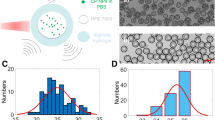
Abstract
Molecular imaging is a crucial technique in clinical diagnostics but it relies on radioactive tracers or strong magnetic fields that are unsuitable for many patients, particularly infants and pregnant women. Ultra-high-frequency radio-frequency acoustic (UHF-RF-acoustic) imaging using non-ionizing RF pulses allows deep-tissue imaging with sub-millimetre spatial resolution. However, lack of biocompatible and targetable contrast agents has prevented the successful in vivo application of UHF-RF-acoustic imaging. Here we report our development of targetable nanodroplets for UHF-RF-acoustic molecular imaging of cancers. We synthesize all-liquid nanodroplets containing hypertonic saline that are stable for at least 2 weeks and can produce high-intensity UHF-RF-acoustic signals. Compared with concentration-matched iron oxide nanoparticles, our nanodroplets produce at least 1,600 times higher UHF-RF-acoustic signals at the same imaging depth. We demonstrate in vivo imaging using the targeted nanodroplets in a prostate cancer xenograft mouse model expressing gastrin release protein receptor (GRPR), and show that targeting specificity is increased by more than 2-fold compared with untargeted nanodroplets or prostate cancer cells not expressing this receptor.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Gambhir, S. S., Ge, T. J., Vermesh, O. & Spitler, R. Toward achieving precision health. Sci. Transl. Med. 10, eaao3612 (2018).
Hamilton, W., Walter, F. M., Rubin, G. & Neal, R. D. Improving early diagnosis of symptomatic cancer. Nat. Rev. Clin. Oncol. 13, 740–749 (2016).
Gambhir, S. S. Molecular imaging of cancer with positron emission tomography. Nat. Rev. Cancer 2, 683–693 (2002).
Weissleder, R. Molecular imaging in cancer. Science 312, 1168–1171 (2006).
Weissleder, R. & Mahmood, U. Molecular imaging. Radiology 219, 316–333 (2001).
Hussain, T. & Nguyen, Q. T. Molecular imaging for cancer diagnosis and surgery. Adv. Drug Deliv. Rev. 66, 90–100 (2014).
Willmann, J. K., van Bruggen, N., Dinkelborg, L. M. & Gambhir, S. S. Molecular imaging in drug development. Nat. Rev. Drug Discov. 7, 591–607 (2008).
Pysz, M. A., Gambhir, S. S. & Willmann, J. K. Molecular imaging: current status and emerging strategies. Clin. Radiol. 65, 500–516 (2010).
Lin, E. C. Radiation risk from medical imaging. Mayo Clin. Proc. 85, 1142–1146 (2010).
James, M. L. & Gambhir, S. S. A molecular imaging primer: modalities, imaging agents, and applications. Physiological Rev. 92, 897–965 (2012).
Loskutova, K., Grishenkov, D. & Ghorbani, M. Review on acoustic droplet vaporization in ultrasound diagnostics and therapeutics. BioMed. Res. Int. 2019, 9480193 (2019).
Jokerst, J. V. & Gambhir, S. S. Molecular imaging with theranostic nanoparticles. Acc. Chem. Res. 44, 1050–1060 (2011).
Weber, J., Beard, P. C. & Bohndiek, S. E. Contrast agents for molecular photoacoustic imaging. Nat. Methods 13, 639–650 (2016).
Bowen, T. in Proc. 1981 IEEE Ultrasonics Symposium 817–822 (IEEE, 1981).
Kruger, R. A. et al. Thermoacoustic CT with radio waves: a medical imaging paradigm. Radiology 211, 275–278 (1999).
Ku, G. et al. Thermoacoustic and photoacoustic tomography of thick biological tissues toward breast imaging. Technol. Cancer Res. Treat. 4, 559–565 (2005).
Kruger, R. A., Reinecke, D. R. & Kruger, G. A. Thermoacoustic computed tomography—technical considerations. Med. Phys. 26, 1832–1837 (1999).
Wen, L. W., Yang, S. H., Zhong, J. P., Zhou, Q. & Xing, D. Thermoacoustic imaging and therapy guidance based on ultra-short pulsed microwave pumped thermoelastic effect induced with superparamagnetic iron oxide nanoparticles. Theranostics 7, 1976–1989 (2017).
