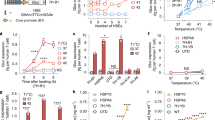
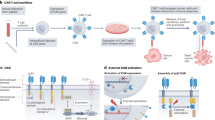
Abstract
Chimeric antigen receptor (CAR) T cell-based immunotherapy, approved by the US Food and Drug Administration, has shown curative potential in patients with haematological malignancies. However, owing to the lack of control over the location and duration of the anti-tumour immune response, CAR T cell therapy still faces safety challenges arising from cytokine release syndrome and on-target, off-tumour toxicity. Herein, we present the design of light-switchable CAR (designated LiCAR) T cells that allow real-time phototunable activation of therapeutic T cells to precisely induce tumour cell killing. When coupled with imaging-guided, surgically removable upconversion nanoplates that have enhanced near-infrared-to-blue upconversion luminescence as miniature deep-tissue photon transducers, LiCAR T cells enable both spatial and temporal control over T cell-mediated anti-tumour therapeutic activity in vivo with greatly mitigated side effects. Our nano-optogenetic immunomodulation platform not only provides a unique approach to interrogate CAR-mediated anti-tumour immunity, but also sets the stage for developing precision medicine to deliver personalized anticancer therapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Supplementary Information is available online. The plasmids and reagents are available from the corresponding authors upon request. Source data are provided with this paper.
References
June, C. H., O’Connor, R. S., Kawalekar, O. U., Ghassemi, S. & Milone, M. C. CAR T cell immunotherapy for human cancer. Science 359, 1361–1365 (2018).
Chmielewski, M., Hombach, A. A. & Abken, H. Antigen-specific T-cell activation independently of the MHC: chimeric antigen receptor-redirected T cells. Front. Immunol. 4, 371 (2013).
Grupp, S. A. et al. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N. Engl. J. Med. 368, 1509–1518 (2013).
Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
Sadelain, M., Brentjens, R. & Riviere, I. The basic principles of chimeric antigen receptor design. Cancer Discov. 3, 388–398 (2013).
Kochenderfer, J. N. et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119, 2709–2720 (2012).
Neelapu, S. S. et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N. Engl. J. Med. 377, 2531–2544 (2017).
Kennedy, M. J. et al. Rapid blue-light-mediated induction of protein interactions in living cells. Nat. Methods 7, 973–975 (2010).
Guntas, G. et al. Engineering an improved light-induced dimer (iLID) for controlling the localization and activity of signaling proteins. Proc. Natl Acad. Sci. USA 112, 112–117 (2015).
He, L. et al. Near-infrared photoactivatable control of Ca2+ signaling and optogenetic immunomodulation. Elife https://doi.org/10.7554/eLife.10024 (2015).
Wu, J. et al. An activating immunoreceptor complex formed by NKG2D and DAP10. Science 285, 730–732 (1999).
Wu, C. Y., Roybal, K. T., Puchner, E. M., Onuffer, J. & Lim, W. A. Remote control of therapeutic T cells through a small molecule-gated chimeric receptor. Science 350, aab4077 (2015).
Irving, B. A., Chan, A. C. & Weiss, A. Functional characterization of a signal transducing motif present in the T cell antigen receptor zeta chain. J. Exp. Med. 177, 1093–1103 (1993).
Zimmerman, S. P. et al. Tuning the binding affinities and reversion kinetics of a light inducible dimer allows control of transmembrane protein localization. Biochemistry 55, 5264–5271 (2016).
Chen, J. et al. NR4A transcription factors limit CAR T cell function in solid tumours. Nature 567, 530–534 (2019).
Maraskovsky, E., Chen, W. F. & Shortman, K. IL-2 and IFN-gamma are two necessary lymphokines in the development of cytolytic T cells. J. Immunol. 143, 1210–1214 (1989).
Yu, N. et al. Near-infrared-light activatable nanoparticles for deep-tissue-penetrating wireless optogenetics. Adv. Healthc. Mater. 8, e1801132 (2019).
Tan, P., He, L., Han, G. & Zhou, Y. Optogenetic immunomodulation: shedding light on antitumor immunity. Trends Biotechnol. 35, 215–226 (2017).
