Abstract
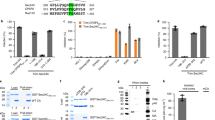
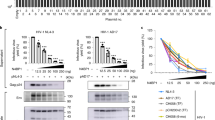
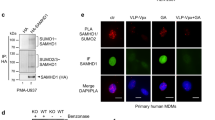
In this study, we report that the tetraspanin CD81 enhances human immunodeficiency virus (HIV)-1 reverse transcription in HIV-1-infected cells. This is enabled by the direct interaction of CD81 with the deoxynucleoside triphosphate phosphohydrolase SAMHD1. This interaction prevents endosomal accumulation and favours the proteasome-dependent degradation of SAMHD1. Consequently, CD81 depletion results in SAMHD1 increased expression, decreasing the availability of deoxynucleoside triphosphates (dNTP) and thus HIV-1 reverse transcription. Conversely, CD81 overexpression, but not the expression of a CD81 carboxy (C)-terminal deletion mutant, increases cellular dNTP content and HIV-1 reverse transcription. Our results demonstrate that the interaction of CD81 with SAMHD1 controls the metabolic rate of HIV-1 replication by tuning the availability of building blocks for reverse transcription, namely dNTPs. Together with its role in HIV-1 entry and budding into host cells, the data herein indicate that HIV-1 uses CD81 as a rheostat that controls different stages of the infection.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Blumenthal, R., Durell, S. & Viard, M. HIV entry and envelope glycoprotein-mediated fusion. J. Biol. Chem. 287, 40841–40849 (2012).
Arhel, N. Revisiting HIV-1 uncoating. Retrovirology 7, 96 (2010).
Ambrose, Z. & Aiken, C. HIV-1 uncoating: connection to nuclear entry and regulation by host proteins. Virology 454–455, 371–379 (2014).
Thali, M. The roles of tetraspanins in HIV-1 replication. Curr. Top. Microbiol. Immunol. 339, 85–102 (2009).
Rocha-Perugini, V., Gordon-Alonso, M. & Sanchez-Madrid, F. PIP: choreographer of actin-adaptor proteins in the HIV-1 dance. Trends Microbiol. 22, 379–388 (2014).
Yanez-Mo, M. et al. Tetraspanin-enriched microdomains: a functional unit in cell plasma membranes. Trends Cell Biol. 19, 434–446 (2009).
Levy, S. & Shoham, T. The tetraspanin web modulates immune-signalling complexes. Nat. Rev. Immunol. 5, 136–148 (2005).
van Spriel, A. B. & Figdor, C. G. The role of tetraspanins in the pathogenesis of infectious diseases. Microbes Infect. 12, 106–112 (2010).
Rocha-Perugini, V., Sanchez-Madrid, F. & Martinez Del Hoyo, G. Function and dynamics of tetraspanins during antigen recognition and immunological synapse formation. Front. Immunol. 6, 653 (2015).
Grigorov, B. et al. A role for CD81 on the late steps of HIV-1 replication in a chronically infected T cell line. Retrovirology 6, 28 (2009).
Ono, A. Relationships between plasma membrane microdomains and HIV-1 assembly. Biol. Cell 102, 335–350 (2010).
Thali, M. Tetraspanin functions during HIV-1 and influenza virus replication. Biochem. Soc. Trans. 39, 529–531 (2011).
Krementsov, D. N. et al. Tetraspanins regulate cell-to-cell transmission of HIV-1. Retrovirology 6, 64 (2009).
Sato, K. et al. Modulation of human immunodeficiency virus type 1 infectivity through incorporation of tetraspanin proteins. J. Virol. 82, 1021–1033 (2008).
Weng, J. et al. Formation of syncytia is repressed by tetraspanins in human immunodeficiency virus type 1-producing cells. J. Virol. 83, 7467–7474 (2009).
Yoshida, T. et al. A CD63 mutant inhibits T-cell tropic human immunodeficiency virus type 1 entry by disrupting CXCR4 trafficking to the plasma membrane. Traffic 9, 540–558 (2008).
Gordon-Alonso, M. et al. Tetraspanins CD9 and CD81 modulate HIV-1-induced membrane fusion. J. Immunol. 177, 5129–5137 (2006).
Lambele, M. et al. Vpu is the main determinant for tetraspanin downregulation in HIV-1-infected cells. J. Virol. 89, 3247–3255 (2015).
