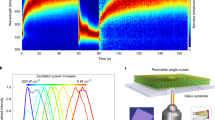
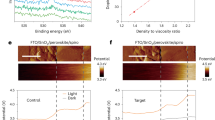
Abstract
Metal halide perovskites have become a popular material system for fabricating photovoltaics and various optoelectronic devices. However, long-term reliability must be assured. Instabilities are manifested as light-induced ion migration and segregation, which can lead to material degradation. Discordant reports have shown a beneficial role of ion migration under illumination, leading to defect healing. By combining ab initio simulations with photoluminescence measurements under controlled conditions, we demonstrate that photo-instabilities are related to light-induced formation and annihilation of defects acting as carrier trap states. We show that these phenomena coexist and compete. In particular, long-living carrier traps related to halide defects trigger photoinduced material transformations, driving both processes. Defect formation can be controlled by blocking under-coordinated surface sites, which act as a defect reservoir. By use of a passivation strategy we are thus able to stabilize the perovskite layer, leading to improved optoelectronic material quality and enhanced photostability in solar cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors on reasonable request.
References
NREL Best Research-Cell Efficiencies https://www.nrel.gov/pv/assets/images/efficiency-chart.png (2018).
Xu, W. et al. Rational molecular passivation for high-performance perovskite light-emitting diodes. Nat. Photon. https://doi.org/10.1038/s41566-019-0390-x (2019).
Lin, Q., Armin, A., Lyons, D. M., Burn, P. L. & Meredith, P. Low noise, IR-blind organohalide perovskite photodiodes for visible light detection and imaging. Adv. Mater. 27, 2060–2064 (2015).
Wei, H. et al. Sensitive X-ray detectors made of methylammonium lead tribromide perovskite single crystals. Nat. Photon. 10, 333–339 (2016).
Venugopalan, V. et al. High-detectivity perovskite light detectors printed in air from benign solvents. Chem 5, 868–880 (2019).
Zhu, H. et al. Lead halide perovskite nanowire lasers with low lasing thresholds and high quality factors. Nat. Mater. 14, 636–642 (2015).
Deschler, F. et al. High photoluminescence efficiency and optically pumped lasing in solution-processed mixed halide perovskite semiconductors. J. Phys. Chem. Lett. 5, 1421–1426 (2014).
Stranks, S. D. et al. Electron–hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science 342, 341–344 (2013).
Johnston, M. B. & Herz, L. M. Hybrid perovskites for photovoltaics: charge-carrier recombination, diffusion and radiative efficiencies. Acc. Chem. Res. 49, 146–154 (2016).
deQuilettes, D. W. et al. Photoluminescence lifetimes exceeding 8 μs and quantum yields exceeding 30% in hybrid perovskite thin films by ligand passivation. ACS Energy Lett. 1, 438–444 (2016).
Ball, J. M. & Petrozza, A. Defects in perovskite-halides and their effects in solar cells. Nat. Energy 1, 16149 (2016).
Meggiolaro, D. et al. Iodine chemistry determines the defect tolerance of lead-halide perovskites. Energy Environ. Sci. 11, 702–713 (2018).
Hoke, E. T. et al. Reversible photo-induced trap formation in mixed-halide hybrid perovskites for photovoltaics. Chem. Sci. 6, 613–617 (2014).
Sanchez, R. S. et al. Slow dynamic processes in lead halide perovskite solar cells. Characteristic times and hysteresis. J. Phys. Chem. Lett. 5, 2357–2363 (2014).
Leijtens, T. et al. Mapping electric field-induced switchable poling and structural degradation in hybrid lead halide perovskite thin films. Adv. Energy Mater. 5, 1–11 (2015).
Gottesman, R. et al. Photoinduced reversible structural transformations in free-standing CH3NH3PbI3 perovskite films. J. Phys. Chem. Lett. 6, 2332–2338 (2015).
Gottesman, R. & Zaban, A. Perovskites for photovoltaics in the spotlight: photoinduced physical changes and their implications. Acc. Chem. Res. 49, 320–329 (2016).
Motti, S. G. et al. Photoinduced emissive trap states in lead halide perovskite semiconductors. ACS Energy Lett. 1, 726–730 (2016).
Xing, J. et al. Ultrafast ion migration in hybrid perovskite polycrystalline thin films under light and suppression in single crystals. Phys. Chem. Chem. Phys. 18, 30484–30490 (2016).
