abstract
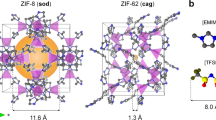
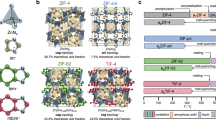
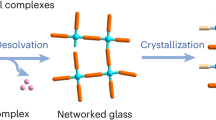
Metal–organic frameworks (MOFs) are microporous materials with huge potential for chemical processes. Structural collapse at high pressure, and transitions to liquid states at high temperature, have recently been observed in the zeolitic imidazolate framework (ZIF) family of MOFs. Here, we show that simultaneous high-pressure and high-temperature conditions result in complex behaviour in ZIF-62 and ZIF-4, with distinct high- and low-density amorphous phases occurring over different regions of the pressure–temperature phase diagram. In situ powder X-ray diffraction, Raman spectroscopy and optical microscopy reveal that the stability of the liquid MOF state expands substantially towards lower temperatures at intermediate, industrially achievable pressures and first-principles molecular dynamics show that softening of the framework coordination with pressure makes melting thermodynamically easier. Furthermore, the MOF glass formed by melt quenching the high-temperature liquid possesses permanent, accessible porosity. Our results thus imply a route to the synthesis of functional MOF glasses at low temperatures, avoiding decomposition on heating at ambient pressure.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Experimental and computational data supporting the findings of this work are available from the public GitHub online repository at https://github.com/fxcoudert/citable-data.
References
Moghadam, P. Z. et al. Development of a cambridge structural database subset: a collection of metal-organic frameworks for past, present, and future. Chem. Mater. 29, 2618–2625 (2017).
Kim, H. et al. Water harvesting from air with metal-organic frameworks powered by natural sunlight. Science 356, 430–434 (2017).
Mason, J. A. et al. Methane storage in flexible metal–organic frameworks with intrinsic thermal management. Nature 527, 357–361 (2015).
Denny, M. S. Jr, Moreton, J. C., Benz, L. & Cohen, S. M. Metal–organic frameworks for membrane-based separations. Nat. Rev. Mater. 1, 16078 (2016).
Mondloch, J. E. et al. Destruction of chemical warfare agents using metal–organic frameworks. Nat. Mater. 14, 512–516 (2015).
Horike, S., Shimomura, S. & Kitagawa, S. Soft porous crystals. Nat. Chem. 1, 695–704 (2009).
Dissegna, S., Epp, K., Heinz, W. R., Kieslich, G. & Fischer, R. A. Defective metal-organic frameworks. Adv. Mater. 30, 1704501–1704524 (2018).
Schneemann, A. et al. Flexible metal-organic frameworks. Chem. Soc. Rev. 43, 6062–6096 (2014).
Bennett, T. D., Cheetham, A. K., Fuchs, A. H. & Coudert, F. X. Interplay between defects, disorder and flexibility in metal–organic frameworks. Nat. Chem. 9, 11–16 (2016).
Panda, T. et al. Mechanical alloying of metal-organic frameworks. Angew. Chem. Int. Ed. 56, 2413–2417 (2017).
Coudert, F.-X. responsive metal–organic frameworks and framework materials: under pressure, taking the heat, in the spotlight, with friends. Chem. Mater. 27, 1905–1916 (2015).
Krause, S. et al. A pressure-amplifying framework material with negative gas adsorption transitions. Nature 532, 348–352 (2016).
Carrington, E. J. et al. Solvent-switchable continuous-breathing behaviour in a diamondoid metal–organic framework and its influence on CO2 versus CH4 selectivity. Nat. Chem. 9, 882–889 (2017).
Longley, L., Li, N., Wei, F. & Bennett, T. D. Uncovering a reconstructive solid–solid phase transition in a metal–organic framework. R. Soc. Open Sci. 4, 171355 (2017).