Nguyen, N. T. et al. CRAC channel-based optogenetics. Cell Calcium 75, 79–88 (2018).
Huang, K., Jayakumar, M. K. G. & Zhang, Y. Lutetium doping for making big core and core-shell upconversion nanoparticles. J. Mater. Chem. C 3, 10267–10272 (2015).
Barolet, D. Light-emitting diodes (LEDs) in dermatology. Semin Cutan. Med. Surg. 27, 227–238 (2008).
Pan, Y. et al. Mechanogenetics for the remote and noninvasive control of cancer immunotherapy. Proc. Natl Acad. Sci. USA 115, 992–997 (2018).
Ma, Y. et al. Targeting of antigens to B lymphocytes via CD19 as a means for tumor vaccine development. J. Immunol. 190, 5588–5599 (2013).
Dai, H., Wang, Y., Lu, X. & Han, W. Chimeric antigen receptors modified T-cells for cancer therapy. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djv439 (2016)
Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
Maude, S. L. et al. Chimeric antigen receptor T cells for sustained remissions in leukemia. N. Engl. J. Med. 371, 1507–1517 (2014).
Giavridis, T. et al. CAR T cell–induced cytokine release syndrome is mediated by macrophages and abated by IL-1 blockade. Nat. Med. 24, 731–738 (2018).
Allen, M. E. et al. An AND-gated drug and photoactivatable Cre-loxP system for spatiotemporal control in cell-based therapeutics. ACS Synth. Biol. 8, 2359–2371 (2019).
Huang, Z. et al. Engineering light-controllable CAR T cells for cancer immunotherapy. Sci. Adv. 6, eaay9209 (2020).
Acknowledgements
We acknowledge L. Strittmatter and the Electron Microscopy Facility at University of Massachusetts Medical School for EDS sample handling and analysis. We thank S. Siwko for manuscript editing. This work was supported by grants from the National Institutes of Health (R01CA232017 to Y.Z. and G.H., R01GM112003 to Y.Z., R01HL146852 to Y.H., R21GM138824 to Y.H., R01CA240258 to Y.H., R01AI040127 to A.R. and R01AI109842 to A.R.), the Department of Defense (W81XWH-19-1-0379 to G.H. and Y.Z.), the Cancer Prevention and Research Institute of Texas (RR140053, with Y.H. as a CPRIT Scholar in Cancer Research, and RP210070 to Y.Z.), the Welch Foundation (BE-1913-20190330 to Y.Z.) and the American Cancer Society (RSG-16-215-01-TBE to Y.Z. and RSG-18-043-01-LIB to Y.H.).
Author information
Authors and Affiliations
Contributions
Y.Z., G.H. and Y.H. conceived the study; Y.Z., G.H. and N.T.N. designed the experiments; N.T.N., H.Z., J.J., R.W., S.F., Z.H., X.L. and K.H. performed the experiments and data analysis; K.H. synthesized and characterized the nanoparticles, and interpreted the EDS data; G.H., J.C., Y.H., M.J.Y. and A.R. provided intellectual input and scientific feedback, as well as suggestions for animal models; Y.Z., Y.H., N.T.N., K.H. and G.H. wrote the manuscript. All authors provided feedback and helped shape the research, data analysis and the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Y.Z. and G.H. have submitted a patent application to the United States Patent and Trademark Office pertaining to the design and biomedical application aspect(s) of this work (application number 62/942,770). The remaining authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks Betty Kim and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Design and screening of CRY2- and LOV2-based LiCARs.
a, Constructs used to screen and optimize the designed LiCARs (Components I + II). The inset showed confocal images of HeLa cells expressing the PM-embedded constructs A or C (without fluorescent tag) after non-permeabilized immunostaining with an anti-Myc antibody. When Component II was expressed as a cytosolic protein, we also monitored light-inducible cytosol-to-PM translocation to confirm the photo-responsiveness of the optical dimerizer. Scale bar, 5 µm. b-d, Confocal images of HeLa cells expressing the indicated components before and after light illumination. The introduction of ER trafficking/export signals in Component I substantially improved PM targeting; whereas the addition of NES to Component II substantially reduced nuclear accumulation (b/c vs d). Four images for each combination were taken. Scale bar, 5 µm. e-f, Confocal images of HeLa cells co-expressing constructs A + B3 or C + D4. Note that B3 and D4 contained the CD8 transmembrane domain (TM) and were thus embedded in the plasma membrane. Four images for each combination were taken. Scale bar, 5 µm.
