Abstract
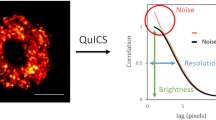
The principles of quantum optics have yielded a plethora of ideas to surpass the classical limitations of sensitivity and resolution in optical microscopy. While some ideas have been applied in proof-of-principle experiments, imaging a biological sample has remained challenging, mainly due to the inherently weak signal measured and the fragility of quantum states of light. In principle, however, these quantum protocols can add new information without sacrificing the classical information and can therefore enhance the capabilities of existing super-resolution techniques. Image scanning microscopy, a recent addition to the family of super-resolution methods, generates a robust resolution enhancement without reducing the signal level. Here, we introduce quantum image scanning microscopy: combining image scanning microscopy with the measurement of quantum photon correlation allows increasing the resolution of image scanning microscopy up to twofold, four times beyond the diffraction limit. We introduce the Q-ISM principle and obtain super-resolved optical images of a biological sample stained with fluorescent quantum dots using photon antibunching, a quantum effect, as a resolution-enhancing contrast mechanism.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The raw data that support the findings of this study are available in the figshare repository under the name ‘QISM_SoftwareAndData_zip’ and with the identifier https://doi.org/10.6084/m9.figshare.7241294.v1.
References
Abbe, E. Beiträge zur Theorie des Mikroskops und der mikroskopischen Wahrnehmung. Arch. für Mikroskopische Anat. 9, 413–418 (1873).
Hell, S. W. & Wichmann, J. Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt. Lett. 19, 780–782 (1994).
Gustafsson, M. G. L. Surpassing the lateral resolution limit by a factor of two using structured illumination microscopy. J. Microsc. 198, 82–87 (2000).
Rust, M. J., Bates, M. & Zhuang, X. Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Methods 3, 793–795 (2006).
Betzig, E. et al. Imaging intracellular fluorescent proteins at nanometer resolution. Science 313, 1642–1645 (2006).
Dertinger, T., Colyer, R., Iyer, G., Weiss, S. & Enderlein, J. Fast, background-free, 3D super-resolution optical fluctuation imaging (SOFI). Proc. Natl Acad. Sci. USA 106, 22287–22292 (2009).
Schwartz, O. et al. Superresolution microscopy with quantum emitters. Nano Lett. 13, 5832–5836 (2013).
Gatto Monticone, D. et al. Beating the Abbe diffraction limit in confocal microscopy via nonclassical photon statistics. Phys. Rev. Lett. 113, 143602 (2014).
Tsang, M. Quantum limits to optical point-source localization. Optica 2, 646–653 (2015).
Tsang, M., Nair, R. & Lu, X.-M. Quantum theory of superresolution for two incoherent optical point sources. Phys. Rev. X 6, 031033 (2016).
Classen, A., von Zanthier, J., Scully, M. O. & Agarwal, G. S. Superresolution via structured illumination quantum correlation microscopy. Optica 4, 580 (2017).
Israel, Y., Tenne, R., Oron, D. & Silberberg, Y. Quantum correlation enhanced super-resolution localization microscopy enabled by a fibre bundle camera. Nat. Commun. 8, 14786 (2017).
Aßmann, M. Quantum-optically enhanced STORM (QUEST) for multi-emitter localization. Sci. Rep. 8, 7829 (2018).
Cui, J.-M., Sun, F.-W., Chen, X.-D., Gong, Z.-J. & Guo, G.-C. Quantum statistical imaging of particles without restriction of the diffraction limit. Phys. Rev. Lett. 110, 153901 (2013).
Sheppard, C. J. R. Superresolution in confocal imaging. Optik 80, 53–54 (1988).
Müller, C. B. & Enderlein, J. Image scanning microscopy. Phys. Rev. Lett. 104, 198101 (2010).
Rego, E. H. et al. Nonlinear structured-illumination microscopy with a photoswitchable protein reveals cellular structures at 50-nm resolution. Proc. Natl Acad. Sci. USA 109, E135–E143 (2012).
Blom, H. & Widengren, J. Stimulated emission depletion microscopy. Chem. Rev. 117, 7377–7427 (2017).
Sheppard, C. J. R., Mehta, S. B. & Heintzmann, R. Superresolution by image scanning microscopy using pixel reassignment. Opt. Lett. 38, 2889 (2013).
Gu, M. & Sheppard, C. J. R. Confocal fluorescent microscopy with a finite-sized circular detector. J. Opt. Soc. Am. A. 9, 151 (1992).
Ward, E. N. & Pal, R. Image scanning microscopy: an overview. J. Microsc. 266, 221–228 (2017).
York, A. G. et al. Instant super-resolution imaging in live cells and embryos via analog image processing. Nat. Methods 10, 1122–1126 (2013).
Roth, S., Sheppard, C. J., Wicker, K. & Heintzmann, R. Optical photon reassignment microscopy (OPRA). Opt. Nanoscopy 2, 5 (2013).
De Luca, G. M. R. et al. Re-scan confocal microscopy: scanning twice for better resolution. Biomed. Opt. Express 4, 2644–2656 (2013).
Laporte, G. P. J., Stasio, N., Sheppard, C. J. R. & Psaltis, D. Resolution enhancement in nonlinear scanning microscopy through post-detection digital computation. Optica 1, 455 (2014).
