Abstract
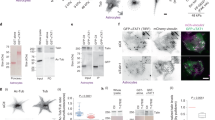
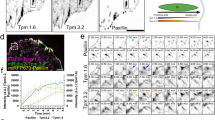
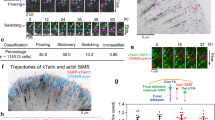
The interrelationship between microtubules and the actin cytoskeleton in mechanoregulation of integrin-mediated adhesions is poorly understood. Here, we show that the effects of microtubules on two major types of cell-matrix adhesion, focal adhesions and podosomes, are mediated by KANK family proteins connecting the adhesion protein talin with microtubule tips. Both total microtubule disruption and microtubule uncoupling from adhesions by manipulations with KANKs trigger a massive assembly of myosin IIA filaments, augmenting focal adhesions and disrupting podosomes. Myosin IIA filaments are indispensable effectors in the microtubule-driven regulation of integrin-mediated adhesions. Myosin IIA filament assembly depends on Rho activation by the RhoGEF GEF-H1, which is trapped by microtubules when they are connected with integrin-mediated adhesions via KANK proteins but released after their disconnection. Thus, microtubule capture by integrin-mediated adhesions modulates the GEF-H1-dependent effect of microtubules on the assembly of myosin IIA filaments. Subsequent actomyosin reorganization then remodels the focal adhesions and podosomes, closing the regulatory loop.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files). Raw datasets generated during and/or analysed during the current study are available from the corresponding authors on reasonable request.
Code availability
Custom-written code used to analyse the data in the current study is available from the corresponding authors on reasonable request.
Change history
30 May 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Kasza, K. E. et al. The cell as a material. Curr. Opin. Cell Biol. 19, 101–107 (2007).
Geiger, B., Spatz, J. P. & Bershadsky, A. D. Environmental sensing through focal adhesions. Nat. Rev. Mol. Cell Biol. 10, 21–33 (2009).
Kanchanawong, P. et al. Nanoscale architecture of integrin-based cell adhesions. Nature 468, 580–584 (2010).
Horton, E. R. et al. The integrin adhesome network at a glance. J. Cell Sci. 129, 4159–4163 (2016).
Bershadsky, A., Chausovsky, A., Becker, E., Lyubimova, A. & Geiger, B. Involvement of microtubules in the control of adhesion-dependent signal transduction. Curr. Biol. 6, 1279–1289 (1996).
Enomoto, T. Microtubule disruption induces the formation of actin stress fibers and focal adhesions in cultured cells: possible involvement of the rho signal cascade. Cell Struct. Funct. 21, 317–326 (1996).
Ezratty, E. J., Partridge, M. A. & Gundersen, G. G. Microtubule-induced focal adhesion disassembly is mediated by dynamin and focal adhesion kinase. Nat. Cell Biol. 7, 581–590 (2005).
Bouchet, B. P. et al. Talin-KANK1 interaction controls the recruitment of cortical microtubule stabilizing complexes to focal adhesions. eLife. 5, e18124 (2016).
Sun, Z. et al. Kank2 activates talin, reduces force transduction across integrins and induces central adhesion formation. Nat. Cell Biol. 18, 941–953 (2016).
Chen, N. P., Sun, Z. & Fassler, R. The Kank family proteins in adhesion dynamics. Curr. Opin. Cell Biol. 54, 130–136 (2018).
van der Vaart, B. et al. CFEOM1-associated kinesin KIF21A is a cortical microtubule growth inhibitor. Dev. Cell 27, 145–160 (2013).
Lansbergen, G. et al. CLASPs attach microtubule plus ends to the cell cortex through a complex with LL5beta. Dev. Cell 11, 21–32 (2006).
Clohisey, S. M., Dzhindzhev, N. S. & Ohkura, H. Kank Is an EB1 interacting protein that localises to muscle-tendon attachment sites in Drosophila. PloS ONE 9, e106112 (2014).
Murphy, D. A. & Courtneidge, S. A. The ‘ins’ and ‘outs’ of podosomes and invadopodia: characteristics, formation and function. Nat. Rev. Mol. Cell Biol. 12, 413–426 (2011).
Cox, S. & Jones, G. E. Imaging cells at the nanoscale. Int. J. Biochem. Cell Biol. 45, 1669–1678 (2013).
Yu, C. H. et al. Integrin-matrix clusters form podosome-like adhesions in the absence of traction forces. Cell Rep. 5, 1456–1468 (2013).
