Abstract
Several vaccines against COVID-19 use nanoparticles to protect the antigen cargo (either proteins or nucleic acids), increase the immunogenicity and ultimately the efficacy. The characterization of these nanomedicines is challenging due to their intrinsic complexity and requires the use of multidisciplinary techniques and competencies. The accurate characterization of nanovaccines can be conceptualized as a combination of physicochemical, immunological and toxicological assays. This will help to address key challenges in the preclinical characterization, will guide the rapid development of safe and effective vaccines for current and future health crises, and will streamline the regulatory process.
Similar content being viewed by others
Main
The unprecedented global effort to develop COVID-19 vaccines has led, in around two years, to more than 30 vaccines approved or in phase 3 clinical trials1. Several of these vaccines use organic nanoparticles (NPs) in their formulations for the protection and delivery of the active substance (AS)2. The AS is either a recombinant spike protein or a nucleic acid that encodes the viral antigen (the SARS-CoV-2 whole-spike protein or its subunits). Two vaccines authorized by the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) use lipid NPs (LNP) to carry the mRNA that codifies for the spike protein of SARS-CoV-2. Owing to the size of the nanocarriers (NCs), usually between 50 and 200 nm, the complete formulation is generally referred to as a nanovaccine (NV). The NC in the formulation contributes to the overall immunomodulatory properties of the vaccine itself, in addition to the delivery and protection of the cargo3.
The evaluation reports of the two LNP–messenger RNA (mRNA) NVs by FDA and EMA highlight the difficulties and challenges in their characterization and the breadth of techniques and considerations needed for a successful clinical translation of these complex systems4,5,6. To avoid costly setbacks, researchers should incorporate early in the design and development of NVs an adequate characterization strategy that utilizes multiple techniques, such as to combine orthogonal measurements4,5 and interdisciplinary expertise.
The first EMA report of the BNT162b2 mRNA vaccine4 (produced by BioNTech, Pfizer, commercially named Comirnaty) highlighted the need for additional analysis of the capped-intact RNA measurement, truncated and/or modified mRNA species fate, translated protein characterization, assessment of lipid-related impurities and biological characterization to give a complete evaluation of the clinical performance.
Limited characterization was shown to be responsible for the erroneous dose administration in the phase 3 clinical trial of the Vaxzevria COVID-19 viral vector vaccine, in which a subset cohort received a half dose of the vaccine as first dose. The mistake was corrected after a change in the methodology for dosage quantification7. This error led to a delay in vaccine permission release and generated scepticism with evident implications in public trust7, which further highlights the critical need for and importance of robust characterization processes and guidelines8. These aspects are of particular importance in emergency situations in which the time period for vaccine development and production is extremely critical.
As they are fully synthetic, LNPs represent flexible technologies that should avoid the risk of antivector immunity experienced toward viral vectors9. NVs bring the hope to reduce and/or avoid carrier immunogenicity, and to permit an efficient prime–boost strategy with the same carrier, which can be used for the further dose administration of a COVID-19 vaccine (as it is for annual influenza) in response to the emerging variants or to exploit the same NC in vaccines against other pathogens.
To design vaccines able to induce an optimal response, it is essential to optimize their immunogenicity without enhancing reactogenicity. To address these challenges, a commonly shared characterization strategy is important during the preclinical development and would also help regulators to better and faster assess the product efficacy and safety.
Nanoparticles in COVID-19 nanovaccines
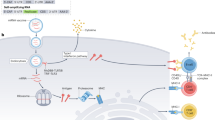
Several COVID-19 NVs have been developed2,10; they can be grouped into three main categories based on their functional components: virus (not addressed here), protein based or nucleic acid based (Fig. 1a). Almost all the NCs share several components, such as ionizable lipids, polyethylene glycol (PEG)–lipids, structural lipids and cholesterol (Fig. 1a), assembled as NPs of 50 to 150 nm in size. The goal for a rational NV design is to elicit an effective adaptive immune response that generates immunological memory. This means a relatively long-lived surveillance system of specific memory B cells able to build up a massive clonal expansion of plasma blasts with subsequent differentiation into plasma cells capable of producing specific antibodies in response to pathogen exposure. These specific antibodies should block interaction between the virus and the host cell (neutralization), and ultimately lead to virus clearance. In addition to the generation of neutralizing antibodies, the activation of cytotoxic CD8+ T cells (CD, cluster of differentiation) is essential to clear host cells infected by the virus.
