Abstract
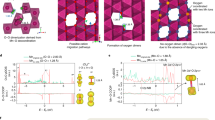
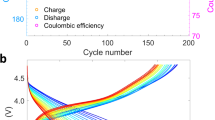
Surfaces, interfaces and grain boundaries are classically known to be sinks of defects generated within the bulk lattice. Here, we report an inverse case by which the defects generated at the particle surface are continuously pumped into the bulk lattice. We show that, during operation of a rechargeable battery, oxygen vacancies produced at the surfaces of lithium-rich layered cathode particles migrate towards the inside lattice. This process is associated with a high cutoff voltage at which an anionic redox process is activated. First-principle calculations reveal that triggering of this redox process leads to a sharp decrease of both the formation energy of oxygen vacancies and the migration barrier of oxidized oxide ions, therefore enabling the migration of oxygen vacancies into the bulk lattice of the cathode. This work unveils a coupled redox dynamic that needs to be taken into account when designing high-capacity layered cathode materials for high-voltage lithium-ion batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data that support the findings of this study are kept at the William R. Wiley Environmental Molecular Sciences Laboratory at PNNL and are available from the corresponding authors upon request.
References
Rozier, P. & Tarascon, J. M. Review—Li-rich layered oxide cathodes for next-generation Li-ion batteries: chances and challenges. J. Electrochem. Soc. 162, A2490–A2499 (2015).
Manthiram, A., Knight, J. C., Myung, S. T., Oh, S. M. & Sun, Y. K. Nickel-rich and lithium-rich layered oxide cathodes: progress and perspectives. Adv. Energy Mater. 6, 1501010 (2016).
Hy, S. et al. Performance and design considerations for lithium excess layered oxide positive electrode materials for lithium ion batteries. Energy Environ. Sci. 9, 1931–1954 (2016).
Seo, D. H. et al. The structural and chemical origin of the oxygen redox activity in layered and cation-disordered Li-excess cathode materials. Nat. Chem. 8, 692–697 (2016).
Luo, K. et al. Charge-compensation in 3D-transition-metal-oxide intercalation cathodes through the generation of localized electron holes on oxygen. Nat. Chem. 8, 684–691 (2016).
McCalla, E. et al. Visualization of O–O peroxo-like dimers in high-capacity layered oxides for Li-ion batteries. Science 350, 1516–1521 (2015).
Sathiya, M. et al. Reversible anionic redox chemistry in high-capacity layered-oxide electrodes. Nat. Mater. 12, 827–835 (2013).
Qiu, B., Zhang, M., Xia, Y., Liu, Z. & Meng, Y. S. Understanding and controlling anionic electrochemical activity in high-capacity oxides for next generation Li-ion batteries. Chem. Mater. 29, 908–915 (2017).
Ceder, G. et al. Identification of cathode materials for lithium batteries guided by first-principles calculations. Nature 392, 694–696 (1998).
Zheng, J. et al. Structural and chemical evolution of Li- and Mn-rich layered cathode material. Chem. Mater. 27, 1381–1390 (2015).
Gu, L., Xiao, D., Hu, Y. S., Li, H. & Ikuhara, Y. Atomic-scale structure evolution in a quasi-equilibrated electrochemical process of electrode materials for rechargeable batteries. Adv. Mater. 27, 2134–2149 (2015).
Gu, M. et al. Nanoscale phase separation, cation ordering and surface chemistry in pristine Li1.2Ni0.2Mn0.6O2 for Li-ion batteries. Chem. Mater. 25, 2319–2326 (2013).
Xu, B., Fell, C. R., Chi, M. F. & Meng, Y. S. Identifying surface structural changes in layered Li-excess nickel manganese oxides in high voltage lithium ion batteries: a joint experimental and theoretical study. Energy Environ. Sci. 4, 2223–2233 (2011).
Zheng, J. et al. Corrosion/fragmentation of layered composite cathode and related capacity/voltage fading during cycling process. Nano Lett. 13, 3824–3830 (2013).
Mohanty, D. et al. Structural transformation of a lithium-rich Li1.2Co0.1Mn0.55Ni0.15O2 cathode during high voltage cycling resolved by in situ X-ray diffraction. J. Power Sources 229, 239–248 (2013).
Yang, X. Q., Sun, X. & McBreen, J. New findings on the phase transitions in Li1-xNiO2: in situ synchrotron X-ray diffraction studies. Electrochem. Commun. 1, 227–232 (1999).
Yoon, W.-S., Chung, K. Y., McBreen, J. & Yang, X.-Q. A comparative study on structural changes of LiCo1/3Ni1/3Mn1/3O2 and LiNi0.8Co0.15Al0.05O2 during first charge using in situ XRD. Electrochem. Commun. 8, 1257–1262 (2006).
