Abstract
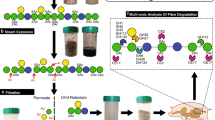
Processed foods often include food additives such as xanthan gum, a complex polysaccharide with unique rheological properties, that has established widespread use as a stabilizer and thickening agent. Xanthan gum’s chemical structure is distinct from those of host and dietary polysaccharides that are more commonly expected to transit the gastrointestinal tract, and little is known about its direct interaction with the gut microbiota, which plays a central role in digestion of other dietary fibre polysaccharides. Here we show that the ability to digest xanthan gum is common in human gut microbiomes from industrialized countries and appears contingent on a single uncultured bacterium in the family Ruminococcaceae. Our data reveal that this primary degrader cleaves the xanthan gum backbone before processing the released oligosaccharides using additional enzymes. Some individuals harbour Bacteroides intestinalis that is incapable of consuming polymeric xanthan gum but grows on oligosaccharide products generated by the Ruminococcaceae. Feeding xanthan gum to germfree mice colonized with a human microbiota containing the uncultured Ruminococcaceae supports the idea that the additive xanthan gum can drive expansion of the primary degrader Ruminococcaceae, along with exogenously introduced B. intestinalis. Our work demonstrates the existence of a potential xanthan gum food chain involving at least two members of different phyla of gut bacteria and provides an initial framework for understanding how widespread consumption of a recently introduced food additive influences human microbiomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All sequencing reads have been deposited at the European Nucleotide Archive under BioProject PRJEB44146. All annotated MAGs are publicly available via Figshare (DOIs: 10.6084/m9.figshare.14494602, 10.6084/m9.figshare.14494536, 10.6084/m9.figshare.14494677, 10.6084/m9.figshare.14494683 and 10.6084/m9.figshare.14494689). The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE101 partner repository with the dataset identifier PXD031522. There are no restrictions on data or biological resource availability. Data and biological resources can be requested from the corresponding authors. Source data are provided with this paper.
Code availability
No new codes were developed or compiled in this study.
References
Chassaing, B. et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519, 92–96 (2015).
Collins, J. et al. Dietary trehalose enhances virulence of epidemic Clostridium difficile. Nature 553, 291–294 (2018).
Laudisi, F. et al. The food additive maltodextrin promotes endoplasmic reticulum stress–driven mucus depletion and exacerbates intestinal inflammation. Cell. Mol. Gastroenterol. Hepatol. 7, 457–473 (2019).
Etienne-Mesmin, L. et al. Experimental models to study intestinal microbes–mucus interactions in health and disease. FEMS Microbiol. Rev. 43, 457–489 (2019).
García-Ochoa, F., Santos, V. E., Casas, J. A. & Gómez, E. Xanthan gum: production, recovery, and properties. Biotechnol. Adv. 18, 549–579 (2000).
Cartmell, A. et al. A surface endogalactanase in Bacteroides thetaiotaomicron confers keystone status for arabinogalactan degradation. Nat. Microbiol. 3, 1314–1326 (2018).
Pudlo, N. A. et al. Diverse events have transferred genes for edible seaweed digestion from marine to human gut bacteria. Cell Host & Microbe 30, 314–328 (2022).
Casas, J. A., Santos, V. E. & García-Ochoa, F. Xanthan gum production under several operational conditions: molecular structure and rheological properties. Enzym. Microb. Technol. 26, 282–291 (2000).
Sworn, G. in Handbook of Hydrocolloids (Third edition), edited by Phillips, G. O. and Williams, P. A. 833–853 (Elsevier, 2021).
King, J. A. et al. Incidence of celiac disease is increasing over time. Am. J. Gastroenterol. https://doi.org/10.14309/ajg.0000000000000523 (2020).
Mortensen, A. et al. Re-evaluation of xanthan gum (E 415) as a food additive. EFSA J. 15, e04909 (2017).
Baxter, N. T. et al. Dynamics of human gut microbiota and short-chain fatty acids in response to dietary interventions with three fermentable fibers. mBio 10, e02566–18 (2019).
Hehemann, J.-H., Kelly, A. G., Pudlo, N. A., Martens, E. C. & Boraston, A. B. Bacteria of the human gut microbiome catabolize red seaweed glycans with carbohydrate-active enzyme updates from extrinsic microbes. Proc. Natl Acad. Sci. USA 109, 19786–19791 (2012).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41, 590–596 (2013).
Goodman, A. L. et al. Extensive personal human gut microbiota culture collections characterized and manipulated in gnotobiotic mice. Proc. Natl Acad. Sci. USA 108, 6252–6257 (2011).
