Abstract
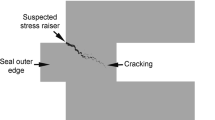
Intergranular stress-corrosion cracking (IGSCC) is a form of environmentally induced crack propagation causing premature failure of elemental metals and alloys. It is believed to require the simultaneous presence of tensile stress and corrosion; however, the exact nature of this synergy has eluded experimental identification. For noble metal alloys such as Ag–Au, IGSCC is a consequence of dealloying corrosion, forming a nanoporous gold layer that is believed to have the ability to transmit cracks into grain boundaries in un-dealloyed parent phase via a pure mechanical process. Here using atomic-scale techniques and statistical characterizations for this alloy system, we show that the separate roles of stress and anodic dissolution can be decoupled and that the apparent synergy exists owing to rapid time-dependent morphology changes at the dealloyed layer/parent phase interface. We discuss the applicability of our findings to the IGSCC of important engineering Fe- and Ni-based alloys in critical applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Scully, J. R. Environment-assisted intergranular cracking. MRS Bull. 24, 36–42 (1999).
Sieradzki, K. & Newman, R. C. Stress-corrosion cracking. J. Phys. Chem. Solids. 48, 1101–1113 (1987).
Parkins, R. N. Mechanistic aspects of intergranular stress corrosion cracking of ferritic steels. Corrosion 52, 363–374 (1996).
Saxena, A., Singh Raman, R. K. & Muddle, B. C. Slow strain rate testing for monitoring cracking of mild steels for vessels and pipes for processing using caustic solutions. Int. J. Pres. Ves. Pip. 83, 399–404 (2006).
Andresen, P. L. & Ford, F. P. Life prediction by mechanistic modeling and system monitoring of environmental cracking of iron and nickel alloys in aqueous systems. Mater. Sci. Eng. A 103, 167–184 (1988).
Lehockey, E. M., Brennenstuhl, A. M. & Thompson, I. On the relationship between grain boundary connectivity, coincident site lattice boundaries, and intergranular stress corrosion cracking. Corros. Sci. 46, 2383–2404 (2004).
Scott, P. M. Stress corrosion cracking in pressurized water reactors—interpretation, modeling, and remedies. Corrosion 56, 771–782 (2000).
King, A., Johnson, G., Engelberg, D., Ludwig, W. & Marrow, J. Observations of intergranular stress corrosion cracking in a grain-mapped polycrystal. Science 321, 382–385 (2008).
Alvarez, A. Corrosion on aircrafts in marine-tropical environments: A technical analysis. Mater. Perform. 36, 33–38 (1997).
King, F., Lilja, C. & Vahanen, M. Progress in the understanding of the long-term corrosion behaviour of copper canisters. J. Nucl. Mater. 438, 228–237 (2013).
Maiya, P. S., Shack, W. J. & Kassner, T. F. Stress-corrosion cracking of candidate material for nuclear waste containers. Corrosion 46, 954–963 (1990).
Kain, V., Sengupta, P., De, P. K. & Banerjee, S. Case reviews on the effect of microstructure on the corrosion behavior of austenitic alloys for processing and storage of nuclear waste. Metall. Mater. Trans. A 36A, 1075–1084 (2005).
Danielson, M. J., Oster, C. A. & Jones, R. H. Crack tip chemistry modeling of intergranular stress corrosion cracks in nickel containing segregated phosphorus and sulfur. Corros. Sci. 32, 1–21 (1991).
Wells, D. B., Stewart, J., Davidson, R., Scott, P. M. & Williams, D. E. The mechanism of intergranular stress corrosion cracking of sensitized austenitic stainless steel in dilute thiosulphate solution. Corros. Sci. 33, 39–71 (1992).
Newman, R. C. & Sieradzki, K. Electrochemical aspects of stress-corrosion cracking of sensitized stainless steels. Corros. Sci. 23, 363–378 (1983).
Staehle, R. W. in The Theory of Stress Corrosion Cracking in Alloys (ed. Scully, J. C.) 223–286 (NATO, Brussels, 1971).
Theus, G. J. & Staehle, R. W. in Stress Corrosion Cracking and Hydrogen Embrittlement of Iron Base Alloys (eds Staehle, R. W. et al.) 845–892 (National Association of Corrosion Engineers (NACE), Houston, 1977).
Erlebacher, J., Aziz, M. J., Karma, A., Dimitrov, N. & Sieradzki, K. Evolution of nanoporosity in dealloying. Nature 410, 450–453 (2001).
