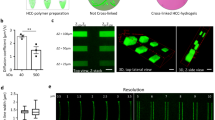
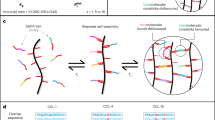
Abstract
Epithelial organoids are most efficiently grown from mouse-tumour-derived, reconstituted extracellular matrix hydrogels, whose poorly defined composition, batch-to-batch variability and immunogenicity limit clinical applications. Efforts to replace such ill-defined matrices for organoid culture have largely focused on non-adaptable hydrogels composed of covalently crosslinked hydrophilic macromolecules. However, the excessive forces caused by tissue expansion in such elastic gels severely restrict organoid growth and morphogenesis. Chemical or enzymatic degradation schemes can partially alleviate this problem, but due to their irreversibility, long-term applicability is limited. Here we report a family of synthetic hydrogels that promote extensive organoid morphogenesis through dynamic rearrangements mediated by reversible hydrogen bonding. These tunable matrices are stress relaxing and thus promote efficient crypt budding in intestinal stem-cell epithelia through increased symmetry breaking and Paneth cell formation dependent on yes-associated protein 1. As such, these well-defined gels provide promising versatile matrices for fostering elaborate in vitro morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets and the statistical analysis results that support the findings of this study are available in Zenodo with the identifier https://doi.org/10.5281/zenodo.5243103.
References
Rossi, G., Manfrin, A. & Lutolf, M. P. Progress and potential in organoid research. Nat. Rev. Genet. 19, 671–687 (2018).
Clevers, H. Modeling development and disease with organoids. Cell 165, 1586–1597 (2016).
Lancaster, M. A. & Knoblich, J. A. Organogenesis in a dish: modeling development and disease using organoid technologies. Science 345, 1247125 (2014).
Sasai, Y. Cytosystems dynamics in self-organization of tissue architecture. Nature 493, 318–326 (2013).
Huch, M., Knoblich, J. A., Lutolf, M. P. & Martinez-Arias, A. The hope and the hype of organoid research. Development 144, 938–941 (2017).
Gjorevski, N., Ranga, A. & Lutolf, M. P. Bioengineering approaches to guide stem cell-based organogenesis. Development 141, 1794–1804 (2014).
Blondel, D. & Lutolf, M. P. Bioinspired hydrogels for 3D organoid culture. Chimia 73, 81–85 (2019).
Wang, H. & Heilshorn, S. C. Adaptable hydrogel networks with reversible linkages for tissue engineering. Adv. Mater. 27, 3717–3736 (2015).
Gjorevski, N. et al. Designer matrices for intestinal stem cell and organoid culture. Nature 539, 560–564 (2016).
DiMarco, R. L., Dewi, R. E., Bernal, G., Kuo, C. & Heilshorn, S. C. Protein-engineered scaffolds for in vitro 3D culture of primary adult intestinal organoids. Biomater. Sci. 3, 1376–1385 (2015).
Ng, S., Tan, W. J., Pek, M. M. X., Tan, M.-H. & Kurisawa, M. Mechanically and chemically defined hydrogel matrices for patient-derived colorectal tumor organoid culture. Biomaterials 219, 119400 (2019).
Ranga, A. et al. Neural tube morphogenesis in synthetic 3D microenvironments. Proc. Natl Acad. Sci. USA 113, E6831–E6839 (2016).
Ye, S. et al. A chemically defined hydrogel for human liver organoid culture. Adv. Funct. Mater. 30, 2000893 (2020).
Rezakhani, S., Gjorevski, N. & Lutolf, M. P. Low-defect thiol-Michael addition hydrogels as Matrigel substitutes for epithelial organoid derivation. Adv. Funct. Mater. 30, 2000761 (2020).
Cruz-Acuña, R. et al. Synthetic hydrogels for human intestinal organoid generation and colonic wound repair. Nat. Cell Biol. 19, 1326–1335 (2017).
Hernandez-Gordillo, V. et al. Fully synthetic matrices for in vitro culture of primary human intestinal enteroids and endometrial organoids. Biomaterials 254, 120125 (2020).
Lutolf, M. P. & Hubbell, J. A. Synthesis and physicochemical characterization of end-linked poly(ethylene glycol)-co-peptide hydrogels formed by Michael-type addition. Biomacromolecules 4, 713–722 (2003).
