Abstract
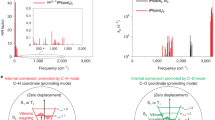
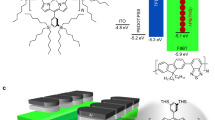
The development of high-performance near-infrared organic light-emitting diodes is hindered by strong non-radiative processes as governed by the energy gap law. Here, we show that exciton delocalization, which serves to decouple the exciton band from highly vibrational ladders in the S0 ground state, can bring substantial enhancements in the photoluminescence quantum yield of emitters, bypassing the energy gap law. Experimental proof is provided by the design and synthesis of a series of new Pt(ii) complexes with a delocalization length of 5–9 molecules that emit at 866–960 nm with a photoluminescence quantum yield of 5–12% in solid films. The corresponding near-infrared organic light-emitting diodes emit light with a 930 nm peak wavelength and a high external quantum efficiency up to 2.14% and a radiance of 41.6 W sr−1 m−2. Both theoretical and experimental results confirm the exciton–vibration decoupling strategy, which should be broadly applicable to other well-aligned molecular solids.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Caspar, J. V. & Meyer, T. J. Application of the energy gap law to nonradiative, excited-state decay. J. Phys. Chem. 87, 952–957 (1983).
Treadway, J. A. et al. Effect of delocalization and rigidity in the acceptor ligand on MLCT excited-state decay. Inorg. Chem. 35, 2242–2246 (1996).
Wilson, J. S. et al. The energy gap law for triplet states in Pt-containing conjugated polymers and monomers. J. Am. Chem. Soc. 123, 9412–9417 (2001).
Whittle, C. E., Weinstein, J. A., George, M. W. & Schanze, K. S. Photophysics of diimine platinum(ii) bis-acetylide complexes. Inorg. Chem. 40, 4053–4062 (2001).
Jelle, B. P., Kalnæs, S. E. & Gao, T. Low-emissivity materials for building applications: a state-of-the-art review and future research perspectives. Energy Buildings 96, 329–356 (2015).
Xiang, H., Cheng, J., Ma, X., Zhou, X. & Chruma, J. J. Near-infrared phosphorescence: materials and applications. Chem. Soc. Rev. 42, 6128–6185 (2013).
Tessler, N., Medvedev, V., Kazes, M., Kan, S. & Banin, U. Efficient near-infrared polymer nanocrystal light-emitting diodes. Science 295, 1506–1508 (2002).
Borek, C. et al. Highly efficient, near-infrared electrophosphorescence from a Pt–metalloporphyrin complex. Angew. Chem. Int. Ed. 46, 1109–1112 (2007).
Cocchi, M., Kalinowski, J., Virgili, D. & Williams, J. A. G. Excimer-based red/near-infrared organic light-emitting diodes with very high quantum efficiency. Appl. Phys. Lett. 92, 113302 (2008).
Graham, K. R. et al. Extended conjugation platinum(ii) porphyrins for use in near-infrared emitting organic light emitting diodes. Chem. Mater. 23, 5305–5312 (2011).
Barbieri, A., Bandini, E., Monti, F., Praveen, V. K. & Armaroli, N. The rise of near-infrared emitters: organic dyes, porphyrinoids and transition metal complexes. Top. Curr. Chem. 374, 47 (2016).
Ai, X. et al. Efficient radical-based light-emitting diodes with doublet emission. Nature 563, 536–540 (2018).
Zhang, Y. et al. Near-infrared emitting materials via harvesting triplet excitons: molecular design, properties and application in organic light emitting diodes. Adv. Opt. Mater. 6, 1800466 (2018).
Özçelik, S. & Akins, D. L. Superradiance of aggregated thiacarbocyanine molecules. J. Phys. Chem. B 103, 8926–8929 (1999).
Arias, D. H. et al. Thermally-limited exciton delocalization in superradiant molecular aggregates. J. Phys. Chem. B 117, 4553–4559 (2013).
Cai, K., Xie, J. & Zhao, D. NIR J-aggregates of hydroazaheptacene tetraimides. J. Am. Chem. Soc. 136, 28–31 (2014).
Qian, G. et al. Simple and efficient near-infrared organic chromophores for light-emitting diodes with single electroluminescent emission above 1,000 nm. Adv. Mater. 21, 111–116 (2009).
Zampetti, A., Minotto, A. & Cacialli, F. Near-infrared (NIR) organic light-emitting diodes (OLEDs): challenges and opportunities. Adv. Funct. Mater. 29, 1807623 (2019).
