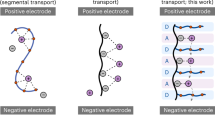
Abstract
A critical challenge for next-generation lithium-based batteries lies in development of electrolytes that enable thermal safety along with the use of high-energy-density electrodes. We describe molecular ionic composite electrolytes based on an aligned liquid crystalline polymer combined with ionic liquids and concentrated Li salt. This high strength (200 MPa) and non-flammable solid electrolyte possesses outstanding Li+ conductivity (1 mS cm−1 at 25 °C) and electrochemical stability (5.6 V versus Li|Li+) while suppressing dendrite growth and exhibiting low interfacial resistance (32 Ω cm2) and overpotentials (≤120 mV at 1 mA cm−2) during Li symmetric cell cycling. A heterogeneous salt doping process modifies a locally ordered polymer–ion assembly to incorporate an inter-grain network filled with defective LiFSI and LiBF4 nanocrystals, strongly enhancing Li+ conduction. This modular material fabrication platform shows promise for safe and high-energy-density energy storage and conversion applications, incorporating the fast transport of ceramic-like conductors with the superior flexibility of polymer electrolytes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated and analysed in this study are included in this published article and its Supplementary Information file and are also available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Wang, Y. et al. Highly conductive and thermally stable ion gels with tunable anisotropy and modulus. Adv. Mater. 28, 2571–2578 (2016) .
Christie, A. M., Lilley, S. J., Staunton, E., Andreev, Y. G. & Bruce, P. G. Increasing the conductivity of crystalline polymer electrolytes. Nature 433, 50–53 (2005).
Famprikis, T., Canepa, P., Dawson, J. A., Islam, M. S. & Masquelier, C. Fundamentals of inorganic solid-state electrolytes for batteries. Nat. Mater. 18, 1278–1291 (2019).
Manthiram, A., Yu, X. & Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2, 16103 (2017).
Lu, Y. Y., Tu, Z. Y. & Archer, L. A. Stable lithium electrodeposition in liquid and nanoporous solid electrolytes. Nat. Mater. 13, 961–969 (2014).
Qian, J. F. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Bruce, P. G., Freunberger, S. A., Hardwick, L. J. & Tarascon, J. M. Erratum: Li–O2 and Li–S batteries with high energy storage. Nat. Mater. 11, 172 (2012).
Krause, A. et al. High area capacity lithium-sulfur full-cell battery with prelitiathed silicon nanowire-carbon anodes for long cycling stability. Sci. Rep. 6, 27982 (2016).
Lu, J. et al. A lithium–oxygen battery based on lithium superoxide. Nature 529, 377–382 (2016).
Maier, J. Nanoionics: ion transport and electrochemical storage in confined systems. Nat. Mater. 4, 805–815 (2005).
Sakuda, A., Hayashi, A. & Tatsumisago, M. Sulfide solid electrolyte with favorable mechanical property for all-solid-state lithium battery. Sci. Rep. 3, 2261 (2013).
Elia, G. A. et al. An advanced lithium–air battery exploiting an ionic liquid-based electrolyte. Nano Lett. 14, 6572–6577 (2014).
Bostwick, J. E. et al. Ion transport and mechanical properties of non-crystallizable molecular ionic composite electrolytes. Macromolecules 53, 1405–1414 (2020).
Fox, R. J. et al. Nanofibrillar ionic polymer composites enable high-modulus ion-conducting membranes. ACS Appl. Mater. Interfaces 11, 40551–40563 (2019).
Yu, Z., He, Y. D., Wang, Y., Madsen, L. A. & Qiao, R. Molecular structure and dynamics of ionic liquids in a rigid-rod polyanion-based ion gel. Langmuir 33, 322–331 (2017).
Wang, Y. et al. Double helical conformation and extreme rigidity in a rodlike polyelectrolyte. Nat. Commun. 10, 801 (2019).
Wang, Y., Gao, J., Dingemans, T. J. & Madsen, L. A. Molecular alignment and ion transport in rigid rod polyelectrolyte solutions. Macromolecules 47, 2984–2992 (2014).
Lodge, T. P. A unique platform for materials design. Science 321, 50–51 (2008).