Shugar, D. & Fox, J. J. Spectrophotometric studies of nucleic acid derivatives and related compounds as a function of pH. I. Pyrimidines. Biochim. Biophys. Acta 9, 199–218 (1952).
Dankers, P. Y. W. et al. Hierarchical formation of supramolecular transient networks in water: a modular injectable delivery system. Adv. Mater. 24, 2703–2709 (2012).
Ye, X. et al. Self-healing pH-sensitive cytosine- and guanosine-modified hyaluronic acid hydrogels via hydrogen bonding. Polymer 108, 348–360 (2017).
Bastings, M. M. C. et al. A fast pH-switchable and self-healing supramolecular hydrogel carrier for guided, local catheter injection in the infarcted myocardium. Adv. Healthc. Mater. 3, 70–78 (2014).
Hamada, N. & Einaga, Y. Effects of hydrophobic chain length on the characteristics of the micelles of octaoxyethylene tetradecyl C14E8, hexadecyl C16E8, and octadecyl C18E8 ethers. J. Phys. Chem. B 109, 6990–6998 (2005).
Génin, F., Quilès, F. & Burneau, A. Infrared and Raman spectroscopic study of carboxylic acids in heavy water. Phys. Chem. Chem. Phys. 3, 932–942 (2001).
Badasyan, A., Mavrič, A., Cigić, I. K., Bencik, T. & Valant, M. Polymer nanoparticle sizes from dynamic light scattering and size exclusion chromatography: the case study of polysilanes. Soft Matter 14, 4735–4740 (2018).
Coleman, M. M., Lee, K. H., Skrovanek, D. J. & Painter, P. C. Hydrogen bonding in polymers. 4. Infrared temperature studies of a simple polyurethane. Macromolecules 19, 2149–2157 (1986).
Cortese, J., Soulié-Ziakovic, C., Cloitre, M., Tencé-Girault, S. & Leibler, L. Order–disorder transition in supramolecular polymers. J. Am. Chem. Soc. 133, 19672–19675 (2011).
Chan, J. W., Hoyle, C. E., Lowe, A. B. & Bowman, M. Nucleophile-initiated thiol-Michael reactions: effect of organocatalyst, thiol, and ene. Macromolecules 43, 6381–6388 (2010).
Peppas, N. A. & Merrill, E. W. Poly(vinyl alcohol) hydrogels: reinforcement of radiation-crosslinked networks by crystallization. J. Polym. Sci. Polym. Chem. Ed. 14, 441–457 (1976).
Chaudhuri, O. Viscoelastic hydrogels for 3D cell culture. Biomater. Sci. 5, 1480–1490 (2017).
Dosh, R. H., Jordan-Mahy, N., Sammon, C. & Maitre, C. L. L. Use of L-pNIPAM hydrogel as a 3D-scaffold for intestinal crypts and stem cell tissue engineering. Biomater. Sci. 7, 4310–4324 (2019).
Babaei, B., Davarian, A., Pryse, K. M., Elson, E. L. & Genin, G. M. Efficient and optimized identification of generalized Maxwell viscoelastic relaxation spectra. J. Mech. Behav. Biomed. Mater. 55, 32–41 (2016).
Chaudhuri, O. et al. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 15, 326–334 (2016).
Zhao, X., Huebsch, N., Mooney, D. J. & Suo, Z. Stress-relaxation behavior in gels with ionic and covalent crosslinks. J. Appl. Phys. 107, 063509 (2010).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Sato, T. & Clevers, H. Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science 340, 1190–1194 (2013).
Yang, Q. et al. Cell fate coordinates mechano-osmotic forces in intestinal crypt formation. Nat. Cell Biol. 23, 733–744 (2021).
Serra, D. et al. Self-organization and symmetry breaking in intestinal organoid development. Nature 569, 66–72 (2019).
Broguiere, N. et al. Growth of epithelial organoids in a defined hydrogel. Adv. Mater. 30, e1801621 (2018).
Lee, H.-P., Gu, L., Mooney, D. J., Levenston, M. E. & Chaudhuri, O. Mechanical confinement regulates cartilage matrix formation by chondrocytes. Nat. Mater. 16, 1243–1251 (2017).
Yin, X. et al. Niche-independent high-purity cultures of Lgr5+ intestinal stem cells and their progeny. Nat. Methods 11, 106–112 (2014).