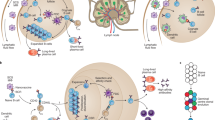
a, Schematic representation of different NPs used for mRNA, DNA and protein-based COVID-19 vaccines. For mRNA, the lipid NCs contain ionizable lipid, PEG–lipid, structural lipid (distearoylphosphatidylcholine, DSPC) and cholesterol. DNA cargos are, instead, protected by either proteolipid NPs, formulated with neutral lipid and the proprietary fusion-associated small transmembrane (FAST) proteins or with polymeric poly(lactic-co-glycolic acid (PLGA) NPs, whereas protein-based NVs use nanoemulsion formulations, such as AS03, Matrix-M and MF59 (oil in water emulsion of α-tocopherol, squalene or saponin), or virus-like particles (chimeric multimers of proteins and phospholipids self-assembled to mimic the viral structure). b, NV immune modulation overview. (1) Inductive phase: IM NV administration induces an inflammatory cytokines release and immune cell recruitment (for example, neutrophils, macrophages and APCs). APCs (for example, DCs) internalise, process and load the antigen on MHC molecules. (2) Antigen-loaded APCs and free NPs (<250 nm) move through the lymphatic vessels to the draining lymph nodes, where subcapsular sinus macrophages uptake the free NPs and present them to B cells. (3) Migratory DC present antigens to the T cells, which differentiate into effector T helper or T cytotoxic cells. (4) Activated T helper cells move to the T/B zone border and interact with antigen-specific B lymphocytes. T cells drive the formation of a germinal centre for B-cell maturation. Some of the B cells differentiate into plasma blasts, whereas others differentiate into long-lived plasma cells and memory B cells. (5) Activated immune cells leave the lymph node and enter the bloodstream. (6) Effector phase: activated immune cells exit the bloodstream and exert their effector function. Plasma cells secrete antibodies and activated cytotoxic and helper T cells carry out cell-mediated responses. Antigen-specific memory B and T cells can be reactivated upon pathogen encounter, leading respectively to antigen-specific antibodies production and killing of the infected cells. c, Schematic overview of NV-processing and antigen-presentation pathways by APCs. mRNA cargo embedded within ionizable lipid NPs (upper right) enters the cell via the endosome and as the endosome maturates, pH reduction induces the protonation of the lipid (lipid changes colour from light to dark pink). The interaction of the positively charged lipids with the endosomal anionic lipids alters the membrane stability and induces fusion and the release of the mRNA payload into the cytoplasm (endosomal escape). Ribosomes translate the mRNA into proteins, which undergo proteosomal degradation and loading onto MHC-I and/or MHC-II for antigen presentation. Some of the mRNA molecules can be sensed by innate immune sensors (Toll-like receptors and retinoic acid-inducible gene I (RIG-I)), which leads to IFN-1 secretion. Proteolipid and polymeric NPs (upper left) deliver the DNA payload through the fusogenic FAST protein or by means of electroporation, respectively. DNA enters the nucleus and undergoes nuclear transcription; the newly synthetized mRNA exits the nucleus and is translated into protein. Nanoemulsions and virus like particles (left) enter the cell, and proteins are directly processed within the endosome and exposed onto MHC-II. NF-κB, nuclear factor-κB. ER, endoplasmic reticulum; TAP, Transporter associated with antigen processing protein complex.
Intramuscular (IM) administration is the primary route of choice for COVID-19 NVs as it both produces a low local reactogenicity and ensures an efficient biodistribution, which contributes to the overall immunogenicity of NVs4. On IM injection, the NV enters the lymphatic vessels and is delivered to the lymph nodes11 (Fig. 1b, (1) and (2)). NVs are also internalized by tissue-resident antigen-presenting cells (APC: dendritic cells (DCs), monocytes and macrophages) and by muscle cells (Fig. 1b, (1)). Regardless of the NP used, the final aim of all these nanoformulations is the efficient delivery and/or expression of the antigen within the cell, its processing, the upload of the epitopes on the major histocompatibility complex (MHC) and the exposure on the APC surface (Fig. 1c) for T-cell activation that supports B-cell antibody production Fig. 1b, (6).
Characterization of nanovaccines
The characterization of NVs is intrinsically different from that of the ‘traditional’ vaccines or from nanomedicines; the NC generally contains several components, the cargo is often a complex biological molecule and the AS delivery, intracellular fate, biodistribution and, ultimately, immunogenicity depend on the NC composition and physicochemical properties.
Here we propose an iterative characterization strategy for the preclinical assessment of the quality, efficacy and safety of NVs based on the measurements of physical, chemical and stability properties, in vitro and in vivo immunogenicity and toxicity (Fig. 2). The emphasis on characterizing immunogenicity is a major difference compared with the safety characterization of intravenously administered nanomedicines for cancer treatments12. In fact, for NVs the primary goal is to promote an effective immune response to the cargo while minimizing the response to the NC; for intravenously administered nanomedicines, on the contrary, there is a need to avoid or reduce interaction with the immune system. Figure 2 highlights the iterative nature of the development process that leads to the optimal formulations with a high potency and reduced toxicity. The methods and the acceptability criteria for every critical parameter must be established based on the mechanism of action of the specific vaccine and on the impact on the performance of every component of the formulation13. In addition, mRNA- or protein-based NVs will require different techniques for the characterization of the AS.
