Abstract
Many pathogenic fungi depend on the development of specialized infection structures called appressoria to invade their hosts and cause disease. Impairing the function of fungal infection structures therefore provides a potential means by which diseases could be prevented. In spite of this extraordinary potential, however, relatively few anti-penetrant drugs have been developed to control fungal diseases, of either plants or animals. In the present study, we report the identification of compounds that act specifically to prevent fungal infection. We found that the organization of septin GTPases, which are essential for appressorium-mediated infection in the rice blast fungus Magnaporthe oryzae, requires very-long-chain fatty acids (VLCFAs), which act as mediators of septin organization at membrane interfaces. VLCFAs promote septin recruitment to curved plasma membranes and depletion of VLCFAs prevents septin assembly and host penetration by M. oryzae. We observed that VLCFA biosynthesis inhibitors not only prevent rice blast disease, but also show effective, broad-spectrum fungicidal activity against a wide range of fungal pathogens of maize, wheat and locusts, without affecting their respective hosts. Our findings reveal a mechanism underlying septin-mediated infection structure formation in fungi and provide a class of fungicides to control diverse diseases of plants and animals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data supporting the findings of the present study are available within this article, Extended Data and the Supplementary Information files. Source data are provided with this paper.
References
Fisher, M. C. et al. Emerging fungal threats to animal, plant and ecosystem health. Nature 484, 186–194 (2012).
Hawkins, N. J. & Fraaije, B. A. Fitness penalties in the evolution of fungicide resistance. Annu. Rev. Phytopathol. 56, 339–360 (2018).
Fisher, M. C., Hawkins, N. J., Sanglard, D. & Gurr, S. J. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 360, 739–742 (2018).
Théry, M. & Bornens, M. Cell shape and cell division. Curr. Opin. Cell Biol. 18, 648–657 (2006).
Keren, K. et al. Mechanism of shape determination in motile cells. Nature 453, 475–480 (2008).
Momany, M. & Talbot, N. J. Septins focus cellular growth for host infection by pathogenic fungi. Front. Cell Dev. Biol. 5, 33 (2017).
Roberts, R. E. & Hallett, M. B. Neutrophil cell shape change: mechanism and signalling during cell spreading and phagocytosis. Int. J. Mol. Sci. 20, 1383 (2019).
Luxenburg, C. & Zaidel-Bar, R. From cell shape to cell fate via the cytoskeleton—insights from the epidermis. Exp. Cell. Res. 378, 232–237 (2019).
Mostowy, S. & Cossart, P. Septins: the fourth component of the cytoskeleton. Nat. Rev. Mol. Cell Biol. 13, 183–194 (2012).
Oh, Y. & Bi, E. Septin structure and function in yeast and beyond. Trends Cell Biol. 21, 141–148 (2011).
Bridges, A. A. & Gladfelter, A. S. Fungal pathogens are platforms for discovering novel and conserved septin properties. Curr. Opin. Microbiol. 20, 42–48 (2014).
Angelis, D. & Spiliotis, E. T. Septin mutations in human cancers. Front. Cell Dev. Biol. 4, 122 (2016).
Bridges, A. A., Jentzsch, M. S., Oakes, P. W., Occhipinti, P. & Gladfelter, A. S. Micron-scale plasma membrane curvature is recognized by the septin cytoskeleton. J. Cell Biol. 213, 23–32 (2016).
Cannon, K. S., Woods, B. L., Crutchley, J. M. & Gladfelter, A. S. An amphipathic helix enables septins to sense micrometer-scale membrane curvature. J. Cell Biol. 218, 1128–1137 (2019).
Spiliotis, E. T. & Gladfelter, A. S. Spatial guidance of cell asymmetry: septin GTPases show the way. Traffic 13, 195–203 (2012).
Bertin, A. et al. Phosphatidylinositol-4,5-bisphosphate promotes budding yeast septin filament assembly and organization. J. Mol. Biol. 404, 711–731 (2010).
Kihara, A. Very long-chain fatty acids: elongation, physiology and related disorders. J. Biochem. 152, 387–395 (2012).