Kim, C. C. et al. Genomic insights from Monoglobus pectinilyticus: a pectin-degrading specialist bacterium in the human colon. ISME J. 13, 1437–1456 (2019).
Ruijssenaars, H. J., de Bont, J. A. M. & Hartmans, S. A pyruvated mannose-specific xanthan lyase involved in xanthan degradation by Paenibacillus alginolyticus XL-1. Appl. Environ. Microbiol. 65, 2446–2452 (1999).
Nankai, H., Hashimoto, W., Miki, H., Kawai, S. & Murata, K. Microbial system for polysaccharide depolymerization: enzymatic route for xanthan depolymerization by Bacillus sp. strain GL1. Appl. Environ. Microbiol. 65, 2520–2526 (1999).
Hashimoto, W., Nankai, H., Mikami, B. & Murata, K. Crystal structure of Bacillus sp. GL1 xanthan lyase, which acts on the side chains of xanthan. J. Biol. Chem. 278, 7663–7673 (2003).
Jensen, P. F. et al. Structure and dynamics of a promiscuous xanthan lyase from Paenibacillus nanensis and the design of variants with increased stability and activity. Cell Chem. Biol. 26, 191–202.e6 (2019).
Aspeborg, H., Coutinho, P. M., Wang, Y., Brumer, H. & Henrissat, B. Evolution, substrate specificity and subfamily classification of glycoside hydrolase family 5 (GH5). BMC Evol. Biol. 12, 1–16 (2012).
Jongkees, S. A. K. & Withers, S. G. Unusual enzymatic glycoside cleavage mechanisms. Acc. Chem. Res. 47, 226–235 (2014).
Rovira, C., Males, A., Davies, G. J. & Williams, S. J. Mannosidase mechanism: at the intersection of conformation and catalysis. Curr. Opin. Struct. Biol. 62, 79–92 (2020).
Kool, M. M. et al. Characterization of an acetyl esterase from Myceliophthora thermophila C1 able to deacetylate xanthan. Carbohydr. Polym. 111, 222–229 (2014).
Grondin, J. M., Tamura, K., Déjean, G., Abbott, D. W. & Brumer, H. Polysaccharide utilization loci: fueling microbial communities. J. Bacteriol. 199, e00860–16 (2017).
Pilgaard, B., Vuillemin, M., Holck, J., Wilkens, C. & Meyer, A. S. Specificities and synergistic actions of novel PL8 and PL7 alginate lyases from the marine fungus Paradendryphiella salina. J. Fungi 7, 80 (2021).
Zhu, B. & Yin, H. Alginate lyase: review of major sources and classification, properties, structure-function analysis and applications. Bioengineered 6, 125–131 (2015).
Terrapon, N. et al. PULDB: the expanded database of polysaccharide utilization loci. Nucleic Acids Res. 46, D677–D683 (2018).
Sun, Z., Liu, H., Wang, X., Yang, F. & Li, X. Proteomic analysis of the xanthan-degrading pathway of Microbacterium sp. XT11. ACS Omega 4, 19096–19105 (2019).
Yang, F. et al. Novel endotype xanthanase from xanthan-degrading Microbacterium sp. strain XT11. Appl. Environ. Microbiol. 85, e01800–18 (2019).
Guillén, D., Sánchez, S. & Rodríguez-Sanoja, R. Carbohydrate-binding domains: multiplicity of biological roles. Appl. Microbiol. Biotechnol. 85, 1241–1249 (2010).
Mistry, J. et al. Pfam: the protein families database in 2021. Nucleic Acids Res. 49, D412–D419 (2021).
Ebbes, M. et al. Fold and function of the InlB B-repeat. J. Biol. Chem. 286, 15496–15506 (2011).
Bleymüller, W. M. et al. MET-activating residues in the B-repeat of the Listeria monocytogenes invasion protein InlB. J. Biol. Chem. 291, 25567–25577 (2016).
Kool, M. M., Gruppen, H., Sworn, G. & Schols, H. A. Comparison of xanthans by the relative abundance of its six constituent repeating units. Carbohydr. Polym. 98, 914–921 (2013).
Moroz, O. V. et al. Structural dynamics and catalytic properties of a multi-modular xanthanase. ACS Catal. 8, 6021–6034 (2018).
Yang, F. et al. Production and purification of a novel xanthan lyase from a xanthan-degrading Microbacterium sp. strain XT11. ScientificWorldJournal 2014, A368434 (2014).
