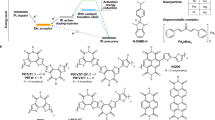
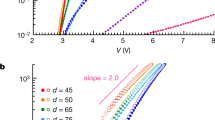
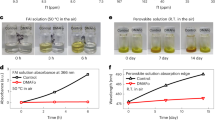
Abstract
Electronic doping of organic semiconductors is essential for their usage in highly efficient optoelectronic devices. Although molecular and metal complex-based dopants have already enabled significant progress of devices based on organic semiconductors, there remains a need for clean, efficient and low-cost dopants if a widespread transition towards larger-area organic electronic devices is to occur. Here we report dimethyl sulfoxide adducts as p-dopants that fulfil these conditions for a range of organic semiconductors. These adduct-based dopants are compatible with both solution and vapour-phase processing. We explore the doping mechanism and use the knowledge we gain to ‘decouple’ the dopants from the choice of counterion. We demonstrate that asymmetric p-doping is possible using solution processing routes, and demonstrate its use in metal halide perovskite solar cells, organic thin-film transistors and organic light-emitting diodes, which showcases the versatility of this doping approach.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The datasets used in this work are available in the Oxford University Research Archive repository48.
References
Granström, M. et al. Laminated fabrication of polymeric photovoltaic diodes. Nature 395, 257–260 (1998).
Halls, J. J. M. et al. Efficient photodiodes from interpenetrating polymer networks. Nature 376, 498–500 (1995).
Sirringhaus, H. et al. Two-dimensional charge transport in self-organized, high-mobility conjugated polymers. Nature 401, 685–688 (1999).
Tang, C. W. & Vanslyke, S. A. Organic electroluminescent diodes. Appl. Phys. Lett. 51, 913–915 (1987).
Yu, G., Gao, J., Hummelen, J. C., Wudl, F. & Heeger, A. J. Polymer photovoltaic cells: enhanced efficiencies via a network of internal donor–acceptor heterojunctions. Science 270, 1789–1791 (1995).
Blochwitz, J., Pfeiffer, M., Fritz, T. & Leo, K. Low voltage organic light emitting diodes featuring doped phthalocyanine as hole transport material. Appl. Phys. Lett. 73, 729–731 (1998).
Maennig, B. et al. Controlled p-type doping of polycrystalline and amorphous organic layers: self-consistent description of conductivity and field-effect mobility by a microscopic percolation mode. Phys. Rev. B 64, 195208 (2001).
Godet, C. Variable range hopping revisited: the case of an exponential distribution of localized states. J. Non-Cryst. Solids 302, 333–338 (2002).
Rubel, O., Baranovskii, S. D., Thomas, P. & Yamasaki, S. Concentration dependence of the hopping mobility in disordered organic solids. Phys. Stat. Sol. 171, 168–171 (2004).
Blochwitz, J. et al. Interface electronic structure of organic semiconductors with controlled doping levels. Org. Electron. 2, 97–104 (2001).
Gao, W. & Kahn, A. Controlled p-doping of zinc phthalocyanine by coevaporation with tetrafluorotetracyanoquinodimethane: a direct and inverse photoemission study. Appl. Phys. Lett. 79, 4040–4042 (2001).
Harada, K. et al. Organic homojunction diodes with a high built-in potential: interpretation of the current–voltage characteristics by a generalized Einstein relation. Phys. Rev. Lett. 94, 36601 (2005).
Chan, C. K., Zhao, W., Barlow, S., Marder, S. & Kahn, A. Decamethylcobaltocene as an efficient n-dopant in organic electronic materials and devices. Org. Electron. 9, 575–581 (2008).
Lin, X. et al. Beating the thermodynamic limit with photo-activation of n-doping in organic semiconductors. Nat. Mater. 16, 1209–1215 (2017).
Tietze, M. L. et al. Elementary steps in electrical doping of organic semiconductors. Nat. Commun. 9, 1182 (2018).
