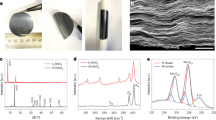
Abstract
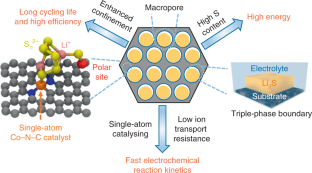
Lithium–sulfur batteries are attractive alternatives to lithium-ion batteries because of their high theoretical specific energy and natural abundance of sulfur. However, the practical specific energy and cycle life of Li–S pouch cells are significantly limited by the use of thin sulfur electrodes, flooded electrolytes and Li metal degradation. Here we propose a cathode design concept to achieve good Li–S pouch cell performances. The cathode is composed of uniformly embedded ZnS nanoparticles and Co–N–C single-atom catalyst to form double-end binding sites inside a highly oriented macroporous host, which can effectively immobilize and catalytically convert polysulfide intermediates during cycling, thus eliminating the shuttle effect and lithium metal corrosion. The ordered macropores enhance ionic transport under high sulfur loading by forming sufficient triple-phase boundaries between catalyst, conductive support and electrolyte. This design prevents the formation of inactive sulfur (dead sulfur). Our cathode structure shows improved performances in a pouch cell configuration under high sulfur loading and lean electrolyte operation. A 1-A-h-level pouch cell with only 100% lithium excess can deliver a cell specific energy of >300 W h kg−1 with a Coulombic efficiency >95% for 80 cycles.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Change history
18 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41565-020-00829-5
References
Ji, X., Lee, K. T. & Nazar, L. F. A highly ordered nanostructured carbon–sulphur cathode for lithium–sulphur batteries. Nat. Mater. 8, 500–506 (2009).
Seh, Z. W., Sun, Y., Zhang, Q. & Cui, Y. Designing high-energy lithium–sulfur batteries. Chem. Soc. Rev. 45, 5605–5634 (2016).
Evers, S. & Nazar, L. F. New approaches for high energy density lithium–sulfur battery cathodes. Acc. Chem. Res. 46, 1135–1143 (2013).
Manthiram, A., Fu, Y., Chung, S., Zu, C. & Su, Y. Rechargeable lithium–sulfur batteries. Chem. Rev. 114, 11751–11787 (2014).
Luo, L., Li, J., Yaghoobnejad Asl, H. & Manthiram, A. In-situ assembled VS4 as a polysulfide mediator for high-loading lithium–sulfur batteries. ACS Energy Lett. 5, 1177–1185 (2020).
Ye, Y. et al. Toward practical high-energy batteries: a modular-assembled oval-like carbon microstructure for thick sulfur electrodes. Adv. Mater. 29, 1700598 (2017).
Xue, W. et al. Intercalation-conversion hybrid cathodes enabling Li–S full-cell architectures with jointly superior gravimetric and volumetric energy densities. Nat. Energy 4, 374–382 (2019).
Wu, F. et al. Sulfur nanodots stitched in 2D “bubble-like” interconnected carbon fabric as reversibility-enhanced cathodes for lithium–sulfur batteries. ACS Nano 11, 4694–4702 (2017).
Zhang, J., Yang, C., Yin, Y., Wan, L. & Guo, Y. Sulfur encapsulated in graphitic carbon nanocages for high-rate and long-cycle lithium–sulfur batteries. Adv. Mater. 28, 9539–9544 (2016).
Wang, W. et al. Lithium phosphorus oxynitride as an efficient protective layer on lithium metal anodes for advanced lithium–sulfur batteries. Energy Storage Mater. 18, 414–422 (2019).
Cheng, X. et al. The gap between long lifespan Li–S coin and pouch cells: the importance of lithium metal anode protection. Energy Storage Mater. 6, 18–25 (2017).
Amine, R. et al. Regulating the hidden solvation‐ion‐exchange in concentrated electrolytes for stable and safe lithium metal batteries. Adv. Energy Mater. 10, 2000901 (2020).
Cha, E. et al. 2D MoS2 as an efficient protective layer for lithium metal anodes in high-performance Li–S batteries. Nat. Nanotechnol. 13, 337–344 (2018).
Lin, D., Liu, Y. & Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 12, 194–206 (2017).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Zheng, J. et al. Electrolyte additive enabled fast charging and stable cycling lithium metal batteries. Nat. Energy 2, 17012 (2017).