Gregg, K. J. et al. Analysis of a new family of widely distributed metal-independent α-mannosidases provides unique insight into the processing of N-linked glycans. J. Biol. Chem. 286, 15586–15596 (2011).
Daly, J., Tomlin, J. & Read, N. W. The effect of feeding xanthan gum on colonic function in man: correlation with in vitro determinants of bacterial breakdown. Br. J. Nutr. 69, 897–902 (1993).
Kiełbasa, S. M., Wan, R., Sato, K., Horton, P. & Frith, M. C. Adaptive seeds tame genomic sequence comparison. Genome Res. 21, 487–493 (2011).
Chen, I. M. A. et al. The IMG/M data management and analysis system v.6.0: new tools and advanced capabilities. Nucleic Acids Res. 49, D751–D763 (2021).
Liang, R. et al. Metabolic capability of a predominant Halanaerobium sp. in hydraulically fractured gas wells and its implication in pipeline corrosion. Front. Microbiol. 7, 988 (2016).
Schnizlein, M. K., Vendrov, K. C., Edwards, S. J., Martens, E. C. & Young, V. B. Dietary xanthan gum alters antibiotic efficacy against the murine gut microbiota and attenuates Clostridioides difficile colonization. Msphere 5, e00708–19 (2020).
Katzbauer, B. Properties and applications of xanthan gum. Polym. Degrad. Stab. 59, 81–84 (1998).
Kozich, J. J., Westcott, S. L., Baxter, N. T., Highlander, S. K. & Schloss, P. D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79, 5112–5120 (2013).
Schloss, P. D. et al. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 75, 7537–7541 (2009).
Core R Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2020).
Wickham, H. Reshaping Data with the reshape Package. J. Stat. Softw. 21, 1–20 (2007).
Neuwirth, E. RColorBrewer: ColorBrewer Palettes (2014); https://cran.r-project.org/package=RColorBrewer
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Second edition). (Springer, 2016).
Martens, E. C. et al. Recognition and degradation of plant cell wall polysaccharides by two human gut symbionts. PLoS Biol. 9, e1001221 (2011).
Pope, P. B. et al. Isolation of Succinivibrionaceae implicated in low methane emissions from Tammar wallabies. Science 333, 646–648 (2011).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 17, 10–12 (2011).
Nurk, S., Meleshko, D., Korobeynikov, A. & Pevzner, P. A. MetaSPAdes: a new versatile metagenomic assembler. Genome Res. 27, 824–834 (2017).
Kang, D. D., Froula, J., Egan, R. & Wang, Z. MetaBAT, an efficient tool for accurately reconstructing single genomes from complex microbial communities. PeerJ 3, e1165 (2015).
Parks, D. H., Imelfort, M., Skennerton, C. T., Hugenholtz, P. & Tyson, G. W. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 25, 1043–1055 (2015).
Chen, I. M. A. et al. IMG/M: integrated genome and metagenome comparative data analysis system. Nucleic Acids Res. 45, D507–D516 (2017).
Lombard, V., Golaconda Ramulu, H., Drula, E., Coutinho, P. M. & Henrissat, B. The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res. 42, 490–495 (2014).
Rodriguez-R, L. M. et al. The Microbial Genomes Atlas (MiGA) webserver: taxonomic and gene diversity analysis of Archaea and Bacteria at the whole genome level. Nucleic Acids Res. 46, W282–W288 (2018).
Chaumeil, P. A., Mussig, A. J., Hugenholtz, P. & Parks, D. H. GTDB-Tk: a toolkit to classify genomes with the genome taxonomy database. Bioinformatics 36, 1925–1927 (2020).
Seemann, T. Prokka: rapid prokaryotic genome annotation. Bioinformatics 30, 2068–2069 (2014).
Koren, S. et al. Canu: scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res. 27, 722–736 (2017).
Li, H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics 34, 3094–3100 (2018).
Vaser, R., Sović, I., Nagarajan, N. & Šikić, M. Fast and accurate de novo genome assembly from long uncorrected reads. Genome Res. 27, 737–746 (2017).
Seppey, M., Manni, M. & Zdobnov, E. M. BUSCO: Assessing Genome Assembly and Annotation Completeness BT - Gene Prediction: Methods and Protocols (Humana Press, 2019).
Jain, C., Rodriguez-R, L. M., Phillippy, A. M., Konstantinidis, K. T. & Aluru, S. High throughput ANI analysis of 90K prokaryotic genomes reveals clear species boundaries. Nat. Commun. 9, 5114 (2018).
