Abstract
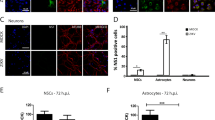
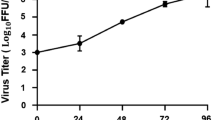
Zika virus (ZIKV) is associated with neonatal microcephaly and Guillain–Barré syndrome1,2. While progress has been made in understanding the causal link between ZIKV infection and microcephaly3,4,5,6,7,8,9, the life cycle and pathogenesis of ZIKV are less well understood. In particular, there are conflicting reports on the role of AXL, a TAM family kinase receptor that was initially described as the entry receptor for ZIKV10,11,12,13,14,15,16,17,18,19,20,21,22. Here, we show that while genetic ablation of AXL protected primary human astrocytes and astrocytoma cell lines from ZIKV infection, AXL knockout did not block the entry of ZIKV. We found, instead, that the presence of AXL attenuated the ZIKV-induced activation of type I interferon (IFN) signalling genes, including several type I IFNs and IFN-stimulating genes. Knocking out type I IFN receptor α chain (IFNAR1) restored the vulnerability of AXL knockout astrocytes to ZIKV infection. Further experiments suggested that AXL regulates the expression of SOCS1, a known type I IFN signalling suppressor, in a STAT1/STAT2-dependent manner. Collectively, our results demonstrate that AXL is unlikely to function as an entry receptor for ZIKV and may instead promote ZIKV infection in human astrocytes by antagonizing type I IFN signalling.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Brasil, P. et al. Zika virus infection in pregnant women in Rio de Janeiro. N. Engl. J. Med. 375, 2321–2334 (2016).
Parra, B. et al. Guillain-Barre syndrome associated with Zika virus infection in Colombia. N. Engl. J. Med. 375, 1513–1523 (2016).
Tang, H. et al. Zika virus infects human cortical neural progenitors and attenuates their growth. Cell Stem Cell 18, 587–590 (2016).
Garcez, P. P. et al. Zika virus impairs growth in human neurospheres and brain organoids. Science 352, 816–818 (2016).
Qian, X. et al. Brain-region-specific organoids using mini-bioreactors for modeling ZIKV exposure. Cell 165, 1238–1254 (2016).
Miner, J. J. et al. Zika virus infection during pregnancy in mice causes placental damage and fetal demise. Cell 165, 1081–1091 (2016).
Cugola, F. R. et al. The Brazilian Zika virus strain causes birth defects in experimental models. Nature 534, 267–271 (2016).
Li, H. et al. Zika virus infects neural progenitors in the adult mouse brain and alters proliferation. Cell Stem Cell 19, 593–598 (2016).
Adams Waldorf, K. M. et al. Fetal brain lesions after subcutaneous inoculation of Zika virus in a pregnant nonhuman primate. Nat. Med. 22, 1256–1259 (2016).
Meertens, L. et al. The TIM and TAM families of phosphatidylserine receptors mediate dengue virus entry. Cell Host Microbe 12, 544–557 (2012).
Miner, J. J. et al. The TAM receptor Mertk protects against neuroinvasive viral infection by maintaining blood-brain barrier integrity. Nat. Med. 21, 1464–1472 (2015).
Hamel, R. et al. Biology of Zika Virus infection in human skin cells. J. Virol. 89, 8880–8896 (2015).
Retallack, H. et al. Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proc. Natl Acad. Sci. USA 113, 14408–14413 (2016).
Savidis, G. et al. Identification of Zika virus and Dengue virus dependency factors using functional genomics. Cell Rep. 16, 232–246 (2016).
Nowakowski, T. J. et al. Expression analysis highlights AXL as a candidate Zika virus entry receptor in neural stem cells. Cell Stem Cell 18, 591–596 (2016).
Tabata, T. et al. Zika virus targets different primary human placental cells, suggesting two routes for vertical transmission. Cell Host Microbe 20, 155–166 (2016).
Ma, W. et al. Zika virus causes testis damage and leads to male infertility in mice. Cell 167, 1511–1524 (2016).
Wells, M. F. et al. Genetic ablation of AXL does not protect human neural progenitor cells and cerebral organoids from Zika virus infection. Cell Stem Cell 19, 703–708 (2016).
Miner, J. J. et al. Zika virus infection in mice causes panuveitis with shedding of virus in tears. Cell Rep. 16, 3208–3218 (2016).
Hastings, A. K. et al. TAM receptors are not required for Zika virus infection in mice. Cell Rep. 19, 558–568 (2017).
Li, F. et al. AXL is not essential for Zika virus infection in the mouse brain. Emerg. Microbes Infect. 6, e16 (2017).
Govero, J. et al. Zika virus infection damages the testes in mice. Nature 540, 438–442 (2016).
Grant, A. et al. Zika virus targets human STAT2 to inhibit type I interferon signaling. Cell Host Microbe 19, 882–890 (2016).
Kumar, A. et al. Zika virus inhibits type-I interferon production and downstream signaling. EMBO Rep. 17, 1766–1775 (2016).
Khan, S. et al. Dampened antiviral immunity to intravaginal exposure to RNA viral pathogens allows enhanced viral replication. J. Exp. Med. 213, 2913–2929 (2016).
Meertens, L. et al. Axl mediates ZIKA virus entry in human glial cells and modulates innate immune responses. Cell Rep. 18, 324–333 (2017).
Rausch, K. et al. Screening bioactives reveals nanchangmycin as a broad spectrum antiviral active against Zika virus. Cell Rep. 18, 804–815 (2017).
Liu, S., DeLalio, L. J., Isakson, B. E. & Wang, T. T. AXL-mediated productive infection of human endothelial cells by Zika virus. Circ. Res. 119, 1183–1189 (2016).