Linder, S., Hufner, K., Wintergerst, U. & Aepfelbacher, M. Microtubule-dependent formation of podosomal adhesion structures in primary human macrophages. J. Cell Sci. 113, 4165–4176 (2000).
Meddens, M. B. et al. Actomyosin-dependent dynamic spatial patterns of cytoskeletal components drive mesoscale podosome organization. Nat. Commun. 7, 13127 (2016).
Monypenny, J. et al. Role of WASP in cell polarity and podosome dynamics of myeloid cells. Eur. J. Cell Biol. 90, 198–204 (2011).
Kaverina, I., Krylyshkina, O. & Small, J. V. Microtubule targeting of substrate contacts promotes their relaxation and dissociation. J. Cell Biol. 146, 1033–1044 (1999).
Hu, S. et al. Long-range self-organization of cytoskeletal myosin II filament stacks. Nat. Cell Biol. 19, 133–141 (2017).
Ito, S. et al. Induced cortical tension restores functional junctions in adhesion-defective carcinoma cells. Nat. Commun. 8, 1834 (2017).
Vicente-Manzanares, M., Zareno, J., Whitmore, L., Choi, C. K. & Horwitz, A. F. Regulation of protrusion, adhesion dynamics, and polarity by myosins IIA and IIB in migrating cells. J. Cell Biol. 176, 573–580 (2007).
Even-Ram, S. et al. Myosin IIA regulates cell motility and actomyosin-microtubule crosstalk. Nat. Cell Biol. 9, 299–309 (2007).
Riveline, D. et al. Focal contacts as mechanosensors: externally applied local mechanical force induces growth of focal contacts by an mDia1-dependent and ROCK-independent mechanism. J. Cell Biol. 153, 1175–1186 (2001).
Ren, X. D., Kiosses, W. B. & Schwartz, M. A. Regulation of the small GTP-binding protein Rho by cell adhesion and the cytoskeleton. EMBO J. 18, 578–585 (1999).
Kakinuma, N., Roy, B. C., Zhu, Y., Wang, Y. & Kiyama, R. Kank regulates RhoA-dependent formation of actin stress fibers and cell migration via 14-3-3 in PI3K-Akt signaling. J. Cell Biol. 181, 537–549 (2008).
Linder, S. The matrix corroded: podosomes and invadopodia in extracellular matrix degradation. Trends Cell Biol. 17, 107–117 (2007).
Wang, Y. & McNiven, M. A. Invasive matrix degradation at focal adhesions occurs via protease recruitment by a FAK-p130Cas complex. J. Cell Biol. 196, 375–385 (2012).
Krendel, M., Zenke, F. T. & Bokoch, G. M. Nucleotide exchange factor GEF-H1 mediates cross-talk between microtubules and the actin cytoskeleton. Nat. Cell Biol. 4, 294–301 (2002).
Kwan, K. M. & Kirschner, M. W. A microtubule-binding Rho-GEF controls cell morphology during convergent extension of Xenopus laevis. Development 132, 4599–4610 (2005).
Jiu, Y. et al. Vimentin intermediate filaments control actin stress fiber assembly through GEF-H1 and RhoA. J. Cell Sci. 130, 892–902 (2017).
Ren, Y., Li, R., Zheng, Y. & Busch, H. Cloning and characterization of GEF-H1, a microtubule-associated guanine nucleotide exchange factor for Rac and Rho GTPases. J. Biol. Chem. 273, 34954–34960 (1998).
Chang, Y. C., Nalbant, P., Birkenfeld, J., Chang, Z. F. & Bokoch, G. M. GEF-H1 couples nocodazole-induced microtubule disassembly to cell contractility via RhoA. Mol. Biol. Cell 19, 2147–2153 (2008).
Gee, H. Y. et al. KANK deficiency leads to podocyte dysfunction and nephrotic syndrome. J. Clin. Invest. 125, 2375–2384 (2015).
Portran, D., Schaedel, L., Xu, Z., Thery, M. & Nachury, M. V. Tubulin acetylation protects long-lived microtubules against mechanical ageing. Nat. Cell Biol. 19, 391–398 (2017).
Xu, Z. et al. Microtubules acquire resistance from mechanical breakage through intralumenal acetylation. Science 356, 328–332 (2017).
Samereier, M. et al. EB1 contributes to proper front-to-back polarity in neutrophil-like HL-60 cells. Eur. J. Cell Biol. 96, 143–153 (2017).
Bershadsky, A., Kozlov, M. & Geiger, B. Adhesion-mediated mechanosensitivity: a time to experiment, and a time to theorize. Curr. Opin. Cell Biol. 18, 472–481 (2006).