Kim, G. Y. et al. Large tunable photoeffect on ion conduction in halide perovskites and implications for photodecomposition. Nat. Mater. 17, 445–449 (2018).
deQuilettes, D. W. et al. Photo-induced halide redistribution in organic–inorganic perovskite films. Nat. Commun. 7, 11683 (2016).
Stranks, S. D. et al. Recombination kinetics in organic–inorganic perovskites: excitons, free charge and subgap states. Phys. Rev. Appl. 2, 034007 (2014).
Mosconi, E., Meggiolaro, D., Snaith, H. J., Stranks, S. D. & De Angelis, F. Light-induced annihilation of Frenkel defects in organo–lead halide perovskites. Energy Environ. Sci. 9, 3180–3187 (2016).
Chen, S. et al. Light illumination induced photoluminescence enhancement and quenching in lead halide perovskite. Sol. RRL 1, 1600001 (2017).
Hong, D. et al. Nature of photo-induced quenching traps in methylammonium lead triiodide perovskite revealed by reversible photoluminescence decline. ACS Photon. 5, 2034–2043 (2018).
Fang, H.-H. et al. Ultrahigh sensitivity of methylammonium lead tribromide perovskite single crystals to environmental gases. Sci. Adv. 2, e1600534 (2016).
Galisteo-López, J. F., Anaya, M., Calvo, M. E. & Míguez, H. Environmental effects on the photophysics of organic–inorganic halide perovskites. J. Phys. Chem. Lett. 6, 2200–2205 (2015).
Tian, Y. et al. Mechanistic insights into perovskite photoluminescence enhancement: light curing with oxygen can boost yield thousandfold. Phys. Chem. Chem. Phys. 17, 24978–24987 (2015).
Meggiolaro, D., Mosconi, E. & De Angelis, F. Mechanism of reversible trap passivation by molecular oxygen in lead–halide perovskites. ACS Energy Lett. 2, 2794–2798 (2017).
Quitsch, W.-A. et al. The role of excitation energy in photobrightening and photodegradation of halide perovskite thin films. J. Phys. Chem. Lett. 9, 2062–2069 (2018).
Leijtens, T. et al. Carrier trapping and recombination: the role of defect physics in enhancing the open circuit voltage of metal halide perovskite solar cells. Energy Environ. Sci. 9, 3472–3481 (2016).
Miyata, K. et al. Large polarons in lead halide perovskites. Sci. Adv. 3, e1701217 (2017).
Tsai, H. et al. Light-induced lattice expansion leads to high-efficiency perovskite solar cells. Science 360, 67–70 (2018).
Azpiroz, J. M., Mosconi, E., Bisquert, J. & De Angelis, F. Defect migration in methylammonium lead iodide and its role in perovskite solar cell operation. Energy Environ. Sci. 8, 2118–2127 (2015).
Gottesman, R. et al. Extremely slow photoconductivity response of CH3NH3PbI3 perovskites suggesting structural changes under working conditions. J. Phys. Chem. Lett. 5, 2662–2669 (2014).
Barker, A. J. et al. Defect-assisted photoinduced halide segregation in mixed-halide perovskite thin films. ACS Energy Lett. 2, 1416–1424 (2017).
Brennan, M. C., Draguta, S., Kamat, P. V. & Kuno, M. Light-induced anion phase segregation in mixed halide perovskites. ACS Energy Lett. 3, 204–213 (2018).
Yoon, S. J., Kuno, M. & Kamat, P. V. Shift happens. How halide ion defects influence photoinduced segregation in mixed halide perovskites. ACS Energy Lett. 2, 1507–1514 (2017).
Eames, C. et al. Ionic transport in hybrid lead iodide perovskite solar cells. Nat. Commun. 6, 7497 (2015).
Meggiolaro, D., Mosconi, E. & De Angelis, F. Modeling the interaction of molecular iodine with MAPbI3 : a probe of lead–halide perovskites defect chemistry. ACS Energy Lett. 3, 447–451 (2018).
Boschloo, G. & Hagfeldt, A. Characteristics of the iodide/triiodide redox mediator in dye-sensitized solar cells. Acc. Chem. Res. 42, 1819–1826 (2009).
Zhang, L. & Sit, P. H.-L. Ab initio study of the role of oxygen and excess electrons in the degradation of CH3NH3PbI3. J. Mater. Chem. A 5, 9042–9049 (2017).
