Abstract
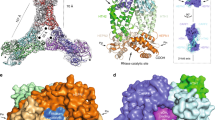
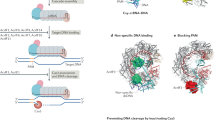
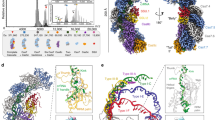
Type IV CRISPR–Cas modules belong to class 1 prokaryotic adaptive immune systems, which are defined by the presence of multisubunit effector complexes. They usually lack the known Cas proteins involved in adaptation and target cleavage, and their function has not been experimentally addressed. To investigate RNA and protein components of this CRISPR–Cas type, we located a complete type IV cas gene locus and an adjacent CRISPR array on a megaplasmid of Aromatoleum aromaticum EbN1, which contains an additional type I-C system on its chromosome. RNA sequencing analyses verified CRISPR RNA (crRNA) production and maturation for both systems. Type IV crRNAs were shown to harbour unusually short 7 nucleotide 5′-repeat tags and stable 3′ hairpin structures. A unique Cas6 variant (Csf5) was identified that generates crRNAs that are specifically incorporated into type IV CRISPR–ribonucleoprotein (crRNP) complexes. Structures of RNA-bound Csf5 were obtained. Recombinant production and purification of the type IV Cas proteins, together with electron microscopy, revealed that Csf2 acts as a helical backbone for type IV crRNPs that include Csf5, Csf3 and a large subunit (Csf1). Mass spectrometry analyses identified protein–protein and protein–RNA contact sites. These results highlight evolutionary connections between type IV and type I CRISPR–Cas systems and demonstrate that type IV CRISPR–Cas systems employ crRNA-guided effector complexes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jansen, R., Embden, J. D., Gaastra, W. & Schouls, L. M. Identification of genes that are associated with DNA repeats in prokaryotes. Mol. Microbiol. 43, 1565–1575 (2002).
Bolotin, A., Quinquis, B., Sorokin, A. & Ehrlich, S. D. Clustered regularly interspaced short palindrome repeats (CRISPRs) have spacers of extrachromosomal origin. Microbiology 151, 2551–2561 (2005).
Mojica, F. J., Diez-Villasenor, C., Garcia-Martinez, J. & Soria, E. Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements. J. Mol. Evol. 60, 174–182 (2005).
Pourcel, C., Salvignol, G. & Vergnaud, G. CRISPR elements in Yersinia pestis acquire new repeats by preferential uptake of bacteriophage DNA, and provide additional tools for evolutionary studies. Microbiology 151, 653–663 (2005).
Barrangou, R. et al. CRISPR provides acquired resistance against viruses in prokaryotes. Science 315, 1709–1712 (2007).
Westra, E. R. et al. The CRISPRs, they are a-changin’: how prokaryotes generate adaptive immunity. Annu. Rev. Genet. 46, 311–339 (2012).
Mojica, F. J., Diez-Villasenor, C., Garcia-Martinez, J. & Almendros, C. Short motif sequences determine the targets of the prokaryotic CRISPR defence system. Microbiology 155, 733–740 (2009).
Nunez, J. K. et al. Cas1–Cas2 complex formation mediates spacer acquisition during CRISPR-Cas adaptive immunity. Nat. Struct. Mol. Biol. 21, 528–534 (2014).
Yosef, I., Goren, M. G. & Qimron, U. Proteins and DNA elements essential for the CRISPR adaptation process in Escherichia coli. Nucleic Acids Res. 40, 5569–5576 (2012).
Swarts, D. C., Mosterd, C., van Passel, M. W. & Brouns, S. J. CRISPR interference directs strand specific spacer acquisition. PLoS ONE 7, e35888 (2012).
Pul, U. et al. Identification and characterization of E. coli CRISPR-cas promoters and their silencing by H-NS. Mol. Microbiol. 75, 1495–1512 (2010).
Pougach, K. et al. Transcription, processing and function of CRISPR cassettes in Escherichia coli. Mol. Microbiol. 77, 1367–1379 (2010).
Brouns, S. J. et al. Small CRISPR RNAs guide antiviral defense in prokaryotes. Science 321, 960–964 (2008).
Jore, M. M. et al. Structural basis for CRISPR RNA-guided DNA recognition by Cascade. Nat. Struct. Mol. Biol. 18, 529–536 (2011).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Hale, C. R. et al. RNA-guided RNA cleavage by a CRISPR RNA-Cas protein complex. Cell 139, 945–956 (2009).
Garneau, J. E. et al. The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature 468, 67–71 (2010).
Westra, E. R. et al. CRISPR immunity relies on the consecutive binding and degradation of negatively supercoiled invader DNA by Cascade and Cas3. Mol. Cell 46, 595–605 (2012).
Makarova, K. S. et al. An updated evolutionary classification of CRISPR-Cas systems. Nat. Rev. Microbiol. 13, 722–736 (2015).
Koonin, E. V., Makarova, K. S. & Zhang, F. Diversity, classification and evolution of CRISPR-Cas systems. Curr. Opin. Microbiol. 37, 67–78 (2017).
Mohanraju, P. et al. Diverse evolutionary roots and mechanistic variations of the CRISPR-Cas systems. Science 353, aad5147 (2016).
Kazlauskiene, M., Tamulaitis, G., Kostiuk, G., Venclovas, C. & Siksnys, V. Spatiotemporal control of Type III-A CRISPR-Cas immunity: coupling DNA degradation with the target RNA recognition. Mol. Cell 62, 295–306 (2016).
