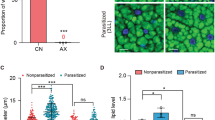
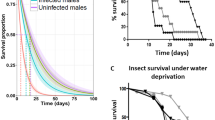
Abstract
Microsporidia are located at the base of the fungal evolutionary tree. They are obligate intracellular parasites and harness host metabolism to fuel their growth and proliferation. However, how the infestation of cells affects the whole organism and how the organism contributes to parasite proliferation remain poorly understood. Here, we have developed a Tubulinosema ratisbonensis systemic infection model in the genetically amenable Drosophila melanogaster host, in which parasite spores obtained in a mammalian cell culture infection system are injected into adult flies. The parasites proliferate within flies and ultimately kill their hosts. As commonly observed for microsporidia infecting insects, T. ratisbonensis preferentially grows in the fat body and ultimately depletes the host metabolic stores. We find that supplementing the fly diet with yeast does not benefit the host but the parasite, which increases its proliferation. Unexpectedly, fatty acids and not carbohydrates or amino acids are the critical components responsible for this phenomenon. Our genetic dissection of host lipid metabolism identifies a crucial compound hijacked by T. ratisbonensis: phosphatidic acid. We propose that phosphatidic acid is a limiting precursor for the synthesis of the parasite membranes and, hence, of its proliferation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data sets, including the 16S rRNA DNA sequences of bacteria retrieved in the haemolymph of control or infected flies, generated and/or analysed during the current study are available in the ‘Phosphatidic acid as a limiting host metabolite for the proliferation of the microsporidium T. ratisbonensis in Drosophila flies’ repository: https://figshare.com/s/c6ef12b7c46e81e58bb1.
References
Weiss, L. M. & Becnel, J. J. Microsporidia (Wiley Blackwell, Oxford, 2014).
Corradi, N. Microsporidia: eukaryotic intracellular parasites shaped by gene loss and horizontal gene transfers. Annu. Rev. Microbiol. 69, 167–183 (2015).
Franzen, C. Microsporidia: how can they invade other cells? Trends Parasitol. 20, 275–279 (2004).
Solter, L. E., Becnel, J. J. & Oi, D. H. in Insect Pathology (eds Vega, F. E. & Kaya, H. K.) 221–263 (Academic Press, San Diego, 2012).
Lom, J. & Dykova, I. Microsporidian xenomas in fish seen in wider perspective. Folia Parasitol. (Praha) 52, 69–81 (2005).
Weiser, J. The Pleistophora debaisieuxi xenoma. Z. Parasitenkd. 48, 263–270 (1976).
Balla, K. M., Luallen, R. J., Bakowski, M. A. & Troemel, E. R. Cell-to-cell spread of microsporidia causes Caenorhabditis elegans organs to form syncytia. Nat. Microbiol. 1, 16144 (2016).
Nakjang, S. et al. Reduction and expansion in microsporidian genome evolution: new insights from comparative genomics. Genome Biol. Evol. 5, 2285–2303 (2013).
Goldberg, A. V. et al. Localization and functionality of microsporidian iron-sulphur cluster assembly proteins. Nature 452, 624–628 (2008).
Tsaousis, A. D. et al. A novel route for ATP acquisition by the remnant mitochondria of Encephalitozoon cuniculi. Nature 453, 553–556 (2008).
Cuomo, C. A. et al. Microsporidian genome analysis reveals evolutionary strategies for obligate intracellular growth. Genome Res. 22, 2478–2488 (2012).
Visser, B. et al. Loss of lipid synthesis as an evolutionary consequence of a parasitic lifestyle. Proc. Natl Acad. Sci. USA 107, 8677–8682 (2010).
Herren, J. K. et al. Insect endosymbiont proliferation is limited by lipid availability. eLife 3, e02964 (2014).
Itoe, M. A. et al. Host cell phosphatidylcholine is a key mediator of malaria parasite survival during liver stage infection. Cell Host Microbe 16, 778–786 (2014).
Arroyo-Olarte, R. D. et al. Phosphatidylthreonine and lipid-mediated control of parasite virulence. PLoS Biol. 13, e1002288 (2015).