Fan, F. Y., Carter, W. C. & Chiang, Y. Mechanism and kinetics of Li2S precipitation in lithium–sulfur batteries. Adv. Mater. 27, 5203–5209 (2015).
Evers, S., Yim, T. & Nazar, L. F. Understanding the nature of absorption/adsorption in nanoporous polysulfide sorbents for the Li–S battery. J. Phys. Chem. C 116, 19653–19658 (2012).
Zhang, Q. et al. Understanding the anchoring effect of two-dimensional layered materials for lithium–sulfur batteries. Nano Lett. 15, 3780–3786 (2015).
Zhou, G. et al. Theoretical calculation guided design of single-atom catalysts toward fast kinetic and long-life Li–S batteries. Nano Lett. 20, 1252–1261 (2020).
Du, Z. et al. Cobalt in nitrogen-doped graphene as single-atom catalyst for high-sulfur content lithium–sulfur batteries. J. Am. Chem. Soc. 141, 3977–3985 (2019).
Pang, Q., Liang, X., Kwok, C. Y. & Nazar, L. F. Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 1, 16132 (2016).
Peng, H. et al. Enhanced electrochemical kinetics on conductive polar mediators for lithium-sulfur batteries. Angew. Chem. Int. Ed. 55, 12990–12995 (2016).
Shen, K. et al. Ordered macro-microporous metal-organic framework single crystals. Science 359, 206–210 (2018).
Wang, J. et al. Design of N-coordinated dual-metal sites: a stable and active Pt-free catalyst for acidic oxygen reduction reaction. J. Am. Chem. Soc. 139, 17281–17284 (2017).
Alves, M. C. M., Dodelet, J. P., Guay, D., Ladouceur, M. & Tourillon, G. Origin of the electrocatalytic properties for oxygen reduction of some heat-treated polyacrylonitrile and phthalocyanine cobalt compounds adsorbed on carbon black as probed by electrochemistry and X-ray absorption spectroscopy. J. Phys. Chem. 96, 10898–10905 (1992).
Lawniczak-Jablonska, K. et al. Local electronic structure of ZnS and ZnSe doped by Mn, Fe, Co, and Ni from X-ray-absorption near-edge structure studies. Phys. Rev. B 53, 1119–1128 (1996).
Cao, L. et al. Identification of single-atom active sites in carbon-based cobalt catalysts during electrocatalytic hydrogen evolution. Nat. Catal. 2, 134–141 (2018).
Curcio, A. L., Bernardi, M. I. B. & Mesquita, A. Local structure and photoluminescence properties of nanostructured Zn1‐xMnxS material. Phys. Status Solidi C 12, 1367–1371 (2015).
Yuan, H. et al. Conductive and catalytic triple-phase interfaces enabling uniform nucleation in high-rate lithium–sulfur batteries. Adv. Energy Mater. 9, 1802768 (2019).
Feng, Z. et al. Unravelling the role of Li2S2 in lithium–sulfur batteries: a first principles study of its energetic and electronic properties. J. Power Sources 272, 518–521 (2014).
Paolella, A. et al. Transient existence of crystalline lithium disulfide Li2S2 in a lithium–sulfur battery. J. Power Sources 325, 641–645 (2016).
Nandasiri, M. I. et al. In situ chemical imaging of solid-electrolyte interphase layer evolution in Li–S batteries. Chem. Mater. 29, 4728–4737 (2017).
Fan, X. et al. Non-flammable electrolyte enables Li-metal batteries with aggressive cathode chemistries. Nat. Nanotechnol. 13, 715–722 (2018).
Liu, J. et al. Pathways for practical high-energy long-cycling lithium metal batteries. Nat. Energy 4, 180–186 (2019).
Zhao, M., Li, B., Zhang, X., Huang, J. & Zhang, Q. A perspective toward practical lithium–sulfur batteries. ACS Cent. Sci. 6, 1095–1104 (2020).
Zhao, C., Xu, G., Zhao, T. & Amine, K. Beyond the polysulfide shuttle and lithium dendrite formation: addressing the sluggish sulfur redox kinetics for practical high-energy Li‐S batteries. Angew. Chem. Int. Ed. 59, 17634–17640 (2020).