Kruger, R. A. et al. Breast cancer in vivo: contrast enhancement with thermoacoustic CT at 434 MHz—feasibility study. Radiology 216, 279–283 (2000).
Omar, M., Kellnberger, S., Sergiadis, G., Razansky, D. & Ntziachristos, V. Near-field thermoacoustic imaging with transmission line pulsers. Med. Phys. 39, 4460–4466 (2012).
Laqua, D., Just, T. & Husar, P. in Proc. 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology Society 1437–1440 (IEEE, 2010).
Ogunlade, O. & Beard, P. Exogenous contrast agents for thermoacoustic imaging: an investigation into the underlying sources of contrast. Med. Phys. 42, 170–181 (2016).
Byrd, D., Hanson, G.W. & Patch, S.K. in Proc. SPIE 7564, Photons Plus Ultrasound: Imaging and Sensing 2010, 756417 (SPIE, 2010).
Wu, D., Huang, L., Jiang, M. S. & Jiang, H. Contrast agents for photoacoustic and thermoacoustic imaging: a review. Int. J. Mol. Sci. 15, 23616 (2014).
Nie, L., Ou, Z., Yang, S. & Xing, D. Thermoacoustic molecular tomography with magnetic nanoparticle contrast agents for targeted tumor detection. Med. Phys. 37, 4193–4200 (2010).
Tamarov, K. et al. Electrolytic conductivity-related radiofrequency heating of aqueous suspensions of nanoparticles for biomedicine. Phys. Chem. Chem. Phys. 19, 11510–11517 (2017).
Weast, R.C. CRC Handbook of Chemistry and Physics 70th edn (CRC Press, 1989).
Wolf, A. V. Aqueous Solutions and Body Fluids (Harper & Row, 1966).
Engel, R. H., Riggi, S. J. & Fahrenbach, M. J. Insulin: intestinal absorption as water-in-oil-in-water emulsions. Nature 219, 856–857 (1968).
Gresham, P. A., Barnett, M., Smith, S. V. & Schneider, R. Use of a sustained-release multiple emulsion to extend the period of radioprotection conferred by cysteamine. Nature 234, 149 (1971).
Garti, N. & Bisperink, C. Double emulsions: progress and applications. Curr. Opin. Colloid Interface Sci. 3, 657–667 (1998).
Muschiolik, G. Multiple emulsions for food use. Curr. Opin. Colloid Interface Sci. 12, 213–220 (2007).
Chong, D. et al. Advances in fabricating double-emulsion droplets and their biomedical applications. Microfluidics Nanofluidics 19, 1071–1090 (2015).
Chen, L. et al. Photoresponsive monodisperse cholesteric liquid crystalline microshells for tunable omnidirectional lasing enabled by a visible light-driven chiral molecular switch. Adv. Optical Mater. 2, 845–848 (2014).
Mezzenga, R., Folmer, B. M. & Hughes, E. Design of double emulsions by osmotic pressure tailoring. Langmuir 20, 3574–3582 (2004).
Wen, L. & Papadopoulos, K. D. Effects of osmotic pressure on water transport in W1/O/W2 emulsions. J. Colloid Interface Sci. 235, 398–404 (2001).
Atzberger, P. J. & Kramer, P. R. Theoretical framework for microscopic osmotic phenomena. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 75, 061125 (2007).
Freire, M. G., Dias, A. M. A., Coelho, M. A. Z., Coutinho, J. A. P. & Marrucho, I. M. Aging mechanisms of perfluorocarbon emulsions using image analysis. J. Colloid Interface Sci. 286, 224–232 (2005).
Hilder, M. H. The solubility of water in edible oils and fats. J. Am. Oil Chem. Soc. 45, 703–707 (1968).
Freire, M. G., Gomes, L., Santos, L. M., Marrucho, I. M. & Coutinho, J. A. Water solubility in linear fluoroalkanes used in blood substitute formulations. J. Phys. Chem. B 110, 22923–22929 (2006).
Rotariu, G., Fraga, D. & Hildebrand, J. The solubility of water in normal perfluoroheptane. J. Am. Chem. Soc. 75, 6357–6357 (1953).
Shpak, O. et al. Acoustic droplet vaporization is initiated by superharmonic focusing. Proc. Natl Acad. Sci. USA 111, 1697–1702 (2014).