Bach, L. & Faure, J.-D. Role of very-long-chain fatty acids in plant development, when chain length does matter. C. R. Biol. 333, 361–370 (2010).
Shang, B. et al. Very-long-chain fatty acids restrict regeneration capacity by confining pericycle competence for callus formation in Arabidopsis. Proc. Natl Acad. Sci. USA 113, 5101–5106 (2016).
Schneiter, R. et al. Identification and biophysical characterization of a very-long-chain-fatty-acid-substituted phosphatidylinositol in yeast subcellular membranes. Biochem. J. 381, 941–949 (2004).
Rezanka, T., Kolouchova, I., Gharwalova, L., Palyzova, A. & Sigler, K. Identification and characterization of phospholipids with very long chain fatty acids in brewer’s yeast. Lipids 52, 1007–1017 (2017).
Ramos, A. P., Lagüe, P., Lamoureux, G. & Lafleur, M. Effect of saturated very long-chain fatty acids on the organization of lipid membranes: a study combining 2H NMR spectroscopy and molecular dynamics simulations. J. Phys. Chem. B 120, 6951–6960 (2016).
Obara, K., Kojima, R. & Kihara, A. Effects on vesicular transport pathways at the late endosome in cells with limited very long-chain fatty acids. J. Lipid Res. 54, 831–842 (2013).
Roudier, F. et al. Very-long-chain fatty acids are involved in polar auxin transport and developmental patterning in Arabidopsis. Plant Cell 22, 364–375 (2010).
Villasmil, M. L., Gallo-Ebert, C., Liu, H. Y., Francisco, J. & Nickels, J. T. Jr. A link between very long chain fatty acid elongation and mating-specific yeast cell cycle arrest. Cell Cycle 16, 2192–2203 (2017).
Koyuncu, E., Purdy, J. G., Rabinowitz, J. D. & Shenk, T. Saturated very long chain fatty acids are required for the production of infectious human cytomegalovirus progeny. PLoS Pathog. 9, e1003333 (2013).
Ramakrishnan, S. et al. The intracellular parasite Toxoplasma gondii depends on the synthesis of long-chain and very long-chain unsaturated fatty acids not supplied by the host cell. Mol. Microbiol. 97, 64–76 (2015).
Dagdas, Y. F. et al. Septin-mediated plant cell invasion by the rice blast fungus, Magnaporthe oryzae. Science 336, 1590–1595 (2012).
Wilson, R. A. & Talbot, N. J. Under pressure: investigating the biology of plant infection by Magnaporthe oryzae. Nat. Rev. Microbiol. 7, 185–195 (2009).
Li, L. et al. A phosphoinositide-specific phospholipase C pathway elicits stress-induced Ca2+ signals and confers salt tolerance to rice. N. Phytol. 214, 1172–1187 (2017).
Noack, L. C. & Jaillais, Y. Precision targeting by phosphoinositides: how PIs direct endomembrane trafficking in plants. Curr. Opin. Plant Biol. 40, 22–33 (2017).
Qin, L. et al. Specific recruitment of phosphoinositide species to the plant–pathogen interfacial membrane underlies Arabidopsis susceptibility to fungal infection. Plant Cell https://doi.org/10.1105/tpc.19.00970 (2020).
Casamayor, A. & Snyder, M. Molecular dissection of a yeast septin: distinct domains are required for septin interaction, localization, and function. Mol. Cell. Biol. 23, 2762–2777 (2003).
Walker, E. H. et al. Structural determinants of phosphoinositide 3-kinase inhibition by wortmannin, LY294002, quercetin, myricetin, and staurosporine. Mol. Cell 6, 909–919 (2000).
Wenk, M. R. et al. Phosphoinositide profiling in complex lipid mixtures using electrospray ionization mass spectrometry. Nat. Biotechnol. 21, 813–817 (2003).
D’Souza, K. & Epand, R. M. Enrichment of phosphatidylinositols with specific acyl chains. BBA-Biomembranes 1838, 1501–1508 (2014).