Li, G. et al. A post-entry role for CD63 in early HIV-1 replication. Virology 412, 315–324 (2011).
Franzolin, E. et al. The deoxynucleotide triphosphohydrolase SAMHD1 is a major regulator of DNA precursor pools in mammalian cells. Proc. Natl Acad. Sci. USA 110, 14272–14277 (2013).
Ballana, E. & Este, J. A. SAMHD1: at the crossroads of cell proliferation, immune responses, and virus restriction. Trends Microbiol. 23, 680–692 (2015).
Baldauf, H. M. et al. SAMHD1 restricts HIV-1 infection in resting CD4+ T cells. Nat. Med. 18, 1682–1687 (2012).
Lahouassa, H. et al. SAMHD1 restricts the replication of human immunodeficiency virus type 1 by depleting the intracellular pool of deoxynucleoside triphosphates. Nat. Immunol. 13, 223–228 (2012).
Ruffin, N. et al. Low SAMHD1 expression following T-cell activation and proliferation renders CD4+ T cells susceptible to HIV-1. Aids 29, 519–530 (2015).
Behrendt, R. et al. Mouse SAMHD1 has antiretroviral activity and suppresses a spontaneous cell-intrinsic antiviral response. Cell Rep. 4, 689–696 (2013).
Rehwinkel, J. et al. SAMHD1-dependent retroviral control and escape in mice. EMBO J. 32, 2454–2462 (2013).
Cribier, A. et al. Phosphorylation of SAMHD1 by cyclin A2/CDK1 regulates its restriction activity toward HIV-1. Cell Rep. 3, 1036–1043 (2013).
White, T. E. et al. The retroviral restriction ability of SAMHD1, but not its deoxynucleotide triphosphohydrolase activity, is regulated by phosphorylation. Cell Host Microbe 13, 441–451 (2013).
Yan, J. et al. CyclinA2-cyclin-dependent kinase regulates SAMHD1 protein phosphohydrolase domain. J. Biol. Chem. 290, 13279–13292 (2015).
Coiras, M. et al. IL-7 Induces SAMHD1 phosphorylation in CD4+ T lymphocytes, improving early steps of HIV-1 life cycle. Cell Rep. 14, 2100–2107 (2016).
Beloglazova, N. et al. Nuclease activity of the human SAMHD1 protein implicated in the Aicardi-Goutieres syndrome and HIV-1 restriction. J. Biol. Chem. 288, 8101–8110 (2013).
Choi, J. et al. SAMHD1 specifically restricts retroviruses through its RNase activity. Retrovirology 12, 46 (2015).
Ryoo, J. et al. The ribonuclease activity of SAMHD1 is required for HIV-1 restriction. Nat. Med. 20, 936–941 (2014).
Antonucci, J. M. et al. SAMHD1-mediated HIV-1 restriction in cells does not involve ribonuclease activity. Nat. Med. 22, 1072–1074 (2016).
Bhattacharya, A. et al. Effects of T592 phosphomimetic mutations on tetramer stability and dNTPase activity of SAMHD1 can not explain the retroviral restriction defect. Sci. Rep. 6, 31353 (2016).
Perez-Hernandez, D. et al. The intracellular interactome of tetraspanin-enriched microdomains reveals their function as sorting machineries toward exosomes. J. Biol. Chem. 288, 11649–11661 (2013).
Sala-Valdes, M. et al. EWI-2 and EWI-F link the tetraspanin web to the actin cytoskeleton through their direct association with ezrin-radixin-moesin proteins. J. Biol. Chem. 281, 19665–19675 (2006).
Tejera, E. et al. CD81 regulates cell migration through its association with Rac GTPase. Mol. Biol. Cell 24, 261–273 (2013).
Rocha-Perugini, V. et al. CD81 controls sustained T cell activation signaling and defines the maturation stages of cognate immunological synapses. Mol. Cell Biol. 33, 3644–3658 (2013).
Welbourn, S. et al. Identification and characterization of naturally occurring splice variants of SAMHD1. Retrovirology 9, 86 (2012).
Mauney, C.H. et al. The SAMHD1 dNTP triphosphohydrolase is controlled by a redox switch. Antioxid. Redox Signal. http://dx.doi.org/10.1089/ars.2016.6888 (2017).