Extended Data Fig. 2 Early T cell activation reported by cell surface expression of CD69.
a, Quantification of the CD69 expression level in Jurkat T cells expressing WT CAR, defective LiCAR (C + D5 lacking the CD3 subunit), or LiCAR (C + D4) before (open box) and after light illumination (blue box; 20 min and then 10 h with the pulse of 10 s ON + 60 s OFF). Cells were co-incubated with hCD19− K562 cells (open box) or hCD19+ Raji cells (red box). b, Quantification of CD69 expression in Jurkat T cells co-cultured with hCD19-negative K562 (open box) or hCD19-positive Raji cells (red box). n = 2 independent biological replicates (mean ± range).
Extended Data Fig. 3 Expression of engineered CARs in human primary CD8+ T cells.
a, Evaluation of the purity of CD8+ T cells isolated from PBMCs of healthy donors. Isolated T cells were stained with anti-CD8-APC. Non-stained CD8+ T cells were used as negative control to aid the gating of cell populations. b, Quantification of WT CAR (GFP-tagged), LiCAR (C-GFP + D4-mCh) or defective LiCAR (C-GFP + D5-mCh) expression in human CD8+ T cells. GFP-positive (for the WT CAR group) or double positive cells (for the LiCAR and the defective LiCAR groups) were used for functional assays. c, Confocal images of human CD8+ T-cells transduced with WT CAR (green, top panel) or the indicated CAR components (C-GFP, green; or D4/D5-mCherry, red; middle and bottom panels). Three images per group were taken. Scale bar, 10 µm. d, Time-lapse imaging of tumour cells (Daudi) co-cultured with human CD8+ T cells (asterisk) expressing either WT CAR (GFP-tagged, top) in the dark or defective LiCAR (C-GFP + D5-mCh, bottom) exposed to blue light. Dying cells were indicated by SYTOX blue staining. Scale bar, 5 µm. Also see Supplementary Videos 3-4.
Extended Data Fig. 4 Optimization and characterization of synthesized UCNPs.
a, Comparison of the the core/shell structures of synthesized UCNPs (top, β-NaYF4:30% Yb, 0.5% Tm@NaYF4; bottom, β-NaYbF4:0.5%Tm@NaYF4) and TEM images (right). Scale bar, 100 nm. b, TEM images and the size distribution (height denoted as ‘H’ and diameter denoted as ‘D’) of the NaYbF4:0.5%Tm core nanoparticles (left), NaYbF4:0.5%Tm@NaYF4 core-shell nanoplates (middle), and silica-coated NaYbF4:0.5%Tm@NaYF4 core-shell nanoplates (right). c, Comparison of the upconversion luminescence spectra of synthesized UCNPs upon NIR light illumination at 980 nm (black, β-NaYF4:30%Yb,0.5%Tm@NaYF4; red, β-NaYbF4:0.5%Tm@NaYF4 nanoplates). Their luminescence intensities were compared at the same condition with the same amounts of total lanthanide ions. d, Blue light emitting from the leftmost cuvette containing UCNPs (β-NaYbF4:0.5%Tm@NaYF4) upon NIR illumination. The UCNP-containing cuvette (leftmost) was placed next to the indicated numbers of H2O-containing plastic cuvettes (labeled as 1, 2 and 3; top) or a cuvette containing dark ink (bottom). The NIR light source (980 nm) was placed on the right. Pictures were taken in a dark room except for the leftmost images. The light intensity was strong enough to illuminate the background after penetrating through cuvettes. e, UCNPs emitted bright blue light locally at the injection site in vivo upon NIR light stimulation (980 nm; 250 mW/cm2). Pictures were taken for the same mouse in the bright field without (left) or with NIR light (middle), or in the dark field with NIR light (right).
