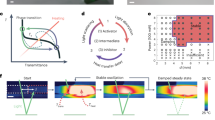
Abstract
The development of synthetic structures that mimic mechanical actuation in living matter such as autonomous translation and shape changes remains a grand challenge for materials science. In living systems the integration of supramolecular structures and covalent polymers contributes to the responsive behaviour of membranes, muscles and tendons, among others. Here we describe hybrid light-responsive soft materials composed of peptide amphiphile supramolecular polymers chemically bonded to spiropyran-based networks that expel water in response to visible light. The supramolecular polymers form a reversibly deformable and water-draining skeleton that mechanically reinforces the hybrid and can also be aligned by printing methods. The noncovalent skeleton embedded in the network thus enables faster bending and flattening actuation of objects, as well as longer steps during the light-driven crawling motion of macroscopic films. Our work suggests that hybrid bonding polymers, which integrate supramolecular assemblies and covalent networks, offer strategies for the bottom-up design of soft matter that mimics living organisms.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within the article and its Supplementary Information and from the corresponding authors upon reasonable request.
References
Roy, D., Brooks, W. L. A. & Sumerlin, B. S. New directions in thermoresponsive polymers. Chem. Soc. Rev. 42, 7214–7243 (2013).
Kocak, G., Tuncer, C. & Butun, V. pH-responsive polymers. Polym. Chem. 8, 144–176 (2017).
Zarzar, L. D. & Aizenberg, J. Stimuli-responsive chemomechanical actuation: a hybrid materials approach. Acc. Chem. Res. 47, 530–539 (2014).
Fukino, T., Yamagishi, H. & Aida, T. Redox-responsive molecular systems and materials. Adv. Mater. 29, 1603888 (2017).
Russew, M. M. & Hecht, S. Photoswitches: from molecules to materials. Adv. Mater. 22, 3348–3360 (2010).
Szymanski, W., Beierle, J. M., Kistemaker, H. A. V., Velema, W. A. & Feringa, B. L. Reversible photocontrol of biological systems by the incorporation of molecular photoswitches. Chem. Rev. 113, 6114–6178 (2013).
Gelebart, A. H. et al. Making waves in a photoactive polymer film. Nature 546, 632–636 (2017).
Wani, O. M., Zeng, H. & Priimagi, A. A light-driven artificial flytrap. Nat. Commun. 8, 15546 (2017).
Lancia, F., Ryabchun, A. & Katsonis, N. Life-like motion driven by artificial molecular machines. Nat. Rev. Chem. 3, 536–551 (2019).
Iwaso, K., Takashima, Y. & Harada, A. Fast response dry-type artificial molecular muscles with [c2]daisy chains. Nat. Chem. 8, 625–632 (2016).
Chen, J. W. et al. Artificial muscle-like function from hierarchical supramolecular assembly of photoresponsive molecular motors. Nat. Chem. 10, 132–138 (2018).
Yu, Z. L. et al. Simultaneous covalent and noncovalent hybrid polymerizations. Science 351, 497–502 (2016).
Chin, S. M. et al. Covalent-supramolecular hybrid polymers as muscle-inspired anisotropic actuators. Nat. Commun. 9, 2395 (2018).
Ionov, L. Hydrogel-based actuators: possibilities and limitations. Mater. Today 17, 494–503 (2014).
Klajn, R. Spiropyran-based dynamic materials. Chem. Soc. Rev. 43, 148–184 (2014).
Satoh, T., Sumaru, K., Takagi, T. & Kanamori, T. Fast-reversible light-driven hydrogels consisting of spirobenzopyran-functionalized poly(N-isopropylacrylamide). Soft Matter 7, 8030–8034 (2011).
Marrink, S. J., Risselada, H. J., Yefimov, S., Tieleman, D. P. & de Vries, A. H. The MARTINI force field: Coarse grained model for biomolecular simulations. J. Phys. Chem. B 111, 7812–7824 (2007).
Ziolkowski, B., Florea, L., Theobald, J., Benito-Lopez, F. & Diamond, D. Self-protonating spiropyran-co-NIPAM-co-acrylic acid hydrogel photoactuators. Soft Matter 9, 8754–8760 (2013).
Le, X. X., Lu, W., Zhang, J. W. & Chen, T. Recent progress in biomimetic anisotropic hydrogel actuators. Adv. Sci. 6, 1801584 (2019).
Satoh, T., Sumaru, K., Takagi, T., Takai, K. & Kanamori, T. Isomerization of spirobenzopyrans bearing electron-donating and electron-withdrawing groups in acidic aqueous solutions. Phys. Chem. Chem. Phys. 13, 7322–7329 (2011).
