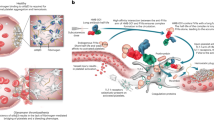
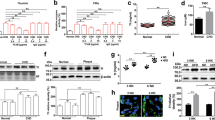
Abstract
Severe infectious diseases are often characterized by an overwhelming and unbalanced systemic immune response to microbial infections. Human antithrombin (hAT) is a crucial coagulation inhibitor with anti-inflammatory activities. Here we identify three hAT-binding proteins (CD13, CD300f and LRP-1) on human monocytes that are involved in blocking the activity of nuclear factor-κB. We found that the modulating effect is primarily restricted to the less abundant β-isoform (hβAT) of hAT that lacks N-glycosylation at position 135. Individuals with a mutation at this position have increased production of hβAT and analysis of their blood, which was stimulated ex vivo with lipopolysaccharide, showed a decreased inflammatory response. Similar findings were recorded when heterozygotic mice expressing hAT or hβAT were challenged with lipopolysaccharide or infected with Escherichia coli bacteria. Our results finally demonstrate that in a lethal E. coli infection model, survival rates increased when mice were treated with hβAT one hour and five hours after infection. The treatment also resulted in a reduction of the inflammatory response and less severe organ damage.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author on request.
References
van der Poll, T., van de Veerdonk, F. L., Scicluna, B. P. & Netea, M. G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 17, 407–420 (2017).
Chaudhry, H. et al. Role of cytokines as a double-edged sword in sepsis. In Vivo 27, 669–684 (2013).
Denstaedt, S. J., Singer, B. H. & Standiford, T. J. Sepsis and nosocomial infection: patient characteristics, mechanisms, and modulation. Front. Immunol. 9, 2446 (2018).
Picard, V., Ersdal-Badju, E. & Bock, S. C. Partial glycosylation of antithrombin III asparagine-135 is caused by the serine in the third position of its N-glycosylation consensus sequence and is responsible for production of the beta-antithrombin III isoform with enhanced heparin affinity. Biochemistry 34, 8433–8440 (1995).
Andersen, O., Flengsrud, R., Norberg, K. & Salte, R. Salmon antithrombin has only three carbohydrate side chains, and shows functional similarities to human beta-antithrombin. Eur. J. Biochem. 267, 1651–1657 (2000).
Allen, D. H. & Tracy, P. B. Human coagulation factor V is activated to the functional cofactor by elastase and cathepsin G expressed at the monocyte surface. J. Biol. Chem. 270, 1408–1415 (1995).
Ghosh, M., Subramani, J., Rahman, M. M. & Shapiro, L. H. CD13 restricts TLR4 endocytic signal transduction in inflammation. J. Immunol. 194, 4466–4476 (2015).
Lee, S. M., Kim, E. J., Suk, K. & Lee, W. H. CD300F blocks both MyD88 and TRIF-mediated TLR signaling through activation of Src homology region 2 domain-containing phosphatase 1. J. Immunol. 186, 6296–6303 (2011).
Gaultier, A. et al. Regulation of tumor necrosis factor receptor-1 and the IKK-NF-kappaB pathway by LDL receptor-related protein explains the antiinflammatory activity of this receptor. Blood 111, 5316–5325 (2008).
Zhang, J., Somani, A. K. & Siminovitch, K. A. Roles of the SHP-1 tyrosine phosphatase in the negative regulation of cell signalling. Semin. Immunol. 12, 361–378 (2000).
Kim, E. J., Lee, S. M., Suk, K. & Lee, W. H. CD300a and CD300f differentially regulate the MyD88 and TRIF-mediated TLR signalling pathways through activation of SHP-1 and/or SHP-2 in human monocytic cell lines. Immunology 135, 226–235 (2012).
Strickland, D. K., Muratoglu, S. C. & Antalis, T. M. Serpin-enzyme receptors LDL receptor-related protein 1. Methods Enzym. 499, 17–31 (2011).
Toldo, S. et al. Low-density lipoprotein receptor-related protein-1 is a therapeutic target in acute myocardial infarction. JACC Basic Transl. Sci. 2, 561–574 (2017).
Janciauskiene, S., Lindgren, S. & Wright, H. T. The C-terminal peptide of alpha-1-antitrypsin increases low density lipoprotein binding in HepG2 cells. Eur. J. Biochem. 254, 460–467 (1998).
Zurhove, K., Nakajima, C., Herz, J., Bock, H. H. & May, P. Gamma-secretase limits the inflammatory response through the processing of LRP1. Sci. Signal. 1, ra15 (2008).
Pasi, K. J. et al. Targeting of antithrombin in hemophilia A or B with RNAi therapy. N. Engl. J. Med. 377, 819–828 (2017).
Corral, J., de la Morena-Barrio, M. E. & Vicente, V. The genetics of antithrombin. Thromb. Res. 169, 23–29 (2018).
Luxembourg, B. et al. Molecular basis of antithrombin deficiency. Thromb. Haemost. 105, 635–646 (2011).
Bayston, T. A. et al. Familial overexpression of beta antithrombin caused by an Asn135Thr substitution. Blood 93, 4242–4247 (1999).
Kalle, M. et al. A peptide of heparin cofactor II inhibits endotoxin-mediated shock and invasive Pseudomonas aeruginosa infection. PLoS ONE 9, e102577 (2014).
Delano, M. J. & Ward, P. A. The immune system’s role in sepsis progression, resolution, and long-term outcome. Immunol. Rev. 274, 330–353 (2016).
