Abstract
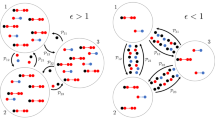
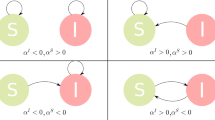
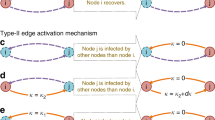
Reaction–diffusion processes1 have been widely used to study dynamical processes in epidemics2,3,4 and ecology5 in networked metapopulations. In the context of epidemics6, reaction processes are understood as contagions within each subpopulation (patch), while diffusion represents the mobility of individuals between patches. Recently, the characteristics of human mobility7, such as its recurrent nature, have been proven crucial to understand the phase transition to endemic epidemic states8,9. Here, by developing a framework able to cope with the elementary epidemic processes, the spatial distribution of populations and the commuting mobility patterns, we discover three different critical regimes of the epidemic incidence as a function of these parameters. Interestingly, we reveal a regime of the reaction–diffussion process in which, counter-intuitively, mobility is detrimental to the spread of disease. We analytically determine the precise conditions for the emergence of any of the three possible critical regimes in real and synthetic networks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Marro, J. & Dickman, R. Nonequilibrium Phase Transitions in Lattice Models (Cambridge Univ. Press, Cambridge, 2005).
Colizza, V., Pastor-Satorras, R. & Vespignani, A. Reaction–diffusion processes and metapopulation models in heterogeneous networks. Nat. Phys. 3, 276–282 (2007).
Colizza, V. & Vespignani, A. Invasion threshold in heterogeneous metapopulation networks. Phys. Rev. Lett. 99, 148701 (2007).
Hufnagel, L., Brockmann, D. & Geisel, T. Forecast and control of epidemics in a globalized world. Proc. Natl Acad. Sci. USA 101, 15124–15129 (2004).
Hanski, I. Metapopulation dynamics. Nature 396, 41–50 (1998).
Pastor-Satorras, R., Castellano, C., Van Mieghem, P. & Vespignani, A. Epidemic processes in complex networks. Rev. Mod. Phys. 87, 925–979 (2015).
Gonzalez, M. C., Hidalgo, C. A. & Barabasi, A.-L. Understanding individual human mobility patterns. Nature 453, 779–782 (2008).
Balcan, D. et al. Multiscale mobility networks and the spatial spreading of infectious diseases. Proc. Natl Acad. Sci. USA 106, 21484–21489 (2009).
Balcan, D. & Vespignani, A. Phase transitions in contagion processes mediated by recurrent mobility patterns. Nat. Phys. 7, 581–586 (2011).
Sattenspiel, L. & Dietz, K. A structured epidemic model incorporating geographic mobility among regions. Math. Biosci. 128, 71–91 (1995).
Grenfell, B. & Harwood, J. (Meta)population dynamics of infectious diseases. Trends Ecol. Evol. 12, 395–399 (1997).
Keeling, M. J. & Rohani, P. Modeling Infectious Diseases in Humans and Animals (Princeton Univ. Press, Princeton, NJ, 2008).
Hanski, I. & Gaggiotti, O. E. Ecology, Genetics, and Evolution of Metapopulations (Princeton Univ. Press, Princeton, NJ, 2004).
Bajardi, P., Barrat, A., Natale, F., Savini, L. & Colizza, V. Dynamical patterns of cattle trade movements. PLoS ONE 6, e19869 (2011).
Ball, F. et al. Seven challenges for metapopulation models of epidemics, including households models. Epidemics 10, 63–67 (2015).
Funk, S. et al. Nine challenges in incorporating the dynamics of behaviour in infectious diseases models. Epidemics 10, 21–25 (2015).
Eubank, S. et al. Modelling disease outbreaks in realistic urban social networks. Nature 429, 180–184 (2004).
Ferguson, N. M. et al. Strategies for mitigating an influenza pandemic. Nature 442, 448–452 (2006).
Van den Broeck, W. et al. The GLEaMviz computational tool, a publicly available software to explore realistic epidemic spreading scenarios at the global scale. BMC Infect. Dis. 11, 37 (2011).
Lofgren, E. T. et al. Opinion: mathematical models: a key tool for outbreak response. Proc. Natl Acad. Sci. USA 111, 18095–18096 (2014).
Scarpino, S. V., Allard, A. & Hebert-Dufresne, L. The effect of a prudent adaptive behaviour on disease transmission. Nat. Phys. 12, 1042–1046 (2016).
Marcus, R. A. Brief comments on perturbation theory of a nonsymmetric matrix: the GF matrix. J. Phys. Chem. A 105, 2612–2616 (2001).
Estrada, E., Meloni, S., Sheerin, M. & Moreno, Y. Epidemic spreading in random rectangular networks. Phys. Rev. E 94, 052316 (2016).
Gross, T., D’Lima, C. J. D. & Blasius, B. Epidemic dynamics on an adaptive network. Phys. Rev. Lett. 96, 208701 (2006).
Schwarzkopf, Y., Rákos, A. & Mukamel, D. Epidemic spreading in evolving networks. Phys. Rev. E 82, 036112 (2010).
Shaw, L. B. & Schwartz, I. B. Fluctuating epidemics on adaptive networks. Phys. Rev. E 77, 066101 (2008).
Barton, N. H. The probability of fixation of a favoured allele in a subdivided population. Genet. Res. 62, 149–157 (1993).
Whitlock, M. C. Fixation probability and time in subdivided populations. Genetics 164, 767–779 (2003).
Colizza, V., Barrat, A., Barthelemy, M., Valleron, A.-J. & Vespignani, A. Modeling the worldwide spread of pandemic influenza: Baseline case and containment interventions. PLoS Med. 4, 0095–0110 (2007).
Kitsak, M. et al. Identification of influential spreaders in complex networks. Nat. Phys. 6, 888–893 (2010).
Escobar-Morales, G. Cali en Cifras 2013 (Departamento Administrativo de Planeación, Santiago de Cali, 2013).
Acknowledgements
We acknowledge S. Meloni for the very many discussions and useful suggestions. J.G.-G. and A.A. acknowledge financial support from MINECO (projects FIS2015-71582-C2 and FIS2014-55867-P) and from the Departamento de Industria e Innovación del Gobierno de Aragón y Fondo Social Europeo (FENOL group E-19). A.A. acknowledges also financial support from the ICREA Academia, the James S. McDonnell Foundation.
Author information
Authors and Affiliations
Contributions
All of the authors wrote the paper and contributed equally to the production of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary notes 1–8, Supplementary figures 1–7, Supplementary Notes
Rights and permissions
About this article
Cite this article
Gómez-Gardeñes, J., Soriano-Paños, D. & Arenas, A. Critical regimes driven by recurrent mobility patterns of reaction–diffusion processes in networks. Nature Phys 14, 391–395 (2018). https://doi.org/10.1038/s41567-017-0022-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41567-017-0022-7
This article is cited by
-
Epidemic dynamics with non-Markovian travel in multilayer networks
Communications Physics (2023)
-
The effects of travel restrictions and detection measures on epidemic spreading in a metapopulation network
Nonlinear Dynamics (2023)
-
Statistical inference links data and theory in network science
Nature Communications (2022)
-
Variation in human mobility and its impact on the risk of future COVID-19 outbreaks in Taiwan
BMC Public Health (2021)
-
Interplay between population density and mobility in determining the spread of epidemics in cities
Communications Physics (2021)