Kuksenok, O. & Balazs, A. C. Stimuli-responsive behavior of composites integrating thermo-responsive gels with photo-responsive fibers. Mater. Horiz. 3, 53–62 (2016).
Kuksenok, O. & Balazs, A. C. Designing polymer gels and composites that undergo bio-inspired phototactic reconfiguration and motion. Bioinspir. Biomim. 13, 035004 (2018).
Maeda, S., Hara, Y., Sakai, T., Yoshida, R. & Hashimoto, S. Self-walking gel. Adv. Mater. 19, 3480–3484 (2007).
Francis, W., Dunne, A., Delaney, C., Florea, L. & Diamond, D. Spiropyran based hydrogels actuators—walking in the light. Sens. Actuat. B Chem. 250, 608–616 (2017).
Ortony, J. H. et al. Water dynamics from the surface to the interior of a supramolecular nanostructure. J. Am. Chem. Soc. 139, 8915–8921 (2017).
Toby, B. H. & von Dreele, R. B. GSAS-II: the genesis of a modern open-source all purpose crystallography software package. J. Appl. Crystallogr. 46, 544–549 (2013).
Ilavsky, J. & Jemian, P. R. Irena: tool suite for modeling and analysis of small-angle scattering. J. Appl. Crystallogr. 42, 347–353 (2009).
Wang, J. M., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004).
Schmidt, M. W. et al. General atomic and molecular electronic-structure system. J. Comput. Chem. 14, 1347–1363 (1993).
Bayly, C. I., Cieplak, P., Cornell, W. D. & Kollman, P. A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 97, 10269–10280 (1993).
Case, D. A. et al. AMBER 2016 (University of California, San Francisco, 2016).
Lee, O. S., Cho, V. & Schatz, G. C. Modeling the self-assembly of peptide amphiphiles into fibers using coarse-grained molecular dynamics. Nano Lett. 12, 4907–4913 (2012).
van Gunsteren, W. F. & Berendsen, H. J. C. Thermodynamic cycle integration by computer simulation as a tool for obtaining free energy differences in molecular chemistry. J. Comput. Aided Mol. Des. 1, 171–176 (1987).
Acknowledgements
This work was supported by the Center for Bio-Inspired Energy Science (CBES), an Energy Frontiers Research Center (EFRC) funded by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under award number DE-SC0000989. Z.A. received postdoctoral support from the Beatriu de Pinós Fellowship 2014 BP‐A 00007 (Agència de Gestió d'Ajust Universitaris i de Recerca, AGAUR) and a Paralyzed Veterans of America (PVA) (grant no. PVA17_RF_0008). This work made use of the Peptide Synthesis Core Facility (peptide synthesis) of the Simpson Querrey Institute at Northwestern University. This facility has current support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS–1542205). The Simpson Querrey Institute, Northwestern University Office for Research, US Army Research Office, and the US Army Medical Research and Materiel Command also provided funding to develop this facility. This work also made use of the EPIC and BioCryo facilities as well as the CryoCluster equipment of Northwestern University’s NUANCE Center, which has received support from the Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS-1542205); the MRSEC Program (NSF DMR-1720139) at the Materials Research Center; the International Institute for Nanotechnology (IIN); the Keck Foundation; and the State of Illinois, through the IIN and the MRI program (NSF DMR-1229693). This work also made use of the MatCI Facility, which receives support from the MRSEC Program (NSF DMR-1720139) of the Materials Research Center at Northwestern University. This work also made use of the IMSERC at Northwestern University, which has received support from the NIH (1S10OD012016-01/1S10RR019071-01A1), Soft and Hybrid Nanotechnology Experimental (SHyNE) Resource (NSF ECCS-1542205), the State of Illinois, and the International Institute for Nanotechnology (IIN). This work also made use of Keck Biophysics Facility at Northwestern University. X-ray experiments were performed at the DuPont–Northwestern–Dow Collaborative Access Team (DND-CAT) located at Sector 5 of the Advanced Photon Source (APS). DND-CAT is supported by Northwestern University, The Dow Chemical Company, and DuPont de Nemours, Inc. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. X-ray scattering data was collected using an instrument funded by the National Science Foundation under Award Number 0960140. We thank Z. Yu for helpful discussion and R. Qiu for 1H nuclear Overhauser effect spectroscopy NMR analysis. We thank M. Seniw for providing schematic illustrations.