Physicochemical properties of the formulation impact on its immunogenicity and safety, for example, the pKa value of the ionizable lipid in the LNP–mRNA vaccine of Moderna is directly linked to its overall potency and safety3. The availability of this information for a wide range of different formulations could enable groups to establish correlations between physicochemical characteristics of a given formulation and its immunogenicity, efficacy and safety. To obtain meaningful correlations with published experimental data there is a need to improve the reproducibility and robustness of reports, as proposed by the MIRIBEL (Minimum Information Reporting in Bio-Nano Experimental Literature) initiative14,15.
Physical characterization
Size or, better, particle-size distribution, is a key property of NVs and influences both the immunomodulation and the biodistribution of the NC–AS system. For example, LNP–mRNA particles of 146 nm induced a statistically significant higher immunoglobulin G (IgG) titre in mice compared with smaller (64 nm) ones16. Dynamic light scattering is probably the technique most used to measure size; it is very simple and easy to use, can be very useful as a preliminary checkpoint for sample screening, but it is not appropriate for polydisperse samples17. Techniques that combine a separation step (based on the size of the different particles) with an online measurement of size (dynamic light scattering and/or multiangle light scattering) can accurately measure the size distribution of challenging samples. In particular, asymmetric-flow field-flow fractionation coupled with multidetectors is well suited for lipid-based NPs18, even if some technical adjustments need to be made for LNP–RNA19. Other techniques, such as NP tracking analysis, tunable resistive pulse sensing and analytical ultracentrifugation20, can also provide the information needed, with various degrees of accuracy as well as other complementary data. Transmission electron microscopy is the technique most used (even if technically demanding) to image LNP–mRNA.
Chemical characterization
The identification and quantification of the different components of both the NC and the AS is the key chemical characterization step for NVs; it usually requires a careful sample preparation, followed by measurements with the appropriate analytical techniques. The most used is generally liquid chromatography (LC) separation coupled to concentration detectors with an appropriate linearity and sensitivity. LC coupled to a charged aerosol detector provides accurate results for both identification and quantification of the lipid NCs of LNP–mRNA authorized vaccines5. Similar results could also be obtained with mass spectrometry (MS) detectors. NMR spectroscopy can be used both to investigate the molecular structure of the novel lipid excipients and to quantify them5, and high-resolution mass spectrometry can give complementary information on the structure of the complex lipid components. The characterization of the AS is also particularly challenging—owing to the complexity of proteins and nucleic acids, it needs to follow their structural hierarchy. Electrophoresis (particularly capillary gel electrophoresis) is a versatile technique that can give information on the nucleic acid and protein integrity, molecular weight and concentration, but also, in the case of proteins, on the aggregation, disulfide bonds and glycosylation. The coupling of an analytical separation method (such as a capillary electrophoresis or LC) to an MS/MS detector reveals useful information on both the sequence and post-translational modifications of proteins21, as well as on the presence of eventual manufacture contaminants (for example, residual DNA). For RNA- and DNA-based vaccines, the sequence can be confirmed by next-generation sequencing.
The capping at the 5′ and the 3′ poly-A tail are important for the stability of mRNA and its translational efficiency, and result in a higher production of the desired antigen22, which potentially leads to a more favourable therapeutic index. The amount of uncapped mRNA can be measured by LC–MS23. Indeed, EMA in its evaluation of the BNT162b2 for marketing authorization required further characterization data for the quality attribute 5′ capped intact RNA4.
The secondary structure of both protein antigens and mRNA can be measured with classic biophysical techniques, such as circular dichroism. The AS content can be measured with the sophisticated analytical techniques described above, but also with simpler ones (such as by ultraviolet absorption and fluorescence) using appropriate labelling probes to increase the sensitivity. Measurement of the free and/or encapsulated cargo is of critical importance. Usual methods combine an ultrafiltration step to separate the free from the bound cargo molecules, followed by quantification with the appropriate analytical technique. Measurement of the ζ potential (the electrostatic potential at the hydrodynamic plane of shear of the NPs) through electrophoretic light scattering is currently the most diffused and straightforward method for surface charge characterization in nanocolloids24. NCs that contain ionizable lipids also require the assessment of their pKa, a key characteristic for NP functionality (especially for endosomal escape, Fig. 1c), which can be assessed by several methods, such as potentiometry and fluorimetry25.
Stability
Given the global spread of COVID-19, the distribution and storage of vaccines has raised great concern; the announcement of the regulatory approval of the BNT162b2 vaccine started a technological race to find more stable and high-temperature-tolerant systems26. General stability testing guidelines can be applied for these NVs (for example, ICH Q1A (R2) and ICH Q5C); however, as this is the debut of such a combination of biotechnological AS and innovative carrier nanotechnologies, special attention is required.