Extended Data Fig. 5 Effects of WT CAR T-cells and UCNPs on tumour growth.
a, WT CAR-expressing CD8+ T-cells selectively destroy CD19-positive melanoma tumours without light stimulation. Left, C57BL/6J mice were intradermally inoculated with 2.5×105 B16-OVA-hCD19 cells in the left flank (red circle) and 2.5×105 B16-OVA cells (CD19-negative tumour as control; blue circle) in the right flank. Two representative mice are shown after treatment with WT CAR T-cells + UCNPs for 8 days. Middle, Tumour sizes at the indicated time points were measured by a digital caliper with the tumour areas calculated in mm2 (length x width). n = 4 biologically independent mice (mean ± s.e.m.). P values were calculated using two-sided unpaired Student’s t-tests. Right, isolated B16-OVA and B16-OVA-hCD19 tumours at day 18. b, The growth curves of B16-OVA-CD19/UCNPs and B16-OVA/UCNPs upon NIR light irradiation. Left, C57BL/6J mice were intradermally inoculated with 2.5×105 B16-OVA-hCD19 cells in the left flank (red circle) and 2.5×105 B16-OVA cells (CD19-negative tumour as control; blue circle) in the right flank. Two representative mice are shown after injection of UCNPs for 8 days without CAR T-cells under NIR treatment. Middle, Tumour sizes at the indicated time points were measured by a digital caliper with the tumour areas calculated in mm2 (length x width). No notable difference in tumour size were observed. P = 0.93 when compared to the B16-OVA group at day 16 (unpaired two-sided Student’s t-test; n = 3 biologically independent mice; mean ± s.e.m.). Right, isolated B16-OVA and B16-OVA-hCD19 tumours at day 16. c, UCNPs did not affect tumour growth. C57BL/6J mice were intradermally inoculated with 2.5×105 B16-OVA cells to each flank. Four representative mice were shown after injection with UCNPs to the right flank tumour. Top right, isolated B16-OVA and B16-OVA-UCNPs tumours before and after UCNP removals at day 18. Bottom left and middle, tumour sizes before tumour surgery (measured from outside the skin) and after UCNP removal, respectively, at day 18 were measured by a digital caliper with the tumour areas calculated in mm2 (length x width). Bottom right, tumour weight after UCNPs removal at day 18. n = 6 biologically independent tumours isolated from 6 mice (mean ± s.e.m.). P values were calculated using two-sided unpaired Student’s t-tests. No statistically significant difference in tumour sizes or weights were observed.
Extended Data Fig. 6 Light-inducible selective killing of CD19+ solid tumours in vivo using LiCAR-expressing T cells.
a, CD8+ LiCAR T-cells selectively destroy CD19-expressing melanoma in response to NIR light illumination. Left, C57BL/6 J mice were intradermally inoculated with 2.5×105 B16-OVA-hCD19 cells in the left flank and 2.5×105 B16-OVA cells (CD19-negative tumour as control) in the right flank. Two representative mice with opened tumour areas are shown after treatment with LiCAR T-cells + UCNPs and exposure to NIR pulses for 9 days. Right, isolated B16-OVA/UCNPs and B16-OVA-hCD19/UCNPs tumours at day 19. Green arrow, tumour cells. Blue arrow, UCNPs injected to tumour sites. The tumour masses after UCNP removal were shown in Fig. 3g. b, LiCAR T-cells permit NIR light-inducible killing of B16-OVA-hCD19 melanoma in selected regions. Left, C57BL/6 J mice were intradermally inoculated at both flanks with 3×105 B16-OVA-hCD19 cells. After injection with the LiCAR T-cells + UCNP mixture, the left flank was exposed to NIR pulses for 8 days, while the right side was protected from NIR light using aluminum foil. Two representative mice with opened tumours are shown at day 18. Right, isolated B16-OVA-hCD19/UCNPs tumours with and without NIR at day 18. Green arrow, tumour cells. Blue arrow, UCNPs injected to tumour sites. The tumour masses after UCNP removal were shown in Fig. 3h.