Lu, Y. Y., Korf, K., Kambe, Y., Tu, Z. Y. & Archer, L. A. Ionic-liquid–nanoparticle hybrid electrolytes: applications in lithium metal batteries. Angew. Chem. Int. Ed. 53, 488–492 (2014).
Wu, F. et al. “Liquid-in-solid” and “solid-in-liquid” electrolytes with high rate capacity and long cycling life for lithium-ion batteries. Chem. Mater. 28, 848–856 (2016).
Armand, M., Endres, F., MacFarlane, D. R., Ohno, H. & Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 8, 621–629 (2009).
Ueki, T. & Watanabe, M. Macromolecules in ionic liquids: progress, challenges, and opportunities. Macromolecules 41, 3739–3749 (2008).
Horowitz, A. I. & Panzer, M. J. High-performance, mechanically compliant silica-based ionogels for electrical energy storage applications. J. Mater. Chem. 22, 16534–16539 (2012).
Lodge, T. P. & Ueki, T. Mechanically tunable, readily processable ion gels by self-assembly of block copolymers in ionic liquids. Acc. Chem. Res 49, 2107–2114 (2016).
le Bideau, J., Ducros, J. B., Soudan, P. & Guyomard, D. Solid-state electrode materials with ionic-liquid properties for energy storage: the lithium solid-state ionic-liquid concept. Adv. Funct. Mater. 21, 4073–4078 (2011).
Bhattacharyya, A. J., Dolle, M. & Maier, J. Improved Li-battery electrolytes by heterogeneous doping of nonaqueous Li-salt solutions. Electrochem. Solid State Lett. 7, A432–A434 (2004).
MacFarlane, D. R. & Forsyth, M. Plastic crystal electrolyte materials: new perspectives on solid state ionics. Adv. Mater. 13, 957–966 (2001).
Alarco, P. J., Abu-Lebdeh, Y. & Armand, M. Highly conductive, organic plastic crystals based on pyrazolium imides. Solid State Ion. 175, 717–720 (2004).
Matsumoto, H. et al. Fast cycling of Li/LiCoO2 cell with low-viscosity ionic liquids based on bis(fluorosulfonyl)imide [FSI]–. J. Power Sources 160, 1308–1313 (2006).
Zaghib, K. et al. Safe Li-ion polymer batteries for HEV applications. J. Power Sources 134, 124–129 (2004).
Basile, A., Bhatt, A. I. & O’Mullane, A. P. Stabilizing lithium metal using ionic liquids for long-lived batteries. Nat. Commun. 7, ncomms11794 (2016).
Yoon, H., Howlett, P. C., Best, A. S., Forsyth, M. & MacFarlane, D. R. Fast charge/discharge of Li metal batteries using an ionic liquid electrolyte. J. Electrochem. Soc. 160, A1629–A1637 (2013).
Yoon, H., Best, A. S., Forsyth, M., MacFarlane, D. R. & Howlett, P. C. Physical properties of high Li-ion content N-propyl-N-methylpyrrolidinium bis(fluorosulfonyl)imide based ionic liquid electrolytes. Phys. Chem. Chem. Phys. 17, 4656–4663 (2015).
Kim, O., Kim, K., Choi, U. H. & Park, M. J. Tuning anhydrous proton conduction in single-ion polymers by crystalline ion channels. Nat. Commun. 9, 5029 (2018).
Patterson, A. L. The Scherrer formula for X-ray particle size determination. Phys. Rev. 56, 978–982 (1939).
Levitt, M. H. Spin Dynamics: Basics of Nuclear Magnetic Resonance (Wiley, 2001).
Marple, M., Aitken, B., Kim, S. & Sen, S. Fast Li-ion dynamics in stoichiometric Li2S–Ga2Se3–GeSe2 glasses. Chem. Mater. 29, 8704–8710 (2017).
Kidd, B. E., Forbey, S. J., Steuber, F. W., Moore, R. B. & Madsen, L. A. Multiscale lithium and counterion transport in an electrospun polymer-gel electrolyte. Macromolecules 48, 4481–4490 (2015).
Kidd, B. E., Lingwood, M. D., Lee, M., Gibson, H. W. & Madsen, L. A. Cation and anion transport in a dicationic imidazolium-based plastic crystal ion conductor. J. Phys. Chem. B 118, 2176–2185 (2014).
Lingwood, M. D. et al. Unraveling the local energetics of transport in a polymer ion conductor. Chem. Commun. 49, 4283–4285 (2013).