D’Souza, K. & Epand, R. M. The phosphatidylinositol synthase-catalyzed formation of phosphatidylinositol does not exhibit acyl chain specificity. Biochemistry 54, 1151–1153 (2015).
Jakobsson, A., Westerberg, R. & Jacobsson, A. Fatty acid elongases in mammals: their regulation and roles in metabolism. Prog. Lipid Res. 45, 237–249 (2006).
Sharma, S. et al. Sphingolipid biosynthetic pathway genes FEN1 and SUR4 modulate amphotericin B resistance. Antimicrob. Agents Chemother. 58, 2409–2414 (2014).
Busi, R. Resistance to herbicides inhibiting the biosynthesis of very-long-chain fatty acids. Pest Manag. Sci. 70, 1378–1384 (2014).
Khan, A., McQuilken, M. & Gladfelter, A. S. Septins and generation of asymmetries in fungal cells. Annu. Rev. Microbiol. 69, 487–503 (2015).
Leonard, A. E., Pereira, S. L., Sprecher, H. & Huang, Y. S. Elongation of long-chain fatty acids. Prog. Lipid Res. 43, 36–54 (2004).
Godwin, J., Norsworthy, J. K. & Scott, R. C. Selectivity of very-long-chain fatty acid-inhibiting herbicides in rice as influenced by application timing and soil texture. Crop Forage Turfgrass Manag. 4, 180016 (2018).
Sassa, T. & Kihara, A. Metabolism of very long-chain fatty acids: genes and pathophysiology. Biomol. Ther. 22, 83–92 (2014).
Talbot, N. J., Ebbole, D. J. & Hamer, J. E. Identification and characterization of MPG1, a gene involved in pathogenicity from the rice blast fungus Magnaporthe grisea. Plant Cell 5, 1575–1590 (1993).
Hou, Y.-P. et al. Baseline sensitivity of Bipolaris maydis to the novel succinate dehydrogenase inhibitor benzovindiflupyr and its efficacy. Pestic. Biochem. Physiol. 149, 81–88 (2018).
Chen, T. et al. Two members of TaRLK family confer powdery mildew resistance in common wheat. BMC Plant Biol. 16, 27 (2016).
Hu, J. & Xia, Y. Increased virulence in the locust-specific fungal pathogen Metarhizium acridum expressing dsRNAs targeting the host F1F0-ATPase subunit genes. Pest Manag. Sci. 75, 180–186 (2019).
He, M. et al. MoSnt2-dependent deacetylation of histone H3 mediates MoTor-dependent autophagy and plant infection by the rice blast fungus Magnaporthe oryzae. Autophagy 14, 1543–1561 (2018).
Li, W. et al. A natural allele of a transcription factor in rice confers broad-spectrum blast resistance. Cell 170, 114–126 (2017).
Lu, J., Cao, H., Zhang, L., Huang, P. & Lin, F. Systematic analysis of Zn2Cys6 transcription factors required for development and pathogenicity by high-throughput gene knockout in the rice blast fungus. PLoS Pathog. 10, e1004432 (2014).
Turgeon, B. G., Condon, B., Liu, J. & Zhang, N. in Molecular and Cell Biology Methods for Fungi Methods in Molecular Biology Vol. 638 (ed. Sharon, A.) 3–19 (Humana Press, 2010).
Hamilton, P. J. et al. PIP2 regulates psychostimulant behaviors through its interaction with a membrane protein. Nat. Chem. Biol. 10, 582–589 (2014).
Lam, S. M. et al. Lipidomic analysis of human tear fluid reveals structure-specific lipid alterations in dry eye syndrome. J. Lipid Res. 55, 299–306 (2014).
Clark, J. et al. Quantification of PtdInsP3 molecular species in cells and tissues by mass spectrometry. Nat. Methods 8, 267–272 (2011).
Wang, Z. et al. LecRK-V, an L-type lectin receptor kinase in Haynaldia villosa, plays positive role in resistance to wheat powdery mildew. Plant Biotechnol. J. 16, 50–62 (2018).
