Abstract
Oral epithelial cells discriminate between pathogenic and non-pathogenic stimuli, and only induce an inflammatory response when they are exposed to high levels of a potentially harmful microorganism. The pattern recognition receptors (PRRs) in epithelial cells that mediate this differential response are poorly understood. Here, we demonstrate that the ephrin type-A receptor 2 (EphA2) is an oral epithelial cell PRR that binds to exposed β-glucans on the surface of the fungal pathogen Candida albicans. Binding of C. albicans to EphA2 on oral epithelial cells activates signal transducer and activator of transcription 3 and mitogen-activated protein kinase signalling in an inoculum-dependent manner, and is required for induction of a proinflammatory and antifungal response. EphA2–/– mice have impaired inflammatory responses and reduced interleukin-17 signalling during oropharyngeal candidiasis, resulting in more severe disease. Our study reveals that EphA2 functions as a PRR for β-glucans that senses epithelial cell fungal burden and is required for the maximal mucosal inflammatory response to C. albicans.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
12 June 2018
In the version of this Article originally published, the authors described the ANT compound used in their study as 4-(2,5-dimethyl-1H-pyrrol-1-yl)-2-hydroxybenzoic acid (ANT). The authors now wish to clarify that the ANT compound used was actually a 2,5-dimethylpyrrolyl benzoic acid derivative1 that has been shown to inhibit not only the enzymatic activity of EphA2, but also several unrelated enzymes2. The description of the compound in the Article has now been changed to 4-(2,5-dimethyl-1H-pyrrol-1-yl)-2-hydroxybenzoic acid derivative (ANT) to reflect this.
13 February 2018
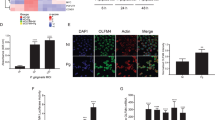
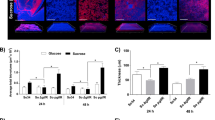
In the version of this Article originally published, technical problems led to errors in Figs. 2d and 5b. In the western blot panel in Fig. 2d, actin bands were misaligned at the bottom of the image and did not line up with the other bands in the panel; the corrected figure is shown below. In Fig. 5b, the upper right panel had an incorrect title of ‘CXCL3/KC’; it should have instead been ‘CXCL1/KC’. These errors have now been corrected in all versions of the Article.
References
Underhill, D. M. & Iliev, I. D. The mycobiota: interactions between commensal fungi and the host immune system. Nat. Rev. Immunol. 14, 405–416 (2014).
Salvatori, O., Puri, S., Tati, S. & Edgerton, M. Innate immunity and saliva in Candida albicans-mediated oral diseases. J. Dent. Res. 95, 365–371 (2016).
Conti, H. R. et al. IL-17 receptor signaling in oral epithelial cells is critical for protection against oropharyngeal candidiasis. Cell Host Microbe 20, 606–617 (2016).
Moyes, D. L. et al. A biphasic innate immune MAPK response discriminates between the yeast and hyphal forms of Candida albicans in epithelial cells. Cell Host Microbe 8, 225–235 (2010).
Moyes, D. L. et al. Protection against epithelial damage during Candida albicans infection is mediated by PI3K/Akt and mammalian target of rapamycin signaling. J. Infect. Dis. 209, 1816–1826 (2014).
Park, H. et al. Role of the fungal Ras-protein kinase A pathway in governing epithelial cell interactions during oropharyngeal candidiasis. Cell. Microbiol. 7, 499–510 (2005).
Wells, J. M., Rossi, O., Meijerink, M. & van Baarlen, P. Epithelial crosstalk at the microbiota-mucosal interface. Proc. Natl Acad. Sci. USA 1, 4607–4614 (2011).
Zhu, W. et al. EGFR and HER2 receptor kinase signaling mediate epithelial cell invasion by Candida albicans during oropharyngeal infection. Proc. Natl Acad. Sci. USA 109, 14194–14199 (2012).
