. S-trityl-l-cysteine is a reversible, tight binding inhibitor of the human kinesin Eg5 that specifically blocks mitotic progression. J. Biol. Chem. 281, 17559–17569 (2006).
William, D. J. & Shah, M. A fast algorithm for active contours and curvature estimation. CVGIP Image Understanding 55, 14–26 (1992).
Nam, S., Hu, K. H., Butte, M. J. & Chaudhuri, O. Strain-enhanced stress relaxation impacts nonlinear elasticity in collagen gels. Proc. Natl Acad. Sci. USA 113, 5492–5497 (2016).
Nam, S., Lee, J., Brownfield, D. G. & Chaudhuri, O. Viscoplasticity enables mechanical remodeling of matrix by cells. Biophys. J. 111, 2296–2308 (2016).
Acknowledgements
The authors thank the members of the Chaudhuri laboratory, J. Nelson (Stanford University) and D. Fletcher (University of California, Berkeley) for helpful discussions, and M. Levenston (Stanford University) for use of the rheometer. This work was supported by a Samsung Scholarship for S.N., and a grant from the National Science Foundation (CMMI-1536736) to O.C.
Author information
Authors and Affiliations
Contributions
S.N. and O.C. designed the experiments and analysed the data. S.N. conducted the experiments and ran the simulations. S.N. and O.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–14, Supplementary Table 1, Supplementary Notes 1–3, Supplementary References 44–49
Videos
Supplementary Video 1: Dividing cells deform the surrounding matrices as they progress mitosis
Single dividing MDA-MB-2.1 cell within a hydrogel exert outward forces and pushes away the surrounding hydrogels along the mitotic axis. Matrix deformation was visualized with microbeads embedded in the hydrogels. White arrows point to specific microbeads, which are notably displaced. Images were shown at the indicated mitotic stages. Scale bar is 10µm.
Supplementary Video 2: Another example of matrix deformation during mitosis
Another movie of cell division within a hydrogel. Scale bar is 10µm.
Supplementary Video 3: Matrix deformation associated with mitotic swelling.
Cells undergoing mitotic swelling expand their size and generate outward forces, consequently deforming the surrounding matrices. Cells are arrested at mitosis by introducing S-trityl-L-cysteine. Matrix deformation was visualized by microbeads embedded in the hydrogels. White arrows point microbeads, which are notably displaced. Scale bar is 10µm.
Supplementary Video 4: Cell at metaphase does not progress through division in stiff and elastic 3D hydrogels.
Single dividing MDA-MB-2.1 cell within stiff and elastic hydrogels does not progress through mitosis, failing to divide. Matrix deformation was visualized with microbeads embedded in the hydrogels. White arrows point to specific microbeads, which are notably displaced inwardly. Scale bar is 10µm.
Supplementary Video 5: Another example of failure of division of cell at metaphase in stiff and elastic 3D hydrogels.
Single dividing MDA-MB-2.1 cell within stiff and elastic hydrogels does not progress through mitosis, failing to divide. Matrix deformation was visualized with microbeads embedded in the hydrogels. White arrows point to specific microbeads, which are notably displaced inwardly. Scale bar is 10µm.
Supplementary Video 6: Cells dividing in stiff and elastic hydrogels often undergo cell death.
Example of cell at metaphase in a stiff and elastic gel, which fails to undergo mitosis and later undergoes apoptosis. Scale bar is 10µm.
Supplementary Video 7: 3D reconstruction of a dividing cell.
Time-lapse images of a dividing cell were three-dimensionally reconstructed. Scale bar is 10µm.
Supplementary Video 8: Demonstration of outward force generation by lateral contraction using water balloon.
A macroscopic analogy to the outward forces generated by lateral contraction of the cytokinetic ring would be squeezing of a spherical water balloon at its equator, which leads to longitudinal expansion at the poles. In the movie, a green balloon contains water inside and is wrapped with a cable. The green balloon and the cable represent a cell and cytokinetic ring. The cable was pulled by a hand. Red arrows indicate ingression of the balloon equator due to pulling of the cable. Black arrows indicate longitudinal expansion of the balloon due to the water flow induced by pulling of the cable.
Supplementary Video 9: Buckled interpolar spindles observed for cells dividing in 3D hydrogels.
Cells dividing in 3D hydrogels were confined by the surrounding hydrogels and often exhibited very curved spindles at the end of cell division, indicative of buckling under compression. Yellow arrows indicate compressive reaction forces from hydrogels, in response to spindle-driven forces. White arrow points high curvature of interpolar spindle. Scale bar is 10µm.
Supplementary Video 10: Cells dividing in 2D culture exhibit straight interpolar spindles.
Cells dividing on 2D cell culture plates were free from confinement and did not show curved spindles, in contrast to cells dividing in 3D hydrogels. Scale bar is 10µm.
Supplementary Video 11: Relaxation of hydrogel deformation after interpolar spindle ablation.
Laser ablation was used to sever the interpolar spindles for cells entering anaphase. Relaxation of hydrogel deformation was visualized by microbeads embedded in the hydrogels. The position of beads retracts immediately in the direction of the cell after the spindles were ablated. White line represents the region of laser ablation, and white arrowheads indicate spindles ablated. White arrows point to specific microbeads, which are notably displaced inwardly. Scale bar is 10µm.
Supplementary Video 12: Another example of laser ablation at anaphase, imaging from metaphase.
Single dividing cell within a hydrogel generate outward forces and pushes away beads embedded in the hydrogel along the mitotic axis, when the cell progresses mitosis from metaphase to anaphase B. However, the position of the beads immediately retracts in the direction of the cell after the spindle was severed by laser ablation. White line represents the region of laser ablation, and white arrowheads indicate spindles ablated. White arrows point to specific microbeads, which are notably displaced. Images were shown at the indicated mitotic stages. Scale bar is 10µm.
Rights and permissions
About this article
Cite this article
Nam, S., Chaudhuri, O. Mitotic cells generate protrusive extracellular forces to divide in three-dimensional microenvironments. Nature Phys 14, 621–628 (2018). https://doi.org/10.1038/s41567-018-0092-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41567-018-0092-1
This article is cited by
-
Cell–extracellular matrix mechanotransduction in 3D
Nature Reviews Molecular Cell Biology (2023)
-
Dynamic and reconfigurable materials from reversible network interactions
Nature Reviews Materials (2022)
-
Enhanced substrate stress relaxation promotes filopodia-mediated cell migration
Nature Materials (2021)
-
Netrin-4 regulates stiffness and metastasis
Nature Materials (2021)
-
Osmotic pressure modulates single cell cycle dynamics inducing reversible growth arrest and reactivation of human metastatic cells
Scientific Reports (2021)