Acknowledgements
We thank H. Pan and X. Zhang from Jilin University for their generous help over B. maydis transformation. We also thank W. Zhang from Nanjing Agricultural University for providing the PHPLCδ1 and PHFAPP1 plasmids. This work was supported by grants from: the National Natural Science Foundation of China (NSFC) (grant nos. 31825022 and 31772153), National Key Research and Development Plan of China (grant no. 2016YFD0100600) and Tencent Exploration Award to X.W.C.; NSFC (grant no. 31871920), Open Funds of the State Key Laboratory of Crop Genetics and Germplasm Enhancement (grant no. ZW201803), International Cooperation and Exchange Program of Sichuan Science and Technology Department (grant no. 2020YFH0137) and Science Foundation of Education Department of Sichuan Province (grant no. 17ZA0313) to M.H.; NSFC (grant no. 31772152) to W.T.L.; NSFC (grant no. 31401351) to J.W.; NSFC (grant no. 31601290) to J.J.Y.; and NSFC (grant no. 31701779) to X.B.Z.. M.C. was supported by NSF (grant no. 1237975), NIH (grant no. GM59962) and USDA NIFA (grant no. 2017-67013-26590). Research in the laboratory of N.J.T. was supported by the Biotechnology and Biological Sciences Research Council Institute Strategic Grant in Plant Health (grant no. BB/N009959/1) and the Gatsby Charitable Foundation.
Author information
Authors and Affiliations
Contributions
M.H. and X.W.C. conceived and designed the study. M.H., J.S., Y.P.X., J.H.C., M.L.L., T.Q., K.K.Z., Y.Y.C., Y.L., W.W.L., J.W., Z.K.W., L.S.R., B.Z.T., S.M.L., G.X.P. and X.F.S. performed experiments with phenotypical and biochemical assays. M.O.R., I.E., X.Y., J.J.Y., X.B.Z., B.T.M., J.C.W., J.L.L., H.Q., L.S., L.W., Q.Q.H., P.Q., Y.L., J.F., D.Q.L., Y.P.W., X.W., L.J., G.S.G., F.H., G.H.S., Y.X.X., W.M.W., X.J.W., P.L., L.H.Z. and S.G.L. contributed to reagents, plant and fungal materials. M.H., J.S., Y.P.X., J.H.C., L.M.L., T.Q., Z.K.W. and X.W.C. collected data. M.H., M.S.C., N.J.T. and X.W.C. analysed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
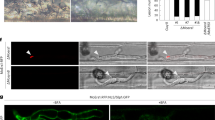
Extended Data Fig. 1 Sep3 associates with PIPs in M. oryzae.
a, Pulldown analysis on the interaction between Sep3 and PIPs. Total extracts of transformants of M. oryzae expressing Sep3-GFP or GFP only were immunoprecipitated by anti-PIP antibody and then subjected to immunoblot analysis by anti-GFP antibody. n.s. represents non-specific protein. The experiment was repeated twice with similar results. b, Role of PB domain for Sep3 binding PIPs determined by the protein lipid overlay assay. Bacterially expressed GST-Sep3 and GST-Sep36Q was respectively incubated with the commercial lipid strip. GST antibody was then used to detect proteins bound on the lipid strip. The experiment was repeated twice with similar results.
Extended Data Fig. 2 Generation of RNAi transformants for Knocking down the PI-3K encoding gene VPS34 of M. oryzae.
a, Schematic representation of VPS34 RNAi constructs pSilent-Vps34. Promoter of gene RP27 was used to drive the transcription of a self-complementary hairpin RNA targeting VPS34. IT represents the intron 2 of M. oryzae cutinase encoding gene (MGG_01943). b, RT-qPCR analysis on expression levels of VPS34 RNAi transformants. pSilent1 represents the empty RNAi vector transformant, whereas pSilent1-Vps34.1 and pSilent1-Vps34.2 represent two independent VPS34 RNAi transformants. Data from n = 3 biological replicates are depicted as bar plots showing the mean ± s.d. overlaid with the individual data points. Two-sided Student’s t-test was used to calculate statistical significance.