Ahn, J. Functional organization of human SAMHD1 and mechanisms of HIV-1 restriction. Biol. Chem. 397, 373–379 (2016).
Pauls, E. et al. Cell cycle control and HIV-1 susceptibility are linked by CDK6-dependent CDK2 phosphorylation of SAMHD1 in myeloid and lymphoid cells. J. Immunol. 193, 1988–1997 (2014).
Welbourn, S. & Strebel, K. Low dNTP levels are necessary but may not be sufficient for lentiviral restriction by SAMHD1. Virology 488, 271–277 (2016).
Seamon, K. J. et al. SAMHD1 is a single-stranded nucleic acid binding protein with no active site-associated nuclease activity. Nucleic Acids Res. 43, 6486–6499 (2015).
Diamond, T. L. et al. Macrophage tropism of HIV-1 depends on efficient cellular dNTP utilization by reverse transcriptase. J. Biol. Chem. 279, 51545–51553 (2004).
Kyei, G. B., Cheng, X., Ramani, R. & Ratner, L. Cyclin L2 is a critical HIV dependency factor in macrophages that controls SAMHD1 abundance. Cell Host Microbe 17, 98–106 (2015).
Morrissey, C. et al. The eukaryotic elongation factor eEF1A1 interacts with SAMHD1. Biochem. J. 466, 69–76 (2015).
Yanez-Mo, M. et al. Regulation of endothelial cell motility by complexes of tetraspan molecules CD81/TAPA-1 and CD151/PETA-3 with α3β1 integrin localized at endothelial lateral junctions. J. Cell Biol. 141, 791–804 (1998).
Gordon-Alonso, M. et al. Actin-binding protein drebrin regulates HIV-1-triggered actin polymerization and viral infection. J. Biol. Chem. 288, 28382–28397 (2013).
Acknowledgements
The authors thank M. Vicente-Manzanares (Hospital de la Princesa, UAM, Spain) for critical reading of the manuscript. Microscopy was performed at CNIC Microscopy & Dynamic Imaging Unit. This work was supported by grants to S.L. (Translational Cancer Award from Stanford Cancer Institute, SPARK at Stanford, and the Breast Cancer Research program from the Department of Defense grant W81XWH-14-1-0397); to B.K. (R01 GM104198; R01 AI049784); to M.A.M.-F. (RD16/0025/0019; PI16/01863; CYTED 214RT0482); to F.S.-M. (SAF2014-55579-R; INDISNET-S2011/BMD-2332; ERC-2011-AdG 294340-GENTRIS; PIE13/00041) and to MY-M (BFU2014-55478-R; Fundación Ramón Areces; RYC-2012-11025); and was co-funded by Fondo Europeo de Desarrollo Regional (FEDER). The CNIC is supported by the Spanish Ministry of Economy and Competitiveness (MINECO) and the Pro CNIC Foundation. FV-C was supported by The American Association of Immunologist through a Careers in Immunology Fellowship; and HS by a FPI-UAM Fellowship.
Author information
Authors and Affiliations
Contributions
V.R.P., F.S.M. and M.Y.M. conceived and designed research. V.R.P., H.S., S.A., S.L.M., G.L. and M.Y.M. performed experimental work. S.A., F.V.C., S.L., B.K., M.A.M.F., F.S.M. and M.Y.M. provided reagents. V.R.P. and M.Y.M. analysed the data. V.R.P. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information
Supplementary Figures 1–8.
Rights and permissions
About this article
Cite this article
Rocha-Perugini, V., Suárez, H., Álvarez, S. et al. CD81 association with SAMHD1 enhances HIV-1 reverse transcription by increasing dNTP levels. Nat Microbiol 2, 1513–1522 (2017). https://doi.org/10.1038/s41564-017-0019-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41564-017-0019-0
This article is cited by
-
SAMHD1-induced endosomal FAK signaling promotes human renal clear cell carcinoma metastasis by activating Rac1-mediated lamellipodia protrusion
Experimental & Molecular Medicine (2023)
-
Human cytomegalovirus UL24 and UL43 products participate in SAMHD1 subcellular localization
VirusDisease (2022)
-
CD81, a new actor in the development of preeclampsia
Cellular & Molecular Immunology (2021)
-
Tetraspanins in the regulation of mast cell function
Medical Microbiology and Immunology (2020)
-
Tetraspanin CD81 regulates HSV-1 infection
Medical Microbiology and Immunology (2020)