Huo, Y. et al. Structures of CRISPR Cas3 offer mechanistic insights into Cascade-activated DNA unwinding and degradation. Nat. Struct. Mol. Biol. 21, 771–777 (2014).
Sashital, D. G., Wiedenheft, B. & Doudna, J. A. Mechanism of foreign DNA selection in a bacterial adaptive immune system. Mol. Cell 46, 606–615 (2012).
Voloshin, O. N. & Camerini-Otero, R. D. The DinG protein from Escherichia coli is a structure-specific helicase. J. Biol. Chem. 282, 18437–18447 (2007).
Gleditzsch, D. et al. Modulating the Cascade architecture of a minimal Type I-F CRISPR-Cas system. Nucleic Acids Res. 44, 5872–5882 (2016).
Pausch, P. et al. Structural variation of Type I-F CRISPR RNA guided DNA surveillance. Mol. Cell 67, 622–632 (2017).
Nam, K. H. et al. Cas5d protein processes pre-crRNA and assembles into a cascade-like interference complex in subtype I-C/Dvulg CRISPR-Cas system. Structure 20, 1574–1584 (2012).
Hochstrasser, M. L., Taylor, D. W., Kornfeld, J. E., Nogales, E. & Doudna, J. A. DNA targeting by a minimal CRISPR RNA-guided cascade. Mol. Cell 63, 840–851 (2016).
Garside, E. L. et al. Cas5d processes pre-crRNA and is a member of a larger family of CRISPR RNA endonucleases. RNA 18, 2020–2028 (2012).
Hochstrasser, M. L. & Doudna, J. A. Cutting it close: CRISPR-associated endoribonuclease structure and function. Trends Biochem. Sci. 40, 58–66 (2015).
Plagens, A. et al. In vitro assembly and activity of an archaeal CRISPR-Cas type I-A Cascade interference complex. Nucleic Acids Res. 42, 5125–5138 (2014).
Reeks, J. et al. Structure of a dimeric crenarchaeal Cas6 enzyme with an atypical active site for CRISPR RNA processing. Biochem. J. 452, 223–230 (2013).
Shmakov, S. A. et al. The CRISPR spacer space is dominated by sequences from species-specific mobilomes. mBio 8, e01397-17 (2017).
Rabus, R. & Widdel, F. Anaerobic degradation of ethylbenzene and other aromatic hydrocarbons by new denitrifying bacteria. Arch. Microbiol. 163, 96–103 (1995).
Grant, T., Rohou, A. & Grigorieff, N. cisTEM, user-friendly software for single-particle image processing. elife 7, e35383 (2018).
Miyatake, H., Hasegawa, T. & Yamano, A. New methods to prepare iodinated derivatives by vaporizing iodine labelling (VIL) and hydrogen peroxide VIL (HYPER-VIL). Acta Crystallogr. D 62, 280–289 (2006).
Gabadinho, J. et al. MxCuBE: a synchrotron beamline control environment customized for macromolecular crystallography experiments. J. Synchrotron. Radiat. 17, 700–707 (2010).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D 67, 235–242 (2011).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Kramer, K. et al. Photo-cross-linking and high-resolution mass spectrometry for assignment of RNA-binding sites in RNA-binding proteins. Nat. Methods 11, 1064–1070 (2014).
Yang, B. et al. Identification of cross-linked peptides from complex samples. Nat. Methods 9, 904–906 (2012).
Combe, C. W., Fischer, L. & Rappsilber, J. xiNET: cross-link network maps with residue resolution. Mol. Cell Proteomics 14, 1137–1147 (2015).
Acknowledgements
G.B thanks the LOEWE excellence initiative for financial support. G.B. and P.P. acknowledge the always excellent support by the European Synchrotron Radiation Facility (ESRF) in Grenoble, France. This work was supported by the DFG (FOR1680 to L.R.), SPP2141 (to L.R. and G.B.), TÜBİTAK (to A.Ö.) and the Max Planck Society (to L.R.).
Author information
Authors and Affiliations
Contributions
A.Ö. and P.P. purified proteins. P.P. determined the crystal structure. A.L., A.W. and H.U. performed mass spectrometry analyses. K.S. and J.H. cultured A. aromatoleum strains. T.H. performed transmission electron microscopy analyses. L.R and A.Ö. conceived the experiments. L.R. wrote the manuscript with support from A.Ö., P.P., G.B., J.H., T.H. and H.U.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–6, Supplementary Tables 1, 2, 4 and 5.
Supplementary Table 3
Overview of crRNP protein–protein cross-links. Contains a structured representation of all identified protein–protein cross-links (cross-linked peptide spectrum matches (CSM)) of the recombinant type IV crRNP of Aromatoleum aromaticum. A detailed legend is included as a separate worksheet.
Rights and permissions
About this article
Cite this article
Özcan, A., Pausch, P., Linden, A. et al. Type IV CRISPR RNA processing and effector complex formation in Aromatoleum aromaticum. Nat Microbiol 4, 89–96 (2019). https://doi.org/10.1038/s41564-018-0274-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-018-0274-8
This article is cited by
-
An Update on the Application of CRISPR Technology in Clinical Practice
Molecular Biotechnology (2024)
-
Transposon-encoded nucleases use guide RNAs to promote their selfish spread
Nature (2023)
-
Alternative functions of CRISPR–Cas systems in the evolutionary arms race
Nature Reviews Microbiology (2022)
-
Characterization of the self-targeting Type IV CRISPR interference system in Pseudomonas oleovorans
Nature Microbiology (2022)
-
CRISPR-Cas-Systeme der Klasse 1: Genome Engineering und Silencing
BIOspektrum (2022)