In protein-based vaccines, the structural unfolding of the protein antigen can reduce the potency; the stability and thermal unfolding of protein antigens bound to the NC can be monitored by circular dichroism27 and by differential scanning calorimetry28.
The chemical, physical and biological identity of the formulation must be assessed at different time points by treating the samples according to the real-life conditions of use and storage, which include cycles of freeze–thaw and the manipulation time at room temperature. The translation from the laboratory to the real-life application goes through a batch-to-batch consistency assessment. Owing to the variety of existing products and the diversity of their physicochemical characteristics, it is important to select specific quality attributes for each formulation, evaluate their reproducibility within established ranges and carry this out for an appropriate number of batches29.
In vitro testing
In vitro immunostimulation
The evaluation of in vitro delivery efficiency, the interaction with immune cells, the immunomodulatory properties and how modifications of the NP composition can affect these properties is essential in the characterization of novel NP-based vaccines (Fig. 2). NPs play an important role in the activation of APCs, which determine the initiation and polarization of the immune response, an essential step to shape and optimize the humoral and cellular vaccine responses (Fig. 1b, (3) and (4)). Indeed, although NP internalization occurs even in muscle cells on IM injection, DCs are the principal and most efficient target for NV delivery (Fig. 1c), even if other professional APCs, such as macrophages and B cells, can present the antigen to T cells. The interaction and antigen delivery of NVs to DCs is therefore critical and must be assessed to optimize the NV for the subsequent induction of humoral and cellular vaccine responses (Fig. 1b, (3) and (4)). Monocyte-derived DCs isolated from human peripheral blood mononuclear cells30 can be used to assess NV uptake, cell activation, antigen processing and MHC class I and II presentations (Fig. 1c). The kinetics of NV uptake by target cells can be followed by optical microscopy of fluorescent stained NPs and/or cargo. DC maturation can be monitored by analysing the expression of activation markers (T-costimulatory molecules, such as CD40, CD80 and CD86)31 using multiparametric flow cytometry, and cytokines production (whose level should be balanced not to overstimulate the inflammatory pathway) with multiplex assay and intracellular flow cytometry.
Nowadays, novel technologies are implemented to study the immune response in vitro. Of note, tissue engineering (for example, three-dimensional organoids) aims to recreate an in vitro version of human organs, which are a very promising replacement of animal models to appreciate the mechanism of NP localization and accumulation, and to carry out toxicology32 and immunological studies33.
In vitro toxicology
After having evaluated the sterility (ISO 11737-2:2019), bioburden (EN ISO 11737-1:2018) and endotoxin presence (ISO 29701:201029 or in alternative functional Toll-like receptor 4 (TLR4)-based assays34), another important step in the NV characterization is the establishment of the toxicological profile, a prerequisite for nanoformulation biocompatibility. Toxicology can be assessed in vitro following the available standards (ASTM E2526-08 and ISO 10993-22); however, it is recommended to also use more relevant target cells, such as whole blood and peripheral blood mononuclear cells as well as specific immune cell subtypes, such as CEM (T cell), Raji (B lymphocyte) and THP-1 (human monocyte) cell lines so as to define clear exposure–response relationships35. Peripheral blood mononuclear cells are suitable cells for a micronucleus test, which is required for the risk assessment of every new NV component and excipient13. Although NVs are generally administered IM they could, however, interact with blood. Thus, haematotoxicity and blood immunotoxicity, which include haemolysis and complement activation, should be evaluated. For this scope, standard in vitro test methods designed for NPs and medical devices (ASTM E2524-08, ISO 10993-4 and ISO/TR 10993-22:2017) provide a structured test-selection system and references that are useful to also tailor blood compatibility analysis for NVs, and are quicker and less costly than the in vivo methods. Dose–response systemic complement activation studies are fundamental, especially in the case of liposome-based or biological-based formulations, due to potential hypersensitivity reactions36.
The coagulation and the clotting cascade is another critical point as alteration of this cascade system can have important adverse effects, such as thrombosis. Vaccine-induced thrombotic–thrombocytopenia, mediated by the presence of platelet-activating antibodies against factor 4, has emerged as a rare idiosyncratic reaction to adenovirus-based COVID-19 vaccines37,38. An AstraZeneca case on the risk of correlation between adenovirus COVID-19 vaccine and thrombosis has sparked great concern, which underlines the importance of this assessment. The effects on white blood cells also need to be assessed as adverse interactions with nanomaterials can impair the body’s natural immune defence.
In vivo preclinical testing
Animal models play a critical role in the assessment of immunogenicity, efficacy and safety of vaccine candidates (Fig. 2) and preclinical data are mandatory to advance them to the clinical phases. Based on the World Health Organization guidelines on the non-clinical evaluation of vaccines39, preclinical studies in animal models should be conducted in strict compliance with national and international regulations for the protection of laboratory animals40 and biosafety requirements41. Importantly, the administration route should correspond to that intended for the clinical studies. Small-animal models, such as mice and rats, are useful for a preliminary evaluation of safety, immunogenicity and dosing of the vaccine candidates. Non-human primates (that is, rhesus macaques) are recognized, so far, as the best models to assess the protective efficacy of vaccine candidates as they are phylogenetically related to humans (their immune system, respiratory system anatomy and tissue structure are very similar).
