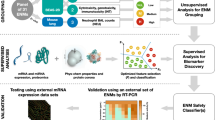
Abstract
Engineered nanomaterials (ENMs) have tremendous potential to produce beneficial technological impact in numerous sectors in society. Safety assessment is, of course, of paramount importance. However, the myriad variations of ENM properties makes the identification of specific features driving toxicity challenging. At the same time, reducing animal tests by introducing alternative and/or predictive in vitro and in silico methods has become a priority. It is important to embrace these new advances in the safety assessment of ENMs. Indeed, remarkable progress has been made in recent years with respect to mechanism-based hazard assessment of ENMs, including systems biology approaches as well as high-throughput screening platforms, and new tools are also emerging in risk assessment and risk management for humans and the environment across the whole life-cycle of nano-enabled products. Here, we highlight some of the key advances in the hazard and risk assessment of ENMs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Maynard, A. D. et al. Safe handling of nanotechnology. Nature 444, 267–269 (2006).
Krug, H. F. Nanosafety research--are we on the right track? Angew. Chem. Int. Ed. Engl. 53, 12304–12319 (2014).
Valsami-Jones, E. & Lynch, I. NANOSAFETY. How safe are nanomaterials? Science 350, 388–389 (2015).
Sayes, C. M. & Warheit, D. B. Characterization of nanomaterials for toxicity assessment. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 1, 660–670 (2009).
Fadeel, B., Fornara, A., Toprak, M. S. & Bhattacharya, K. Keeping it real: the importance of material characterization in nanotoxicology. Biochem. Biophys. Res. Commun. 468, 498–503 (2015).
Walkey, C. D. & Chan, W. C. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev. 41, 2780–2799 (2012).
Monopoli, M. P., Åberg, C., Salvati, A. & Dawson, K. A. Biomolecular coronas provide the biological identity of nanosized materials. Nat. Nanotech. 7, 779–786 (2012).
Cohen, Y., Rallo, R., Liu, R. & Liu, H. H. In silico analysis of nanomaterials hazard and risk. Acc. Chem. Res. 46, 802–812 (2013).
Winkler, D. A. Recent advances, and unresolved issues, in the application of computational modelling to the prediction of the biological effects of nanomaterials. Toxicol. Appl. Pharmacol. 299, 96–100 (2016).
Fadeel, B. et al. There’s plenty of room at the forum: potential risks and safety assessment of engineered nanomaterials. Nanotoxicology 1, 73–84 (2007).
Savolainen, K. et al. Risk assessment of engineered nanomaterials and nanotechnologies--a review. Toxicology 269, 92–104 (2010).
Hussain, S. M. et al. At the crossroads of nanotoxicology in vitro: past achievements and current challenges. Toxicol. Sci. 147, 5–16 (2015).
Valsami-Jones, E., Lynch, I. & Charitidis, C. A. Nanomaterial ontologies for nanosafety: a rose by any other name…. J. Nanomed. Res. 3, 00070 (2016).
Worth, A. et al. Evaluation of the Availability and Applicability of Computational Approaches in the Safety Assessment of Nanomaterials (Joint Research Centre, 2017); https://doi.org/10.2760/248139
Hastings, J. et al. eNanoMapper: harnessing ontologies to enable data integration for nanomaterial risk assessment. J. Biomed. Semant. 6, 1–15 (2015).
Jeliazkova, N. et al. The eNanoMapper database for nanomaterial safety information. Beilstein J. Nanotechnol. 6, 1609–1634 (2015).
Farcal, L. et al. Comprehensive in vitro toxicity testing of a panel of representative oxide nanomaterials: first steps towards an intelligent testing strategy. PLoS ONE 10, e0127174 (2015).
Collins, A. R. et al. High throughput toxicity screening and intracellular detection of nanomaterials. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 9, e1413 (2017).
Richard, A. M. et al. ToxCast chemical landscape: paving the road to 21st century toxicology. Chem. Res. Toxicol. 29, 1225–1251 (2016).
Nel, A. E. & Malloy, T. F. Policy reforms to update chemical safety testing. Science 355, 1016–1018 (2017).
Anguissola, S., Garry, D., Salvati, A., O’Brien, P. J. & Dawson, K. A. High content analysis provides mechanistic insights on the pathways of toxicity induced by amine-modified polystyrene nanoparticles. PLoS ONE 9, e108025 (2014).
Harris, G. et al. Iron oxide nanoparticle toxicity testing using high-throughput analysis and high-content imaging. Nanotoxicology 9, 87–94 (2015).