Klaus, D. R., Keene, M., Silchenko, S., Berezin, M. & Gerasimchuk, N. 1D polymeric platinum cyanoximate: a strategy toward luminescence in the near-infrared region beyond 1,000 nm. Inorg. Chem. 54, 1890–1900 (2015).
Ly, K. T. et al. Near-infrared organic light-emitting diodes with very high external quantum efficiency and radiance. Nat. Photon. 11, 63–68 (2017).
Zhang, Y. et al. Achieving NIR emission for donor–acceptor type platinum(ii) complexes by adjusting coordination position with isomeric ligands. Inorg. Chem. 57, 14208–14217 (2018).
Yam, V. W.-W., Wong, K. M.-C. & Zhu, N. Solvent-induced aggregation through metal···metal/π···π interactions: large solvatochromism of luminescent organoplatinum(ii) terpyridyl complexes. J. Am. Chem. Soc. 124, 6506–6507 (2002).
Yu, C., Wong, K. M.-C., Chan, K. H.-Y. & Yam, V. W.-W. Polymer-induced self-assembly of alkynylplatinum(ii) terpyridyl complexes by metal–metal/pi–pi interactions. Angew. Chem. Int. Ed. 44, 791–794 (2005).
Wong, K. M.-C. & Yam, V. W.-W. Self-assembly of luminescent alkynylplatinum(ii) terpyridyl complexes: modulation of photophysical properties through aggregation behavior. Acc. Chem. Res. 44, 424–434 (2011).
Hong, Y., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission. Chem. Soc. Rev. 40, 5361–5388 (2011).
Englman, R. & Jortner, J. The energy gap law for radiationless transitions in large molecules. Mol. Phys. 18, 145–164 (1970).
Herrera, F. & Spano, F. C. Cavity-controlled chemistry in molecular ensembles. Phys. Rev. Lett. 116, 238301 (2016).
Spano, F. C., Silvestri, L., Spearman, P., Raimondo, L. & Tavazzi, S. Reclassifying exciton–phonon coupling in molecular aggregates: evidence of strong nonadiabatic coupling in oligothiophene crystals. J. Chem. Phys. 127, 184703 (2007).
Spano, F. C. The spectral signatures of Frenkel polarons in H- and J-aggregates. Acc. Chem. Res. 43, 429–439 (2010).
Spano, F. C. & Yamagata, H. Vibronic coupling in J-aggregates and beyond: a direct means of determining the exciton coherence length from the photoluminescence spectrum. J. Phys. Chem. B 115, 5133–5143 (2011).
Chen, W.-C., Chou, P.-T. & Cheng, Y.-C. Low internal reorganization energy of the metal–metal-to-ligand charge transfer emission in dimeric Pt(ii) complexes. J. Phys. Chem. C 123, 10225–10236 (2019).
Hestand, N. J. & Spano, F. C. Interference between Coulombic and CT-mediated couplings in molecular aggregates: H- to J-aggregate transformation in perylene-based π-stacks. J. Chem. Phys. 143, 244707 (2015).
Yersin, H., Rausch, A. F., Czerwieniec, R., Hofbeck, T. & Fischer, T. The triplet state of organo-transition metal compounds. Triplet harvesting and singlet harvesting for efficient OLEDs. Coord. Chem. Rev. 255, 2622–2652 (2011).
Li, K. et al. Highly phosphorescent platinum(ii) emitters: photophysics, materials and biological applications. Chem. Sci. 7, 1653–1673 (2016).
Hutchison, G. R., Ratner, M. A. & Marks, T. J. Hopping transport in conductive heterocyclic oligomers: reorganization energies and substituent effects. J. Am. Chem. Soc. 127, 2339–2350 (2005).
Robinson, G. W. & Frosch, R. P. Electronic excitation transfer and relaxation. J. Phys. Chem. 38, 1187–1203 (1963).
Wysokiński, R., Hernik, K., Szostak, R. & Michalska, D. Electronic structure and vibrational spectra of cis-diammine(orotato)platinum(ii), a potential cisplatin analogue: DFT and experimental study. Chem. Phys. 333, 37–48 (2007).
Juliá, F. & González-Herrero, P. Aromatic C–H activation in the triplet excited state of cyclometalated platinum(ii) complexes using visible light. J. Am. Chem. Soc. 138, 5276–5282 (2016).