Extended Data Fig. 3 Phylogenetic analysis of Elo homologues and generation of targeted ELO1 gene deletion mutants.
a, Phylogenetic tree of Elo homologues. b, Transmembrane domain analysis of M. oryzae Elo1 and yeast (Sc) Elo proteins by the TMHMM software. c, ClustalOmega alignment of Elo homologues. d, Schematic representation of the targeted deletion of ELO1 in M. oryzae. e, PCR analysis of the targeted deletion in the Δelo1 mutants. Genomic DNA was analyzed by PCR with primer pairs indicated in panel (d). f, RT-PCR analysis of the Δelo1 mutants.
Extended Data Fig. 4 ELO1 encodes an ER-membrane associated protein required for VLCFAs biosynthesis.
a, Subcellular localization of Elo1-GFP on ER membrane as shown by co-localization with the ER maker fusion protein mRFP-HDEL. White dotted lines show scans used to generate corresponding GFP and mRFP fluorescence intensity distribution in the linescan graph. Scale bar, 10 μm. The experiment was repeated twice with similar results. b, RT-qPCR analysis on the expression profiles of ELO1 in Guy11 at different developmental processes (n = 3 biological replicates, mean ± s.d., two-sided Student’s t-test). c, Quantitation of fatty acids of different carbon lengths in M. oryzae. Fatty acids were extracted from Guy11 and a Δelo1 mutant, and subjected to UPLC-MS/MS analysis (n = 3 biologically independent samples, mean ± s.d., two-sided Student’s t-test). d, Quantitation of PIP by UPLC-MS/MS on M. oryzae (n = 3 biologically independent samples, mean ± s.d., two-sided Student’s t-test). e, Quantitation of PIP2 by UPLC-MS/MS on M. oryzae (n = 3 biologically independent samples, mean ± s.d., two-sided Student’s t-test).
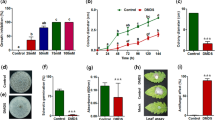
Extended Data Fig. 5 VLCFA biosynthesis is crucial for host penetration by M. oryzae.
a, Infection of detached rice leaves with spores in spotting inoculation assay. Mean length of blast lesions was calculated after five-days infection (n = 15 independent lesions, mean ± s.d., two-sided Student’s t-test). Fungal biomass in infected leaves was measured based on qPCR analysis of POT2 (n = 3 biological replicates, mean ± s.d., two-sided Student’s t-test). b, Infection structure development on onion epidermis at 24 hpi. Black arrows and arrowheads indicate conidium and appressorium respectively; white arrowheads indicate invasive hypha. Scale bar, 10 μm. Appressorium and penetration peg formation were quantitated from at least 100 spores (n = 3 biologically independent samples, mean ± s.d., two-sided Student’s t-test). c, Infection structure development on rice leaf sheath at 24 hpi. Scale bar, 10 μm. The experiment was repeated three times with similar results. d, Detached leaves infected with Guy11 in spotting inoculation assay with 500 μM VLCFAs biosynthesis inhibitors. The solvent DMSO was used as a negative control. Length of blast lesion was calculated five days after infection (n = 15 independent lesions, mean ± s.d., two-sided Student’s t-test). e, Rice leaves infected with Guy11 in spore spraying inoculation in the presence of 500 μM inhibitor. Lesion density was calculated from infected leaf segments of 5 cm in length (n = 21 independent leaves for DMSO, n = 28 for metazachlor, n = 37 for cafenstrole, n = 33 for diallate; mean ± s.d., two-sided Student’s t-test). f, TEM analysis on the penetration of onion epidermis by M. oryzae in the presence or absence of an inhibitor at 24 hpi. Scale bar, 2 μm. The experiment was repeated twice with similar results.
Extended Data Fig. 6 Effects on inhibition of M. oryzae pathogenicity are proportional to the concentrations of the VLCFA biosynthesis inhibitors, and combination of VLCFAs inhibitors improves efficacy of carbendazim in reducing pathogenicity.