Kunath, B. J. et al. From proteins to polysaccharides: lifestyle and genetic evolution of Coprothermobacter proteolyticus. ISME J. 13, 603–617 (2019).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Kopylova, E., Noé, L. & Touzet, H. SortMeRNA: fast and accurate filtering of ribosomal RNAs in metatranscriptomic data. Bioinformatics 28, 3211–3217 (2012).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Massie, H. R. & Zimm, B. H. The use of hot phenol in preparing DNA. Proc. Natl Acad. Sci. USA 54, 1641–1643 (1965).
Nie, X. Relationships Between Dietary Fiber Structural Features and Growth and Utilization Patterns of Human Gut Bacteria. Doctoral dissertation, Purdue University (2016).
Tuncil, Y. E., Thakkar, R. D., Marcia, A. D. R., Hamaker, B. R. & Lindemann, S. R. Divergent short-chain fatty acid production and succession of colonic microbiota arise in fermentation of variously-sized wheat bran fractions. Sci. Rep. 8, 16655 (2018).
Arnal, G., Attia, M. A., Asohan, J. & Brumer, H. in Protein-Carbohydrate Interactions. Methods and Protocols (eds Abbott, D. W. & Lammerts van Bueren, A.) 209–214 (Springer, 2017).
Speer, M. A. Development of a Genetically Modified Silage Inoculant for the Biological Pretreatment of Lignocellulosic Biomass (Pennsylvania State Univ., 2013).
Anders, S. et al. Count-based differential expression analysis of RNA sequencing data using R and Bioconductor. Nat. Protoc. 8, 1765–1786 (2013).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Anders, S., Pyl, P. T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2009).
Thorvaldsdóttir, H., Robinson, J. T. & Mesirov, J. P. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief. Bioinform. 14, 178–192 (2013).
Wang, J. et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 490, 55–60 (2012).
Yu, J. et al. Metagenomic analysis of faecal microbiome as a tool towards targeted non-invasive biomarkers for colorectal cancer. Gut 66, 70–78 (2017).
Liu, R. et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat. Med. 23, 859–868 (2017).
Gu, Y. et al. Analyses of gut microbiota and plasma bile acids enable stratification of patients for antidiabetic treatment. Nat. Commun. 8, 1–12 (2017).
He, Q. et al. Two distinct metacommunities characterize the gut microbiota in Crohn’s disease patients. Gigascience 6, 1–11 (2017).
Zhang, X. et al. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 21, 895–905 (2015).
Nishijima, S. et al. The gut microbiome of healthy Japanese and its microbial and functional uniqueness. DNA Res. 23, 125–133 (2016).
Lloyd-Price, J. et al. Strains, functions and dynamics in the expanded Human Microbiome Project. Nature 550, 61–66 (2017).
Le Chatelier, E. et al. Richness of human gut microbiome correlates with metabolic markers. Nature 500, 541–546 (2013).
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Smits, S. A. et al. Seasonal cycling in the gut microbiome of the Hadza hunter-gatherers of Tanzania. Science 357, 802–805 (2017).
Conteville, L. C., Oliveira-Ferreira, J. & Vicente, A. C. P. Gut microbiome biomarkers and functional diversity within an Amazonian semi-nomadic hunter-gatherer group. Front. Microbiol. 10, 1743 (2019).
Boratyn, G. M., Thierry-Mieg, J., Thierry-Mieg, D., Busby, B. & Madden, T. L. Magic-BLAST, an accurate RNA-seq aligner for long and short reads. BMC Bioinformatics 20, 405 (2019).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Clum, A. et al. DOE JGI Metagenome Workflow. Msystems 6, e00804–20 (2021).
Schnizlein, M. K., Vendrov, K. C., Edwards, S. J., Martens, E. C. & Young, V. B. Dietary xanthan gum alters antibiotic efficacy against the murine gut microbiota and attenuates Clostridioides difficile colonization. mSphere 5, e00708–19 (2020).
Desai, M. S. et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167, 1339–1353.e21 (2016).
Larsbrink, J. et al. A discrete genetic locus confers xyloglucan metabolism in select human gut Bacteroidetes. Nature 506, 498–502 (2014).
Foley, M. H., Martens, E. C. & Koropatkin, N. M. SusE facilitates starch uptake independent of starch binding in B. thetaiotaomicron. Mol. Microbiol. 108, 551–566 (2018).
Luis, A. S. et al. A single sulfatase is required to access colonic mucin by a gut bacterium. Nature 598, 332–337 (2021).
Perez-Riverol, Y. et al. The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res. 50, D543–D552 (2022).