Panciera, T., Azzolin, L., Cordenonsi, M. & Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 18, 758–770 (2017).
Lukonin, I. et al. Phenotypic landscape of intestinal organoid regeneration. Nature 586, 275–280 (2020).
Chaudhuri, O. et al. Substrate stress relaxation regulates cell spreading. Nat. Commun. 6, 6364 (2015).
Broguiere, N., Formica, F. A., Barreto, G. & Zenobi-Wong, M. Sortase A as a cross-linking enzyme in tissue engineering. Acta Biomater. 77, 182–190 (2018).
Valdez, J. et al. On-demand dissolution of modular, synthetic extracellular matrix reveals local epithelial-stromal communication networks. Biomaterials 130, 90–103 (2017).
Fujii, M. et al. Human intestinal organoids maintain self-renewal capacity and cellular diversity in niche-inspired culture condition. Cell Stem Cell 23, 787–793 (2018).
He, S. et al. Stiffness regulates intestinal stem cell fate. Preprint at https://doi.org/10.1101/2021.03.15.435410 (2021).
Zhang, J. et al. Physically associated synthetic hydrogels with long-term covalent stabilization for cell culture and stem cell transplantation. Adv. Mater. 23, 5098–5103 (2011).
Shin, S. R. et al. Cell-laden microengineered and mechanically tunable hybrid hydrogels of gelatin and graphene oxide. Adv. Mater. 25, 6385–6391 (2013).
Hong, S. et al. 3D printing of highly stretchable and tough hydrogels into complex, cellularized structures. Adv. Mater. 27, 4035–4040 (2015).
Miyoshi, H. & Stappenbeck, T. S. In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat. Protoc. 8, 2471–2482 (2013).
Chen, I., Dorr, B. M. & Liu, D. R. A general strategy for the evolution of bond-forming enzymes using yeast display. Proc. Natl Acad. Sci. USA 108, 11399–11404 (2011).
Acknowledgements
We thank M. Meyer for help with stem-cell colony and organoid quantification, S. Gerber and B. Muriel for advice on the synthesis of hybrid gels, O. Mitrofanova for expanding hSI organoids and A. Manfrin for the advice on qPCR primer design. We thank M. Zenobi-Wong and D. Fercher for sortase production and the Université de Lausanne (UNIL) facility for peptide production for peptide synthesis. We acknowledge support from the Ecole Polytechnique Fédérale de Lausanne Bio Imaging & Optics Core Facility for image analysis, and from D. Pioletti and the DLL-Engineering facility at Ecole Polytechnique Fédérale de Lausanne for rheometer use. We thank H.-A. Klok for FTIR and DLS use and the Ecole Polytechnique Fédérale de Lausanne Gene Expression Core Facility for qPCR instrument use. We thank G. Schwank for providing the third hSI organoid line. This work was funded by the National Center of Competence in Research Bio-Inspired Materials for A.C. and D.B., the Swiss 3R Competence Center for S.R. and N.B., the EU Horizon 2020 research programme INTENS (http://www.intens.info/; no. 668294-2), the Personalized Health and Related Technologies Initiative from the Eidgenössische Technische Hochschule (ETH) Board and the Swiss National Science Foundation research grant no. 310030_179447.
Author information
Authors and Affiliations
Contributions
M.P.L. and D.B. conceived the initial idea. A.C., D.B. and M.P.L. designed experiments, analysed data and interpreted results. A.C. performed all key experiments for revisions. S.R. was involved in the human organoid culture. N.B. was involved in designing dithiol sortase peptide, sortase synthesis and the force displacement field measurement analysis. All authors read and provided feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks Eric Appel, Sina Bartfeld, Melissa Little and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Oscillatory rheological measurement of hydrogels.