Liu, R. et al. Classification NanoSAR development for cytotoxicity of metal oxide nanoparticles. Small 7, 1118–1126 (2011).
Rallo, R. et al. Self-organizing map analysis of toxicity-related cell signaling pathways for metal and metal oxide nanoparticles. Environ. Sci. Technol. 45, 1695–1702 (2011).
George, S. et al. Use of a high-throughput screening approach coupled with in vivo zebrafish embryo screening to develop hazard ranking for engineered nanomaterials. ACS Nano 5, 1805–1817 (2011).
Liu, R. et al. Automated phenotype recognition for zebrafish embryo based in vivo high throughput toxicity screening of engineered nano-materials. PLoS ONE 7, e35014 (2012).
Marchese Robinson, R. L. et al. How should the completeness and quality of curated nanomaterial data be evaluated? Nanoscale 8, 9919–9943 (2016).
Nel, A. et al. Nanomaterial toxicity testing in the 21st century: use of a predictive toxicological approach and high-throughput screening. Acc. Chem. Res. 46, 607–621 (2013).
Briffa, S. M. et al. Development of scalable and versatile nanomaterial libraries for nanosafety studies: polyvinylpyrrolidone (PVP) capped metal oxide nanoparticles. RSC Adv. 7, 3894–3906 (2017).
Hansjosten, I. et al. Microscopy-based high-throughput assays enable multi-parametric analysis to assess adverse effects of nanomaterials in various cell lines. Arch. Toxicol. 92, 633–649 (2018).
Gallud, A. et al. Cytotoxicity screening of a panel of 31 nanomaterials in the human monocytic cell line THP.1 versus primary human monocyte-derived macrophages: assessing the role of surface modification. In New Tools and Approaches for Nanomaterial Safety Assessment: Book of Abstracts (2017); https://doi.org/10.13140/RG.2.2.26504.62722
Hongisto, V. et al. High-throughput screening approach evaluated toxicity of 31 engineered nanomaterials generated for the NANOSOLUTIONS project. In New Tools and Approaches for Nanomaterial Safety Assessment : Book of Abstracts (2017); https://doi.org/10.13140/RG.2.2.26504.62722
Walkey, C. D. et al. Protein corona fingerprinting predicts the cellular interaction of gold and silver nanoparticles. ACS Nano 8, 2439–2455 (2014).
Collins, F. S., Gray, G. M. & Bucher, J. R. TOXICOLOGY. Transforming environmental health protection. Science 319, 906–907 (2008).
Sturla, S. J. et al. Systems toxicology: from basic research to risk assessment. Chem. Res. Toxicol. 27, 314–329 (2014).
Hartung, T. et al. Systems toxicology: real world applications and opportunities. Chem. Res. Toxicol. 30, 870–882 (2017).
Costa, P. M. & Fadeel, B. Emerging systems biology approaches in nanotoxicology: towards a mechanism-based understanding of nanomaterial hazard and risk. Toxicol. Appl. Pharmacol. 299, 101–111 (2016).
Nymark, P. et al. A data fusion pipeline for generating and enriching adverse outcome pathway descriptions. Toxicol. Sci. 162, 264–275 (2018).
Fortino, V. & Greco, D. ENM SAFETY CLASSIFIER – a multi-view feature selection and classification algorithm for prediction of engineered nanomaterials (ENM) safety. In New Tools and Approaches for Nanomaterial Safety Assessment : Book of Abstracts (2017); https://doi.org/10.13140/RG.2.2.26504.62722
Kinaret, P. et al. Network analysis reveals similar transcriptomic responses to intrinsic properties of carbon nanomaterials in vitro and in vivo. ACS Nano 11, 3786–3796 (2017).
Bornholdt, J. et al. Identification of gene transcription start sites and enhancers responding to pulmonary carbon nanotube exposure in vivo. ACS Nano 11, 3597–3613 (2017).
Hristozov, D. R., Gottardo, S., Critto, A. & Marcomini, A. Risk assessment of engineered nanomaterials: a review of available data and approaches from a regulatory perspective. Nanotoxicology 6, 880–898 (2012).
Park, M. et al. Hazard evaluation in GUIDENANO: a web-based guidance tool for risk assessment and mitigation of nano-enabled products. In New Tools and Approaches for Nanomaterial Safety Assessment: Book of Abstracts (2017); https://doi.org/10.13140/RG.2.2.26504.62722
Subramanian, V. et al. Sustainable nanotechnology decision support system: bridging risk management, sustainable innovation and risk governance. J. Nanopart. Res. 8, 1–13 (2016).