Hou, J. B., Zhang, Z. Y. & Madsen, L. A. Cation/anion associations in ionic liquids modulated by hydration and ionic medium. J. Phys. Chem. B 115, 4576–4582 (2011).
Simons, T. J. et al. Influence of Zn2+ and water on the transport properties of a pyrrolidinium dicyanamide ionic liquid. J. Phys. Chem. B 118, 4895–4905 (2014).
Jin, L. et al. Structure and transport properties of a plastic crystal ion conductor: diethyl(methyl)(isobutyl)phosphonium hexafluorophosphate. J. Am. Chem. Soc. 134, 9688–9697 (2012).
Zhu, H., MacFarlane, D. R., Pringle, J. M. & Forsyth, M. Organic ionic plastic crystals as solid-state electrolytes. Trends Chem. 1, 126–140 (2019).
MacFarlane, D. R. et al. Ionic liquids and their solid-state analogues as materials for energy generation and storage. Nat. Rev. Mater. 1, 15005 (2016).
Han, X. et al. Negating interfacial impedance in garnet-based solid-state Li metal batteries. Nat. Mater. 16, 572–579 (2017).
Kerner, M., Plylahan, N., Scheers, J. & Johansson, P. Thermal stability and decomposition of lithium bis(fluorosulfonyl)imide (LiFSI) salts. RSC Adv. 6, 23327–23334 (2016).
Zhao, Q., Liu, X., Stalin, S., Khan, K. & Archer, L. A. Solid-state polymer electrolytes with in-built fast interfacial transport for secondary lithium batteries. Nat. Energy 4, 365–373 (2019).
Gao, J. W. et al. Water and sodium transport and liquid crystalline alignment in a sulfonated aramid membrane. J. Membr. Sci. 489, 194–203 (2015).
Li, J., Park, J. K., Moore, R. B. & Madsen, L. A. Linear coupling of alignment with transport in a polymer electrolyte membrane. Nat. Mater. 10, 507–511 (2011).
Acknowledgements
This work was supported primarily by the US National Science Foundation under awards DMR 1507764 and 1810194 and in part by the US Department of Energy under award EE0008860. We also gratefully thank C. Slebodnick at the Virginia Tech Crystallography Lab for assistance with X-ray diffraction analysis.
Author information
Authors and Affiliations
Contributions
Y.W. designed and executed all major experiments and composed and edited article draughts. X.W., R.K., L.J. and M.F. performed and assisted with electrochemistry and impedance experiments and contributed written sections and editing to the article. C.J.Z. performed SSNMR experiments and contributed written sections and editing to the article. W.H.K. analysed the X-ray diffraction data and contributed written sections to the article. T.J.D. modified and supplied the polymer, conceived experiments and contributed written sections and editing to the article. L.A.M. conceived ideas, oversaw experiments, and composed and edited the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information: Nature Materials thanks Fannie Alloin and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–11, Notes 1–12 and Tables 1–4.
Source data
Source Data Fig. 2e,f
X-ray diffraction pattern and refinement results.
Source Data Fig. 3d–f,h
Diffusion and activation energy data.
Source Data Fig. 4a–f
Electrochemical data.
Source Data Fig. 5a,b
Thermal and mechanical data.
Source Data Fig. 6c,f
Li symmetric cell and energy-dispersive X-ray data.
Rights and permissions
About this article
Cite this article
Wang, Y., Zanelotti, C.J., Wang, X. et al. Solid-state rigid-rod polymer composite electrolytes with nanocrystalline lithium ion pathways. Nat. Mater. 20, 1255–1263 (2021). https://doi.org/10.1038/s41563-021-00995-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-00995-4
This article is cited by
-
Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries
Nature Energy (2024)
-
Self-assembled hydrated copper coordination compounds as ionic conductors for room temperature solid-state batteries
Nature Communications (2024)
-
Solid polymer electrolytes based on poly(ionic liquid-co-ethylene oxide) for room-temperature solid-state lithium batteries
Journal of Solid State Electrochemistry (2024)
-
Machine learning-guided discovery of ionic polymer electrolytes for lithium metal batteries
Nature Communications (2023)
-
Sequencing polymers to enable solid-state lithium batteries
Nature Materials (2023)