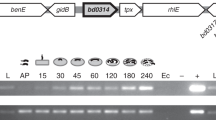
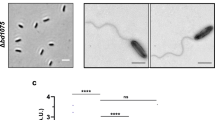
Abstract
Modification of essential bacterial peptidoglycan (PG)-containing cell walls can lead to antibiotic resistance; for example, β-lactam resistance by l,d-transpeptidase activities. Predatory Bdellovibrio bacteriovorus are naturally antibacterial and combat infections by traversing, modifying and finally destroying walls of Gram-negative prey bacteria, modifying their own PG as they grow inside prey. Historically, these multi-enzymatic processes on two similar PG walls have proved challenging to elucidate. Here, with a PG-labelling approach utilizing timed pulses of multiple fluorescent d-amino acids, we illuminate dynamic changes that predator and prey walls go through during the different phases of bacteria:bacteria invasion. We show formation of a reinforced circular port-hole in the prey wall, l,d-transpeptidaseBd-mediated d-amino acid modifications strengthening prey PG during Bdellovibrio invasion, and a zonal mode of predator elongation. This process is followed by unconventional, multi-point and synchronous septation of the intracellular Bdellovibrio, accommodating odd- and even-numbered progeny formation by non-binary division.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
08 January 2018
In the original version of this Article, a grant number and acknowledgement were omitted. The Acknowledgements section should have stated that one of the 3D SIM microscopes used for this research was supported by Medical Research Council UK grant (MR/K015753/1) to S. Foster, University of Sheffield, UK, and that the authors thank C. Walther and S. Foster for the access and their kind help with this. This has now been corrected in all versions of the Article.
References
Mainardi, J. L. et al. A novel peptidoglycan cross-linking enzyme for a β-lactam-resistant transpeptidation pathway. J. Biol. Chem. 280, 38146–38152 (2005).
Cava, F., de Pedro, M. A., Lam, H., Davis, B. M. & Waldor, M. K. Distinct pathways for modification of the bacterial cell wall by non-canonical d-amino acids. EMBO J. 30, 3442–3453 (2011).
Magnet, S. et al. Specificity of l,d-transpeptidases from Gram-positive bacteria producing different peptidoglycan chemotypes. J. Biol. Chem. 282, 13151–13159 (2007).
Fura, J. M., Kearns, D. & Pires, M. M. d-amino acid probes for penicillin binding protein-based bacterial surface labeling. J. Biol. Chem. 290, 30540–30550 (2015).
Gupta, R. et al. The Mycobacterium tuberculosis protein LdtMt2 is a nonclassical transpeptidase required for virulence and resistance to amoxicillin. Nat. Med. 16, 466–469 (2010).
Peltier, J. et al. Clostridium difficile has an original peptidoglycan structure with a high level of N-acetylglucosamine deacetylation and mainly 3-3 cross-links. J. Biol. Chem. 286, 29053–29062 (2011).
Lam, H. et al. d-amino acids govern stationary phase cell wall remodeling in bacteria. Science 325, 1552–1555 (2009).
Radkov, A. D. & Moe, L. A. Bacterial synthesis of d-amino acids. Appl. Microbiol. Biotechnol. 98, 5363–5374 (2014).
Kuru, E., Tekkam, S., Hall, E., Brun, Y. V. & Van Nieuwenhze, M. S. Synthesis of fluorescent d-amino acids and their use for probing peptidoglycan synthesis and bacterial growth in situ. Nat. Protoc. 10, 33–52 (2015).
Fleurie, A. et al. MapZ marks the division sites and positions FtsZ rings in Streptococcus pneumoniae. Nature 516, 259–262 (2014).
Tsui, H. C. et al. Pbp2x localizes separately from Pbp2b and other peptidoglycan synthesis proteins during later stages of cell division of D39. Mol. Microbiol. 94, 21–40 (2014).
Pilhofer, M. et al. Discovery of chlamydial peptidoglycan reveals bacteria with murein sacculi but without FtsZ. Nat. Commun. 4, 2856 (2013).
Stolp, H. & Starr, M. P. Bdellovibrio bacteriovorus gen. et sp. n., a predatory, ectoparasitic, and bacteriolytic microorganism. Antonie van Leeuwenhoek 29, 217–248 (1963).
Sockett, R. E. Predatory lifestyle of Bdellovibrio bacteriovorus. Annu. Rev. Microbiol. 63, 523–539 (2009).