Phan, Q. T. et al. Als3 is a Candida albicans invasin that binds to cadherins and induces endocytosis by host cells. PLoS Biol. 5, e64 (2007).
Swidergall, M. & Filler, S. G. Oropharyngeal candidiasis: fungal invasion and epithelial cell responses. PLoS Pathog. 13, e1006056 (2017).
Pasquale, E. B. Eph-ephrin bidirectional signaling in physiology and disease. Cell 133, 38–52 (2008).
Pitulescu, M. E. & Adams, R. H. Eph/ephrin molecules–a hub for signaling and endocytosis. Genes Dev. 24, 2480–2492 (2010).
Gantner, B. N., Simmons, R. M. & Underhill, D. M. Dectin-1 mediates macrophage recognition of Candida albicans yeast but not filaments. Embo J. 24, 1277–1286 (2005).
Wang, X. D. et al. Identification of candidate predictive and surrogate molecular markers for dasatinib in prostate cancer: rationale for patient selection and efficacy monitoring. Genome Biol. 8, R255 (2007).
Van Hoecke, A. et al. EPHA4 is a disease modifier of amyotrophic lateral sclerosis in animal models and in humans. Nat. Med. 18, 1418–1422 (2012).
Solis, N. V., Swidergall, M., Bruno, V. M., Gaffen, S. L. & Filler, S. G. The aryl hydrocarbon receptor governs epithelial cell invasion during oropharyngeal candidiasis. m Bio 8, e00025-17 (2017).
Larsen, A. B. et al. Activation of the EGFR gene target EphA2 inhibits epidermal growth factor-induced cancer cell motility. Mol. Cancer Res. 5, 283–293 (2007).
Naglik, J. R., Richardson, J. P. & Moyes, D. L. Candida albicans pathogenicity and epithelial immunity. PLoS Pathog. 10, e1004257 (2014).
Swidergall, M. & Ernst, J. F. Interplay between Candida albicans and the antimicrobial peptide armory. Eukaryot. Cell 13, 950–957 (2014).
Conti, H. R. et al. Th17 cells and IL-17 receptor signaling are essential for mucosal host defense against oral candidiasis. J. Exp. Med. 206, 299–311 (2009).
Li, A. et al. IL-22 up-regulates β-defensin-2 expression in human alveolar epithelium via STAT3 but not NF-κB signaling pathway. Inflammation 38, 1191–1200 (2015).
Siddiquee, K. et al. Selective chemical probe inhibitor of Stat3, identified through structure-based virtual screening, induces antitumor activity. Proc. Natl Acad. Sci. USA 104, 7391–7396 (2007).
Gantner, B. N., Simmons, R. M., Canavera, S. J., Akira, S. & Underhill, D. M. Collaborative induction of inflammatory responses by Dectin-1 and Toll-like Receptor 2. J. Exp. Med. 197, 1107–1117 (2003).
Ferwerda, G., Meyer-Wentrup, F., Kullberg, B. J., Netea, M. G. & Adema, G. J. Dectin-1 synergizes with TLR2 and TLR4 for cytokine production in human primary monocytes and macrophages. Cell. Microbiol. 10, 2058–2066 (2008).
Solis, N. V. & Filler, S. G. Mouse model of oropharyngeal candidiasis. Nat. Protoc. 7, 637–642 (2012).
Finney, A. C. et al. EphA2 expression regulates inflammation and fibroproliferative remodeling in atherosclerosis. Circulation 9, 026644 (2017).
Goupil, M. et al. Defective IL-17- and IL-22-dependent mucosal host response to Candida albicans determines susceptibility to oral candidiasis in mice expressing the HIV-1 transgene. BMC Immunol. 15, 49 (2014).
Trautwein-Weidner, K., Gladiator, A., Nur, S., Diethelm, P. & LeibundGut-Landmann, S. IL-17-mediated antifungal defense in the oral mucosa is independent of neutrophils. Mucosal Immunol. 8, 221–231 (2015).
