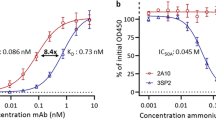
Abstract
The circumsporozoite protein (CSP) is the major surface protein of malaria sporozoites (SPZs), the motile and invasive parasite stage inoculated in the host skin by infected mosquitoes. Antibodies against the central CSP repeats of different plasmodial species are known to block SPZ infectivity1,2,3,4,5, but the precise mechanism by which these effectors operate is not completely understood. Here, using a rodent Plasmodium yoelii malaria model, we show that sterile protection mediated by anti-P. yoelii CSP humoral immunity depends on the parasite inoculation into the host skin, where antibodies inhibit motility and kill P. yoelii SPZs via a characteristic ‘dotty death’ phenotype. Passive transfer of an anti-repeat monoclonal antibody (mAb) recapitulates the skin inoculation-dependent protection, in a complement- and Fc receptor γ-independent manner. This purified mAb also decreases motility and, notably, induces the dotty death of P. yoelii SPZs in vitro. Cytotoxicity is species-transcendent since cognate anti-CSP repeat mAbs also kill Plasmodium berghei and Plasmodium falciparum SPZs. mAb cytotoxicity requires the actomyosin motor-dependent translocation and stripping of the protective CSP surface coat, rendering the parasite membrane susceptible to the SPZ pore-forming-like protein secreted to wound and traverse the host cell membrane6. The loss of SPZ fitness caused by anti-P. yoelii CSP repeat antibodies is thus a dynamic process initiated in the host skin where SPZs either stop moving7, or migrate and traverse cells to progress through the host tissues7,8,9 at the eventual expense of their own life.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Yoshida, N., Nussenzweig, R. S., Potocnjak, P., Nussenzweig, V. & Aikawa, M. Hybridoma produces protective antibodies directed against the sporozoite stage of malaria parasite. Science 207, 71–73 (1980).
Nardin, E. H. et al. Circumsporozoite proteins of human malaria parasites Plasmodium falciparum and Plasmodium vivax. J. Exp. Med. 156, 20–30 (1982).
Cochrane, A. H., Santoro, F., Nussenzweig, V., Gwadz, R. W. & Nussenzweig, R. S. Monoclonal antibodies identify the protective antigens of sporozoites of Plasmodium knowlesi. Proc. Natl Acad. Sci. USA 79, 5651–5655 (1982).
Charoenvit, Y. et al. Inability of malaria vaccine to induce antibodies to a protective epitope within its sequence. Science 251, 668–671 (1991).
Charoenvit, Y. et al. Monoclonal, but not polyclonal, antibodies protect against Plasmodium yoelii sporozoites. J. Immunol. 146, 1020–1025 (1991).
Ishino, T., Chinzei, Y. & Yuda, M. A Plasmodium sporozoite protein with a membrane attack complex domain is required for breaching the liver sinusoidal cell layer prior to hepatocyte infection. Cell Microbiol. 7, 199–208 (2005).
Vanderberg, J. P. & Frevert, U. Intravital microscopy demonstrating antibody-mediated immobilisation of Plasmodium berghei sporozoites injected into skin by mosquitoes. Int. J. Parasitol. 34, 991–996 (2004).
Amino, R. et al. Quantitative imaging of Plasmodium transmission from mosquito to mammal. Nat. Med. 12, 220–224 (2006).
Amino, R. et al. Host cell traversal is important for progression of the malaria parasite through the dermis to the liver. Cell Host Microbe 3, 88–96 (2008).
Ménard, R. et al. Circumsporozoite protein is required for development of malaria sporozoites in mosquitoes. Nature 385, 336–340 (1997).
Pancake, S. J., Holt, G. D., Mellouk, S. & Hoffman, S. L. Malaria sporozoites and circumsporozoite proteins bind specifically to sulfated glycoconjugates. J. Cell Biol. 117, 1351–1357 (1992).
Sidjanski, S. P., Vanderberg, J. P. & Sinnis, P. Anopheles stephensi salivary glands bear receptors for region I of the circumsporozoite protein of Plasmodium falciparum. Mol. Biochem. Parasitol. 90, 33–41 (1997).
Cerami, C. et al. The basolateral domain of the hepatocyte plasma membrane bears receptors for the circumsporozoite protein of Plasmodium falciparum sporozoites. Cell 70, 1021–1033 (1992).
Tewari, R., Spaccapelo, R., Bistoni, F., Holder, A. A. & Crisanti, A. Function of region I and II adhesive motifs of Plasmodium falciparum circumsporozoite protein in sporozoite motility and infectivity. J. Biol. Chem. 277, 47613–47618 (2002).