NP biodistribution and persistence can be assessed by in vivo imaging techniques, which allow us to optimize the NV by facilitating the transport and accumulation to targeted organs42 (secondary lymphatic organs) while limiting the adverse events frequency and severity by reducing local reactions (off-target effects).
SARS-CoV-2 spike and receptor-binding domain (RBD)-specific antibody induction and persistence overtime should be assessed as indicators of virus-specific humoral responses. The functionality of the induced antibody (especially the capacity to prevent host-cell infection by virus neutralization) can be assessed using pseudovirus type or live virus plaque reduction neutralization assays4 and with the SARS-CoV-2 surrogate virus neutralization test based on an antibody-mediated blockage of the ACE2–spike protein–protein interaction (hACE2/RBD inhibition assay; hACE2, human angiotensin-converting enzyme 2)43. Given that the SARS-CoV-2 virus primarily infects the mucosal surface of the respiratory tract, it is particularly important to assess the mucosal immune responses of secretory IgA antibodies and local IgG in mucosal samples collected in the respiratory tract (lung and nasal washes) and antigen-specific B- and T-cell responses elicited in the rodent nasal mucosa-associated lymphoid tissue (which resembles the human Waldeyer’s ring) or in cervical draining lymph nodes44,45.
The induction of memory B cells is a critical outcome of an effective stimulation of the immune system by vaccination. SARS-CoV-2 spike and RBD-specific memory B cells can be analysed by multiparametric flow cytometry using a fluorescent spike antigen as the probe combined with different surface marker antibodies specific for the memory phenotype46. To assess the effector function of antigen-positive B cells on vaccination, the frequency of antigen-specific antibody secreting cells can be determined by an enzyme-linked immune absorbent spot assay on antigen restimulation.
The immunogenicity of NV formulation is also measured by the ability to skew T-helper (TH) cells toward TH1, TH2 and/or TH17 responses, analysing the T-cell cytokine profile (for example, interferon γ (IFN-γ), interleukin 2 (IL-2), tumour necrosis factor α, IL-4, IL-5, IL-10, IL-13 and IL-17), which can be measured with flow cytometry intracellular cytokine staining (ICS) or multiplex cytokine assay. The MHC–peptide tetramers represent a powerful technology for the detection of antigen-specific T cells by specific binding to their T-cell receptor, and their combination with functional assays, that is, intracellular cytokine staining, can be instrumental to characterize the effector antigen-specific T-cell response47. In this context, the computational analysis of complex multiparametric flow cytometry data represents a powerful tool to dissect all the possible cell phenotypes in an operator-independent unbiased way and can be conducted to deeply characterize multiple B- and T-cell subsets48.
For COVID-19 vaccines, the efficacy mainly relies on neutralizing antibodies. Although there is still no unanimous protective threshold established for humans49, in vivo challenge studies in mice could demonstrate the protective role and safety of the NV50. The animal models of SARS-CoV-2 infection currently used are hACE2 transgenic mice51 and non-human primates52 (the latter, although not explicitly required in regulatory guidance documents, are a critical animal model for the immune evaluation of NVs) infected intranasally or intratracheally. Challenge–protection studies that monitor the absence of pathogen replication in target organs (for example, lungs and nasal turbinate for COVID-19 disease) and eventual body weight loss or other associated pathological conditions should be conducted. Controlled human infection models, as the ones already used in influenza or salmonella studies, have also been proposed as a possible approach to accelerate SARS-CoV2 vaccine development.
In vivo toxicological studies are required to advance to clinical trials and should be conducted according to the WHO guidelines39. Toxicity studies should address the potential of the vaccine for causing local inflammatory reactions, and possible effects on the draining lymph nodes, systemic toxicity and on the immune system, together with routine clinical signs observations (such as body weight, posture, gait, and food consumption).
In the public assessment reports of the EMA, for the respective NV, Pfizer/BioNTech and Moderna provided vaccine immunological data on humoral (antigen-specific IgG antibody response, antibody–antigen binding affinity and neutralizing antibody titre) and cellular (CD4+ and CD8+ T-cell subsets, and characterization of the immunophenotype of B and T cells in mice lymph nodes) immune response characterization and cytokines profiling in two animal models (mouse and non-human primates)4,5 and conducted challenge studies in rhesus macaques as a non-clinical proof of concept, whereas preclinical toxicology tests, for safety assessment, were conducted in rats.
Conclusions and perspective
The unprecedented speed of advance of COVID-19 vaccines has changed the conventional strategy for the development of vaccines (for example, safety and efficacy studies were conducted simultaneously, traditional test protocols were shortened and regulation procedures were conducted in parallel in multiple countries), and highlights their strategic role in fighting pandemics.