Pernas, L., Bean, C., Boothroyd, J. C. & Scorrano, L. Mitochondria restrict growth of the intracellular parasite Toxoplasma gondii by limiting its uptake of fatty acids. Cell Metab. 27, 886–897 (2018).
Nolan, S. J., Romano, J. D. & Coppens, I. Host lipid droplets: an important source of lipids salvaged by the intracellular parasite Toxoplasma gondii. PLoS Pathog. 13, e1006362 (2017).
Caradonna, K. L., Engel, J. C., Jacobi, D., Lee, C. H. & Burleigh, B. A. Host metabolism regulates intracellular growth of Trypanosoma cruzi. Cell Host Microbe 13, 108–117 (2013).
Palm, W. et al. Lipoproteins in Drosophila melanogaster-assembly, function, and influence on tissue lipid composition. PLoS Genet. 8, e1002828 (2012).
Bi, J. et al. Opposite and redundant roles of the two Drosophila perilipins in lipid mobilization. J. Cell Sci. 125, 3568–3577 (2012).
Lehmann, M. Endocrine and physiological regulation of neutral fat storage in Drosophila. Mol. Cell. Endocrinol. 461, 165–177 (2018).
Franzen, C., Fischer, S., Schroeder, J., Scholmerich, J. & Schneuwly, S. Morphological and molecular investigations of Tubulinosema ratisbonensis gen. nov., sp. nov. (Microsporidia: Tubulinosematidae fam. nov.), a parasite infecting a laboratory colony of Drosophila melanogaster (Diptera: Drosophilidae). J. Eukaryot. Microbiol. 52, 141–152 (2005).
Vijendravarma, R. K., Godfray, H. C. & Kraaijeveld, A. R. Infection of Drosophila melanogaster by Tubulinosema kingi: stage-specific susceptibility and within-host proliferation. J. Invertebr. Pathol. 99, 239–241 (2008).
Niehus, S., Giammarinaro, P., Liegeois, S., Quintin, J. & Ferrandon, D. Fly culture collapse disorder: detection, prophylaxis and eradication of the microsporidian parasite Tubulinosema ratisbonensis infecting Drosophila melanogaster. Fly (Austin) 6, 193–204 (2012).
Ayyaz, A., Giammarinaro, P., Liegeois, S., Lestradet, M. & Ferrandon, D. A negative role for MyD88 in the resistance to starvation as revealed in an intestinal infection of Drosophila melanogaster with the Gram-positive bacterium Staphylococcus xylosus. Immunobiology 218, 635–644 (2013).
Shin, S. C. et al. Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling. Science 334, 670–674 (2011).
Kamareddine, L., Robins, W. P., Berkey, C. D., Mekalanos, J. J. & Watnick, P. I. The Drosophila immune deficiency pathway modulates enteroendocrine function and host metabolism. Cell Metab. 28, 449–462 (2018).
Guo, Y., Cordes, K. R., Farese, R. V. Jr & Walther, T. C. Lipid droplets at a glance. J. Cell Sci. 122, 749–752 (2009).
Kuhnlein, R. P. Thematic review series: lipid droplet synthesis and metabolism: from yeast to man. Lipid droplet-based storage fat metabolism in Drosophila. J. Lipid Res. 53, 1430–1436 (2012).
Sieber, M. H. & Thummel, C. S. Coordination of triacylglycerol and cholesterol homeostasis by DHR96 and the Drosophila LipA homolog magro. Cell Metab. 15, 122–127 (2012).
Connerth, M. et al. Intramitochondrial transport of phosphatidic acid in yeast by a lipid transfer protein. Science 338, 815–818 (2012).
Aurrecoechea, C. et al. AmoebaDB and microsporidiadb: functional genomic resources for amoebozoa and microsporidia species. Nucleic Acids Res. 39, D612–D619 (2011).
Wu, H., Carvalho, P. & Voeltz, G. K. Here, there, and everywhere: the importance of ER membrane contact sites. Science 361, eaan5835 (2018).
Dumoux, M. & Hayward, R. D. Membrane contact sites between pathogen-containing compartments and host organelles. Biochim. Biophys. Acta 1861, 895–899 (2016).