Wang, S., Jiang, Y., Juarez-Perez, E. J., Ono, L. K. & Qi, Y. Accelerated degradation of methylammonium lead iodide perovskites induced by exposure to iodine vapour. Nat. Energy 2, 16195 (2016).
Sadoughi, G. et al. Observation and mediation of the presence of metallic lead in organic–inorganic perovskite films. ACS Appl. Mater. Interfaces 7, 13440–13444 (2015).
Noel, N. K. et al. Enhanced photoluminescence and solar cell performance via Lewis base passivation of organic–inorganic lead halide perovskites. ACS Nano 8, 9815–9821 (2014).
Ling, Y. et al. Enhanced optical and electrical properties of polymer-assisted all-inorganic perovskites for light-emitting diodes. Adv. Mater. 28, 8983–8989 (2016).
Wang, Z. et al. Efficient and stable pure green all-inorganic perovskite CsPbBr3 light-emitting diodes with a solution-processed NiOx interlayer. J. Phys. Chem. C 121, 28132–28138 (2017).
Kim, M., Motti, S. G., Sorrentino, R. & Petrozza, A. Enhanced solar cell stability by hygroscopic polymer passivation of metal halide perovskite thin film. Energy Environ. Sci. 11, 2609–2619 (2018).
Xu, J. et al. Perovskite–fullerene hybrid materials suppress hysteresis in planar diodes. Nat. Commun. 6, 7081 (2015).
Shao, Y., Xiao, Z., Bi, C., Yuan, Y. & Huang, J. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nat. Commun. 5, 5784 (2014).
Cho, H. et al. Overcoming the electroluminescence efficiency limitations of perovskite light-emitting diodes. Science 350, 1222–1225 (2015).
Xiao, M. et al. A fast deposition–crystallization procedure for highly efficient lead iodide perovskite thin-film solar cells. Angew. Chem. Int. Ed. 126, 10056–10061 (2014).
Acknowledgements
This work has been funded by the European Union project PERT PV under grant no. 763977, ERC project SOPHY under grant no. 771528 and PERSEO ‘PERrovskite-based solar cells: towards high efficiency and long-term stability’ (Bando PRIN 2015—Italian Ministry of University and Scientific Research (MIUR) Decreto Direttoriale 4 Novembre 2015 no. 2488, project no. 20155LECAJ). The Ministero Istruzione dell’Università e della Ricerca (MIUR) and the University of Perugia are acknowledged for the financial support through the program “Dipartimenti di Eccellenza 2018-2022” (Grant AMIS) to F.D.A. S.G.M. thanks the CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico—Brasil) for a scholarship (206502/2014-1). M.K. acknowledges funding from the European Union’s Horizon 2020 research and innovation programme under Marie Skłodowska-Curie grant no. 797546 of the FASTEST project. The authors thank G. Paternò for his support in setting up the transient Voc characterization.
Author information
Authors and Affiliations
Contributions
S.G.M. performed the photoluminescence measurements. D.M. and E.M. performed the first-principles calculations. C.A.R.P., J.M.B., M.G. and M.K. were responsible for fabrication of the thin films. M.K. fabricated the solar cell devices and M.K and A.J.B. characterized the solar cell. A.P., S.G.M., A.J.B., D.M. and F.D.A. analysed the data. S.G.M., F.D.A. and A.P. wrote the first draft of the manuscript and all authors contributed to the discussions and finalized the manuscript. A.P. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Simulations and temporal dynamics.
Rights and permissions
About this article
Cite this article
Motti, S.G., Meggiolaro, D., Barker, A.J. et al. Controlling competing photochemical reactions stabilizes perovskite solar cells. Nat. Photonics 13, 532–539 (2019). https://doi.org/10.1038/s41566-019-0435-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-019-0435-1
This article is cited by
-
Surface-defect-passivation-enabled near-unity charge collection efficiency in bromide-based perovskite gamma-ray spectrum devices
Nature Photonics (2024)
-
Nanosurface-reconstructed perovskite for highly efficient and stable active-matrix light-emitting diode display
Nature Nanotechnology (2024)
-
Multifunctional sulfonium-based treatment for perovskite solar cells with less than 1% efficiency loss over 4,500-h operational stability tests
Nature Energy (2024)
-
Ion-induced field screening as a dominant factor in perovskite solar cell operational stability
Nature Energy (2024)
-
Stabilization of photoactive phases for perovskite photovoltaics
Nature Reviews Chemistry (2023)