Extended Data Fig. 7 UCNPs are well confined within the injection site.
a, Visualizing UCNPs after s.c. injection into the tumour sites under the indicated conditions. Images were acquired in the same mouse under three conditions: bright field without NIR light (left), bright field with NIR light illumination (middle) at the injection site (red arrow), or in the dark room with NIR light (right). Top, in situ images; middle, the UCNP-containing skin tissues were surgically exposed; bottom, after surgical removal. The NIR excitation showed a pink color in the camera if blue emission was not detected. Zoomed-in view on the right (orange box): The UCNP-containing skin/tumour tissues (top) and well-confined UCNPs isolated from the tissue (bottom). b, UCNPs did not spread to other major organs within the experimental window. Major organs were isolated from the mouse shown in panel a and then subjected to NIR light illumination. Only pink color was noted without blue emission, suggesting the absence of UCNPs in these tissues.
Extended Data Fig. 8 Blue light did not induce statistically significant changes in tumour killing.
a, Cartoon illustrating the experimental setup. C57BL/6 J mice were intradermally inoculated in the left flank with 3×105 B16-OVA-hCD19 cells. After injection with the LiCAR T-cells + UCNP mixture, mice were either exposed to blue light (470 nm; 40 mW/cm2, 2 hours per day) for 8 days, or kept in the dark. b, Three representative mice from each group. The lower panel showed mice with opened tumours at day 18. Green arrow, tumour injection sites. Blue arrow, UCNPs in the tumour sites. c, Measurements of tumour sizes at the indicated time points by a digital caliper. Tumour areas were calculated in mm2 (length x width). n = 4 (dark group), n = 3 (light group) biologically independent mice (mean ± s.e.m.). P values were calculated using two-sided unpaired Student’s t-tests. P = 0.25 when compared to the dark group at day 18. d, Representative images of isolated B16-OVA-hCD19 tumours (shown in panel b) with and without blue light at day 18.
Extended Data Fig. 9 Biotinylated LiCAR T-cells remain fully functional and can be coupled with UCNPs that are surface coated with streptavidin (Stv-UCNPs).
a, Schematic illustration of surface biotinylation of engineered CAR T-cells and coupling with our previously-described UCNPs coated with streptavidin (Stv). b, Flow cytometry analysis of biotinylation efficiency of Jurkat T cells expressing WT CAR and LiCAR. Engineered T cells were biotinylated with a high efficiency of over 98%. c, Biotinylation did not affect engineered CAR T-cell activation. Quantification of NFAT-dependent luciferase (NFAT-Luc) reporter activity in engineered Jurkat T cells. LiCAR-transduced T cells engaged tumour cells bearing cognate antigen (hCD19+ Raji cells) under dark (open box) or lit conditions (blue box). Blue light (40 mW/cm2 at 470 nm) was applied for 20 min and then in pulsed cycles of 30 sec ON + 100 sec OFF for 8 h. n = 5 independent biological replicates (mean ± s.e.m.). P values were calculated using two-sided unpaired Student’s t-tests.
Extended Data Fig. 10 Intraperitoneal injection of mLiCAR T-cells to reduce ‘on-target off-tumour’ side effects in a syngeneic mouse model of melanoma.
a, Schematic illustration of the mouse model used to evaluate ‘on-target off-tumour’ effects by intraperitoneal (i.p.) injection of B16-OVA-mCD19 tumour cells (3×106). After tumour growth for 3 weeks, WT CAR T cells/UCNPs or LiCAR-T/UCNPs cells that could engage mCD19-B16-OVA cells were subsequently injected into the tumour sites. LiCAR (combination of C + D4.1) T-cells treated mice were subjected to pulsed NIR light stimulation for 3 days (980 nm at a power density of 250 mW/cm2; pulses of 20 sec ON, 5 minutes OFF; 2 h/day). On day 0 and day 3, blood was collected from the retro-orbital sinus by glass capillary from anesthetized mice for B cell quantification. b, On-target off-tumour effects of mWT CAR and mLiCAR T-cells evaluated by the degree of B cell aplasia. Peripheral blood B cells from the WT mCAR or mLiCAR T-cell treated groups were counted and compared on day 0 and day 3. B cells from peripheral blood of healthy mice were used as control. n = 7 biologically independent mice (mean ± s.e.m.). P values were calculated using two-sided unpaired Student’s t-tests.
