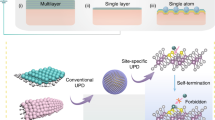
Abstract
The stability of single-atom catalysts is critical for their practical applications. Although a high temperature can promote the bond formation between metal atoms and the substrate with an enhanced stability, it often causes atom agglomeration and is incompatible with many temperature-sensitive substrates. Here, we report using controllable high-temperature shockwaves to synthesize and stabilize single atoms at very high temperatures (1,500–2,000 K), achieved by a periodic on–off heating that features a short on state (55 ms) and a ten-times longer off state. The high temperature provides the activation energy for atom dispersion by forming thermodynamically favourable metal–defect bonds and the off-state critically ensures the overall stability, especially for the substrate. The resultant high-temperature single atoms exhibit a superior thermal stability as durable catalysts. The reported shockwave method is facile, ultrafast and universal (for example, Pt, Ru and Co single atoms, and carbon, C3N4 and TiO2 substrates), which opens a general route for single-atom manufacturing that is conventionally challenging.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors upon reasonable request.
References
Qiao, B. et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nat. Chem. 3, 634–641 (2011).
Jones, J. et al. Thermally stable single-atom platinum-on-ceria catalysts via atom trapping. Science 353, 150–154 (2016).
Nie, L. et al. Activation of surface lattice oxygen in single-atom Pt/CeO2 for low-temperature CO oxidation. Science 358, 1419–1423 (2017).
Li, H. et al. Synergetic interaction between neighbouring platinum monomers in CO2 hydrogenation. Nat. Nanotechnol. 13, 411–417 (2018).
Wei, S. et al. Direct observation of noble metal nanoparticles transforming to thermally stable single atoms. Nat. Nanotechnol. 13, 856–861 (2018).
Liu, P. et al. Photochemical route for synthesizing atomically dispersed palladium catalysts. Science 352, 797–800 (2016).
Lucci, F. R. et al. Selective hydrogenation of 1,3-butadiene on platinum–copper alloys at the single-atom limit. Nat. Commun. 6, 8550 (2015).
Fei, H. et al. General synthesis and definitive structural identification of MN4C4 single-atom catalysts with tunable electrocatalytic activities. Nat. Catal. 1, 63–72 (2018).
Tiwari, J. N. et al. Multicomponent electrocatalyst with ultralow Pt loading and high hydrogen evolution activity. Nat. Energy 3, 773–782 (2018).
Wei, H. et al. Iced photochemical reduction to synthesize atomically dispersed metals by suppressing nanocrystal growth. Nat. Commun. 8, 1490 (2017).
Duchesne, P. N. et al. Golden single-atomic-site platinum electrocatalysts. Nat. Mater. 17, 1033–1039 (2018).
Yang, H. Bin et al. Atomically dispersed Ni(i) as the active site for electrochemical CO2 reduction. Nat. Energy 3, 140–147 (2018).
Qin, R., Liu, P., Fu, G. & Zheng, N. Strategies for stabilizing atomically dispersed metal catalysts. Small Methods 2, 1700286 (2018).
Chen, Y. et al. Single-atom catalysts: synthetic strategies and electrochemical applications. Joule 2, 1242–1264 (2018).
Liu, J. Catalysis by supported single metal atoms. ACS Catal. 7, 34–59 (2017).
Pelletier, J. D. A. & Basset, J. M. Catalysis by design: well-defined single-site heterogeneous catalysts. Acc. Chem. Res. 49, 664–677 (2016).
Zhang, Z. et al. Thermally stable single atom Pt/m-Al2O3 for selective hydrogenation and CO oxidation. Nat. Commun. 8, 16100 (2017).
Hansen, T. W., Delariva, A. T., Challa, S. R. & Datye, A. K. Sintering of catalytic nanoparticles: particle migration or Ostwald ripening? Acc. Chem. Res. 46, 1720–1730 (2013).
Risse, T., Shaikhutdinov, S., Nilius, N., Sterrer, M. & Freund, H. J. Gold supported on thin oxide films: from single atoms to nanoparticles. Acc. Chem. Res. 41, 949–956 (2008).
Kim, Y. T. et al. Fine size control of platinum on carbon nanotubes: from single atoms to clusters. Angew. Chem. Int. Ed. 45, 407–411 (2006).
Sehested, J., Gelten, J. A. P., Remediakis, I. N., Bengaard, H. & Nørskov, J. K. Sintering of nickel steam-reforming catalysts: effects of temperature and steam and hydrogen pressures. J. Catal. 223, 432–443 (2004).
Li, X. et al. Single-atom Pt as Co-catalyst for enhanced photocatalytic H2 evolution. Adv. Mater. 28, 2427–2431 (2016).
Kwak, J. H. et al. Coordinatively unsaturated Al3+ centers as binding sites for active catalyst phases of platinum on γ-Al2O3. Science 325, 1670–1673 (2009).
Choi, C. H. et al. Tuning selectivity of electrochemical reactions by atomically dispersed platinum catalyst. Nat. Commun. 7, 10922 (2016).
Dvořák, F. et al. Creating single-atom Pt–ceria catalysts by surface step decoration. Nat. Commun. 7, 10801 (2016).
Qiu, H. J. et al. Nanoporous graphene with single-atom nickel dopants: an efficient and stable catalyst for electrochemical hydrogen production. Angew. Chem. Int. Ed. 54, 14031–14035 (2015).
Liu, L. et al. Generation of subnanometric platinum with high stability during transformation of a 2D zeolite into 3D. Nat. Mater. 16, 132–138 (2017).