Gilson, P. R. et al. Identification and stoichiometry of glycosylphosphatidylinositol-anchored membrane proteins of the human malaria parasite Plasmodium falciparum. Mol. Cell. Proteomics 5, 1286–1299 (2006).
Olotu, A. et al. Seven-year efficacy of RTS,S/AS01 malaria vaccine among young African children. N. Engl. J. Med. 374, 2519–2529 (2016).
White, M. T. et al. The relationship between RTS,S vaccine-induced antibodies, CD4+ T cell responses and protection against Plasmodium falciparum infection. PLoS ONE 8, e61395 (2013).
Stewart, M. J., Nawrot, R. J., Schulman, S. & Vanderberg, J. P. Plasmodium berghei sporozoite invasion is blocked in vitro by sporozoite-immobilizing antibodies. Infect. Immun. 51, 859–864 (1986).
Hollingdale, M. R., Zavala, F., Nussenzweig, R. S. & Nussenzweig, V. Antibodies to the protective antigen of Plasmodium berghei sporozoites prevent entry into cultured cells. J. Immunol. 128, 1929–1930 (1982).
Hollingdale, M. R., Nardin, E. H., Tharavanij, S., Schwartz, A. L. & Nussenzweig, R. S. Inhibition of entry of Plasmodium falciparum and P. vivax sporozoites into cultured cells; an in vitro assay of protective antibodies. J. Immunol. 132, 909–913 (1984).
Potocnjak, P., Yoshida, N., Nussenzweig, R. S. & Nussenzweig, V. Monovalent fragments (Fab) of monoclonal antibodies to a sporozoite surface antigen (Pb44) protect mice against malarial infection. J. Exp. Med. 151, 1504–1513 (1980).
Egan, J. E. et al. Efficacy of murine malaria sporozoite vaccines: implications for human vaccine development. Science 236, 453–456 (1987).
Kisalu, N. K. et al. A human monoclonal antibody prevents malaria infection by targeting a new site of vulnerability on the parasite. Nat. Med. 24, 408–416 (2018).
Sack, B. K. et al. Model for in vivo assessment of humoral protection against malaria sporozoite challenge by passive transfer of monoclonal antibodies and immune serum. Infect. Immun. 82, 808–817 (2014).
Sedegah, M. et al. Evaluation of vaccines designed to induce protective cellular immunity against the Plasmodium yoelii circumsporozoite protein: vaccinia, pseudorabies, and salmonella transformed with circumsporozoite gene. Bull. World Health Organ. 68, 9–114 (1990).
Collins, W. E. et al. Immunization of Saimiri sciureus boliviensis with recombinant vaccines based on the circumsporozoite protein of Plasmodium vivax. Am. J. Trop. Med. Hyg. 40, 455–464 (1989).
Lal, A. A. et al. In vivo testing of subunit vaccines against malaria sporozoites using a rodent system. Proc. Natl Acad. Sci. USA 84, 8647–8651 (1987).
Belmonte, M. et al. The infectivity of Plasmodium yoelii in different strains of mice. J. Parasitol. 89, 602–603 (2003).
Gueirard, P. et al. Development of the malaria parasite in the skin of the mammalian host. Proc. Natl Acad. Sci. USA 107, 18640–18645 (2010).
Bongfen, S. E. et al. The N-terminal domain of Plasmodium falciparum circumsporozoite protein represents a target of protective immunity. Vaccine 27, 328–335 (2009).
Herrera, R. et al. Reversible conformational change in the Plasmodium falciparum circumsporozoite protein masks its adhesion domains. Infect. Immun. 83, 3771–3780 (2015).
Espinosa, D. A. et al. Proteolytic cleavage of the Plasmodium falciparum circumsporozoite protein is a target of protective antibodies. J. Infect. Dis. 212, 1111–1119 (2015).
Noris, M. & Remuzzi, G. Overview of complement activation and regulation. Semin. Nephrol. 33, 479–492 (2013).
Nimmerjahn, F. & Ravetch, J. V. Fcγ receptors: old friends and new family members. Immunity 24, 19–28 (2006).
Persson, C. et al. Cutting edge: a new tool to evaluate human pre-erythrocytic malaria vaccines: rodent parasites bearing a hybrid Plasmodium falciparum circumsporozoite protein. J. Immunol. 169, 6681–6685 (2002).
Vanderberg, J. P. Studies on the motility of Plasmodium sporozoites. J. Protozool. 21, 527–537 (1974).
Stewart, M. J. & Vanderberg, J. P. Malaria sporozoites release circumsporozoite protein from their apical end and translocate it along their surface. J. Protozool. 38, 411–421 (1991).