Tietze, M. L., Burtone, L., Riede, M., Lüssem, B. & Leo, K. Fermi level shift and doping efficiency in p-doped small molecule organic semiconductors: a photoelectron spectroscopy and theoretical study. Phys. Rev. B 035320, 1–12 (2012).
Li, J. et al. Measurement of small molecular dopant F4TCNQ and C60F36 diffusion in organic bilayer architectures. ACS Appl. Mater. Interfaces 7, 28420–28428 (2015).
Kolesov, V. A. et al. Solution-based electrical doping of semiconducting polymer films over a limited depth. Nat. Mater. 16, 474–481 (2017).
Yusubov, M. S., Filimonov, V. D. & Ogorodnikov, V. D. Dimethyl sulfoxide–hydrobromic acid as a novel reagent for convenient oxidation on a preparative scale of stilbenes and some derivatives of diphenylethane to benzils. Bull. Acad. Sci. USSR Div. Chem. Sci. 40, 766–770 (1991).
Lee, T. V. Oxidation adjacent to oxygen of alcohols by activated DMSO methods. Compr. Org. Synth. 7, 291–303 (1991).
Floyd, M. B., Du, M. T., Fabio, P. F., Jacob, L. A. & Johnson, B. D. The oxidation of acetophenones to arylglyoxals with aqueous hydrobromic acid in dimethyl sulfoxide. J. Org. Chem. 50, 5022–5027 (1985).
Sakai, N. et al. Solution-processed cesium hexabromopalladate(iv), Cs2PdBr6, for optoelectronic applications. J. Am. Chem. Soc. 139, 6030–6033 (2017).
Cappel, U. B., Daeneke, T. & Bach, U. Oxygen-induced doping of spiro-MeOTAD in solid-state dye-sensitized solar cells and its impact on device performance. Nano Lett. 12, 4925–4931 (2012).
Burschka, J. et al. Tris(2-(1H-pyrazol-1-yl)pyridine)cobalt(iii) as p-type dopant for organic semiconductors and Its application in highly efficient solid-state dye-sensitized solar cells. J. Am. Chem. Soc. 133, 18042–18045 (2011).
Planells, M. et al. Diacetylene bridged triphenylamines as hole transport materials for solid state dye sensitized solar cells. J. Mater. Chem. A 1, 6949–6960 (2013).
Abate, A. et al. Protic ionic liquids as p-dopant for organic hole transporting materials and their application in high efficiency hybrid solar cells. J. Am. Chem. Soc. 135, 13538–13548 (2013).
Pellaroque, A. et al. Efficient and stable perovskite solar cells using molybdenum tris(dithiolene)s as p-dopants for spiro-OMeTAD. ACS Energy Lett. 2, 2044–2050 (2017).
Nguyen, W. H., Bailie, C. D., Unger, E. L. & McGehee, M. D. Enhancing the hole-conductivity of spiro-OMeTAD without oxygen or lithium salts by using spiro(TFSI)2 in perovskite and dye-sensitized solar cells. J. Am. Chem. Soc. 136, 10996–11001 (2014).
Chen, C. et al. Cu(ii) complexes as p-type dopants in efficient perovskite solar cells. ACS Energy Lett. 2, 497–503 (2017).
Ono, L. K. et al. Air-exposure-induced gas-molecule incorporation into spiro-MeOTAD films. J. Phys. Chem. Lett. 5, 1374–1379 (2014).
Fawcett, W. R. & Kloss, A. A. Solvent-induced frequency shifts in the infrared spectrum of dimethyl sulfoxide in organic solvents. J. Phys. Chem. 100, 2019–2024 (1996).
Wallace, V. M., Dhumal, N. R., Zehentbauer, F. M., Kim, H. J. & Kiefer, J. Revisiting the aqueous solutions of dimethyl sulfoxide by spectroscopy in the mid- and near-infrared: experiments and Car–Parrinello simulations. J. Phys. Chem. B 119, 14780–14789 (2015).