Rittenberg, S. C. & Shilo, M. Early host damage in the infection cycle of Bdellovibrio bacteriovorus. J. Bacteriol. 102, 149–160 (1970).
Abram, D., Castro e Melo, J. & Chou, D. Penetration of Bdellovibrio bacteriovorus into host cells. J. Bacteriol. 118, 663–680 (1974).
Abram, D. & Davis, B. K. Structural properties and features of parasitic Bdellovibrio bacteriovorus. J. Bacteriol. 104, 948–965 (1970).
Lerner, T. R. et al. Specialized peptidoglycan hydrolases sculpt the intra-bacterial niche of predatory Bdellovibrio and increase population fitness. PLoS Pathog. 8, e1002524 (2012).
Lambert, C. et al. Ankyrin-mediated self-protection during cell invasion by the bacterial predator Bdellovibrio bacteriovorus. Nat. Commun. 6, 8884 (2015).
Lambert, C. et al. Interrupting peptidoglycan deacetylation during Bdellovibrio predator-prey interaction prevents ultimate destruction of prey wall, liberating bacterial-ghosts. Sci. Rep. 6, 26010 (2016).
Evans, K. J., Lambert, C. & Sockett, R. E. Predation by Bdellovibrio bacteriovorus HD100 requires type IV pili. J. Bacteriol. 189, 4850–4859 (2007).
Koval, S. F. et al. Bdellovibrio exovorus sp. nov., a novel predator of Caulobacter crescentus. Int. J. Syst. Evol. Microbiol. 63, 146–151 (2013).
Thomashow, M. F. & Rittenberg, S. C. Intraperiplasmic growth of Bdellovibrio bacteriovorus 109J: solubilization of Escherichia coli peptidoglycan. J. Bacteriol. 135, 998–1007 (1978).
Volle, C. B., Ferguson, M. A., Aidala, K. E., Spain, E. M. & Nunez, M. E. Quantitative changes in the elasticity and adhesive properties of Escherichia coli ZK1056 prey cells during predation by Bdellovibrio bacteriovorus 109J. Langmuir 24, 8102–8110 (2008).
Lambert, C., Chang, C. Y., Capeness, M. J. & Sockett, R. E. The first bite-profiling the predatosome in the bacterial pathogen Bdellovibrio. PLoS ONE 5, e8599 (2010).
Sanders, A. N. & Pavelka, M. S. Phenotypic analysis of Escherichia coli mutants lacking l,d-transpeptidases. Microbiology 159, 1842–1852 (2013).
Magnet, S. et al. Identification of the l,d-transpeptidases responsible for attachment of the Braun lipoprotein to Escherichia coli peptidoglycan. J. Bacteriol. 189, 3927–3931 (2007).
Pilizota, T. & Shaevitz, J. W. Origins of Escherichia coli growth rate and cell shape changes at high external osmolality. Biophys. J. 107, 1962–1969 (2014).
Pilizota, T. & Shaevitz, J. W. Plasmolysis and cell shape depend on solute outer-membrane permeability during hyperosmotic shock in E. coli. Biophys. J. 104, 2733–2742 (2013).
Fenton, A. K., Kanna, M., Woods, R. D., Aizawa, S. I. & Sockett, R. E. Shadowing the actions of a predator: backlit fluorescent microscopy reveals synchronous nonbinary septation of predatory Bdellovibrio inside prey and exit through discrete bdelloplast pores. J. Bacteriol. 192, 6329–6335 (2010).
Araki, Y. & Ruby, E. G. A soluble enzyme activity that attaches free diaminopimelic acid to bdelloplast peptidoglycan. Biochemistry 27, 2624–2629 (1988).
Shilo, M. Morphological and physiological aspects of the interaction of bdellovibrio with host bacteria. Curr. Top. Microbiol. Immunol. 50, 174–204 (1969).
Iida, Y. et al. Roles of multiple flagellins in flagellar formation and flagellar growth post bdelloplast lysis in Bdellovibrio bacteriovorus. J. Mol. Biol. 394, 1011–1021 (2009).
Eksztejn, M. & Varon, M. Elongation and cell division in Bdellovibrio bacteriovorus. Arch. Microbiol. 114, 175–181 (1977).
Kuru, E. et al. In situ probing of newly synthesized peptidoglycan in live bacteria with fluorescent d-amino acids. Angew. Chem. 51, 12519–12523 (2012).
Lambert, C. et al. Characterizing the flagellar filament and the role of motility in bacterial prey-penetration by Bdellovibrio bacteriovorus. Molec. Microbiol. 60, 274–286 (2006).