Three VLCFA biosynthesis inhibitors and DMSO control were individually applied in a field trial. Rice leaves were sprayed with Guy11 spore suspension supplemented with an inhibitor at concentrations of 0, 20, 50, 100, 200, 500, 700 and 1000 μΜ, respectively. Blast lesion density was quantitated five-days post infection. a, Metazachlor (n = 27 independent leaves for 0 μΜ, n = 29 for 20 μΜ, n = 29 for 50 μΜ, n = 30 for 100 μΜ, n = 29 for 200 μΜ, n = 29 for 500 μΜ, n = 30 for 700 μΜ, n = 31 for 1000 μΜ; mean ± s.d.). b, Cafenstrole (n = 29 independent leaves for 0 μΜ, n = 29 for 20 μΜ, n = 28 for 50 μΜ, n = 29 for 100 μΜ, n = 29 for 200 μΜ, n = 28 for 500 μΜ, n = 29 for 700 μΜ, n = 29 for 1000 μΜ; mean ± s.d.). c, Diallate (n = 32 independent leaves for 0 μΜ, n = 30 for 20 μΜ, n = 29 for 50 μΜ, n = 30 for 100 μΜ, n = 31 for 200 μΜ, n = 31 for 500 μΜ, n = 31 for 700 μΜ, n = 30 for 1000 μΜ; mean ± s.d.). d, Effects of combination of three VLCFAs inhibitors on pathogenicity of M. oryzae. The three inhibitors were combined in equal amount to a final concentration of 500 μM in Guy11 spore suspension. Then the spore suspension containing combined inhibitors or the three separate inhibitors was inoculated onto rice leaves. DMSO was used as solvent control. Blast lesion density was quantitated after five days of infection (n = 30 independent leaves for DMSO, n = 30 for metazachlor, n = 31 for cafenstrole, n = 30 for diallate, n = 32 for combination of three inhibitors; mean ± s.d.). e, Effects of combination of each VLCFAs inhibitor with carbendazim on pathogenicity of M. oryzae. The three inhibitors were separately combined with carbendazim to produce Guy11 spore suspension which contains 500 μM VLCFAs inhibitor and 8 mM carbendazim. The spore suspension was inoculated onto rice leaves. Carbendazim, metazachlor, cafenstrole and diallate is respectively abbreviated as Car, Met, Caf and Dia. Blast lesion density was quantitated after five-days of infection (n = 31 independent leaves for DMSO, n = 32 for carbendazim, n = 31 for metazachlor, n = 31 for cafenstrole, n = 30 for diallate, n = 29 for combination of carbendazim with metazachlor, n = 31 for combination of carbendazim with cafenstrole, n = 32 for combination of carbendazim with diallate; mean ± s.d.).
Extended Data Fig. 7 Analysis of septin ring formed by Sep4-GFP and Sep5-GFP in M. oryzae.
Microscopic analysis and quantitation of septin ring formation at 24 hpi in the presence or absence of 500 μM inhibitor. DMSO was used as solvent control. Percentage of septin ring formation was calculated from 100 spores at 24 hpi (n = 3 biologically independent samples, mean ± s.d., two-sided Student’s t-test). Scale bar, 10 μm.
Extended Data Fig. 8 Inhibitors of VLCFA biosynthesis have no obvious effects on autophagic progression and conidial cell death during appressorium development of M. oryzae.
a, Microscopic images and quantitation of autophagosomes during appressorium development. Spore suspension of Guy11 expressing GFP-Atg8 was allowed to undergo appressorium development on hydrophobic surface in exposure to each of the three inhibitors at a concentration 500 μΜ or the solvent control DMSO. Mean autophagosome numbers was quantitated in conidium, germ tube and appressorium (n = 20 independent spores, mean ± s.d.). Scale bar, 10 μm. b, Autophagic conidial cell death of Guy11 at 24 hpi of appressorium development in the presence of inhibitors or solvent DMSO. Autophagic clearance of GFP in the conidial three cells of spores was quantitated from 100 spores (n = 3 independent samples, mean ± s.d.). Scale bar, 10 μm.