Author information
Authors and Affiliations
Contributions
C.L. designed and performed most of the experiments and wrote the manuscript. A.I. performed the CG simulations and wrote the manuscript. H.S. carried out X-ray scattering and Cryo-SEM, K.S. and S.M.C. collected Cryo-TEM, N.A.S. performed 3D printing and Z.A. collected confocal microscopy data. L.C.P., G.C.S. and S.I.S. wrote the manuscript and supervised the research.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Videos 1–21 legends, methods, Figs. 1–48, Tables 1–5 and references.
Supplementary Video 1
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran with 0.5 mm thickness bends towards a top light source.
Supplementary Video 2
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran with 0.2 mm thickness bends towards a top light source.
Supplementary Video 3
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran with 1.0 mm thickness bends towards a top light source.
Supplementary Video 4
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran with 2.0 mm thickness bends towards a top light source.
Supplementary Video 5
Soft covalent polymer hydrogel (2.8% crosslinking) made of nitro-spiropyran exhibits a low degree of bending (<5°).
Supplementary Video 6
Stiff covalent polymer hydrogel (10% crosslinking) made of nitro-spiropyran exhibits a slower response to light.
Supplementary Video 7
Glass fibre (5 wt%) reinforced covalent polymer hydrogel (2.8% crosslinking) made of nitro-spiropyran exhibits a slower response to light.
Supplementary Video 8
PA (0.5 wt%) hybrid hydrogel made of nitro-spiropyran with 0.5 mm thickness bends towards a top light source.
Supplementary Video 9
PA (1.5 wt%) hybrid hydrogel made of nitro-spiropyran with 0.5 mm thickness bends towards a top light source.
Supplementary Video 10
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran (0.5 mm thickness) with aligned PA fibres made by 3D printing bends towards a top light source.
Supplementary Video 11
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran (0.5 mm thickness) with non-aligned PA fibres made by casting bends towards a top light source.
Supplementary Video 12
PA (1 wt%) hybrid hydrogel made of nitro-spiropyran walks unidirectionally on ratcheted surface with a step length of 2.2 mm.
Supplementary Video 13
Soft covalent polymer hydrogel (2.8% crosslinking) made of nitro-spiropyran walks unidirectionally on ratcheted surface with a step length of 0.5 mm.
Supplementary Video 14
PA (1 wt%) hybrid hydrogel made of spiropyran without nitro substituent walks unidirectionally on ratcheted surface with a step length of 2.6 mm.
Supplementary Video 15
Soft covalent polymer hydrogel (2.8% crosslinking) made of spiropyran without nitro substituent walks unidirectionally on ratcheted surface with a step length of 1.1 mm.
Supplementary Video 16
Five steps of unidirectional motion by the PA (1 wt%) hybrid actuator made of nitro-spiropyran after alternating periods of light on (30 min) and light off (10 h).
Supplementary Video 17
Five steps of unidirectional motion by the PA (1 wt%) hybrid actuator made of a spiropyran without nitro substituent after alternating periods of light on (20 min) and light off (1.5 h).
Supplementary Video 18
A PA (1 wt%) hybrid crawler made of nitro-spiropyran with PA fibres aligned parallel to the crawling direction gives a step length of 4.7 mm.
Supplementary Video 19
A PA (1 wt%) hybrid crawler made of nitro-spiropyran with PA fibres aligned perpendicular to the crawling direction gives a step length of 1.8 mm.
Supplementary Video 20
A PA (1 wt%) hybrid crawler made of nitro-spiropyran with PA fibres non-aligned gives a step length of 3.5 mm.
Supplementary Video 21
Rotational motion of a PA (1 wt%) hybrid hydrogel actuator made of nitro-spiropyran.
Rights and permissions
About this article
Cite this article
Li, C., Iscen, A., Sai, H. et al. Supramolecular–covalent hybrid polymers for light-activated mechanical actuation. Nat. Mater. 19, 900–909 (2020). https://doi.org/10.1038/s41563-020-0707-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-020-0707-7
This article is cited by
-
Self-regulated underwater phototaxis of a photoresponsive hydrogel-based phototactic vehicle
Nature Nanotechnology (2024)
-
Reconfiguring hydrogel assemblies using a photocontrolled metallopolymer adhesive for multiple customized functions
Nature Chemistry (2024)
-
Self-regulated reversal deformation and locomotion of structurally homogenous hydrogels subjected to constant light illumination
Nature Communications (2024)
-
Self-sustainable autonomous soft actuators
Communications Chemistry (2024)
-
Highly robust supramolecular polymer networks crosslinked by a tiny amount of metallacycles
Nature Communications (2024)