Zhao, Y. H., Abraham, M. H. & Zissimos, A. M. Fast calculation of van der Waals volume as a sum of atomic and bond contributions and its application to drug compounds. J. Org. Chem. 68, 7368–7373 (2003).
Kang, K. et al. 2D coherent charge transport in highly ordered conducting polymers doped by solid state diffusion. Nat. Mater. 15, 896–902 (2016).
Ávila, J. et al. High voltage vacuum-deposited CH3NH3PbI3–CH3NH3PbI3 tandem solar cells. Energy Environ. Sci. 11, 3292–3297 (2018).
Abdi-Jalebi, M. et al. Charge extraction via graded doping of hole transport layers gives highly luminescent and stable metal halide perovskite devices. Sci. Adv. 5, eaav2012 (2019).
Wanlass, M. Systems and methods for advanced ultra-high-performance InP solar cells. US patent 9590131B2 (2017).
Walzer, K., Maennig, B., Pfeiffer, M. & Leo, K. Highly efficient organic devices based on electrically doped transport layers. Chem. Rev. 107, 1233–1271 (2007).
Pfeiffer, M., Beyer, A., Fritz, T. & Leo, K. Controlled doping of phthalocyanine layers by cosublimation with acceptor molecules: a systematic Seebeck and conductivity study. Appl. Phys. Lett. 73, 3202–3204 (1998).
Nollau, A., Pfeiffer, M., Fritz, T. & Leo, K. Controlled n-type doping of a molecular organic semiconductor: naphthalenetetracarboxylic dianhydride (NTCDA) doped with bis(ethylenedithio)-tetrathiafulvalene (BEDT-TTF). J. Appl. Phys. 87, 4340–4343 (2000).
Kirchartz, T. et al. Sensitivity of the Mott–Schottky analysis in organic solar cells. J. Phys. Chem. C. 116, 7672–7680 (2012).
Deledalle, F. et al. Understanding the effect of unintentional doping on transport optimization and analysis in efficient organic bulk-heterojunction solar cells. Phys. Rev. X 5, 11032 (2015).
Zonno, I., Martinez-Otero, A., Hebig, J. & Kirchartz, T. Understanding Mott–Schottky measurements under illumination in organic bulk heterojunction solar cells. Phys. Rev. Appl. 7, 034018 (2017).
Lin, Y.-H. et al. Deciphering photocarrier dynamics for tuneable high-performance perovskite–organic semiconductor heterojunction phototransistors. Nat. Commun. 10, 4475 (2019).
Lin, Y.-H. et al. Hybrid organic–metal oxide multilayer channel transistors with high operational stability. Nat. Electron. 2, 587–595 (2019).
Anaraki, E. H. et al. Highly efficient and stable planar perovskite solar cells by solution-processed tin oxide. Energy Environ. Sci. 9, 3128–3134 (2016).
Huang, Q. et al. Highly efficient top emitting organic light-emitting diodes with organic outcoupling enhancement layers. Appl. Phys. Lett. 88, 113515 (2006).