Oakes, P. W., Beckham, Y., Stricker, J. & Gardel, M. L. Tension is required but not sufficient for focal adhesion maturation without a stress fiber template. J. Cell Biol. 196, 363–374 (2012).
Choi, C. K. et al. Actin and alpha-actinin orchestrate the assembly and maturation of nascent adhesions in a myosin II motor-independent manner. Nat. Cell Biol. 10, 1039–1050 (2008).
Oakes, P. W. & Gardel, M. L. Stressing the limits of focal adhesion mechanosensitivity. Curr. Opin. Cell Biol. 30, 68–73 (2014).
Rafiq, N. B. et al. Podosome assembly is controlled by the GTPase ARF1 and its nucleotide exchange factor ARNO. J. Cell Biol. 216, 181–197 (2017).
Elliott, H. et al. Myosin II controls cellular branching morphogenesis and migration in three dimensions by minimizing cell-surface curvature. Nat. Cell Biol. 17, 137–147 (2015).
Vasiliev, J. M. et al. Effect of colcemid on the locomotory behaviour of fibroblasts. J. Embryol. Exp. Morphol. 24, 625–640 (1970).
Small, J. V., Geiger, B., Kaverina, I. & Bershadsky, A. How do microtubules guide migrating cells? Nat. Rev. Mol. Cell Biol. 3, 957–964 (2002).
Jones, G. E., Zicha, D., Dunn, G. A., Blundell, M. & Thrasher, A. Restoration of podosomes and chemotaxis in Wiskott-Aldrich syndrome macrophages following induced expression of WASp. Int. J. Biochem. Cell Biol. 34, 806–815 (2002).
Ballestrem, C., Magid, N., Zonis, J., Shtutman, M. & Bershadsky, A. in Cell Motility (eds Ridley, A., Peckham, M. & Clark, P.) 75–99 (Wiley, 2004).
Weng, S., Shao, Y., Chen, W. & Fu, J. Mechanosensitive subcellular rheostasis drives emergent single-cell mechanical homeostasis. Nat. Mater. 15, 961–967 (2016).
Ballestrem, C., Hinz, B., Imhof, B. A. & Wehrle-Haller, B. Marching at the front and dragging behind: differential αVβ3-integrin turnover regulates focal adhesion behavior. J. Cell Biol. 155, 1319–1332 (2001).
Rid, R., Schiefermeier, N., Grigoriev, I., Small, J. V. & Kaverina, I. The last but not the least: the origin and significance of trailing adhesions in fibroblastic cells. Cell Motil. Cytoskel. 61, 161–171 (2005).
Pan, C. Q., Liou, Y.-c. & Low, B. C. Active Mek2 as a regulatory scaffold that promotes Pin1 binding to BPGAP1 to suppress BPGAP1-induced acute Erk activation and cell migration. J. Cell Sci. 123, 903–916 (2010).
Sabass, B., Gardel, M. L., Waterman, C. M. & Schwarz, U. S. High resolution traction force microscopy based on experimental and computational advances. Biophys. J. 94, 207–220 (2008).
Liu, J. et al. Talin determines the nanoscale architecture of focal adhesions. Proc. Natl Acad. Sci. USA 112, E4864–E4873 (2015).
Zhang, Z., Nishimura, Y. & Kanchanawong, P. Extracting microtubule networks from superresolution single-molecule localization microscopy data. Mol. Biol. Cell 28, 333–345 (2017).
Acknowledgements
We thank A. Akhmanova (Utrecht University, Netherlands), R. Fäasler (Max Plank Institute for Biochemistry, Martinsried, Germany), C. M. Waterman (National Institutes of Health, USA), R. Zaidel-Bar (Mechanobiology Institute, Singapore) and M. Dodding for providing constructs used in this study. We are grateful to A. Akhmanova for useful discussions and constructive criticism. We thank D. Pitta de Araujo (MBI Science Communications Unit) for help with Fig. 6. This research is supported by the National Research Foundation, Prime Minister’s Office, Singapore, and the Ministry of Education under the Research Centres of Excellence programme through the Mechanobiology Institute, Singapore (ref no. R-714-006-006-271) (A.D.B., N.B.M.R., V.T. and M.N.) and Singapore Ministry of Education Academic Research Fund Tier 3 MOE grant no. MOE2016-T3-1-002 (A.D.B., Y.N.). A.D.B. also acknowledges support from a Maimonides Israeli–France grant (Israeli Ministry of Science Technology and Space) and EU Marie Skłodowska-Curie Network InCeM (project ID 642866) at the Weizmann Institute of Science. N.B.M.R. is also funded by a joint National University of Singapore–King’s College London graduate studentship. G.E.J. is supported by the Medical Research Council, UK (G1100041, MR/K015664) and the generous provision of a visiting professorship from the Mechanobiology Institute, Singapore. P.K. and Z.Z. are funded by the Ministry of Education Academic Research Fund Tier 2 (MOE-T2-1-124), the Mechanobiology Institute seed funding, the National Research Foundation Fellowship (NRF-NRFF-2011-04) and the National Research Foundation Competitive Research Programme (NRF2012NRF-CRP001-084).