Gregor, I. et al. Rapid nonlinear image scanning microscopy. Nat. Methods 14, 1087–1089 (2017).
Glauber, R. J. The quantum theory of optical coherence. Phys. Rev. 130, 2529–2539 (1963).
Gerry, C. C. & Knight, P. L. Introductory Quantum Optics (Cambridge University Press, Cambridge, 2005).
Kimble, H. J., Dagenais, M. & Mandel, L. Photon antibunching in resonance fluorescence. Phys. Rev. Lett. 39, 691–695 (1977).
Basche’, T., Moerner, W. E., Orrit, M. & Talon, H. Photon antihunching in the fluorescence of a single dye molecule trapped in a solid. Phys. Rev. Lett. 69, 1516–1519 (1992).
Michler, P. et al. A quantum dot single-photon turnstile device. Science 290, 2282–2285 (2000).
Brouri, R., Beveratos, A., Poizat, J.-P. & Grangier, P. Photon antibunching in the fluorescence of individual color centers in diamond. Opt. Lett. 25, 1294–1296 (2000).
Grußmayer, K. S. & Herten, D.-P. Time-resolved molecule counting by photon statistics across the visible spectrum. Phys. Chem. Chem. Phys. 19, 8962–8969 (2017).
Roider, C., Ritsch-Marte, M. & Jesacher, A. High-resolution confocal Raman microscopy using pixel reassignment. Opt. Lett. 41, 3825–3828 (2016).
Portaluppi, D., Conca, E. & Villa, F. 32 x 32 CMOS SPAD imager for gated imaging, photon timing, and photon coincidence. IEEE J. Sel. Top. Quantum Electron. 24, 3800706 (2018).
Castello, M. et al. Image scanning microscopy with single-photon detector array. Preprint at https://doi.org/10.1101/335596 (2018).
Antolovic, I. M., Burri, S., Bruschini, C., Hoebe, R. A. & Charbon, E. SPAD imagers for super resolution localization microscopy enable analysis of fast fluorophore blinking. Sci. Rep. 7, 44108 (2017).
York, A. G. et al. Resolution doubling in live, multicellular organisms via multifocal structured illumination microscopy. Nat. Methods 9, 749–754 (2012).
Schwartz, O. et al. Colloidal quantum dots as saturable fluorophores. ACS Nano 6, 8778–8782 (2012).
Sýkora, J. et al. Exploring fluorescence antibunching in solution to determine the stoichiometry of molecular complexes. Anal. Chem. 79, 4040–4049 (2007).
Eisaman, M. D., Fan, J., Migdall, A. & Polyakov, S. V. Invited review article: Single-photon sources and detectors. Rev. Sci. Instrum. 82, 071101 (2011).
Hänninen, P. E., Schrader, M., Soini, E. & Hell, S. W. Two‐photon excitation fluorescence microscopy using a semiconductor laser. Bioimaging 3, 70–75 (1995).
Hell, S. W., Soukka, J. & Hänninen, P. E. Two‐ and multiphoton detection as an imaging mode and means of increasing the resolution in far‐field light microscopy: a study based on photon‐optics. Bioimaging 3, 64–69 (1995).
Acknowledgements
The authors thank Y. Ebenstein for the preparation of biological samples and S. Itzhakov for synthesizing the quantum dots used in this work. This work was supported by ERC consolidator grant ColloQuanto, ERC grant QUAMI, the ERC-POC project ‘SFICAM’, the ICore program of the ISF, the Crown Photonics Center and the Israeli ministry of science Tashtiyot program. R.L. and A.K.-P. acknowledge the hospitality of the Weizmann Institute of Science and the support of National Science Centre (Poland) grants nos. 2015/17/D/ST2/03471 and 2015/16/S/ST2/00424, the Polish Ministry of Science and Higher Education, and the Foundation for Polish Science under the FIRST TEAM project ‘Spatiotemporal photon correlation measurements for quantum metrology and super-resolution microscopy’ cofinanced by the European Union under the European Regional Development Fund.
Author information
Authors and Affiliations
Contributions
R.T., Y.I., Y.S. and D.O. proposed and designed the experiment. R.T., U.R., B.R., Y.I. and R.L. performed the experimental work. R.T., U.R., B.R., A.K.-P. and R.L. performed the data analysis. R.T. wrote the manuscript with significant contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors delcare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
This file contains more information about the work and Supplementary Figures 1–9.
Rights and permissions
About this article
Cite this article
Tenne, R., Rossman, U., Rephael, B. et al. Super-resolution enhancement by quantum image scanning microscopy. Nature Photon 13, 116–122 (2019). https://doi.org/10.1038/s41566-018-0324-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41566-018-0324-z
This article is cited by
-
Coincidence imaging for Jones matrix with a deep-learning approach
npj Nanophotonics (2024)
-
Quantum enhanced non-interferometric quantitative phase imaging
Light: Science & Applications (2023)
-
Machine learning assisted quantum super-resolution microscopy
Nature Communications (2023)
-
Focus image scanning microscopy for sharp and gentle super-resolved microscopy
Nature Communications (2022)
-
Quantum light in complex media and its applications
Nature Physics (2022)