Lapidus, S. H., Halder, G. J., Chupas, P. J. & Chapman, K. W. Exploiting high pressures to generate porosity, polymorphism, and lattice expansion in the nonporous molecular framework Zn(CN)2. J. Am. Chem. Soc. 135, 7621–7628 (2013).
Robert M. Hazen, R. T. D. High-Temperature and High-Pressure Crystal Chemistry (Reviews in Mineralogy and Geochemistry Vol. 41, Mineralogical Society of America, 2003).
Dove, M. T. et al. Crystal structure of the high-pressure monoclinic phase-II of cristobalite, SiO2. Mineral. Mag. 64, 569–576 (2000).
Hemley, R. J., Jephcoat, A. P., Mao, H. K., Ming, L. C. & Manghnani, M. H. Pressure-induced amorphization of crystalline silica. Nature 334, 52–54 (1988).
Natarajan, S. & Mahata, P. Metal-organic framework structures - how closely are they related to classical inorganic structures? Chem. Soc. Rev. 38, 2304–2318 (2009).
Decurtins, S., Schmalle, H. W., Schneuwly, P., Ensling, J. & Guetlich, P. A concept for the synthesis of 3-dimensional homo-and bimetallic oxalate-bridged networks [M2(ox)3]n. Structural, Moessbauer, and magnetic studies in the field of molecular-based magnets. J. Am. Chem. Soc. 116, 9521–9528 (1994).
Sadakiyo, M., Yamada, T., Honda, K., Matsui, H. & Kitagawa, H. Control of crystalline proton-conducting pathways by water-induced transformations of hydrogen-bonding networks in a metal–organic framework. J. Am. Chem. Soc. 136, 7701–7707 (2014).
Huskić, I., Pekov, I. V., Krivovichev, S. V. & Friščić, T. Minerals with metal-organic framework structures. Sci. Adv. 2, e1600621–e1600629 (2016).
Banerjee, R. et al. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture. Science 319, 939–943 (2008).
Park, K. S. et al. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl Acad. Sci. USA 103, 10186–10191 (2006).
McKellar, S. C. & Moggach, S. A. Structural studies of metal-organic frameworks under high pressure. Acta Crystallogr. Sect. B 71, 587–607 (2015).
Cruciani, G. Zeolites upon heating: factors governing their thermal stability and structural changes. J. Phys. Chem. Solids 67, 1973–1994 (2006).
Hazen, R. M. Zeolite molecular sieve 4A: anomalous compressibility and volume discontinuities at high pressure. Science 219, 1065–1067 (1983).
Chapman, K. W., Sava, D. F., Halder, G. J., Chupas, P. J. & Nenoff, T. M. Trapping guests within a nanoporous metal–organic framework through pressure-induced amorphization. J. Am. Chem. Soc. 133, 18583–18585 (2011).
Zhao, P. et al. Pressure-induced oversaturation and phase transition in zeolitic imidazolate frameworks with remarkable mechanical stability. Dalt. Trans. 44, 4498–4503 (2015).
Henke, S. et al. Pore closure in zeolitic imidazolate frameworks under mechanical pressure. Chem. Sci. 9, 1654–1660 (2018).
Bennett, T. D. et al. Thermal amorphization of zeolitic imidazolate frameworks. Angew. Chemie Int. Ed. 50, 3067–3071 (2011).
Bennett, T. D. et al. Melt-quenched glasses of metal–organic frameworks. J. Am. Chem. Soc. 138, 3484–3492 (2016).
Gaillac, R. et al. Liquid metal–organic frameworks. Nat. Mater. 16, 1149–1155 (2017).
Bennett, T. D. et al. Hybrid glasses from strong and fragile metal-organic framework liquids. Nat. Commun. 6, 8079–8086 (2015).
Qiao, A. et al. A metal-organic framework with ultrahigh glass-forming ability. Sci. Adv. 4, 6827–6834 (2018).