Lambert, C. & Sockett, R. Nucleases in Bdellovibrio bacteriovorus contribute towards efficient self-biofilm formation and eradication of pre-formed prey biofilms. FEMS Microbiol. Lett. 320, 109–116 (2013).
Magnet, S., Dubost, L., Marie, A., Arthur, M. & Gutmann, L. Identification of the l,d-transpeptidases for peptidoglycan cross-linking in Escherichia coli. J. Bacteriol. 190, 4782–4785 (2008).
Baba, T. et al. Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol. Syst. Biol. 2, 2006.0008 (2006).
Thomason, L. C., Sawitzke, J. A., Li, X., Constantino, N. & Court, D. L. Recombineering: genetic engineering in bacteria using homologous recombination. Curr. Protoc. Mol. Biol. 14, 1–39 (2014).
Datsenko, K. A. & Wanner, B. L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl Acad. Sci. USA 97, 6640–6645 (2000).
Fenton, A. K., Lambert, C., Wagstaff, P. C. & Sockett, R. E. Manipulating each MreB of Bdellovibrio bacteriovorus gives diverse morphological and predatory phenotypes. J. Bacteriol. 192, 1299–1311 (2010).
Ducret, A., Quardokus, E. M. & Brun, Y. V. MicrobeJ, a tool for high throughput bacterial cell detection and quantitative analysis. Nat. Microbiol. 1, 16077 (2016).
Acknowledgements
We thank D. Kearns and his laboratory (Indiana University, USA) for facilities and hospitality to culture B. bacteriovorus, A. Lovering (University of Birmingham, UK) for insights and assistance with the alignment of l,d-transpeptidase protein sequences in B. bacteriovorus, T. Pilizota (University of Edinburgh, UK) for advice on osmotic stress conditions, and R. Lowry (University of Nottingham, UK) for assistance in image acquisition. One of the 3D SIM microscopes used for this research was supported by Medical Research Council UK grant (MR/K015753/1) to S. Foster, University of Sheffield, UK, and we thank C. Walther and S. Foster for the access and their kind help with this. This work was supported by BBSRC grant [BB/M010325/1] to C.L., a Leverhulme Trust (UK) Research Leave Fellowship RF-2013-348 to R.E.S., NIH GM113172 grant to M.V.N. and Y.V.B. and R35GM122556 and GM51986 to Y.V.B. A.De. was supported by an EMBO long-term fellowship, and W.V. was supported by funds from the Wellcome Trust (101824/Z/13/Z).
Author information
Authors and Affiliations
Contributions
E.K. and R.E.S. conceived the study and carried out the experiments along with C.L. using reagents constructed by M.V.N. and J.R., and bacterial strains constructed by R.T. and A. De. J.G. and J.B. performed muropeptide analysis in the laboratory of W.V. A. Du. wrote code and aided C.L. and E.K. with image analysis. Y.V.B. provided microscopy facilities and with M.V.N. and W.V. provided helpful comments. E.K., C.L. and R.E.S. wrote the manuscript with input and comments from the other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A correction to this article is available online at https://doi.org/10.1038/s41564-017-0087-1.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–9, Supplementary Tables 1–3, Supplementary References.
Supplementary Video 1
3D-project of 3D-SIM scan of BADA-labelled Bdellovibrio cells (false coloured in red) preying upon E. coli cells pulse-labelled with HADA (false coloured in cyan), 15 minutes post-mixing.
Rights and permissions
About this article
Cite this article
Kuru, E., Lambert, C., Rittichier, J. et al. Fluorescent D-amino-acids reveal bi-cellular cell wall modifications important for Bdellovibrio bacteriovorus predation. Nat Microbiol 2, 1648–1657 (2017). https://doi.org/10.1038/s41564-017-0029-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-017-0029-y
This article is cited by
-
Omics for deciphering oral microecology
International Journal of Oral Science (2024)
-
Bdellovibrio bacteriovorus uses chimeric fibre proteins to recognize and invade a broad range of bacterial hosts
Nature Microbiology (2024)
-
Prey killing without invasion by Bdellovibrio bacteriovorus defective for a MIDAS-family adhesin
Nature Communications (2024)
-
Bdellovibrio predation cycle characterized at nanometre-scale resolution with cryo-electron tomography
Nature Microbiology (2023)
-
Learning with Bdellovibrio
Nature Microbiology (2023)