Acknowledgements
We thank S. Theide for growth curve analysis suggestions; T. Johannessen and A. Lysberg for help with Nanopore metagenomics; the University of Michigan Proteomics Resource Facility, Germfree Animal Core, Microbiome Core, and Natural Products Discovery Core for their support in completion of this project; the University of Michigan Center for Gastrointestinal Research (UMCGR, NIDDK 5P30DK034933) for financial support with proteomics. This work was supported by the US National Institutes of Health (DK118024, DK125445 to E.C.M. and UL1TR002240 to M.P.O. and AI124255 to V.B.Y.) and the Research Council of Norway (FRIPRO programme, P.B.P. and S.L.L.R.: 250479; L.H.H.: 302639). The work conducted by the US Department of Energy Joint Genome Institute, a DOE Office of Science User Facility, was supported under Contract No. DE-AC02-05CH11231.
Author information
Authors and Affiliations
Contributions
M.P.O., S.L.L.R., P.B.P. and E.C.M. designed experiments and wrote the manuscript. T.M.S. provided access to cohort faecal samples for initial enrichment cultures by D.B. and E.C.M. M.P.O. and E.C.M. ran all additional culture experiments. S.L.L.R., B.J.K., L.H.H. and P.B.P. carried out metagenomic and metatranscriptomic analysis on cultured microbes. T.Y. and B. Hamaker carried out neutral monosaccharide analysis. M.P.O., S.L.L.R. and G.F. carried out recombinant enzyme studies. M.P.O., A.R. and A.T. isolated pure tetrasaccharide and A.R. characterized it with NMR. J.L., S.P.M. and H.B. provided isomeric tetrasaccharide from P. nanensis GH9. M.P.O., L.Q., G.P. and N.A.P. carried out qPCR and RNA-seq. M.K.S. and V.B.Y. provided faecal samples from mice fed xanthan gum. N.T., V.L. and B. Henrissat carried out CAZyme annotation. N.J.V., G.P., S.L.L.R., M.P.O., P.B.P. and E.A.E.-F. carried out bioinformatic searches for loci of interest in metagenomes and genomes from global gut and environmental samples. M.P.O. and E.C.M. carried out mouse experiments. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
E.C.M., P.B.P., M.P.O., S.L.L.R. and B.J.K. are listed as inventors on a patent application (number PCT/US2021/050494) from the Norwegian University of Life Sciences and The Regents of the University of Michigan. Recombinant enzyme constructs for processing xanthan gum and oligosaccharides are covered in this patent application and discussed in this paper. E.C.M. serves as consultant and Scientific Advisory Board member of Novome Biotechnologies. All other authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks Harry Gilbert, Seth Rakoff-Nahoum and the other anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Dilution to extinction suggests that xanthan gum degradation is a multi-species phenotype.
An active xanthan culture was diluted in 2x defined media without a major carbon source, then divided and diluted 1:1 with either 2x xanthan gum or 2x monosaccharide mix (2:2:1 mannose;glucose;glucuronic acid), then aliquoted into 200 uL cultures in 96-well plates. Each datapoint represents the fraction of cultures (out of 96) growing above OD600 0.7 at each dilution, grown in either xanthan gum or monosaccharide mix media. Data were fit to the Hill equation to calculate a 50% growth dilution factor (GDF 50) at which half of the cultures would grow above OD 0.7. Across 5 independent experiments, there was an average GDF50 difference of 1.8 (standard deviation = 0.4, SEM = 0.2). This demonstrates that at comparable dilutions, microbes were present that could grow on monosaccharides but were unable to grow using XG, suggesting that several microbes are required in this media to allow growth on XG. In the schematic representation of 96-well plates, yellow wells represent non-growing media while gray wells represent a growing culture.
Extended Data Fig. 2 Sample 0 growth and metatranscriptomic analysis.
a, Two distinct replicates of the Sample 0 culture were grown and sampled at multiple timepoints for b, metatranscriptomic analysis of annotated CAZymes in each of the MAGs (completeness value > 75%) reconstructed from metagenomic data from the enrichment culture. Values are transcripts per million (TPM). MAG taxonomy (Supplementary Table 2) is indicated in parentheticals. An ‘#’ indicates a low AAI%.
Extended Data Fig. 3 Culture supernatant contains enzymes capable of depolymerizing xanthan gum, while intracellular contents are required for complete saccharification.