Dickson, M. A. et al. Human keratinocytes that express hTERT and also bypass ap16(INK4a)-enforced mechanism that limits life span become immortal yet retain normal growth and differentiation characteristics. Mol. Cell. Biol. 20, 1436–1447 (2000).
Subbarayal, P. et al. EphrinA2 receptor (EphA2) is an invasion and intracellular signaling receptor for Chlamydia trachomatis. PLoS Pathog. 11, e1004846 (2015).
Noberini, R. et al. Small molecules can selectively inhibit ephrin binding to the EphA4 and EphA2 receptors. J. Biol. Chem. 283, 29461–29472 (2008).
Purvis, H. A., Anderson, A. E., Young, D. A., Isaacs, J. D. & Hilkens, C. M. A negative feedback loop mediated by STAT3 limits human Th17 responses. J. Immunol. 193, 1142–1150 (2014).
Bertuzzi, M. et al. The pH-responsive PacC transcription factor of Aspergillus fumigatus governs epithelial entry and tissue invasion during pulmonary aspergillosis. PLoS Pathog. 10, e1004413 (2014).
Fu, Y. et al. Investigation of the function of Candida albicans Als3 by heterologous expression in Candida glabrata. Infect. Immun. 81, 2528–2535 (2013).
Ariyachet, C. et al. SR-like RNA-binding protein Slr1 affects Candida albicans filamentation and virulence. Infect. Immun. 81, 1267–1276 (2013).
Phan, Q. T. & Filler, S. G. Endothelial cell stimulation by Candida albicans. Methods Mol. Biol. 470, 313–326 (2009).
Liu, H. et al. Aspergillus fumigatus CalA binds to integrin α5β1 and mediates host cell invasion. Nat. Microbiol. 2, 211 (2016).
Break, T. J. et al. CX(3)CR1 is dispensable for control of mucosal Candida albicans infections in mice and humans. Infect. Immun. 83, 958–965 (2015).
Acknowledgements
This work was supported in part by NIH grants R01DE022600 and R01AI124566 to S.G.F. and grant K99DE026856 to M.S. We are grateful to A.W. Orr and A.C. Finney for providing the EphA2–/– mice. We thank S.W. French and E. Vitocruz for histopathology; A.S. Ibrahim, M.R. Yeaman and J. Naglik for providing strains; and members of the Division of Infectious Diseases at Harbor-UCLA Medical Center for critical suggestions.
Author information
Authors and Affiliations
Contributions
M.S., N.V.S., M.S.L. and S.G.F. designed the experiments. M.S. and N.V.S. performed the experiments. M.S., N.V.S., M.S.L. and S.G.F. analysed the data. M.S. and S.G.F. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
S.G.F. is a co-founder of and shareholder in NovaDigm Therapeutics, Inc., a company that is developing a vaccine against mucosal and invasive Candida infections.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables 1–3, Supplementary Figures 1–34.
Rights and permissions
About this article
Cite this article
Swidergall, M., Solis, N.V., Lionakis, M.S. et al. EphA2 is an epithelial cell pattern recognition receptor for fungal β-glucans. Nat Microbiol 3, 53–61 (2018). https://doi.org/10.1038/s41564-017-0059-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-017-0059-5
This article is cited by
-
Immune responses to human fungal pathogens and therapeutic prospects
Nature Reviews Immunology (2023)
-
Morphogenic plasticity: the pathogenic attribute of Candida albicans
Current Genetics (2023)
-
IL-23 signaling prevents ferroptosis-driven renal immunopathology during candidiasis
Nature Communications (2022)
-
Immunosurveillance of Candida albicans commensalism by the adaptive immune system
Mucosal Immunology (2022)
-
TAGAP instructs Th17 differentiation by bridging Dectin activation to EPHB2 signaling in innate antifungal response
Nature Communications (2020)