Moliner, M. et al. Reversible transformation of Pt nanoparticles into single atoms inside high-silica chabazite zeolite. J. Am. Chem. Soc. 138, 15743–15750 (2016).
Li, Z. et al. Platinum–nickel frame within metal–organic framework fabricated in situ for hydrogen enrichment and molecular sieving. Nat. Commun. 6, 1–8 (2015).
Yin, P. et al. Single cobalt atoms with precise N-coordination as superior oxygen reduction reaction catalysts. Angew. Chem. Int. Ed. 55, 10800–10805 (2016).
Xie, P. et al. Nanoceria-supported single-atom platinum catalysts for direct methane conversion. ACS Catal. 8, 4044–4048 (2018).
Yao, Y. et al. Carbon welding by ultrafast Joule heating. Nano Lett. 16, 7282–7289 (2016).
Yao, Y. et al. Carbothermal shock synthesis of high-entropy-alloy nanoparticles. Science 359, 1489–1494 (2018).
Bugaris, D. E., Smith, M. D. & Zur Loye, H. C. Hydroflux crystal growth of platinum group metal hydroxides: Sr6NaPd2(OH)17, Li2Pt(OH)6, Na2Pt(OH)6, Sr2Pt(OH)8, and Ba2Pt(OH)8. Inorg. Chem. 52, 3836–3844 (2013).
Cheng, N. et al. Platinum single-atom and cluster catalysis of the hydrogen evolution reaction. Nat. Commun. 7, 13638 (2016).
Sun, X., Han, P., Li, B. & Zhao, Z. Tunable catalytic performance of single Pt atom on doped graphene in direct dehydrogenation of propane by rational doping: a density functional theory study. J. Phys. Chem. C 122, 1570–1576 (2018).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Kresse, G. & Hafner, J. Ab initio molecular dynamics for liquid metals. Phys. Rev. B 47, 558 (1993).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Sanz-Navarro, C. F. et al. Molecular dynamics simulations of the interactions between platinum clusters and carbon platelets. J. Phys. Chem. A 112, 1392–1402 (2008).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Gerceker, D. et al. Methane conversion to ethylene and aromatics on PtSn catalysts. ACS Catal. 7, 2088–2100 (2017).
Guo, X. et al. Direct, nonoxidative conversion of methane to ethylene, aromatics, and hydrogen. Science 344, 616–619 (2014).
Marcano, D. et al. Improved synthesis of graphene oxide. ACS Nano 4, 4806–4814 (2010).
Jacob, R. J., Kline, D. J. & Zachariah, M. R. High speed 2-dimensional temperature measurements of nanothermite composites: probing thermal vs. gas generation effects. J. Appl. Phys. 123, 115902 (2018).
Acknowledgements
This project is not directly funded. We acknowledge the support of the Maryland Nanocenter, its Surface Analysis Center and AIMLab and the University of Maryland supercomputing resources (http://hpcc.umd.edu). Z.H. and R.S.-Y. acknowledge the financial support from NSF-DMR award no. 1809439. P.X. and Chao Wang thank the support from the Advanced Research Projects Agency—Energy (ARPA-E), Department of Energy (DOE) and the Petroleum Research Fund, American Chemical Society. L.M., T.W. and J.L. acknowledge the financial support from the US Department of Energy under Contract DE-AC02-06CH11357. Research conducted at beamline 9-BM used resources of the Advanced Photon Source, an Office of Science User Facility operated for the US DOE by Argonne National Laboratory under Contract no. DE-AC02-06CH11357. Chongmin Wang thanks the support of LDRD of PNNL and the in situ ETEM was conducted in the William R. Wiley Environmental Molecular Sciences Laboratory (EMSL), a national scientific user facility sponsored by DOE’s Office of Biological and Environmental Research and located at PNNL. PNNL is operated by Battelle for the Department of Energy under Contract DE-AC05-76RLO1830.
Author information
Authors and Affiliations
Contributions
L.H. and Y.Y. contributed to the idea and experimental design. Y.Y., T.L., M.J. and Z.L. conducted the experiments and materials preparation. Z.H. and R.S.-Y. performed the high-resolution microscopy. P.X. and Chao Wang contributed to the catalysis evaluation. L.W., Z.P. and T.L. conducted the simulation analysis. L.M., T.W. and J.L. contributed to the X-ray absorption measurements and analysis. Y.H. and Chongmin Wang performed the in situ environmental microscopy. D.J.K. and M.R.Z. performed the temperature characterization and thermal gravimetric analysis. L.H. and Y.Y. wrote the paper and all the authors commented on the final manuscript.
Corresponding authors
Additional information
Peer review information: Nature Nanotechnology thanks Abhaya (Krishna) Datye, Frédéric Jaouen and Yadong Li for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary Figs. S1–S22.
Rights and permissions
About this article
Cite this article
Yao, Y., Huang, Z., Xie, P. et al. High temperature shockwave stabilized single atoms. Nat. Nanotechnol. 14, 851–857 (2019). https://doi.org/10.1038/s41565-019-0518-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-019-0518-7
This article is cited by
-
Continuous and low-carbon production of biomass flash graphene
Nature Communications (2024)
-
Challenging thermodynamics: combining immiscible elements in a single-phase nano-ceramic
Nature Communications (2024)
-
The reformation of catalyst: From a trial-and-error synthesis to rational design
Nano Research (2024)
-
Emerging Atomically Precise Metal Nanoclusters and Ultrasmall Nanoparticles for Efficient Electrochemical Energy Catalysis: Synthesis Strategies and Surface/Interface Engineering
Electrochemical Energy Reviews (2024)
-
Data-driven rational design of single-atom materials for hydrogen evolution and sensing
Nano Research (2024)