Representative thin layer chromatography of xanthan gum incubated with different fractions of an active xanthan gum culture (supernatant, washed cell pellet, lysed cell pellet, or lysed culture). Negative controls were prepared by heating fractions at 95 C for 15 minutes prior to initiating with xanthan gum. EDTA was added to a final concentration of ~50 mM to determine the necessity of divalent cations for enzyme activity. Strong color development in circles at baseline is undigested polysaccharide while bands that migrated with solvent are digested oligosaccharides and monosaccharides. Repeated experiments showed similar results.
Extended Data Fig. 4 Activity of R.UCG13 GH5 enzymes on various polysaccharides.
a, SDS-PAGE gel of purified GH5 constructs and their resultant activity as assessed by TLC on b, xanthan gum, b-c, carboxymethyl cellulose (CMC), c, hydroxyethyl cellulose (HEC), d, barley β-glucan, d-e, yeast β-glucan, e, tamarind xyloglucan, f, xylan, and f-g, wheat arabinoxylan. Enzymes are 1, RuGH5b (GH5 only); 2, RuGH5b (GH5 with UD-A); 3, RuGH5b (GH5 with UD-A/B); 4, RuGH5b (full protein); 5, RuGH5a (GH5 only); 6, RuGH5a (GH5 with UD-A); 7, RuGH5a (GH5 with UD-A/B); 8, RuGH5a (GH5 with UD-A/B/C); 9, RuGH5a (full protein); 10, replicate of 8. Strong color development in circles at baseline is undigested polysaccharide while bands or streaking that migrated with solvent are digested oligosaccharides and monosaccharides. Although minor streaking appears in some substrates due to residual oligosaccharides, comparing untreated substrate with enzyme incubated substrate allows determination of enzyme activity. RuGH5a constructs with all 3 UDs (8-10) showed clear activity on XG. Repeated experiments showed similar results.
Extended Data Fig. 5 Activity of R.UCG13 and B. intestinalis enzymes.
Enzyme activities were tested by incubating different combinations of enzymes with 2.5 mg/mL XGOs (generated by RuGH5a) at 37 °C for 16 hours. LC-MS analysis was used to track relative increases and decreases of intermediate oligosaccharides with the addition of enzymes, verifying their abilities to successively cleave XG pentasaccharides to their substituent monosaccharides. Integrated extracted ion counts (mean of n=4 distinct enzyme reactions and error bars displaying SEM; individual reactions shown as open circles) that correlate with compound abundance are shown for a, acetylated pentasaccharide (M-H ions: 883.26, 953.26, 925.27), b, deacetylated pentasaccharide (M-H ions: 841.25, 911.25), c, acetylated tetrasaccharide (2M-H ion: 1407.39), d, tetrasaccharide (M-H ion: 661.18), e, acetylated trisaccharide (M+Cl ion: 581.15), f, trisaccharide (M+Cl ion: 539.14), g, cellobiose (M+Cl ion: 377.09), and h, pyruvylated mannose (M-H ion: 249.06). Reactions were carried out using xanthan oligosaccharides produced by the RuGH5a to test activities of the R.UCG13 (R0-R8) and B. intestinalis (B0-B5) enzymes. R.UCG13 enzymes were tested in reactions that included (R0) no enzyme, (R1) R.UCG13 CE-A, (R2) R.UCG13 CE-B, (R3) R.UCG13 PL8, (R4) R.UCG13 PL8 and CE-A, (R5) R.UCG13 PL8 and CE-B, (R6) R.UCG13 PL8, both CEs, and GH88, (R7) R.UCG13 PL8, both CEs, GH88, and GH38-A, (R8) R.UCG13 PL8, both CEs, GH88, and GH38-B. B. intestinalis enzymes were tested in reactions that included (B0) no enzyme, (B1) Bi PL-only, (B2) Bi PL-CE, (B3) Bi PL-CE and Bacillus PL8, (B4) Bi PL-CE and GH88 and Bacillus PL8, (B5) Bi PL-CE, GH88, and GH92 and Bacillus PL8. i, Legend of enzymes included in each reaction. Recombinant enzymes were purified and analyzed for expression and purity by SDS-PAGE. Proteins generally expressed well with a single dense band indicating overexpression of the enzyme at its predicted molecular weight as compared to a size ladder. Exceptions included the R.UCG13 GH88 and CE-A, both of which had bands at the