The ability of NCs to modulate humoral and cellular responses, critically important in the case of a compromised or senescent immune system, such as in the elder population53,54, together with the proven success in systemic and mucosal administration, make them particularly useful for vaccine design. In fact, they can incorporate diverse antigens and immunostimulants compared with conventional subunit and/or adjuvant vaccines, and in addition improve the stability and shelf life of the AS.
The current pandemic emergency has given an exceptional boost to the clinical testing of these nanotechnologies, which serves as a global test bed that is leading us to rethink the conventional parameters of nanomedicine and adapt the characterization processes accordingly. The implementation of a structured characterization strategy will contribute to improve vaccine performance and reduce development times, a critical aspect, especially in response to emerging pathogen outbreaks and antimicrobial resistance, an already declared ‘future’ challenge55. Finding the right workflow means we must critically evaluate each measurement technique and eventually innovate the methods when needed. The different techniques (and especially the most innovative ones) will need both reference materials and standard methods to ensure the required confidence in the measurements. The optimized characterization will play a key role in the fast development of the ‘next generation’ NVs that ideally will be stable at room temperature, and so require less stringent storage conditions and therefore be simpler to produce and deliver.
The numerous vaccine platforms proposed require very distinct production lines, which has increased investment uncertainty, and thus some candidates rely on proven vaccine approaches adapted by large pharmaceutical companies with proven regulatory and manufacturing experience. Smaller companies and academic labs were shown to be key players in the development of COVID-19 vaccines, particularly suited for the development and characterization of NVs, but many lack the large spectrum of competences and measurement techniques, and extensive experience in the regulatory process. They could particularly benefit from a predefined vaccine characterization process that could guide them through the characterization steps to be conducted, in addition to external services for the analysis that cannot be performed in house. There are, indeed, several characterization laboratories and research infrastructures, such as the US National Cancer Institute Nanotechnology Characterization Laboratory (https://ncl.cancer.gov/), the European Union Nanomedicine Characterization Laboratory (www.euncl.org), the TRANSVAC consortium (www.transvac.org) and specialized contract research organizations that could provide the necessary analysis once guided by a defined characterization process. This would be particularly relevant for regulators to support the assessment of novel vaccines and speed up the approval process, especially in emergencies.
Given the scale and urgency, international collaboration has a key role to develop, validate and apply measurement methods and standards in NVs. Research scientists from academia, industry and regulatory agencies should join forces to identify and prioritize specific measurements and standards needed to facilitate the preclinical development of these innovative vaccines.
References
COVID-19 Vaccine Tracker and Landscape (WHO, accessed 4 August 2021); https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines
Kisby, T., Yilmazer, A. & Kostarelos, K. Reasons for success and lessons learnt from nanoscale vaccines against COVID-19. Nat. Nanotechnol. 16, 843–850 (2021).
Hassett, K. J. et al. Optimization of lipid nanoparticles for intramuscular administration of mRNA vaccines. Mol. Ther. Nucleic Acids 15, 1–11 (2019).
Comirnaty Assessment Report EMA\/707383\/2020 (EMA, 2021); https://www.ema.europa.eu/en/documents/assessment-report/comirnaty-epar-public-assessment-report_en.pdf
COVID-19 Vaccine Moderna Assessment Report EMA/15689/2021 (EMA, 2021); https://www.ema.europa.eu/en/documents/assessment-report/spikevax-previously-covid-19-vaccine-moderna-epar-public-assessment-report_en.pdf
Pfizer-BioNTech COVID-19 FDA Briefing Document (FDA, 2020); https://www.fda.gov/media/144245/download
Mahase, E. Covid-19: UK government asks regulator to assess Oxford vaccine as questions are raised over interim data. Brit. Med. J. 371, m4670 (2020).
Knoll, M. D. & Wonodi, C. Oxford–AstraZeneca COVID-19 vaccine efficacy. The Lancet 397, 72–74 (2021).
Rappuoli, R. & De Gregorio, E. Editorial overview: vaccines: novel technologies for vaccine development. Curr. Opin. Immunol. 41, v–vii (2016).
Calzolai, L., Gioria, S. & Magrì, D. Nanoparticle-Based Vaccines in Clinical Trial/Use for COVID-19 and Licensed for Other Pathogens (European Commission, 2021); https://data.jrc.ec.europa.eu/dataset/1575f3b3-f8e6-4f6c-a296-77e8d1be4ee1#citation
Lindsay, K. E. et al. Visualization of early events in mRNA vaccine delivery in non-human primates via PET–CT and near-infrared imaging. Nat. Biomed. Eng. 3, 371–380 (2019).
Hall, J. B., Dobrovolskaia, M. A., Patri, A. K. & McNeil, S. E. Characterization of nanoparticles for therapeutics. Nanomedicine 2, 789–803 (2007).