Sakai, N. et al. Adduct-Based p-Doping of Organic Semiconductors (Oxford University Research Archive, 2021); https://doi.org/10.5287/bodleian:zrMDxRzzB
Acknowledgements
This research has mainly received funding from the European Commission (PERTPV- agreement no. 763977) and EPSRC (EP/M005143/1 and EP/S004947/1). M.R. has received funding from the EC FP 7 MSCA—Career Integration Grant (630864) and M.R. and S.V.K. acknowledge funding from the EPSRC WAFT project (EP/M015173/1). R.W. is supported by EPSRC CDT Plastic Electronics (EP/L016702/1). P.K.N. acknowledges support from the Department of Atomic Energy, Government of India, under Project Identification no. RTI 4007 and SERB India core research grant (CRG/2020/003877). F.Z., X.L. and A.K. acknowledge funding from National Science Foundation under grants DMR-1506097 and DMR-1807797. S.N. acknowledges Marie Skłodowska-Curie Actions individual fellowships (grant agreement no. 659306) and a start-up grant from CSIR-IMMT, India. T.M. and V.G. acknowledge funding from European Regional Development Fund (project no. 01.2.2-LMT-K-718-03-0040) under a grant agreement with the Research Council of Lithuania (LMTLT). T.D.A. and A.B. are grateful to King Abdullah University of Science and Technology (KAUST), KAUST Solar Centre and KAUST Office for Sponsored Research (OSR) for the financial support under award no: OSR-2019-CRG8-4095, no. OSR-2018-CARF/CCF-3079. J.L. and C.G. are grateful for support for the NanoSIMS facility from EPSRC under grant EP/M018237/1. We thank I. McPherson for his help in mass spectrometry measurements and M. Heeney for providing the C16IDT-BT polymer.
Author information
Authors and Affiliations
Contributions
N.S. and P.K.N. conceived and executed the initial proof-of-concept experiments and unravelled the mechanism of doping. P.K.N. proposed the dopant system. H.J.S. proposed the asymmetric doping and N.S. designed and performed the experiments. N.S. and R.W. performed the conductivity measurements. N.S. and R.W. performed the doping stability test under the supervision of M.R. S.V.K. performed the ellipsometry measurements, analyses and simulation under the supervision of M.R. R.W. fabricated the OLEDs under the supervision of M.R. and N.S. fabricated all the other the devices used in this work. S.N. and P.K.N. performed the attenuated total reflection FTIR measurements. F.Z. and X.L. did the UPS, XPS and Kelvin probe measurements under the supervision of A.K. F.Z. did the AFM measurements. J.L. did the nanosecondary ion mass spectrometry measurements with inputs from P.K.N. and N.S. C.G. planned and helped interpret the nanosecondary ion mass spectrometry measurements. N.S. and Y.-H.L. performed the capacitance–voltage measurements. H.S.B. performed the quantum chemical calculations. T.M. conducted the synthesis of the HTM V886 and V.G. supervised the synthesis. A.B. fabricated the OTFTs and performed the electrical characterization under the supervision of T.D.A. T.D.A., Y.-H.L. and A.B. interpreted the results and provided the analysis of the OTFTs. N.S. and P.K.N. wrote the first draft. All the authors contributed to the analysis of the results, discussion of the content and revisions of the manuscript. P.K.N. and H.J.S. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
A patent based on this work has been filed (international application number PCT/GB2018/053014) by the University of Oxford. H.J.S. is a co-founder of Oxford PV Ltd and Helio Display Materials. The remaining authors declare no competing interests.
Additional information
Peer review information Nature Materials thanks Adam Moule and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Characterization, Supplementary Figs. 1–42, Notes 1–9 and Tables 1–6.
Rights and permissions
About this article
Cite this article
Sakai, N., Warren, R., Zhang, F. et al. Adduct-based p-doping of organic semiconductors. Nat. Mater. 20, 1248–1254 (2021). https://doi.org/10.1038/s41563-021-00980-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-021-00980-x
This article is cited by
-
Photoexcitation dynamics and energy engineering in supramolecular doping of organic conjugated molecules
Light: Science & Applications (2023)
-
Investigating the electrical crosstalk effect between pixels in high-resolution organic light-emitting diode microdisplays
Scientific Reports (2023)
-
Highly conductive tissue-like hydrogel interface through template-directed assembly
Nature Communications (2023)
-
Gelation of Hole Transport Layer to Improve the Stability of Perovskite Solar Cells
Nano-Micro Letters (2023)
-
Improved efficiency of red perovskite quantum dots-based light-emitting diodes by thickness tuning of PEDOT:PSS
Journal of Materials Science: Materials in Electronics (2022)