Carey, A. F. et al. Calcium dynamics of Plasmodium berghei sporozoite motility. Cell Microbiol. 16, 768–783 (2014).
Risco-Castillo, V. et al. Malaria sporozoites traverse host cells within transient vacuoles. Cell Host Microbe 18, 593–603 (2015).
Ishino, T., Yano, K., Chinzei, Y. & Yuda, M. Cell-passage activity is required for the malarial parasite to cross the liver sinusoidal cell layer. PLoS Biol. 2, E4 (2004).
Kariu, T., Ishino, T., Yano, K., Chinzei, Y. & Yuda, M. CelTOS, a novel malarial protein that mediates transmission to mosquito and vertebrate hosts. Mol. Microbiol. 59, 1369–1379 (2006).
Jimah, J. R. Malaria parasite CelTOS targets the inner leaflet of cell membranes for pore-dependent disruption. eLife 5, e20621 (2016).
Fine, E., Aikawa, M., Cochrane, A. H. & Nussenzweig, R. S. Immuno-electron microscopic observations on Plasmodium knowlesi sporozoites: localization of protective antigen and its precursors. Am. J. Trop. Med. Hyg. 33, 220–226 (1984).
Posthuma, G. et al. Immunogold determination of Plasmodium falciparum circumsporozoite protein in Anopheles stephensi salivary gland cells. Eur. J. Cell Biol. 49, 66–72 (1989).
Wang, R. et al. Induction of protective polyclonal antibodies by immunization with a Plasmodium yoelii circumsporozoite protein multiple antigen peptide vaccine. J. Immunol. 154, 2784–2793 (1995).
Yilmaz, B. et al. Gut microbiota elicits a protective immune response against malaria transmission. Cell 159, 1277–1289 (2014).
Bouharoun-Tayoun, H., Oeuvray, C., Lunel, F. & Druilhe, P. Mechanisms underlying the monocyte-mediated antibody-dependent killing of Plasmodium falciparum asexual blood stages. J. Exp. Med. 182, 409–418 (1995).
Weinbaum, F. I., Evans, C. B. & Tigelaar, R. E. An in vitro assay for T cell immunity to malaria in mice. J. Immunol. 116, 1280–1283 (1976).
Ono, T., Tadakuma, T. & Rodriguez, A. Plasmodium yoelii yoelii 17XNL constitutively expressing GFP throughout the life cycle. Exp. Parasitol. 115, 310–313 (2007).
Manzoni, G. et al. A rapid and robust selection procedure for generating drug-selectable marker-free recombinant malaria parasites. Sci. Rep. 4, 4760 (2014).
Mwakingwe, A. et al. Noninvasive real-time monitoring of liver-stage development of bioluminescent Plasmodium parasites. J. Infect. Dis. 200, 1470–1478 (2009).
Ishino, T., Orito, Y., Chinzei, Y. & Yuda, M. A calcium-dependent protein kinase regulates Plasmodium ookinete access to the midgut epithelial cell. Mol. Microbiol. 59, 1175–1184 (2006).
Sturm, A. et al. Alteration of the parasite plasma membrane and the parasitophorous vacuole membrane during exo-erythrocytic development of malaria parasites. Protist 160, 51–63 (2009).
Demarta-Gatsi, C. et al. Immunological memory to blood-stage malaria infection is controlled by the histamine releasing factor (HRF) of the parasite. Sci. Rep. 7, 9129 (2017).
Ponnudurai, T., Leeuwenberg, A. D. & Meuwissen, J. H. Chloroquine sensitivity of isolates of Plasmodium falciparum adapted to in vitro culture. Trop. Geogr. Med. 33, 50–54 (1981).
Gu, H., Zou, Y. R. & Rajewsky, K. Independent control of immunoglobulin switch recombination at individual switch regions evidenced through Cre-loxP-mediated gene targeting. Cell 73, 1155–1164 (1993).
Wessels, M. R. et al. Studies of group B streptococcal infection in mice deficient in complement component C3 or C4 demonstrate an essential role for complement in both innate and acquired immunity. Proc. Natl Acad. Sci. USA 92, 11490–11494 (1995).
Takai, T., Li, M., Sylvestre, D. & Clynes, R. FcR γ chain deletion results in pleiotrophiceffector cell defects.Cell 76, 519–529 (1994).
Ponnudurai, T., Lensen, A. H. & Meuwissen, J. H. An automated large-scale culture system of Plasmodium falciparum using tangential flow filtration for medium change. Parasitology 87(Pt 3), 439–445 (1983).
Liu, S. et al. Removal of endotoxin from recombinant protein preparations. Clin. Biochem. 30, 455–463 (1997).