Fang, Z. et al. Mesoporous carbon nanotube aerogel-sulfur cathodes: a strategy to achieve ultrahigh areal capacity for lithium–sulfur batteries via capillary action. Carbon 166, 183–192 (2020).
Salihoglu, O. & Demir-Cakan, R. Factors affecting the proper functioning of a 3Ah Li-S pouch cell. J. Electrochem. Soc. 164, A2948–A2955 (2017).
Qu, C. et al. LiNO3-free electrolyte for Li-S battery: a solvent of choice with low Ksp of polysulfide and low dendrite of lithium. Nano Energy 39, 262–272 (2017).
Shi, L. et al. Reaction heterogeneity in practical high-energy lithium–sulfur pouch cells. Energy Environ. Sci. 13, 3620–3632 (2020).
Weller, C., Thieme, S., Haertel, P., Althues, H. & Kaskel, S. Intrinsic shuttle suppression in lithium–sulfur batteries for pouch cell application. J. Electrochem. Soc. 164, A3766–A3771 (2017).
Niu, C. et al. High-energy lithium metal pouch cells with limited anode swelling and long stable cycles. Nat. Energy 4, 551–559 (2019).
Kang, N. et al. Cathode porosity is a missing key parameter to optimize lithium–sulfur battery energy density. Nat. Commun. 10, 4597 (2019).
Sander, J. S., Erb, R. M., Li, L., Gurijala, A. & Chiang, Y. M. High-performance battery electrodes via magnetic templating. Nat. Energy 1, 16099 (2016).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Harl, J. & Kresse, G. Accurate bulk properties from approximate many-body techniques. Phys. Rev. Lett. 103, 056401 (2009).
Acknowledgements
Research at the Argonne National Laboratory was funded by the US Department of Energy (DOE), Vehicle Technologies Office. Support from T. Duong of the US DOE’s Office of Vehicle Technologies Program is gratefully acknowledged. Use of the Advanced Photon Source, an Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory, was supported by DOE under contract no. DE-AC02-06CH11357. This work was also supported by a grant from the Research Grants Council of the Hong Kong Special Administrative Region, China (project no. T23-601/17-R). K.A. and G.X. also thank the support from Clean Vehicles, US-China Clean Energy Research Centre (CERC-CVC2). K.A. and G.-L.X. thank S. Ahmed at Argonne National Laboratory for the simulation on the cell specific energy of Li–S and NMC811–graphite pouch cells.
Author information
Authors and Affiliations
Contributions
G.-L.X., K.A. and T.Z. initiated this research project. G.-L.X. conceived the idea and designed the experiment under the supervision of K.A. and T.Z.; C.Z. synthesized and characterized the structures of the materials and tested the half-cells and pouch cells with the assistance of L.Z. and Yuxun Ren; G.-L.X., I.H., C.-J.S., Yang Ren and X.Z. conducted synchrotron X-ray measurements and analysis. Z.Y. and L.C. conducted the calculation simulation. Y.-X.M., J.-T.L. and S.-G.S. conducted the in situ UV-vis measurement. C.Z., G.-L.X., K.A. and T.Z. prepared the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks Long Qie, Yunxiao Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–33, Materials and Methods, Discussion, Tables 1–6 and refs. 1–75.
Rights and permissions
About this article
Cite this article
Zhao, C., Xu, GL., Yu, Z. et al. A high-energy and long-cycling lithium–sulfur pouch cell via a macroporous catalytic cathode with double-end binding sites. Nat. Nanotechnol. 16, 166–173 (2021). https://doi.org/10.1038/s41565-020-00797-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-020-00797-w
This article is cited by
-
Standardized cycle life assessment of batteries using extremely lean electrolytic testing conditions
Communications Materials (2024)
-
Developing high-power Li||S batteries via transition metal/carbon nanocomposite electrocatalyst engineering
Nature Nanotechnology (2024)
-
Analytical noncovalent electrochemistry for battery engineering
Nature Chemical Engineering (2024)
-
Metal electrodes for next-generation rechargeable batteries
Nature Reviews Electrical Engineering (2024)
-
Interface-induced polymerization strategy for constructing titanium dioxide embedded carbon porous framework with enhanced chemical immobilization towards lithium polysulfides
Nano Research (2024)