Supplementary information
Supplementary Information
Supplementary Notes 1–5, Figs. 1–10, Tables 1–4, captions for videos and references.
Supplementary Video 1
Light-induced recruitment of cytosolic construct D2 (mCherry-tagged) toward construct C localized in the plasma membrane. Two repeated dark–light cycles (470 nm, 40 mW cm–2) were applied to visualize the reversible cytosol-to-PM translocation of construct D2. Related to Fig. 1d. Scale bar, 5 µm.
Supplementary Video 2
Light-induced recruitment of cytosolic construct D4.2 (mCherry-tagged) toward construct C localized in the plasma membrane. Two repeated dark–light cycles (470 nm, 40 mW cm–2) were applied to visualize the reversible cytosol-to-PM translocation of construct D4.2. Related to Fig. 1e. Scale bar, 5 µm.
Supplementary Video 3
Time-lapse imaging of Daudi tumour cell killing mediated by human CD8+ T cells transduced with WT CAR (GFP-tagged; green) in the dark. T cells (indicated by ‘T’) were surrounded by co-cultured Daudi cells (indicated by ‘D’). SYTOX blue was used to stain dying Daudi tumour cells with compromised plasma membranes. Related to Supplementary Fig. 5d. Scale bar, 5 µm.
Supplementary Video 4
Time-lapse imaging of Daudi (indicated by ‘D’) tumour cells co-cultured with human CD8+ T cells (indicated by ‘T’) expressing defective LiCAR (C-GFP + D5-mCh). The mixture was exposed to blue light illumination (470 nm, 40 mW cm–2, for 5 h). No appreciable SYTOX blue staining was observed. Related to Supplementary Fig. 5d. Scale bar, 5 µm.
Supplementary Video 5
Time-lapse imaging of light-induced tumour cell killing mediated by human CD8+ T cells expressing LiCAR (C-GFP + D4-mCh). LiCAR T cells (T) were surrounded by Daudi lymphoma cells (D) and subjected to photostimulation (470 nm, 40 mW cm–2, for 5 h). Daudi cells started to die at 15 min, as indicated by positive SYTOX blue staining. Within 5 h, we noticed the death of all three surrounding Daudi cells. Related to Fig. 2h. Scale bar, 5 µm.
Supplementary Video 6
Time-lapse imaging of Daudi cells (D) co-cultured with human CD8+ T cells (T) expressing LiCAR (C-GFP + D4-mCh) in the dark for more than 5 h. The LiCAR T cells did not induce any tumour cell death in the absence of blue light, as reflected by negative SYTOX blue staining. Related to Fig. 2h. Scale bar, 5 µm.
Additional Supplementary Files 1
Source data for Supplementary Figs. 1, 2, 4 and 5.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Rights and permissions
About this article
Cite this article
Nguyen, N.T., Huang, K., Zeng, H. et al. Nano-optogenetic engineering of CAR T cells for precision immunotherapy with enhanced safety. Nat. Nanotechnol. 16, 1424–1434 (2021). https://doi.org/10.1038/s41565-021-00982-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-021-00982-5
This article is cited by
-
Synthetic biology approaches for improving the specificity and efficacy of cancer immunotherapy
Cellular & Molecular Immunology (2024)
-
Development of an optogenetics tool, Opto-RANK, for control of osteoclast differentiation using blue light
Scientific Reports (2024)
-
Triplet–triplet annihilation photon upconversion-mediated photochemical reactions
Nature Reviews Chemistry (2024)
-
In vivo manufacture and manipulation of CAR-T cells for better druggability
Cancer and Metastasis Reviews (2024)
-
Challenges and new technologies in adoptive cell therapy
Journal of Hematology & Oncology (2023)