Erlebacher, J. & Sieradzki, K. Pattern formation during dealloying. Scripta Mater. 49, 991–996 (2003).
Rugolo, J., Erlebacher, J. & Sieradzki, K. Length scales in alloy dissolution and measurement of absolute interfacial free energy. Nat. Mater. 5, 946–949 (2006).
Dursun, A., Pugh, D. V. & Corcoran, S. G. Probing the dealloying critical potential: Morphological characterization and steady-state current behavior. J. Electrochem. Soc. 152, B65–B72 (2005).
Li, R. & Sieradzki, K. Ductile–brittle transition in random porous Au. Phys. Rev. Lett. 68, 1168–1171 (1992).
Briot, N. J., Kennerknecht, T., Eberl, C. & Balk, T. J. Mechanical properties of bulk single crystalline nanoporous gold investigated by millimetre-scale tension and compression testing. Phil. Mag. 94, 847–866 (2014).
Jin, H. J. et al. Deforming nanoporous metal: Role of lattice coherency. Acta Mater. 57, 2665–2672 (2009).
Huber, N., Viswanath, R. N., Mameka, N., Markmann, J. & Weißmüller, J. Scaling laws of nanoporous metals under uniaxial compression. Acta Mater. 67, 252–265 (2014).
Badwe, N., Chen, X. & Sieradzki, K. Mechanical properties of nanoporous gold in tension. Acta Mater. 129, 251–258 (2017).
Sun, S., Chen, X., Badwe, N. & Sieradzki, K. Potential-dependent dynamic fracture of nanoporous gold. Nat. Mater. 14, 894–898 (2015).
Serebrinsky, S. A. & Galvele, J. R. Effect of the strain rate on stress corrosion crack velocities in face-centred cubic alloys. A mechanistic interpretation. Corros. Sci. 46, 591–612 (2004).
Newman, R. C. & Senior, N. A. A revised interpretation of ultra-fast stress corrosion cracking experiments by Serebrinsky and Galvele. Corros. Sci. 52, 1541–1544 (2010).
Stewart, J., Wells, D. B., Scott, P. M. & Williams, D. E. Electrochemical noise measurements of stress corrosion cracking of sensitized austenitic stainless steel in high-purity oxygenated water at 288 °C. Corros. Sci. 33, 73–88 (1992).
Manahan, M. P., Macdonald, D. D. & Peterson, A. J. Determination of the fate of the current in the stress corrosion cracking of sensitized 304SS in high temperature aqueous systems. Corros. Sci. 37, 189–208 (1995).
Bruemmer, S. M. & Thomas, L. E. High-resolution analytical electron microsopy characterization of corrosion and cracking at buried interfaces. Surf. Interface Anal. 31, 571–581 (2001).
Schreiber, D. K., Olszta, M. J. & Bruemmer, S. M. Grain boundary depletion and migration during the selective oxidation of Cr in a Ni–5Cr alloy exposed to high-temperature hydrogenated water. Scipta Mater. 89, 41–44 (2014).
Bruemmer, S. M., Olszta, M. J., Toloczko, M. B. & Schreiber, D. K. Grain boundary selective oxidation and stress corrosion crack growth of high-purity nickel binary alloys in high-temperature hydrogenated water. Corros. Sci. 131, 310–323 (2018).
Shen, Z., Arioka, K. & Lozano-Perez, S. A mechanistic study of SCC in Alloy 600 through high-resolution characterization. Corros. Sci. 132, 244–259 (2018).
Forty, A. J. in Physical Metallurgy of Stress Corrosion Fracture (ed. Rhodin, T. N.) 99-120 (Interscience, New York, 1959).
Fritz, J. D., Parks, B. W. & Pickering, H. W. Stress corrosion cracking of Cu–18% Au in 1N Na2SO4–0.01N H2SO4. Scripta Metall. 22, 1063–1068 (1988).
Kelly, R. G., Frost, A. J., Shahrabi, T. & Newman, R. C. Brittle-fracture of an Au/Ag alloy induced by a surface-film. Metall. Trans. A 22A, 531–541 (1991).
Chen, J. S., Slameron, M. & Devine, T. M. Intergranular and transgranular stress corrosion cracking of Cu–30Au. Corros. Sci. 34, 2071–2097 (1993).
Saito, M., Smith, G. S. & Newman, R. C. Testing the film-induced cleavage model of stress-corrosion cracking. Corros. Sci. 35, 411–413 (1993).