a, Evolution of shear moduli over time for Hybrid50 gel at 37 °C (5% strain and 1 Hz) at different PEG content. Data were shown as mean ± S.D (shaded area) from (n = 3) independently prepared gels. b, Determination of gel point for Matrigel and Hybrid50 gel (3% w/v) defined as the time when G’ and G” crossed also phase angle below 45°, indicated by black arrow. The data was representative of (n = 3) independent measurements (5% strain and 1 Hz). c,d, Amplitude sweep (c) and frequency sweep (d) measurement for Matrigel, Hybrid50 and covalent gels at three different stiffnesses at 37 °C. Amplitude sweeps were done at 1 Hz from 1% to 300% strain. Frequency sweeps were performed at 5% strain from 0.01 to 10 Hz. Dotted line in amplitude sweep data (a) indicated the strain used for frequency sweep, still within linear viscoelastic region. Data were shown as mean ± S.D (shaded area) from (n = 3) independently prepared gels. Covalent and hybrid50 gels were swollen prior to measurement while Matrigel was measured subsequent to in-situ gelation. e, Post normalization analysis of frequency sweep results, revealing the influence of different response of hydrogels upon deformation at different time-scale. Data were shown as mean from (n = 3) independently prepared gels.
Extended Data Fig. 2 Stress relaxation curve fitting to generalized Maxwell-Wiechert 3 elements model.
a, Generalized Maxwell-Wiechert 3 elements model fitted perfectly in most of experimental data, determined by coefficient of determination (R2). Data shown as individual data points (Round grey) and curve fit of experimental data mean (triangle black) from (n = 3) independently prepared gels. b, Example of fitted experimental data and curve fit in various hydrogels system.
Extended Data Fig. 3 Stress relaxation curve fitting parameters.
a, Determination of curve fitting parameters for elastic component (σ0) and decay half-time (Td1/2). b-g, Elastic components and decay half-time for Matrigel (b), different cytosine functionalization (Hybrid25 and Hybrid65 gels) (c), different PEG precursors (Aniline50 and COOH50 gels) (d), Cytidine treatment of Hybrid50 gels (e), covalent gels at different stiffnesses (f) and hybrid50 gels at different stiffnesses (g). Data shown as individual data points (Round grey) and curve fit of experimental data mean (triangle black) from (n = 3) independently prepared gels. Statistical analysis was evaluated with one-ways ANOVA followed by Tukey post-hoc analysis (b-f) and unpaired two-tailed student t-test (g), P-values were presented above the data points.
Extended Data Fig. 4 Effect of RGD on mechanical properties of hydrogels.
a, A schematic illustrating functionalization the hydrogel with RGD motif through Michael-type addition between VS and cysteine. b, Effect of RGD to the bulk hydrogel stiffness for Covalent and Hybrid50 gels. We noticed drop of stiffness due to less crosslinking VS-TH at the given polymer content for both covalent and Hybrid50 gels. Data shown as individual data points and line represented mean ± S.D from (n = 3) independently prepared gels from frequency sweep measurement (5% strain and 0.01-10 Hz). c, Stress relaxation profile of the hydrogels with addition of RGD motif. Data were shown as mean ± S.D (shaded area) from (n = 3) independently prepared gels. d, Curve fitting parameters of stress relaxation profile. The stress relaxation (10% strain) of covalent hydrogel remained identical with and without RGD, Hybrid50 gels demonstrated more stress relaxation from 10% to 15% stress relaxed because of addition of 8-PEG-Cytosine50 precursors to satisfy 0.5 mM excess of VS group, but did not alter decay characteristic time. Data shown as individual data points (Round grey) and curve fit of experimental data mean (triangle black) from (n = 3) independently prepared gels. 0.5 mM of RGDSP were used on all experiments. Statistical analysis was evaluated with two-ways ANOVA followed by Tukey (b) and Bonferroni post-hoc analysis (d), P-values were presented above the data points.
Extended Data Fig. 5 3D displacement field of fluorophore beads during mouse intestinal organoid culture in covalent and Hybrid50 gels.
a, A schematic illustrating how the physical remodelling of the hybrid gel during the onset of budding was monitored using fluorescent beads. b, Representative images of displacement fields induced during the collapse and budding of cystic colonies. Orange arrows show a colony collapse before bud formation, and red arrows point to a collapsed colony breaking symmetry and extending an initial bud / bulge. c, The xy components of the displacement field in this plane were used for analysis of the displacement against distance decay curves. Various characteristic events were captured such as collapsing organoid, growth and bud extension. The yellow arrows indicated the direction of bead displacement around the mouse intestinal organoids. The brightfield images represented two different time points earlier and later respectively. The experiments were repeated (n = 2) independent experiments. Scale bar, 50 µm.