Zhou, C. et al. Metal-organic framework glasses with permanent accessible porosity. Nat. Commun. 9, 5042–5051 (2018).
Wenqian, C. et al. Glass formation of a coordinationpolymer crystal for enhanced proton conductivity and material flexibility. Angew. Chemie Int. Ed. 55, 5195–5200 (2016).
Kniep, R., Mootz, D. & Vegas, A. Variscite. Acta Crystallogr. B33, 263–265 (1977).
Song, Y., Zavalij, P. Y., Suzuki, M. & Whittingham, M. S. New iron(III) phosphate phases: crystal structure and electrochemical and magnetic properties. Inorg. Chem. 41, 5778–5786 (2002).
Deiseroth, H. J. & Müller -Buschbaum, H. Über Erdalkalimetalloxogallate. III: Untersuchung des Aufbaus von CaGa O . Z. Anorg. Allg. Chem. 396, 157–164 (1973).
Lazić, B., Kahlenberg, V. & Konzett, J. Rietveld analysis of a high pressure modification of monocalcium oxogallate (CaGa2O4). Z. Anorg. Allg. Chem. 631, 2411–2415 (2005).
Bennett, T. D. et al. Reversible pressure-induced amorphization of a zeolitic imidazolate framework (ZIF-4). Chem. Commun. 47, 7983–7985 (2011).
Bouëssel du Bourg, L., Ortiz, A. U., Boutin, A. & Coudert, F. X. Thermal and mechanical stability of zeolitic imidazolate frameworks polymorphs. APL Mater. 2, 124110–124118 (2014).
Yang, S. et al. Cation-induced kinetic trapping and enhanced hydrogen adsorption in a modulated anionic metal–organic framework. Nat. Chem. 1, 487–493 (2009).
Thornton, A. W. et al. Porosity in metal-organic framework glasses. Chem. Commun. 52, 3750–3753 (2016).
Ortiz, A. U., Boutin, A., Fuchs, A. H. & Coudert, F.-X. Investigating the pressure-induced amorphization of zeolitic imidazolate framework ZIF-8: mechanical instability due to shear mode softening. J. Phys. Chem. Lett. 4, 1861–1865 (2013).
Greaves, G. N. et al. Zeolite collapse and polyamorphism. J. Phys. Condens. Matter 19, 415102–415119 (2007).
Greaves, G. N. & Meneau, F. Probing the dynamics of instability in zeolitic materials. J. Phys. Condens. Matter 16, 3459–3472 (2004).
Armand, M., Endres, F., MacFarlane, D. R., Ohno, H. & Scrosati, B. Ionic-liquid materials for the electrochemical challenges of the future. Nat. Mater. 8, 621–629 (2009).
Lima, T. A., Paschoal, V. H., Faria, L. F. O. & Ribeiro, M. C. C. Unraveling the stepwise melting of an ionic liquid. J. Phys. Chem. B 121, 4650–4655 (2017).
McGreevy, R. L. & Pusztai, L. The structure of molten salts. Proc. Math. Phys. Sci. 430, 241–261 (1990).
Yuan, S. et al. PCN-250 under pressure: sequential phase transformation and the implications for MOF densification. Joule 1, 806–815 (2017).
Mączka, M. et al. Temperature- and pressure-induced phase transitions in the metal formate framework of [ND4][Zn(DCOO)3] and [NH4][Zn(HCOO)3]. Inorg. Chem. 53, 9615–9624 (2014).
Gustafsson, M. & Zou, X. Crystal formation and size control of zeolitic imidazolate frameworks with mixed imidazolate linkers. J. Porous Mater. 20, 55–63 (2013).
Filik, J. et al. Processing two-dimensional X-ray diffraction and small-angle scattering data in DAWN 2. J. Appl. Crystallogr. 50, 959–966 (2017).
Dorogokupets, P. I. & Dewaele, A. Equations of state of MgO, Au, Pt, NaCl-B1, and NaCl-B2: Internally consistent high-temperature pressure scales. High Press. Res. 27, 431–446 (2007).