Author information
Authors and Affiliations
Contributions
A.D.B. conceived and designed the project together with V.V., P.K. and G.E.J. N.B.M.R. and Y.N. equally designed and performed all experiments and prepared the manuscript; S.V.P., V.T., Z.Z., S.S. and M.N. provided assistance in carrying out experiments and discussed results. A.D.B., N.B.M.R. and Y.N., together with V.V., P.K. and G.E.J., discussed results and prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Notes, Supplementary Figs. 1–13, Supplementary Table 1, Supplementary Video Legends 1–17, Supplementary Refs. 1–39
Supplementary Video 1
The microtubule plus ends of an HT1080 cell transfected with control siRNA are accumulated at focal adhesions concentrated within adhesive islands
Supplementary Video 2
The microtubule plus ends were randomly distributed over adhesive islands and spaces between them in an HT1080 cell depleted of KANK1 and KANK2
Supplementary Video 3
Microtubule outgrowth induced transient disassembly of focal adhesions in an HT1080 cell
Supplementary Video 4
Rapamycin-induced linking of two parts of the KANK1 molecule triggered disassembly of focal adhesions in an HT1080 cell
Supplementary Video 5
Microtubule disruption triggered massive assembly of myosin II filaments and disassembly of podosomes in a THP-1 cell
Supplementary Video 6
Microtubule disruption triggered massive assembly of myosin II filaments and augmentation of focal adhesion in an HT1080 cell
Supplementary Video 7
Microtubule outgrowth induced disassembly of myosin II filaments in an HT1080 cell
Supplementary Video 8
Transient decrease in traction forces after microtubule outgrowth in an HT1080 cell
Supplementary Video 9
KANK2 knockdown suppressed disassembly of myosin II filaments induced by microtubule outgrowth in an HT1080 cell
Supplementary Video 10
Activation of RhoA by CN03 augmented myosin II filament formation and podosome disruption in a THP-1 cell
Supplementary Video 11
Dynamics of podosomes and myosin II filaments in a control THP-1 cell
Supplementary Video 12
ROCK inhibition by Y-27632 did not affect podosome dynamics in a THP-1 cell
Supplementary Video 13
Inhibition of ROCK by Y-27632 disrupted myosin II filaments and prevented nocodazole-induced disassembly of podosomes in a THP-1 cell
Supplementary Video 14
Disruption of myosin II filaments by Y-27632 resulted in recovery of podosomes in a THP-1 cell pretreated with nocodazole
Supplementary Video 15
Disruption of myosin II filaments by Y-27632 resulted in recovery of podosomes in a KANK1-depleted THP-1 cell
Supplementary Video 16
Constitutively active RhoA and ROCK prevented the disruption of focal adhesions on microtubule outgrowth in HT1080 cells
Supplementary Video 17
GEF-H1 knockdown suppressed disruption of myosin II filaments and prevented disassembly of focal adhesions on microtubule outgrowth in an HT1080 cell
Rights and permissions
About this article
Cite this article
Rafiq, N.B.M., Nishimura, Y., Plotnikov, S.V. et al. A mechano-signalling network linking microtubules, myosin IIA filaments and integrin-based adhesions. Nat. Mater. 18, 638–649 (2019). https://doi.org/10.1038/s41563-019-0371-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0371-y
This article is cited by
-
Biomaterial-based mechanical regulation facilitates scarless wound healing with functional skin appendage regeneration
Military Medical Research (2024)
-
Focal adhesions contain three specialized actin nanoscale layers
Nature Communications (2024)
-
Talin2 and KANK2 functionally interact to regulate microtubule dynamics, paclitaxel sensitivity and cell migration in the MDA-MB-435S melanoma cell line
Cellular & Molecular Biology Letters (2023)
-
Organization, dynamics and mechanoregulation of integrin-mediated cell–ECM adhesions
Nature Reviews Molecular Cell Biology (2023)
-
Compressive forces stabilize microtubules in living cells
Nature Materials (2023)