predicted enzyme size but also showed bands of comparable density at other sizes resulting from either proteolysis or co-purification of undesired E. coli proteins. j, SDS-PAGE gel of purified enzymes with 4.5 µg loaded, including (1-2) ladder, (3) B. intestinalis GH3, (4) B. intestinalis GH5, (5) B. intestinalis PL-only, (6) B. intestinalis PL-CE, (7) B. intestinalis GH88, (8) B. intestinalis GH92, (9) R. UCG 13 GH38-A, (10) R.UCG13 GH38-B, (11) R.UCG13 GH94, (12) R.UCG13 PL8, (13) R.UCG13 CE-A. Repeated purifications showed similar results. k, SDS-PAGE gel of purified enzymes with 4.5 µg loaded, including (1) ladder, (2) B. intestinalis PL-only, (3) B. intestinalis PL-CE, (4) B. intestinalis GH88, (5) B. intestinalis GH92, (6) R.UCG13 GH38-A, (7) R.UCG13 GH38-B, (8) R.UCG13 CE-A, (9) R.UCG13 GH88, (10) R.UCG13 CE-B, (11) R.UCG13 PL8. Repeated purifications showed similar results. l, TLC analysis showed that R.UCG13 GH94 and B. intestinalis GH3 are active on cellobiose. From left to right lane show (A) RuGH5b (full protein), (B) RuGH5a (full protein), (C) B. intestinalis GH3, (D) B. intestinalis GH5, (E) R.UCG13 GH94, (F) odd standards, (G) even standards, (H) cellobiose. Odd and even standards are maltooligosaccharides with 1, 3, 5, and 7 hexoses or 2, 4, and 6 hexoses, respectively. While the B. intestinalis GH3 only produced one product, the R.UCG13 GH94 produced two, one matching the approximate Rf of glucose while the other had a much lower Rf which presumably is phosphorylated glucose (matching the known phosphorylase activity of the GH94 family). Repeated experiments showed similar results.
Extended Data Fig. 6 Expression and growth analysis of isolates and cultures grown on various substrates.
a, Traces of RNA-seq expression data from n=3 biological replicates of the Sample 0 culture grown on either XG or polygalacturonic acid (PGA), illustrating overexpression of the XG PUL. b, Bacteroides clarus and c, Parabacteroides distasonis isolated from the Sample 0 culture did not grow on XGOs. d, Bacteroides intestinalis did not grow on tetramer generated with P. nanensis GH9 and PL8 (Psp Tetramer) even in the presence of 1 mg/mL RuGH5a generated XGOs to activate the PUL. Growth on glucose confirmed that the Psp Tetramer was not inherently toxic to cells. e, Bacteroides salyersiae grew on lyase treated XGOs but not on full XGOs and f, overexpressed a PUL homologous to the B. intestinalis PUL when grown on lyase treated XGOs. All substrates were used at 5 mg/mL unless otherwise noted. Growths are n ≥ 2 (for b and c, n=2 biological replicates on xanthan oligosaccharides and n=4 biological replicates on water and glucose; for d n=3 biological replicates, for e, n=6 biological replicates, for f n=3 biological replicates), error bars show SEM (in most cases, smaller than the marker). g, Traces of RNA-seq expression data from n=3 biological replicates of B. intestinalis grown on either glucose (Glc) or XG oligosaccharides (XGOs), illustrating overexpression of the XGO PUL.
Extended Data Fig. 7 B. intestinalis has a lyase that removes the terminal mannose from XGOs.
B. intestinalis was grown on XGOs then fractionated into supernatant, washed cell pellets (Pellet), and washed then lysed cells (Lysed), then incubated with fresh XGOs. Timepoints were taken at 30 minutes, 60 minutes, and overnight and analyzed for the formation of a, lyase-produced acetyl tetrasaccharide and b, tetrasaccharide. Integrated extracted ion counts (mean of n=3 distinct enzyme reactions and error bars displaying SEM; individual reactions shown as open circles) that correlate with compound abundance are shown. Relative to boiled controls, lysed cells and pelleted cells showed clear increases in lyase-produced tetrasaccharide.
Extended Data Fig. 8 Metagenomic and bioinformatic analysis of additional xanthan gum degrading loci.