Podeszwa, R., Bukowski, R. & Szalewicz, K. Potential energy surface for the benzene dimer and perturbational analysis of π–π interactions. J. Phys. Chem. A 110, 10345–10354 (2006).
Pullerits, T., Chachisvilis, M. & Sundström, V. Exciton delocalization length in the B850 antenna of Rhodobacter sphaeroides. J. Phys. Chem. 100, 10787–10792 (1996).
Chesnut, D. B. & Suna, A. Fermion behavior of one‐dimensional excitons. J. Chem. Phys. 39, 146–149 (1963).
van Burgel, M., Wiersma, D. A. & Duppen, K. The dynamics of one‐dimensional excitons in liquids. J. Chem. Phys. 102, 20–33 (1995).
Kim, K.-H. et al. Crystal organic light-emitting diodes with perfectly oriented non-doped Pt-based emitting layer. Adv. Mater. 28, 2526–2532 (2016).
Sommer, J. R. et al. Efficient near-infrared polymer and organic light-emitting diodes based on electrophosphorescence from (tetraphenyltetranaphtho[2,3]porphyrin)platinum(ii). ACS Appl. Mater. Interfaces 1, 274–278 (2009).
Nagata, R., Nakanotani, H. & Adachi, C. Near-infrared electrophosphorescence up to 1.1 µm using a thermally activated delayed fluorescence molecule as triplet sensitizer. Adv. Mater. 29, 1604265 (2017).
Kim, D.-H. et al. High-efficiency electroluminescence and amplified spontaneous emission from a thermally activated delayed fluorescent near-infrared emitter. Nat. Photon. 12, 98–104 (2018).
Minotto, A. et al. Efficient near-infrared electroluminescence at 840 nm with ‘metal-free’ small-molecule:polymer blends. Adv. Mater. 30, 1706584 (2018).
Chang, C.-H. et al. A new class of sky-blue-emitting Ir(iii) phosphors assembled using fluorine-free pyridyl pyrimidine cyclometalates: application toward high-performance sky-blue- and white-emitting OLEDs. ACS Appl. Mater. Interfaces 5, 7341–7351 (2013).
Liu, X. et al. Syntheses, crystal structure and photophysical property of iridium complexes with 1,3,4-oxadiazole and 1,3,4-thiadiazole derivatives as ancillary ligands. J. Organomet. Chem. 785, 11–18 (2015).
Acknowledgements
This research was supported by funding from the Ministry of Science and Technology (MOST), the Featured Areas Research Program within the framework of the Higher Education Sprout Project administered by the Ministry of Education (MOE), National Taiwan University, Soochow University and the Research Grant Council and City University of Hong Kong. We are also grateful to the National Center for High-Performance Computing (NCHC) and National Synchrotron Radiation Research Center (NSRRC) for computer time and facilities, respectively.
Author information
Authors and Affiliations
Contributions
Y.-C.W., D.-G.C., K.-H.C., C.-W.W. and S.-H.L. performed optical measurements, simulations and calculations. S.F.W., W.-H.C. and J.-L.L. conducted the synthesis and characterization of Pt(ii) complexes. Y.H., L.-S.L. and W.-Y.H. executed OLED fabrications and analysed data. Y.-C.W. and P.-T.Chou developed the theoretical approach and prepared the manuscript. Y.C. designed the Pt(ii) complexes and interpreted the spectroscopic data. T.-H.W., P.-T.Chen and H.-F.H. performed GIXD experiments and data analysis. All authors discussed the results and contributed to the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–11 and Tables 1–5.
Crystallographic Data
Crystal structure of 4dMp.
Rights and permissions
About this article
Cite this article
Wei, YC., Wang, S.F., Hu, Y. et al. Overcoming the energy gap law in near-infrared OLEDs by exciton–vibration decoupling. Nat. Photonics 14, 570–577 (2020). https://doi.org/10.1038/s41566-020-0653-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-020-0653-6
This article is cited by
-
Laser-driven broadband near-infrared light source with watt-level output
Nature Photonics (2024)
-
Bright and stable near-infrared lead-free perovskite light-emitting diodes
Nature Photonics (2024)
-
In vivo NIR-II fluorescence imaging for biology and medicine
Nature Photonics (2024)
-
Tuning polymer-backbone coplanarity and conformational order to achieve high-performance printed all-polymer solar cells
Nature Communications (2024)
-
Physical insights into non-fullerene organic photovoltaics
Nature Reviews Physics (2024)