Rana, J. et al. Structural changes in Li2MnO3 cathode material for Li-ion batteries. Adv. Energy Mater. 4, 1300998 (2014).
Lin, F. et al. Surface reconstruction and chemical evolution of stoichiometric layered cathode materials for lithium-ion batteries. Nat. Commun. 5, 3529 (2014).
Yabuuchi, N., Yoshii, K., Myung, S. T., Nakai, I. & Komaba, S. Detailed studies of a high-capacity electrode material for rechargeable batteries, Li2MnO3–LiCo1/3Ni1/3Mn1/3O2. J. Am. Chem. Soc. 133, 4404–4419 (2011).
Yan, P. F. et al. Intragranular cracking as a critical barrier for high-voltage usage of layer-structured cathode for lithium-ion batteries. Nat. Commun. 8, 14101 (2017).
Pearce, P. E. et al. Evidence for anionic redox activity in a tridimensional-ordered Li-rich positive electrode beta-Li2IrO3. Nat. Mater. 16, 580–586 (2017).
Lee, E. & Persson, K. A. Structural and chemical evolution of the layered Li-excess LixMnO3 as a function of Li content from first-principles calculations. Adv. Energy Mater. 4, 1400498 (2014).
Armstrong, A. R. et al. Demonstrating oxygen loss and associated structural reorganization in the lithium battery cathode Li[Ni0.2Li0.2Mn0.6]O2. J. Am. Chem. Soc. 128, 8694–8698 (2006).
Lu, Z. H., Beaulieu, L. Y., Donaberger, R. A., Thomas, C. L. & Dahn, J. R. Synthesis, structure and electrochemical behavior of Li[NixLi1/3-2x/3Mn2/3-x/3]O2. J. Electrochem. Soc. 149, A778–A791 (2002).
Hausbrand, R. et al. Fundamental degradation mechanisms of layered oxide Li-ion battery cathode materials: methodology, insights and novel approaches. Mater. Sci. Eng. B 192, 3–25 (2015).
Reimers, J. N. & Dahn, J. R. Electrochemical and in situ X-ray diffraction studies of lithium intercalation in LixCoO2. J. Electrochem. Soc. 139, 2091–2097 (1992).
Hoang, K. Defect physics, delithiation mechanism, and electronic and ionic conduction in layered lithium manganese oxide cathode materials. Phys. Rev. Appl. 3, 024013 (2015).
Gent, W. E. et al. Coupling between oxygen redox and cation migration explains unusual electrochemistry in lithium-rich layered oxides. Nat. Commun. 8, 2091 (2017).
Yan, P. et al. Atomic-resolution visualization of distinctive chemical mixing behavior of Ni, Co and Mn with Li in layered lithium transition metal oxide cathode materials. Chem. Mater. 27, 5393–5401 (2015).
Yan, P., Zheng, J., Zhang, J.-G. & Wang, C. Atomic resolution structural and chemical imaging revealing the sequential migration of Ni, Co and Mn upon the battery cycling of layered cathode. Nano Lett. 17, 3946–3951 (2017).
Gu, M. et al. Conflicting roles of nickel in controlling cathode performance in lithium ion batteries. Nano Lett. 12, 5186–5191 (2012).
Yan, P. et al. Evolution of lattice structure and chemical composition of the surface reconstruction layer in Li1.2Ni0.2Mn0.6O2 cathode material for lithium ion batteries. Nano Lett. 15, 514–522 (2015).
Qian, D., Xu, B., Chi, M. & Meng, Y. S. Uncovering the roles of oxygen vacancies in cation migration in lithium excess layered oxides. Phys. Chem. Chem. Phys. 16, 14665–14668 (2014).
Fell, C. R. et al. Correlation between oxygen vacancy, microstrain and cation distribution in lithium-excess layered oxides during the first electrochemical cycle. Chem. Mater. 25, 1621–1629 (2013).
Sathiya, M. et al. Origin of voltage decay in high-capacity layered oxide electrodes. Nat. Mater. 14, 230–238 (2015).
Mohanty, D. et al. Unraveling the voltage-fade mechanism in high-energy-density lithium-ion batteries: origin of the tetrahedral cations for spinel conversion. Chem. Mater. 26, 6272–6280 (2014).
Abdellahi, A., Urban, A., Dacek, S. & Ceder, G. The effect of cation disorder on the average Li intercalation voltage of transition-metal oxides. Chem. Mater. 28, 3659–3665 (2016).
Lee, J. et al. Unlocking the potential of cation-disordered oxides for rechargeable lithium batteries. Science 343, 519–522 (2014).
Saubanere, M., McCalla, E., Tarascon, J. M. & Doublet, M. L. The intriguing question of anionic redox in high-energy density cathodes for Li-ion batteries. Energy Environ. Sci. 9, 984–991 (2016).