Friedersdorf, F. & Sieradzki, K. Film-induced brittle intergranular cracking of silver–gold alloys. Corrosion 52, 331–336 (1996).
Barnes, A., Senior, N. A. & Newman, R. C. Film-induced cleavage of Ag–Au alloys. Metall. Mater. Trans. A 40A, 58–68 (2009).
Oberdorfer, C., Sebastien, M. E., Lütkemeyer, M. & Schmitz, G. Applications of a versatile modelling approach to 3D atom probe simulations. Ultramicroscopy 159, 184–194 (2015).
Gordon, G. M. in Stress Corrosion Cracking and Hydrogen Embrittlement of Iron Base Alloys (eds Staehle, R. W. et al.) 893–945 (National Association of Corrosion Engineers (NACE), Houston, 1977).
Dugdale, H., Armstrong, D. E. J., Tarleton, E., Roberts, S. J. & Lozano-Perez, S. How oxidized grain boundaries fail. Acta Mater. 61, 4707–4713 (2013).
Sridhar, N. et al. Stress corrosion cracking and localized corrosion of carbon steel in nitrate solutions. Corrosion 72, 927–942 (2016).
Knight, S. P., Birbilis, N., Muddle, B. C., Truemen, A. R. & Lynch, S. P. Correlations between intergranular stress corrosion cracking, grain-boundary microchemistry, and grain-boundary electrochemistry for Al–Zn–Mg–Cu alloys. Corros. Sci. 52, 4073–4080 (2010).
Kerns, G. E., Wang, M. T. & Staehle, R. W. in Stress Corrosion Cracking and Hydrogen Embrittlement of Iron Base Alloys (eds Staehle, R. W. et al.) 703–735 (National Association of Corrosion Engineers (NACE), Houston, 1977).
Lynch, S. P. Progression markings, striations, and crack-arrest markings on fracture surfaces. Mater. Sci. Eng., A 468–470, 74–80 (2007).
Turnbull, A. Modelling of environment assisted cracking. Corros. Sci. 34, 921–960 (1993).
Acknowledgements
K.S. gratefully acknowledges the support of this work by the US Department of Energy (DOE), Office of Science, Basic Energy Sciences (BES) under award DE-SC0008677. N.B. acknowledges assistance from John Mardinly and Toshihiro Aoki with STEM characterization. D.K.S., M.J.O., N.R.O. and S.M.B. acknowledge separate support from the US DOE BES Materials Science and Engineering Division. Some of the work (APT and FIB) was performed using EMSL, a DOE Office of Science User Facility sponsored by the Office of Biological and Environmental Research at the Pacific Northwest National Laboratory (PNNL). PNNL is operated by Battelle for the DOE under contract DE-AC05-76RL01830.
Author information
Authors and Affiliations
Contributions
N.B. performed the control sample and crack injection experiments, prepared FIB milled samples, participated in the APT characterization and performed the STEM characterization at ASU. X.C. performed the crack injection experiments that resulted in sample fracture and SEM characterization. D.K.S. performed FIB specimen preparation (for APT and STEM) and led APT characterizations. M.J.O. performed STEM characterization, and together with N.R.O. determined the grain boundary misorientation for the sample shown in Fig. 2 of the manuscript. E.K.K., X.C. and A.Y.T. performed the control sample and crack injection experiments and statistical analysis of the data. S.M.B. directed the research team at PNNL and K.S. designed and supervised the research at ASU. All authors contributed to writing and editing this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Tables: Table 1, Supplementary Figures: Figures 1–15, Supplementary References: 1–14
Rights and permissions
About this article
Cite this article
Badwe, N., Chen, X., Schreiber, D.K. et al. Decoupling the role of stress and corrosion in the intergranular cracking of noble-metal alloys. Nature Mater 17, 887–893 (2018). https://doi.org/10.1038/s41563-018-0162-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-018-0162-x
This article is cited by
-
One dimensional wormhole corrosion in metals
Nature Communications (2023)
-
Crystallographic effects on transgranular chloride-induced stress corrosion crack propagation of arc welded austenitic stainless steel
npj Materials Degradation (2022)
-
Evolution of micro-pores in Ni–Cr alloys via molten salt dealloying
Scientific Reports (2022)
-
Metal matrix nanocomposites in tribology: Manufacturing, performance, and mechanisms
Friction (2022)
-
Improved anticorrosion performance of mixed valence Mn-modified ZnO dilute magnetic solid solution with multilevel self-assembled network structure
Nano Research (2022)