Foquet, L. et al. Vaccine-induced monoclonal antibodies targeting circumsporozoite protein prevent Plasmodium falciparum infection. J. Clin. Invest. 124, 140–144 (2014).
Schmidt, N. W., Butler, N. S., Badovinac, V. P. & Harty, J. T. Extreme CD8 T cell requirements for anti-malarial liver-stage immunity following immunization with radiation attenuated sporozoites. PLoS Pathog. 6, e1000998 (2010).
Jaeger, B. N. et al. Neutrophil depletion impairs natural killer cell maturation, function, and homeostasis. J. Exp. Med. 209, 565–580 (2012).
Amino, R. et al. Imaging malaria sporozoites in the dermis of the mammalian host. Nat. Protoc. 2, 1705–1712 (2007).
Köhler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Eichinger, D. J., Arnot, D. E., Tam, J. P., Nussenzweig, V. & Enea, V. Circumsporozoite protein of Plasmodium berghei: gene cloning and identification of the immunodominant epitopes. Mol. Cell. Biol. 6, 3965–3972 (1986).
Zavala, F. et al. Rationale for development of a synthetic vaccine against Plasmodium falciparum malaria. Science 228, 1436–1440 (1985).
Prudêncio, M., Rodrigues, C. D., Ataíde , R. & Mota, M. M. Dissecting in vitro host cell infection by Plasmodium sporozoites using flow cytometry. Cell. Microbiol. 10, 218–224 (2008).
Acknowledgements
We thank the team of the Centre of Production and Infection of Anopheles, Institut Pasteur, in particular M. Szatanik, C. Thouvenot, S. Golba, J. Pham and A. Lorthiois for providing mosquitoes and P. falciparum SPZs; the team of the Platform of Dynamic Imaging, Institut Pasteur, in particular Dr S. Shorte and M.-A. Nicola for the access to the confocal microscopes and IVIS system; Dr K. Kim from the Albert Einstein College of Medicine for providing the P. yoelii YM GFP-luciferase; Dr V. Nussenzweig from New York University for providing the P. berghei and falciparumnized P. berghei parasites; Dr M. Soares from the Instituto Gulbenkian for providing the JHT−/− mice; Dr P.-M. Lledo and Dr P. Bruhns from the Institut Pasteur for providing, respectively, the C3−/− and the FcRγ−/− mice; the team of the Clinical Investigation and Access to BioResources, in particular M.-N. Ungeheuer for providing the erythrocytes for the P. falciparum culture; Dr P. Baldacci and Dr P. Formaglio for their critical reading of the manuscript. This work was supported by funds from Institut Pasteur, Paris, the French National Research Agency (grant no. ANR-14-CE16-Im3alaria), the French Government’s ‘Investissement d’Avenir’ program, Laboratoire d’Excellence ‘Integrative Biology of Emerging Infectious Diseases’ (grant no. ANR-10-LABX-62-IBEID) and ‘ParaFrap’ (grant no. ANR-11-LABX-0024), the São Paulo Research Foundation (FAPESP, grant no. 2014/50631-0), the National Council for Scientific and Technological Development (CNPq), the BNP Paribas CIB, the Portuguese Science and Technology Foundation (FCT, grant no. IF/00881/2012/CP0158) and the European Social Fund (Human Potential Operational Programme).
Author information
Authors and Affiliations
Contributions
R.A., S.B.B., E.A. and J.T. conceived and designed the experiments. E.A., J.T., S.T. and R.H.P performed the experiments. M.M.Y., S.D. and F.T. generated the hybridomas. O.S., T.I. and M.Y. contributed with the transgenic parasites. R.A., S.B.B., E.A., J.T., S.T. and R.H.P. analysed the data. R.A, E.A, J.T. and S.B.B. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–5 and Supplementary Table 1.
Rights and permissions
About this article
Cite this article
Aliprandini, E., Tavares, J., Panatieri, R.H. et al. Cytotoxic anti-circumsporozoite antibodies target malaria sporozoites in the host skin. Nat Microbiol 3, 1224–1233 (2018). https://doi.org/10.1038/s41564-018-0254-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-018-0254-z
This article is cited by
-
Sporozoite motility as a quantitative readout for anti-CSP antibody inhibition
Scientific Reports (2022)
-
Limited Plasmodium sporozoite gliding motility in the absence of TRAP family adhesins
Malaria Journal (2021)
-
Mechanisms and targets of Fcγ-receptor mediated immunity to malaria sporozoites
Nature Communications (2021)
-
An epitope-based malaria vaccine targeting the junctional region of circumsporozoite protein
npj Vaccines (2021)
-
A universal vaccine candidate against Plasmodium vivax malaria confers protective immunity against the three PvCSP alleles
Scientific Reports (2021)