Drug Products, Including Biological Products, that Contain Nanomaterials—Guidance for Industry (FDA, 2017); https://www.fda.gov/regulatory-information/search-fda-guidance-documents/drug-products-including-biological-products-contain-nanomaterials-guidance-industry
Faria, M. et al. Minimum information reporting in bio–nano experimental literature. Nat. Nanotechnol. https://doi.org/10.1038/s41565-018-0246-4 (2018).
Leong, H. S. et al. On the issue of transparency and reproducibility in nanomedicine. Nat. Nanotechnol. 14, 629–635 (2019).
Hassett, K. J. et al. Impact of lipid nanoparticle size on mRNA vaccine immunogenicity. J. Control. Release 335, 237–246 (2021).
Caputo, F. et al. Measuring particle size distribution and mass concentration of nanoplastics and microplastics: addressing some analytical challenges in the sub-micron size range. J. Colloid Interface Sci. https://doi.org/10.1016/j.jcis.2020.12.039 (2021).
Parot, J., Caputo, F., Mehn, D., Hackley, V. A. & Calzolai, L. Physical characterization of liposomal drug formulations using multi-detector asymmetrical-flow field flow fractionation. J. Control. Release 320, 495–510 (2020).
Mildner, R. et al. Improved multidetector asymmetrical-flow field-flow fractionation method for particle sizing and concentration measurements of lipid-based nanocarriers for RNA delivery. Eur. J. Pharm. Biopharm. 163, 252–265 (2021).
Mehn, D., Capomaccio, R., Gioria, S., Gilliland, D. & Calzolai, L. Analytical ultracentrifugation for measuring drug distribution of doxorubicin loaded liposomes in human serum. J. Nanopart. Res. 22, 1–7 (2020).
Tran, J. C. et al. Mapping intact protein isoforms in discovery mode using top-down proteomic. Nature 480, 254–258 (2011).
Tanguay, R. L. & Gallie, D. R. Translational efficiency is regulated by the length of the 3′ untranslated region. Mol. Cell. Biol. 16, 146–156 (1996).
Beverly, M., Dell, A., Parmar, P. & Houghton, L. Label-free analysis of mRNA capping efficiency using RNase H probes and LC–MS. Anal. Bioanal. Chem. 408, 5021–5030 (2016).
Varenne, F. et al. Standardization and validation of a protocol of zeta potential measurements by electrophoretic light scattering for nanomaterial characterization. Colloids Surf. https://www.sciencedirect.com/science/article/abs/pii/S0927775715301862 (2015).
Reijenga, J., Van Hoof, A., Van Loon, A. & Teunissen, B. Development of methods for the determination of pKa values. Anal. Chem. Insights https://journals.sagepub.com/doi/full/10.4137/ACI.S12304 (2013).
Capua, I. & Giaquinto, C. The unsung virtue of thermostability. Lancet 397, 1346 (2021).
Guerrini, G. et al. Physicochemical characterization cascade of nanoadjuvant–antigen systems for improving vaccines. Vaccines 9, 544 (2021).
\Durowoju, I. B., Bhandal, K. S., Hu, J., Carpick, B. & Kirkitadze, M. Differential scanning calorimetry—a method for assessing the thermal stability and conformation of protein antigen. J. Vis. Exp. https://www.jove.com/t/55262/differential-scanning-calorimetry-method-for-assessing-thermal (2017).
Crist, R. M. et al. Common pitfalls in nanotechnology: lessons learned from NCI’s Nanotechnology Characterization Laboratory. Integr. Biol. 5, 66–73 (2013).
Sallusto, F. & Lanzavecchia, A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J. Exp. Med. 179, 1109–1118 (1994).
Hubo, M. et al. Costimulatory molecules on immunogenic versus tolerogenic human dendritic cells. Front. Immunol. 4, 82 (2013).
Cupedo, T., Stroock, A. D. & Coles, M. C. Application of tissue engineering to the immune system: development of artificial lymph nodes. Front. Immunol. 3, 3389 (2012).
Wagar, L. E. et al. Modeling human adaptive immune responses with tonsil organoids. Nat. Med. 27, 125–135 (2021).
Haile, L. A. et al. Cell based assay identifies TLR2 and TLR4 stimulating impurities in interferon beta. Sci. Rep. 7, 10490 (2017).
Della Camera, G. et al. A step-by-step approach to improve clinical translation of liposome-based nanomaterials, a focus on innate immune and inflammatory responses. Int. J. Mol. Sci 22, 820 (2021).
Szebeni, J. & Moghimi, S. M. Liposome triggering of innate immune responses: a perspective on benefits and adverse reactions. J. Liposome Res. 19, 85–90 (2009).
Huynh, A., Kelton, J. G., Arnold, D. M., Daka, M. & Nazy, I. Antibody epitopes in vaccine-induced immune thrombotic thrombocytopaenia. Nature 596, 565–569 (2021).