Escoll, P., Mondino, S., Rolando, M. & Buchrieser, C. Targeting of host organelles by pathogenic bacteria: a sophisticated subversion strategy. Nat. Rev. Microbiol. 14, 5–19 (2016).
Reinke, A. W., Balla, K. M., Bennett, E. J. & Troemel, E. R. Identification of microsporidia host-exposed proteins reveals a repertoire of rapidly evolving proteins. Nat. Commun. 8, 14023 (2017).
Jiang, Y. et al. Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science 356, 1172–1175 (2017).
Luginbuehl, L. H. et al. Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant. Science 356, 1175–1178 (2017).
Gazos-Lopes, F., Martin, J. L., Dumoulin, P. C. & Burleigh, B. A. Host triacylglycerols shape the lipidome of intracellular trypanosomes and modulate their growth. PLoS Pathog. 13, e1006800 (2017).
Nagajyothi, F. et al. Crucial role of the central leptin receptor in murine Trypanosoma cruzi (Brazil strain) infection. J. Infect. Dis. 202, 1104–1113 (2010).
Franzen, C. et al. In vitro cultivation of an insect microsporidian Tubulinosema ratisbonensis in mammalian cells. J. Eukaryot. Microbiol. 52, 349–355 (2005).
Thibault, S. T. et al. A complementary transposon tool kit for Drosophila melanogaster using P and piggyBac. Nat. Genet. 36, 283–287 (2004).
Dietzl, G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, 151–156 (2007).
Ni, J. Q. et al. A genome-scale shRNA resource for transgenic RNAi in Drosophila. Nat. Methods 8, 405–407 (2011).
Gargano, J. W., Martin, I., Bhandari, P. & Grotewiel, M. S. Rapid iterative negative geotaxis (RING): a new method for assessing age-related locomotor decline in Drosophila. Exp. Gerontol. 40, 386–395 (2005).
Ja, W. W. et al. Prandiology of Drosophila and the CAFE assay. Proc. Natl Acad. Sci. USA 104, 8253–8256 (2007).
Rera, M. et al. Modulation of longevity and tissue homeostasis by the Drosophila PGC-1 homolog. Cell Metab. 14, 623–634 (2011).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Guo, L., Karpac, J., Tran, S. L. & Jasper, H. PGRP-SC2 promotes gut immune homeostasis to limit commensal dysbiosis and extend lifespan. Cell 156, 109–122 (2014).
Belkorchia, A. et al. In vitro propagation of the microsporidian pathogen Brachiola algerae and studies of its chromosome and ribosomal DNA organization in the context of the complete genome sequencing project. Parasitol. Int. 57, 62–71 (2008).
Acknowledgements
We are grateful to M. Yamba and M.-C. Lacombe for expert technical assistance. We thank F. Delbac, A. Gould, B. Lemaitre and P. Léopold for informative discussions and suggestions, and S. Liégeois for critical reading of the manuscript. Transgenic fly stocks were obtained from the VDRC (www.vdrc.at). Stocks obtained from the Bloomington Drosophila Stock Center (NIH P40OD018537) were used in this study. This work has been funded by CNRS, University of Strasbourg, Fondation pour la Recherche Médicale (Equipe FRM to D.F. and FDT20140930952 to A.F.), Agence Nationale de la Recherche (BEELOSS, ANR-11-EQPX-0022) and the Institut d’Etude Avancée de l’Université de Strasbourg.
Author information
Authors and Affiliations
Contributions
A.F. performed most of the experimental work described in this study, with assistance from G.C. for the natural infestation experiment and the ingestion/injection of phosphatidic acid into control or microsporidia-infected flies. S.N. developed the infection model and all techniques required to maintain parasite cultures, as well as those for infection of flies; he participated in all aspects of the early stages of this study. A.F. and D.F. designed the experiments, analysed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–9, Supplementary Tables 1 and 2.
Rights and permissions
About this article
Cite this article
Franchet, A., Niehus, S., Caravello, G. et al. Phosphatidic acid as a limiting host metabolite for the proliferation of the microsporidium Tubulinosema ratisbonensis in Drosophila flies. Nat Microbiol 4, 645–655 (2019). https://doi.org/10.1038/s41564-018-0344-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-018-0344-y