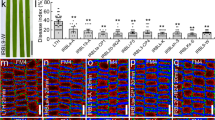
Extended Data Fig. 9 Inhibitors of VLCFAs biosynthesis impaired septin organization and septin-dependent pathogenicity of B. maydis.
a, Microscopic analysis and quantitation of septin puncta detected by BmSep3-GFP at hyphal tips infecting maize leaf at 24 hpi. B. maydis spore suspension was supplemented with 500 μM of each VLCFAs inhibitor and then used for inoculation of maize leaf. DMSO was the solvent control. Percentage of septin dot formation was calculated from 50 hypal tip at 24 hpi (n = 3 independent samples, mean ± s.d., two-sided Student’s t-test). Scale bar, 10 μm. b, Microscopic analysis and quantitation of septin dot detected by BmSep4-GFP from 50 hyphal tips infecting maize leaf at 24 hpi (n = 3 independent samples, mean ± s.d., two-sided Student’s t-test). Scale bar, 10 μm. c, Schematic representation of the targeted deletion of BmSEP3 and BmSEP4 in B. maydis. d, PCR analysis of the targeted deletion in the ΔBmsep3 and ΔBmsep4 mutants. Genomic DNA was analyzed by PCR with primer pairs indicated in panel C. e, Maize leaf infected with B. maydis by a spore suspension spraying assay (n = 7 independent leaves, mean ± s.d., two-sided Student’s t-test).
Extended Data Fig. 10 Inhibitors of VLCFA biosynthesis have no observable effects on host growth or health.
a, Seedlings of rice after seven days of spray with 500 μM inhibitor. b, Yields of rice plants sprayed with inhibitor. Plants were sprayed with 500 μM inhibitor for the first time at the heading stage, and sprayed for the second time at the booting stage. Agronomic traits including seed setting rate, 1000-seed-weight and grain weight per plant were collected from reprehensive plants (n = 6 independent plants, mean ± s.d.). c, Wheat seedlings after seven days of spray with 500 μM inhibitor. d, Maize after seven days of spray with 500 μM inhibitor. e, Locust after seven days of spray with 500 μM inhibitor. f, Quantitative analysis on daily food consumption and feces production behavior of locust after inoculation with 500 μM inhibitor (n = 3 biological replicates, mean ± s.d.).
Supplementary information
Supplementary Table 1
Supplementary Tables 1 and 2.
Supplementary Video 1
Three-dimensional reconstruction of co-localization between Sep3–GFP and a pool of PHFAPP1/PHPLCδ1–mRFP in Guy11 appressorium.
Supplementary Video 2
Three-dimensional reconstruction of the septin ring at the base of appressorium in Guy11 and Δelo1.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 1
Unprocessed western blots.
Source Data Extended Data Fig. 3
Unprocessed gels.
Source Data Extended Data Fig. 9
Unprocessed gels.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Source Data Extended Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 7
Statistical source data.
Source Data Extended Data Fig. 8
Statistical source data.
Source Data Extended Data Fig. 9
Statistical source data.
Source Data Extended Data Fig. 10
Statistical source data.
Rights and permissions
About this article
Cite this article
He, M., Su, J., Xu, Y. et al. Discovery of broad-spectrum fungicides that block septin-dependent infection processes of pathogenic fungi. Nat Microbiol 5, 1565–1575 (2020). https://doi.org/10.1038/s41564-020-00790-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-020-00790-y
This article is cited by
-
Early molecular events in the interaction between Magnaporthe oryzae and rice
Phytopathology Research (2024)
-
Targeting Magnaporthe oryzae effector MoErs1 and host papain-like protease OsRD21 interaction to combat rice blast
Nature Plants (2024)
-
Fimbrin associated with Pmk1 to regulate the actin assembly during Magnaporthe oryzae hyphal growth and infection
Stress Biology (2024)
-
Septin-dependent invasive growth by the rice blast fungus Magnaporthe oryzae
Journal of Plant Diseases and Protection (2024)
-
A molecular mechanosensor for real-time visualization of appressorium membrane tension in Magnaporthe oryzae
Nature Microbiology (2023)