Zabeo, A. et al. SUNDS, a multi-criteria decision support system for nanotechnology risk assessment and management. In New Tools and Approaches for Nanomaterial Safety Assessment : Book of Abstracts (2017); https://doi.org/10.13140/RG.2.2.26504.62722
Dekkers, S. et al. Towards a nanospecific approach for risk assessment. Regul. Toxicol. Pharmacol. 80, 46–59 (2016).
Oomen, A. G. et al. Grouping and read-across approaches for risk assessment of nanomaterials. Int. J. Environ. Res. Public Health 12, 13415–13434 (2015).
González-Gálvez, D., Janer, G., Vilar, G., Vílchez, A. & Vázquez-Campos, S. The life cycle of engineered nanoparticles. Adv. Exp. Med. Biol. 947, 41–69 (2017).
Fernández-Rosas, E. et al. Influence of nanomaterial compatibilization strategies on polyamide nanocomposites properties and nanomaterial release during the use phase. Environ. Sci. Technol. 50, 2584–2594 (2016).
Mitrano, D. M., Lombi, E., Dasilva, Y. A. & Nowack, B. Unraveling the complexity in the aging of nanoenhanced textiles: a comprehensive sequential study on the effects of sunlight and washing on silver nanoparticles. Environ. Sci. Technol. 50, 5790–5799 (2016).
Wohlleben, W. et al. A pilot interlaboratory comparison of protocols that simulate aging of nanocomposites and detect released fragments. Environ. Chem. 11, 402–418 (2014).
Nowack, B. et al. Meeting the needs for released nanomaterials required for further testing - the SUN approach. Environ. Sci. Technol. 50, 2747–2753 (2016).
Tsang, M. P., Kikuchi-Uehara, E., Sonnemann, G. W., Aymonier, C. & Hirao, M. Evaluating nanotechnology opportunities and risks through integration of life-cycle and risk assessment. Nat. Nanotech. 12, 734–739 (2017).
Linkov, I. et al. Integrate life-cycle assessment and risk analysis results, not methods. Nat. Nanotech. 12, 740–743 (2017).
Guinée, J. B., Heijungs, R., Vijver, M. G. & Peijnenburg, W. J. G. M. Setting the stage for debating the roles of risk assessment and life-cycle assessment of engineered nanomaterials. Nat. Nanotech. 12, 727–733 (2017).
Bishop, L. et al. In vivo toxicity assessment of occupational components of the carbon nanotube life cycle to provide context to potential health effects. ACS Nano 11, 8849–8863 (2017).
Rasmussen, K. et al. Review of achievements of the OECD Working Party on Manufactured Nanomaterials’ Testing and Assessment Programme. From exploratory testing to test guidelines. Regul. Toxicol. Pharmacol. 74, 147–160 (2016).
Fadeel, B. Systems biology in nanosafety research. Nanomed. (Lond.) 10, 1039–1041 (2015).
Acknowledgements
The authors and their laboratories were supported, in part, by the European Commission through the Seventh Framework Programme (FP7-eNANOMAPPER, grant no. 604134; FP7-GUIDENANO, grant no. 604387; FP7-NANOMILE, grant no. 310451; FP7-NANOSOLUTIONS, grant no. 309329; FP7-SUN, grant no. 604305). We thank all the project partners for invaluable contributions to these projects, and the project officers for their guidance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fadeel, B., Farcal, L., Hardy, B. et al. Advanced tools for the safety assessment of nanomaterials. Nature Nanotech 13, 537–543 (2018). https://doi.org/10.1038/s41565-018-0185-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-018-0185-0
This article is cited by
-
Analytical methods for assessing antimicrobial activity of nanomaterials in complex media: advances, challenges, and perspectives
Journal of Nanobiotechnology (2023)
-
Principles and requirements for nanomaterial representations to facilitate machine processing and cooperation with nanoinformatics tools
Journal of Cheminformatics (2023)
-
Human and environmental safety of carbon nanotubes across their life cycle
Nature Reviews Materials (2023)
-
A tool for preliminary health and safety risk assessment of activities involved with nanomaterials: design and validation
Journal of Nanoparticle Research (2023)
-
Recent Advances in Multifunctional Reticular Framework Nanoparticles: A Paradigm Shift in Materials Science Road to a Structured Future
Nano-Micro Letters (2023)