Abstract
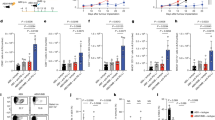
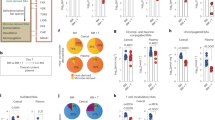
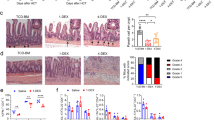
Host NOD-like receptor family pyrin domain-containing 6 (NLRP6) regulates innate immune responses and gastrointestinal homeostasis. Its protective role in intestinal colitis and tumorigenesis is dependent on the host microbiome. Host innate immunity and microbial diversity also play a role in the severity of allogeneic immune-mediated gastrointestinal graft-versus-host disease (GVHD), the principal toxicity after allogeneic haematopoietic cell transplantation. Here, we examined the role of host NLRP6 in multiple murine models of allogeneic bone marrow transplantation. In contrast to its role in intestinal colitis, host NLRP6 aggravated gastrointestinal GVHD. The impact of host NLRP6 deficiency in mitigating GVHD was observed regardless of co-housing, antibiotic treatment or colonizing littermate germ-free wild-type and NLRP6-deficient hosts with faecal microbial transplantation from specific pathogen-free wild-type and Nlrp6−/− animals. Chimaera studies were performed to assess the role of NLRP6 expression on host haematopoietic and non-haematopoietic cells. The allogeneic [B6Ly5.2 → Nlrp6−/−] animals demonstrated significantly improved survival compared to the allogeneic [B6Ly5.2 → B6] animals, but did not alter the therapeutic graft-versus-tumour effects after haematopoietic cell transplantation. Our results unveil an unexpected, pathogenic role for host NLRP6 in gastrointestinal GVHD that is independent of variations in the intestinal microbiome and in contrast to its well-appreciated microbiome-dependent protective role in intestinal colitis and tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The raw sequencing reads have been deposited at the NCBI Short Read Archive under BioProject ID PRJNA491725. Metabolomic data, mass spectral analytical parameters and spectral raw data from the study and metadata have been deposited in the NIH Common Fund’s Data Repository and Coordinating Center under Metabolomics Workbench Project ID PR000728.
References
Staffas, A., Burgos da Silva, M. & van den Brink, M. R. The intestinal microbiota in allogeneic hematopoietic cell transplant and graft-versus-host disease. Blood 129, 927–933 (2017).
Zeiser, R. & Blazar, B. R. Acute graft-versus-host disease—biologic process, prevention, and therapy. N. Engl. J. Med. 377, 2167–2179 (2017).
Elinav, E. et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145, 745–757 (2011).
Levy, M., Shapiro, H., Thaiss, C. A. & Elinav, E. NLRP6: a multifaceted innate immune sensor. Trends Immunol. 38, 248–260 (2017).
Hu, B. et al. Microbiota-induced activation of epithelial IL-6 signaling links inflammasome-driven inflammation with transmissible cancer. Proc. Natl Acad. Sci. USA 110, 9862–9867 (2013).
Wlodarska, M. et al. NLRP6 inflammasome orchestrates the colonic host–microbial interface by regulating goblet cell mucus secretion. Cell 156, 1045–1059 (2014).
Anand, P. K. et al. NLRP6 negatively regulates innate immunity and host defence against bacterial pathogens. Nature 488, 389–393 (2012).
Ferrara, J. L., Levine, J. E., Reddy, P. & Holler, E. Graft-versus-host disease. Lancet 373, 1550–1561 (2009).
Mathewson, N. D. et al. Gut microbiome-derived metabolites modulate intestinal epithelial cell damage and mitigate graft-versus-host disease. Nat. Immunol. 17, 505–513 (2016).
Jenq, R. R. et al. Regulation of intestinal inflammation by microbiota following allogeneic bone marrow transplantation. J. Exp. Med. 209, 903–911 (2012).
Chen, G. Y., Liu, M., Wang, F., Bertin, J. & Nunez, G. A functional role for Nlrp6 in intestinal inflammation and tumorigenesis. J. Immunol. 186, 7187–7194 (2011).
Normand, S. et al. Nod-like receptor pyrin domain-containing protein 6 (NLRP6) controls epithelial self-renewal and colorectal carcinogenesis upon injury. Proc. Natl Acad. Sci. USA 108, 9601–9606 (2011).