Yan, S. B., Helterbrand, J. D., Hartman, D. L., Wright, T. J. & Bernard, G. R. Low levels of protein C are associated with poor outcome in severe sepsis. Chest 120, 915–922 (2001).
Van Den Boogaard, F. E. et al. Recombinant human tissue factor pathway inhibitor exerts anticoagulant, anti-inflammatory and antimicrobial effects in murine pneumococcal pneumonia. J. Thromb. Haemost. 9, 122–132 (2011).
Levy, J. H., Sniecinski, R. M., Welsby, I. J. & Levi, M. Antithrombin: anti-inflammatory properties and clinical applications. Thromb. Haemost. 115, 712–728 (2016).
Shorr, A. F. et al. Protein C concentrations in severe sepsis: an early directional change in plasma levels predicts outcome. Crit. Care 10, R92 (2006).
Gando, S. et al. Tissue factor production not balanced by tissue factor pathway inhibitor in sepsis promotes poor prognosis. Crit. Care Med. 30, 1729–1734 (2002).
Wiedermann, C. J. et al. High-dose antithrombin III in the treatment of severe sepsis in patients with a high risk of death: efficacy and safety. Crit. Care Med. 34, 285–292 (2006).
Poole, D., Bertolini, G. & Garattini, S. Withdrawal of ‘Xigris’ from the market: old and new lessons. J. Epidemiol. Community Health 66, 571–572 (2012).
Laterre, P. F. et al. A clinical evaluation committee assessment of recombinant human tissue factor pathway inhibitor (tifacogin) in patients with severe community-acquired pneumonia. Crit. Care 13, R36 (2009).
Allingstrup, M., Wetterslev, J., Ravn, F. B., Moller, A. M. & Afshari, A. Antithrombin III for critically ill patients. Cochrane Database Syst. Rev. 2, CD005370 (2016).
Kerschen, E. J. et al. Endotoxemia and sepsis mortality reduction by non-anticoagulant activated protein C. J. Exp. Med. 204, 2439–2448 (2007).
Minnema, M. C. et al. Recombinant human antithrombin III improves survival and attenuates inflammatory responses in baboons lethally challenged with Escherichia coli. Blood 95, 1117–1123 (2000).
Rello, J. et al. Towards precision medicine in sepsis: a position paper from the European Society of Clinical Microbiology and Infectious Diseases. Clin. Microbiol. Infect. 24, 1264–1272 (2018).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
de la Morena-Barrio, M. E. et al. A new method to quantify beta-antithrombin glycoform in plasma reveals increased levels during the acute stroke event. Thromb. Res. 136, 634–641 (2015).
Abdillahi, S. M. et al. Collagen VI is upregulated in COPD and serves both as an adhesive target and a bactericidal barrier for Moraxella catarrhalis. J. Innate Immun. 7, 506–517 (2015).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 490–505 (2015).
Yang, H., Wang, H. & Jaenisch, R. Generating genetically modified mice using CRISPR/Cas-mediated genome engineering. Nat. Protoc. 9, 1956–1968 (2014).
Martin Gonzalez, J. et al. Embryonic stem cell culture conditions support distinct states associated with different developmental stages and potency. Stem Cell Rep. 7, 177–191 (2016).
Linder, A., Christensson, B., Herwald, H., Bjorck, L. & Akesson, P. Heparin-binding protein: an early marker of circulatory failure in sepsis. Clin. Infect. Dis. 49, 1044–1050 (2009).
Acknowledgements
This work was supported in part by the Alfred Österlund Foundation (to P.P. and H.H.), the Crafoord Foundation (grant no. 20180506 to P.P.), the Knut and Alice Wallenberg Foundation (grant no. 2011.0037 to H.H.), the Medical Faculty at Lund University (to H.H.), the Swedish Foundation for Strategic Research (grant no. SB12-0019 to A.E. and H.H.), the Swedish Research Council (grant no. 2013-3078 to A.E. and grant no. 2016-01104 to H.H.). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. The authors thank R. Bhongir for performing the microarray assays, M. Baumgarten for her help with the electron microscopy experiments, J. Martin for his support in the generation of mice expressing hAT and hβAT and Lund University Bioimaging Centre.
Author information
Authors and Affiliations
Contributions
P.P., M.R., F.D.H., C.Naudin., E.S., J.W. and G.Kassety. performed the in vitro and in vivo experiments. S.V. generated heat maps. A.A., M.A., C.Novembrino. and I.M. assisted with experiments using blood from hβAT-overexpressing individuals. A.E., I.M.-M., M.E.d.l.M.-B. and J.C. analysed and interpreted the data. C.H.B. designed CRISPR−Cas experiments. A.L. provided patient plasma samples. P.P. and H.H. designed and supervised the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
P.P., A.E., G.K. and H.H. have filed a provisional patent on the possibility of using hβAT as an antimicrobial treatment. All other authors have no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Figs. 1−14 and Tables 1−2.
Rights and permissions
About this article
Cite this article
Papareddy, P., Rossnagel, M., Doreen Hollwedel, F. et al. A human antithrombin isoform dampens inflammatory responses and protects from organ damage during bacterial infection. Nat Microbiol 4, 2442–2455 (2019). https://doi.org/10.1038/s41564-019-0559-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-019-0559-6
This article is cited by
-
FVIII regulates the molecular profile of endothelial cells: functional impact on the blood barrier and macrophage behavior
Cellular and Molecular Life Sciences (2022)