Vernikouskaya, I., Pochert, A., Lindén, M. & Rasche, V. Quantitative 19F MRI of perfluoro-15-crown-5-ether using uniformity correction of the spin excitation and signal reception. Magn. Reson. Mater. Phys., Biol. Med. 32, 25–36 (2019).
Tran, T. D. et al. Clinical applications of perfluorocarbon nanoparticles for molecular imaging and targeted therapeutics. Int. J. Nanomed. 2, 515–526 (2007).
Joseph, G. M. & Noah, W. Poloxamer 188 (P188) as a membrane resealing reagent in biomedical applications. Recent Pat. Biotechnol. 6, 200–211 (2012).
Lifshitz, I. M. & Slyozov, V. V. The kinetics of precipitation from supersaturated solid solutions. J. Phys. Chem. Solids 19, 35–50 (1961).
Sakai, T., Kamogawa, K., Nishiyama, K., Sakai, H. & Abe, M. Molecular diffusion of oil/water emulsions in surfactant-free conditions. Langmuir 18, 1985–1990 (2002).
Doinikov, A. A., Sheeran, P. S., Bouakaz, A. & Dayton, P. A. Vaporization dynamics of volatile perfluorocarbon droplets: a theoretical model and in vitro validation. Med. Phys. 41, 102901 (2014).
Pitt, W. G., Singh, R. N., Perez, K. X., Husseini, G. A. & Jack, D. R. Phase transitions of perfluorocarbon nanoemulsion induced with ultrasound: a mathematical model. Ultrason. Sonochem. 21, 879–891 (2014).
Cornelio, D. B., Roesler, R. & Schwartsmann, G. Gastrin-releasing peptide receptor as a molecular target in experimental anticancer therapy. Ann. Oncol. 18, 1457–1466 (2007).
Acknowledgements
This work was supported in part by grants from NCI CCNE-TD U54 CA199075-04 (S.S.G.), The Canary Foundation (S.S.G.), The Sir Peter Michael Foundation (S.S.G.), Stanford’s Catalyst for Collaborative Solutions (J.D. and S.S.G.), Google Faculty Research Award (Y.-S.C.), The Jump ARCHES endowment through the Health Care Engineering Systems Center (Y.-S.C.), and NIGMS 1R21GM139022-01 (Y.Z.). The authors acknowledge the Stanford Center for Innovation in In Vivo Imaging (SCI3) for assistance with animal imaging and C. Chan for providing insightful discussions. This paper is dedicated to the memory of Dr Sanjiv Sam Gambhir.
Author information
Authors and Affiliations
Contributions
S.S.G. conceived the original idea. Y.-S.C. and S.S.G. designed the experiments. Y.-S.C. developed the contrast agents and performed the in vitro and in vivo imaging. C.B., A.Z. and F.A. assisted in the in vivo experiments. Y.-S.C., Y.Z. and H.W. performed the theoretical study. D.-H.C. contributed to cryo-electron microscopy. C.B., A.Z., E.-C.H., F.A., T.S. and J.D. contributed to the discussion of the data and experimental results. Y.-S.C. and S.S.G. drafted the manuscript and all authors contributed to the writing of the manuscript. S.S.G. supervised the entire study.
Corresponding author
Ethics declarations
Competing interests
S.S.G. declared competing financial interests with Endra Inc and Visualsonics Inc.
Additional information
Peer review information Nature Nanotechnology thanks F. Kiessling, N. van den Berg and M. Tang for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary discussions, methods, Figs. 1–16 and Tables 1 and 2.
Rights and permissions
About this article
Cite this article
Chen, YS., Zhao, Y., Beinat, C. et al. Ultra-high-frequency radio-frequency acoustic molecular imaging with saline nanodroplets in living subjects. Nat. Nanotechnol. 16, 717–724 (2021). https://doi.org/10.1038/s41565-021-00869-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-021-00869-5
This article is cited by
-
In vivo ultrasound-induced luminescence molecular imaging
Nature Photonics (2024)
-
Decorated bacteria-cellulose ultrasonic metasurface
Nature Communications (2023)
-
Nanoparticles with ultrasound-induced afterglow luminescence for tumour-specific theranostics
Nature Biomedical Engineering (2022)
-
Recent advances in aggregation-induced emission luminogens in photoacoustic imaging
European Journal of Nuclear Medicine and Molecular Imaging (2022)
-
Advanced Nanomaterials for Multimodal Molecular Imaging
Chemical Research in Chinese Universities (2021)