Lindqvist, R. et al. Fast type I interferon response protects astrocytes from flavivirus infection and virus-induced cytopathic effects. J. Neuroinflamm. 13, 277 (2016).
Hussmann, K. L., Vandergaast, R., Zheng, K., Hoover, L. I. & Fredericksen, B. L. Structural proteins of West Nile virus are a major determinant of infectious particle production and fitness in astrocytes. J. Gen. Virol. 95, 1991–2003 (2014).
Hussmann, K. L., Samuel, M. A., Kim, K. S., Diamond, M. S. & Fredericksen, B. L. Differential replication of pathogenic and nonpathogenic strains of West Nile virus within astrocytes. J. Virol. 87, 2814–2822 (2013).
Palus, M. et al. Infection and injury of human astrocytes by tick-borne encephalitis virus. J. Gen. Virol. 95, 2411–2426 (2014).
van den Pol, A. N., Mao, G., Yang, Y., Ornaghi, S. & Davis, J. N. Zika virus targeting in the developing brain. J. Neurosci. 37, 2161–2175 (2017).
Deng, Y. Q. et al. Isolation, identification and genomic characterization of the Asian lineage Zika virus imported to China. Sci. China Life Sci. 59, 428–430 (2016).
Perera-Lecoin, M., Meertens, L., Carnec, X. & Amara, A. Flavivirus entry receptors: an update. Viruses 6, 69–88 (2014).
Sasaki, T. et al. Structural basis for Gas6-Axl signalling. EMBO J. 25, 80–87 (2006).
Rothlin, C. V., Ghosh, S., Zuniga, E. I., Oldstone, M. B. & Lemke, G. TAM receptors are pleiotropic inhibitors of the innate immune response. Cell 131, 1124–1136 (2007).
Bhattacharyya, S. et al. Enveloped viruses disable innate immune responses in dendritic cells by direct activation of TAM receptors. Cell Host Microbe 14, 136–147 (2013).
Porritt, R. A. & Hertzog, P. J. Dynamic control of type I IFN signalling by an integrated network of negative regulators. Trends Immunol. 36, 150–160 (2015).
Yockey, L. J. et al. Vaginal exposure to Zika virus during pregnancy leads to fetal brain infection. Cell 166, 1247–1256 e1244 (2016).
Lazear, H. M. et al. A mouse model of Zika virus pathogenesis. Cell Host Microbe 19, 720–730 (2016).
Cho, H., Shrestha, B., Sen, G. C. & Diamond, M. S. A role for IFIT2 in restricting West Nile virus infection in the brain. J. Virol. 87, 8363–8371 (2013).
Wang, Z. Y. et al. Axl is not an indispensable factor for Zika virus infection in mice. J. Gen. Virol. 98, 2061–2068 (2017).
Jagger, B. W. et al. Gestational stage and IFN-λ signaling regulate ZIKV infection in utero. Cell Host Microbe 22, 366–376 e363 (2017).
Wu, T. D. & Nacu, S. Fast and SNP-tolerant detection of complex variants and splicing in short reads. Bioinformatics 26, 873–881 (2010).
Anders, S., Pyl, P. T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
Benjamini, Y. & Hochberg, Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J. R. Stat. Soc. B 57, 289–300 (1995).
Eden, E., Navon, R., Steinfeld, I., Lipson, D. & Yakhini, Z. GOrilla: A tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinform. 10, 48 (2009).
Supek, F., Bosnjak, M., Skunca, N. & Smuc, T. REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS ONE 6, e21800 (2011).
Cheadle, C., Vawter, M. P., Freed, W. J. & Becker, K. G. Analysis of microarray data using Z score transformation. J. Mol. Diagn. 5, 73–81 (2003).
Acknowledgements
This work was supported by the National Science Foundation of China (81430030 to J.X.), the ShanghaiTech University Startup Fund (to B.J. and J.L.), the Shanghai Public Health Clinical Center (to X.Z.), the Developmental Center of Shanghai Shenkang Hospital (SHDC12014104 to X.Z.) and the Shanghai Advanced Biosafety and Pathogen Diagnostic Platform (15DZ2290200 to X.Z.).
Author information
Authors and Affiliations
Contributions
J.X. and J.L. conceived this study. Jian C., Y.-F.Y., B.J., X.Z., J.L. and J.X. designed the experiments. Jian C., Y.-F.Y., Y.Y., Yongquan H., S.-L.S., Y.-R.C., R.B., Y.-J.L. and J.L. performed the experiments. P.Z., Yunwen H. and L.L. prepared the ZIKV. Jun C. analysed the RNA-Seq data. Jian C., Y.-F.Y., J.L. and J.X. prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–11, Supplementary Table 1 and Supplementary Notes.
Rights and permissions
About this article
Cite this article
Chen, J., Yang, Yf., Yang, Y. et al. AXL promotes Zika virus infection in astrocytes by antagonizing type I interferon signalling. Nat Microbiol 3, 302–309 (2018). https://doi.org/10.1038/s41564-017-0092-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-017-0092-4
This article is cited by
-
Therapeutic targeting of the functionally elusive TAM receptor family
Nature Reviews Drug Discovery (2024)
-
Mechanistic insights into bone remodelling dysregulation by human viral pathogens
Nature Microbiology (2024)
-
Zika virus targets human trophoblast stem cells and prevents syncytialization in placental trophoblast organoids
Nature Communications (2023)
-
Revealing the characteristics of ZIKV infection through tissue-specific transcriptome sequencing analysis
BMC Genomics (2022)
-
Long noncoding RNA IRF1-AS is associated with peste des petits ruminants infection
Veterinary Research (2022)