Dewaele, A., Torrent, M., Loubeyre, P. & Mezouar, M. Compression curves of transition metals in the mbar range: experiments and projector augmented-wave calculations. Phys. Rev. B 78, 104102–104115 (2008).
Rekhi, S., Dubrovinsky, L. S. & Saxena, S. K. Temperature-induced ruby fluorescence shifts up to a pressure of 15 GPa in an externally heated diamond anvil cell. HighTemp. High Press. 31, 299–305 (1999).
Coelho, A. A. TOPAS and TOPAS-Academic: an optimization program integrating computer algebra and crystallographic objects written in C++. J. Appl. Crystallogr. 51, 210–218 (2018).
Stinton, G. W. & Evans, J. S. O. Parametric Rietveld refinement. J. Appl. Crystallogr. 40, 87–95 (2007).
Acknowledgements
R.N.W. acknowledges support from the EPSRC in the form of a DTG Graduate Studentship. R.N.W. and A.M.B. thank C. Hunter and his group, at the Department of Chemistry (University of Cambridge), for the use of their HPLC facilities. T.D.B. thanks the Royal Society for a University Research Fellowship and for their support (UF150021). C.Z. acknowledges the financial support of the Elite Research Travel Scholarship from the Danish Ministry of Higher Education and Science. We thank Diamond Light Source for access to beamline I15 (EE16133 and EE19046-1). This work benefitted from the financial support of ANRT (thèse CIFRE 2015/0268) and access to HPC platforms provided by a GENCI grant (A0050807069). Gas sorption on the ZIF-62 glass was supported by a grant from the National Science Foundation, Division of Chemistry under award number CHE-1661655. Synthesis of MOFs was supported by a grant from the National Science Foundation under award number CHE-1359906 (to S.M.C.). L. Friche, N. Bandata and O. T. Qasvini (Massey Unviersity) are thanked for technical assistance, alongside B. R. Pimentel (UCSD).
Author information
Authors and Affiliations
Contributions
T.D.B. and S.A.T.R. designed the project. R.N.W., G.I.L, S.A., S.G.M, A.K.K., M.T.W., S.F., C.Z., C.W. and T.D.B. performed the PXRD experiments. S.G.M. designed and constructed the high-P-T PXRD equipment. R.N.W. performed melting point determinations, Raman spectroscopy and microscopy, and analysed XRD and spectroscopic data. H.P. and C.Z. performed DSC measurements. A.M.B. and R.N.W. performed HPLC measurements. X.Y, S.M.C. and S.G.T. performed gas sorption measurements. R.G. and F.-X.C. designed, performed and analysed the molecular simulations. All authors participated in discussing the data. R.N.W. and T.D.B. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Supplementary Figures 1–22, Supplementary Tables 1–5, Supplementary References 1–10
Supplementary Video 1
In situ video of melting of ZIF-62 crystals at high pressure and temperature, with speed-up factor of ×8.
Rights and permissions
About this article
Cite this article
Widmer, R.N., Lampronti, G.I., Anzellini, S. et al. Pressure promoted low-temperature melting of metal–organic frameworks. Nat. Mater. 18, 370–376 (2019). https://doi.org/10.1038/s41563-019-0317-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-019-0317-4
This article is cited by
-
Melt-quenched glass formation of a family of metal-carboxylate frameworks
Nature Communications (2024)
-
Precise control over gas-transporting channels in zeolitic imidazolate framework glasses
Nature Materials (2024)
-
ZIF-62 glass foam self-supported membranes to address CH4/N2 separations
Nature Materials (2023)
-
Enhancing CO2 adsorption capacity of ZIF-8 by synergetic effect of high pressure and temperature
Scientific Reports (2023)
-
Quantification of gas-accessible microporosity in metal-organic framework glasses
Nature Communications (2022)