Qiu, B. et al. Gas–solid interfacial modification of oxygen activity in layered oxide cathodes for lithium-ion batteries. Nat. Commun. 7, 12108 (2016).
Liu, S. et al. Surface doping to enhance structural integrity and performance of Li-rich layered oxide. Adv. Energy Mater. 8, 1802105 (2018).
Hu, E. et al. Evolution of redox couples in Li- and Mn-rich cathode materials and mitigation of voltage fade by reducing oxygen release. Nat. Energy 3, 690–698 (2018).
Kuppan, S., Shukla, A. K., Membreno, D., Nordlund, D. & Chen, G. Revealing anisotropic spinel formation on pristine Li- and Mn-rich layered oxide surface and its impact on cathode performance. Adv. Energy Mater. 7, 1602010 (2017).
Devaraj, A. et al. Visualizing nanoscale 3D compositional fluctuation of lithium in advanced lithium-ion battery cathodes. Nat. Commun. 6, 8014 (2015).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169–11186 (1996).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Heyd, J., Scuseria, G. E. & Ernzerhof, M. Hybrid functionals based on a screened Coulomb potential. J. Chem. Phys. 118, 8207–8215 (2003); erratum 124, 219906 (2006).
Chen, H. & Islam, M. S. Lithium extraction mechanism in Li-rich Li2MnO3 involving oxygen hole formation and dimerization. Chem. Mater. 28, 6656–6663 (2016).
Van der Ven, A., Aydinol, M. K., Ceder, G., Kresse, G. & Hafner, J. First-principles investigation of phase stability in LixCoO2. Phys. Rev. B 58, 2975–2987 (1998).
Xiao, R., Li, H. & Chen, L. Density functional investigation on Li2MnO3. Chem. Mater. 24, 4242–4251 (2012).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
Chen, H. & Umezawa, N. Hole localization, migration and the formation of peroxide anion in perovskite SrTiO3. Phys. Rev. B 90, 035202 (2014).
Chen, S. & Wang, L.-W. Double-hole-induced oxygen dimerization in transition metal oxides. Phys. Rev. B 89, 014109 (2014).
Acknowledgements
This work was supported by the Assistant Secretary for Energy Efficiency and Renewable Energy, Office of Vehicle Technologies, of the US Department of Energy (DOE) under contract no. DE-AC02-05CH11231, subcontract no. 6951379 under the Batteries for Advanced Battery Materials Research. The work was conducted in the William R. Wiley Environmental Molecular Sciences Laboratory, a national scientific user facility sponsored by DOE’s Office of Biological and Environmental Research and located at PNNL. PNNL is operated by Battelle for DOE under contract DE-AC05-76RLO1830. L.-M.L. was supported by the Science Challenge Project (TZ2018004) of the National Natural Science Foundation of China (nos. 51572016 and U1530401) and the Fundamental Research Funds for the Central Universities, and Newton Advanced Fellowship under grant no. NAFR1180242. P.Y. acknowledges support from the National Natural Science Fund for Innovative Research Groups (grant no. 51621003) and the National Key Research and Development Program of China (grant no. 2016YFB0700700). Z.-K.T. thanks the National Natural Science Foundation of China (grant no. 51602092) for support.
Author information
Authors and Affiliations
Contributions
P.Y. and C.W. conceived the research plan. J.Z., J.-G.Z., K.A. and G.C. synthesized the samples and carried out the cell test. P.Y. conducted the TEM work. Z.-K.T. and L.-M.L. conducted the simulation work. P.Y. and C.W. wrote the manuscript. All authors approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Nanotechnology thanks Jun Chen and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information
Supplementary Figs. 1–10
Supplementary Video 1
Volume rendering and slicing of the 3D reconstruction
Supplementary Video 2
Iso-surface displaying the 3D reconstruction
Rights and permissions
About this article
Cite this article
Yan, P., Zheng, J., Tang, ZK. et al. Injection of oxygen vacancies in the bulk lattice of layered cathodes. Nat. Nanotechnol. 14, 602–608 (2019). https://doi.org/10.1038/s41565-019-0428-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-019-0428-8
This article is cited by
-
Trapped O2 and the origin of voltage fade in layered Li-rich cathodes
Nature Materials (2024)
-
Efficient direct repairing of lithium- and manganese-rich cathodes by concentrated solar radiation
Nature Communications (2024)
-
Structurally robust lithium-rich layered oxides for high-energy and long-lasting cathodes
Nature Communications (2024)
-
Boosting cycling stability by regulating surface oxygen vacancies of LNMO by rapid calcination
Nano Research (2024)
-
Surface-targeted functionalization of nickel-rich cathodes through synergistic slurry additive approach with multi-level impact using minimal quantity
Nano Research (2024)