Jankovic, D. et al. The Nlrp3 inflammasome regulates acute graft-versus-host disease. J. Exp. Med. 210, 1899–1910 (2013).
Chen, S. et al. MicroRNA-155-deficient dendritic cells cause less severe GVHD through reduced migration and defective inflammasome activation. Blood 126, 103–112 (2015).
Koehn, B. H. et al. GVHD-associated, inflammasome-mediated loss of function in adoptively transferred myeloid-derived suppressor cells. Blood 126, 1621–1628 (2015).
Mamantopoulos, M. et al. Nlrp6- and ASC-dependent inflammasomes do not shape the commensal gut microbiota composition. Immunity 47, 339–348 (2017).
Seregin, S. S. et al. NLRP6 protects Il10 −/− mice from colitis by limiting colonization of Akkermansia muciniphila. Cell Rep. 19, 733–745 (2017).
Shono, Y. et al. Increased GVHD-related mortality with broad-spectrum antibiotic use after allogeneic hematopoietic stem cell transplantation in human patients and mice. Sci. Transl Med. 8, 339ra371 (2016).
Shan, M. et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science 342, 447–453 (2013).
Adolph, T. E. et al. Paneth cells as a site of origin for intestinal inflammation. Nature 503, 272–276 (2013).
Thiagarajah, J. R., Zhao, D. & Verkman, A. S. Impaired enterocyte proliferation in aquaporin-3 deficiency in mouse models of colitis. Gut 56, 1529–1535 (2007).
Linden, S. K. et al. MUC1 limits Helicobacter pylori infection both by steric hindrance and by acting as a releasable decoy. PLoS Pathog. 5, e1000617 (2009).
Johansson, M. E. et al. Bacteria penetrate the inner mucus layer before inflammation in the dextran sulfate colitis model. PLoS ONE 5, e12238 (2010).
Wang, F. et al. Isolation and characterization of intestinal stem cells based on surface marker combinations and colony-formation assay. Gastroenterology 145, 383–395 (2013).
Lindemans, C. A. et al. Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature 528, 560–564 (2015).
Wu, S. R. & Reddy, P. Tissue tolerance: a distinct concept to control acute GVHD severity. Blood 129, 1747–1752 (2017).
Wu, S. R. & Reddy, P. Regulating damage from sterile inflammation: a tale of two tolerances. Trends Immunol. 38, 231–235 (2017).
Eriguchi, Y. et al. Graft-versus-host disease disrupts intestinal microbial ecology by inhibiting Paneth cell production of α-defensins. Blood 120, 223–231 (2012).
Schwab, L. et al. Neutrophil granulocytes recruited upon translocation of intestinal bacteria enhance graft-versus-host disease via tissue damage. Nat. Med. 20, 648–654 (2014).
Heimesaat, M. M. et al. MyD88/TLR9 mediated immunopathology and gut microbiota dynamics in a novel murine model of intestinal graft-versus-host disease. Gut 59, 1079–1087 (2010).
Gill, N., Wlodarska, M. & Finlay, B. B. Roadblocks in the gut: barriers to enteric infection. Cell. Microbiol. 13, 660–669 (2011).
Reddy, P. et al. A crucial role for antigen-presenting cells and alloantigen expression in graft-versus-leukemia responses. Nat. Med. 11, 1244–1249 (2005).
Toubai, T. et al. Host-derived CD8+ dendritic cells are required for induction of optimal graft-versus-tumor responses after experimental allogeneic bone marrow transplantation. Blood 121, 4231–4241 (2013).
Reddy, P. et al. Histone deacetylase inhibition modulates indoleamine 2,3-dioxygenase-dependent DC functions and regulates experimental graft-versus-host disease in mice. J. Clin. Invest. 118, 2562–2573 (2008).
Levy, M. et al. Microbiota-modulated metabolites shape the intestinal microenvironment by regulating NLRP6 inflammasome signaling. Cell 163, 1428–1443 (2015).
Cooke, K. R. et al. Tumor necrosis factor-α production to lipopolysaccharide stimulation by donor cells predicts the severity of experimental acute graft-versus-host disease. J. Clin. Invest. 102, 1882–1891 (1998).