Extended Data Fig. 6 Organoids sampling grown in various hydrogels from day 03 to 07.
a, Representative images of sampled organoids from grown in various hydrogels to analyze the presence of Paneth cells (Lyz) and enterocyst (AldoB), also to perform morphological classification. b, Visual quantification of Lyz + (color-coded) over 5 days and clustering of morphologically different population at day 07. The numbers next to dot plot is number of Lyz+ organoids over total number of samples except for enterocyst. The presented data were taken from(n = 3 for day 07 and n = 2 for day 03-06) independent experiment. Scale bar, 50 µm.
Extended Data Fig. 7 Quantitative real-time PCR data of mouse intestinal organoids in synthetic hydrogels.
Heatmap of quantitative real-time PCR data showing the relative expression of mouse intestinal organoids grown in covalent and hybrid50 hydrogels from single cells over four days for Yap1 target genes (Ctgf and Cyr61) and symmetry breaking (Dll1, Atoh1 and Lyz1). The average fold changes were calculated relative to mISCs organoids in covalent gels, derived from (n = 3) independent experiments. Expression level was normalized to Gapdh and relative mRNA expression levels were shown as logarithmic scale (Log2).
Extended Data Fig. 8 Long-term human small intestinal organoids culture.
a, Representative images of hSI organoids (hSI Line 3) over five passages cultured in Hybrid50 hydrogels and Matrigels at day 06 of each passage. b, Quantification of cell number fold changes between day 00 and day 06 at each passage for three different hSI organoids lines expanded in Hybrid50 gels and Matrigel. Data shown as line represented mean ± S.D from (n = 3 hSI lines) with individual data points represented by symbols, hSI line 1 (Circles), hSI line 2 (Squares) and hSI line 3 (Triangles). Scale bar, 50 μm.
Extended Data Fig. 9 Quantitative real-time PCR data of three hSI organoids lines grown in SS-Hybrid50 hydrogels.
Quantitative real-time PCR data showing the relative expression of hSI organoids grown in SS-Hybrid50 gels for differentiated cells markers, stem cells (OLFM4, LGR5 and SOX9), Paneth cells (ATOH1, LYZ, REG3A and DEFA6), Goblet cells (MUC2 and SPINK4), Enteroendocrine cells (CHGA) and Enterocytes (ALDOB, FABP1, FABP2 and VIL1). The data were shown as mean (red line) relative expression to hSI organoids grown in Matrigel mean ± S.E.M (error bars) from three (n = 3) independent experiment (by circles, squares and triangles). Expression level was normalized to GAPDH and relative mRNA expression levels were shown as logarithmic scale (Log2). Statistical analysis was evaluated with two-ways ANOVA followed by Bonferroni post-hoc analysis and P-values were available above the data, statistically significant results were highlighted in red color (below 0.05).
Supplementary information
Supplementary Information
Supplementary Figs. 1–14, Tables 1–3 and descriptions of Videos 1–3.
Supplementary Video 1
Representative images of the budding process of mouse intestinal organoids cultured in the hybrid gel. There are two organoids in the video (left and middle). The left organoid first collapsed before bud generation. The organoid in the middle, which had already collapsed, demonstrated a more advanced budding structure.
Supplementary Video 2
Representative images of the budding structure of mouse intestinal organoid cultured in the hybrid gel. This organoid already had a budding structure, which is in extension.
Supplementary Video 3
Representative images of the mouse intestinal organoid cultured in the covalent gel. Three colonies are present in the frame. All of the colonies collapsed but failed to form any budding structure.
Rights and permissions
About this article
Cite this article
Chrisnandy, A., Blondel, D., Rezakhani, S. et al. Synthetic dynamic hydrogels promote degradation-independent in vitro organogenesis. Nat. Mater. 21, 479–487 (2022). https://doi.org/10.1038/s41563-021-01136-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-01136-7
This article is cited by
-
Hierarchical assembly of tryptophan zipper peptides into stress-relaxing bioactive hydrogels
Nature Communications (2023)
-
Biology-driven material design for ischaemic stroke repair
Nature Reviews Bioengineering (2023)
-
Engineering prostate cancer in vitro: what does it take?
Oncogene (2023)
-
Cell–extracellular matrix mechanotransduction in 3D
Nature Reviews Molecular Cell Biology (2023)
-
Integration of 3D-printed cerebral cortical tissue into an ex vivo lesioned brain slice
Nature Communications (2023)