Greinacher, A. et al. Thrombotic thrombocytopenia after ChAdOx1 nCov-19 vaccination. N. Engl. J. Med. 384, 2092–2101 (2021).
Non-Clinical Evaluation of Vaccines (WHO); https://www.who.int/teams/health-product-policy-and-standards/standards-and-specifications/vaccine-standardization/non-clinical-evaluation-of-vaccines
Manual of Laboratory Methods for Testing of Vaccines used in the WHO Expanded Programme on Immunization (WHO, 1997); https://apps.who.int/iris/handle/10665/63576
Laboratory Biosafety Manual 4th edn (WHO, 2020); https://www.who.int/publications/i/item/9789240011311
Pardi, N. et al. Expression kinetics of nucleoside-modified mRNA delivered in lipid nanoparticles to mice by various routes. J. Control. Release 217, 345–351 (2015).
Tan, C. W. et al. A SARS-CoV-2 surrogate virus neutralization test based on antibody-mediated blockage of ACE2–spike protein–protein interaction. Nat. Biotechnol. 38, 1073–1078 (2020).
Fiorino, F., Pettini, E., Pozzi, G., Medaglini, D. & Ciabattini, A. Prime-boost strategies in mucosal immunization affect local IgA production and the type of the response. Front. Immunol 4, 128 (2003).
Medaglini, D. et al. In vivo activation of naive CD4+ T cells in nasal mucosa-associated lymphoid tissue following intranasal immunization with recombinant Streptococcus gordonii. Infect. Immun. 74, 2760–2766 (2006).
Ciabattini, A. Evidence of SARS-CoV-2-specific memory B cells six months after vaccination with the BNT162b2 mRNA vaccine. Front. Immunol 12, 3389 (2021).
Pastore, G. et al. Optimized protocol for the detection of multifunctional epitope-specific CD4+ T cells combining MHC-II tetramer and intracellular cytokine staining technologies. Front. Immunol. 10, 2304 (2019).
Lucchesi, S. et al. Computational analysis of multiparametric flow cytometric data to dissect B cell subsets in vaccine studies. Cytometry A 97, 259–267 (2020).
Gilbert, P. B. et al. Immune correlates analysis of the mRNA-1273 COVID-19 vaccine efficacy clinical trial. Science 375, 43–50 (2022).
DiPiazza, A. T. et al. COVID-19 vaccine mRNA-1273 elicits a protective immune profile in mice that is not associated with vaccine-enhanced disease upon SARS-CoV-2 challenge. Immunity 54, 1869–1882 (2021).
Jiang, R.-D. et al. Pathogenesis of SARS-CoV-2 in transgenic mice expressing human angiotensin-converting enzyme 2. Cell 182, 50–58 (2020).
Urano, E. et al. COVID-19 cynomolgus macaque model reflecting human COVID-19 pathological conditions. Proc. Natl. Acad. Sci. USA 118, e210484711 (2021).
Ciabattini, A. et al. Vaccination in the elderly: the challenge of immune changes with aging. Semin. Immunol. 40, 83–94 (2018).
Ciabattini, A. et al. Shelter from the cytokine storm: pitfalls and prospects in the development of SARS-CoV-2 vaccines for an elderly population. Semin. Immunopathol. 42, 619–634 (2020).
Jansen, K. U. & Anderson, A. S. The role of vaccines in fighting antimicrobial resistance (AMR). Hum. Vaccin. Immunother. 14, 2142–2149 (2018).
Acknowledgements
The study was partly supported by the EU Joint Research Centre Exploratory Research programme (‘NanoMicrobials’, D.M. and ‘FUTURE’, G.G.) through the Horizon 2020 Framework Programme under the project TRANSVAC2 (grant no. 730694). We thank I. Defaronou for technical assistance with the graphics. We thank A. Ciabattini, D. Boraschi and A. Hoeveler for helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Neill Liptrott and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guerrini, G., Magrì, D., Gioria, S. et al. Characterization of nanoparticles-based vaccines for COVID-19. Nat. Nanotechnol. 17, 570–576 (2022). https://doi.org/10.1038/s41565-022-01129-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-022-01129-w
This article is cited by
-
Subcutaneous drug delivery from nanoscale systems
Nature Reviews Bioengineering (2024)
-
Adjuvant lipidoid-substituted lipid nanoparticles augment the immunogenicity of SARS-CoV-2 mRNA vaccines
Nature Nanotechnology (2023)
-
Active ingredient vs excipient debate for nanomedicines
Nature Nanotechnology (2023)
-
Shortwave-infrared-light-emitting probes for the in vivo tracking of cancer vaccines and the elicited immune responses
Nature Biomedical Engineering (2023)
-
Cholesterol modulates the physiological response to nanoparticles by changing the composition of protein corona
Nature Nanotechnology (2023)