Hill, G. R. et al. Interleukin-11 promotes T cell polarization and prevents acute graft-versus-host disease after allogeneic bone marrow transplantation. J. Clin. Invest. 102, 115–123 (1998).
Toubai, T. et al. Ikaros–Notch axis in host hematopoietic cells regulates experimental graft-versus-host disease. Blood 118, 192–204 (2011).
Toubai, T. et al. Induction of acute GVHD by sex-mismatched H-Y antigens in the absence of functional radiosensitive host hematopoietic-derived antigen-presenting cells. Blood 119, 3844–3853 (2012).
Kozich, J. J., Westcott, S. L., Baxter, N. T., Highlander, S. K. & Schloss, P. D. Development of a dual-index sequencing strategy and curation pipeline for analyzing amplicon sequence data on the MiSeq Illumina sequencing platform. Appl. Environ. Microbiol. 79, 5112–5120 (2013).
Schloss, P. D. et al. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 75, 7537–7541 (2009).
Schloss, P. D. A high-throughput DNA sequence aligner for microbial ecology studies. PLoS ONE 4, e8230 (2009).
Pruesse, E. et al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 35, 7188–7196 (2007).
Schloss, P. D. & Westcott, S. L. Assessing and improving methods used in operational taxonomic unit-based approaches for 16S rRNA gene sequence analysis. Appl. Environ. Microbiol. 77, 3219–3226 (2011).
Westcott, S. L. & Schloss, P. D. De novo clustering methods outperform reference-based methods for assigning 16S rRNA gene sequences to operational taxonomic units. PeerJ 3, e1487 (2015).
Yue, J. C. & Clayton, M. K. A similarity measure based on species proportions. Commun. Stat. Theory Methods 34, 2123–2131 (2005).
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 26, 32–46 (2001).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol. 12, R60 (2011).
Wang, Q., Garrity, G. M., Tiedje, J. M. & Cole, J. R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73, 5261–5267 (2007).
Cole, J. R. et al. Ribosomal Database Project: data and tools for high throughput rRNA analysis. Nucleic Acids Res. 42, D633–D642 (2014).
Acknowledgements
This work was supported by the National Institutes of Health grants AI-075284 (P.R.) and HL-090775 (P.R.), the American Society of Blood and Marrow Transplantation New Investigator Award (T.T.), the JSPS Postdoctoral Fellowships for Research Abroad (H.F.) and the YASUDA Medical Foundation Grants for Research Abroad (H.F.). This research was supported by work performed by the University of Michigan Microbial Systems Molecular Biology Laboratory.
Author information
Authors and Affiliations
Contributions
T.T. designed and performed the experiments, analysed the data and wrote the paper. H.F., C.R. and M.R. performed the experiments, analysed the data and wrote the paper. H.T. and C.Z. performed the experiments and analysed the data. C.L. performed the experiments and histopathological analysis. A.V.M., J.B., K.O.-W., I.M., S.B., Y.S., D.P., J.W., I.H., S.K., J.C., S.S. and S.P. performed the experiments. G.C. analysed the data, contributed reagents and wrote the paper. P.R. designed the experiments, analysed the data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–5.
Supplementary Table 1
A dataset showing the exact P values for all graphs in this manuscript.
Rights and permissions
About this article
Cite this article
Toubai, T., Fujiwara, H., Rossi, C. et al. Host NLRP6 exacerbates graft-versus-host disease independent of gut microbial composition. Nat Microbiol 4, 800–812 (2019). https://doi.org/10.1038/s41564-019-0373-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-019-0373-1
This article is cited by
-
NLRP inflammasomes in health and disease
Molecular Biomedicine (2024)
-
Loss of NLRP6 expression increases the severity of intestinal injury after syngeneic hematopoietic stem cell transplantation
Annals of Hematology (2024)
-
NLRP6 potentiates PI3K/AKT signalling by promoting autophagic degradation of p85α to drive tumorigenesis
Nature Communications (2023)
-
Physiological and pathophysiological functions of NLRP6: pro- and anti-inflammatory roles
Communications Biology (2022)
-
Challenges and opportunities targeting mechanisms of epithelial injury and recovery in acute intestinal graft-versus-host disease
Mucosal Immunology (2022)