a, Metagenomic sequencing of additional 16 cultures (S, human fecal sample) that actively grew on and degraded xanthan gum revealed two architectures of the R.UCG13. The more prevalent locus contained a GH125 insertion. The 10 additional samples with this locus architecture include: S22, S25, S39, S43, S44, S45, S49, S53, S58, and S59. b, The B. intestinalis xanthan locus was present in 3 additional cultures. c, Additional members of the Bacteroideceae family harbor a PUL with a GH88, GH92 and GH3 that could potentially enable utilization of XG-oligosaccharides. d, The GH125-containing version of the R.UCG13 xanthan locus was detected in two mouse fecal samples (M, mouse fecal sample). e, Comparison of the human and mouse RuGH5a aa sequence, showing the annotated signal peptide (SP), GH5 domain, three uncharacterized domains (UDs) with some homology to sugar-binding proteins, and multiple Listeria-Bacteroides repeat domains. f, Genetic organization and aa identity (%) between the B. intestinalis xanthan locus in the human sample and a PUL detected in a fracking water microbial community (FWMC) using LAST-searches. For a-f, the gray box delineates gene architecture and sequences derived from the human-derived Sample 0 culture. g, SDS-PAGE gel of purified enzymes with 4.5 µg loaded, including ladder and the different mouse RuGH5a constructs. A, B, and C are all versions of the GH5 domain alone, D is a construct designed to terminate at a site homologous to the last UD in the human RuGH5a, and E is a full-length construct of the mouse RuGH5a. Repeated purifications showed similar results. h, TLC of each mouse RuGH5a construct incubated with XG and also odd (1, 3, 5, and 7 residues) and even (2, 4, and 6 residues) malto-oligosaccharide standards. The GH5-only constructs did not degrade XG but constructs D and E (with regions homologous to the human RuGH5a UDs) were able to hydrolyze XG. Repeated experiments showed similar results.
Extended Data Fig. 9 Enzymatic model of xanthan degradation.
The RuGH5a enzyme initially depolymerizes XG into XGOs that are then saccharified by additional enzymes in R.UCG13 or B. intestinalis. Although the identified B. intestinalis PL-CE displayed acetylase functionality, we were unable to detect lyase activity with this construct. Lyase activity on XGOs was demonstrated with B. intestinalis pelleted or lysed cells suggesting that one or more unknown lyases can remove the terminal mannose from the pentasaccharide. Lyase activity generates a 4,5 unsaturated glucuronic acid (represented by a pentagon with an A), which is converted back to a standard glucuronic acid upon hydration and release by a GH88. The schematic uses standard nomenclature for glycans (blue circles, glucose; green circles, mannose; white and blue diamonds, glucuronic acid; Ac, acetylation; Py, pyruvylation) with enzymes represented as circles with interior labels.
Supplementary information
Supplementary Information
Supplementary Discussion and figures.
Supplementary Table 1
Relative abundance (%) of bacterial OTUs in human faecal cultures.
Supplementary Table 2
Metagenomic analyses of XG-degrading cultures including taxonomy, with genome and XG locus comparisons.
Supplementary Table 3
Neutral monosaccharide analysis of residual xanthan gum in Sample 0.
Supplementary Table 4
Proteomics hits from 3 activity-guided fractionation experiments with protein queries used to search each sample.
Supplementary Table 5
Hits from LAST search of IMG/M that passed threshold of 70% alignment over the query and 90% nucleotide identity.
Supplementary Table 6
Mouse sample metagenomic analyses and XG locus comparison to human R.UCG13 XG locus.
Supplementary Table 7
Relative abundance (%) of bacterial OTUs in humanized mouse faecal samples
Supplementary Table 8
Plasmids used for recombinant protein expression, and assembly method and primers used.
Supplementary Table 9
Primers used for qPCR of B. intestinalis and B. salyersiae XGO loci.
Source data
Source Data Fig. 1
Source data.
Source Data Fig. 3
Source data.
Source Data Fig. 4
Source data.
Source Data Extended Data Fig. 1
Source data.
Source Data Extended Data Fig. 3
Unprocessed TLC.
Source Data Extended Data Fig. 4
Unprocessed SDS–PAGE and TLCs.
Source Data Extended Data Fig. 5
Source data and unprocessed SDS–PAGE gels and TLC.
Source Data Extended Data Fig. 6
Source data.
Source Data Extended Data Fig. 7
Source data.
Source Data Extended Data Fig. 8
Unprocessed SDS–PAGE gel and TLC.
Rights and permissions
About this article
Cite this article
Ostrowski, M.P., La Rosa, S.L., Kunath, B.J. et al. Mechanistic insights into consumption of the food additive xanthan gum by the human gut microbiota. Nat Microbiol 7, 556–569 (2022). https://doi.org/10.1038/s41564-022-01093-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-022-01093-0
This article is cited by
-
Xanthan: enzymatic degradation and novel perspectives of applications
Applied Microbiology and Biotechnology (2024)
-
Production and application of xanthan gum—prospects in the dairy and plant-based milk food industry: a review
Food Science and Biotechnology (2024)
-
Sustainable plant-based ingredients as wheat flour substitutes in bread making
npj Science of Food (2022)