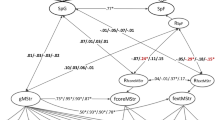
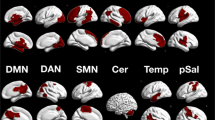
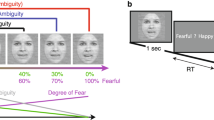
Abstract
Face processing supports our ability to recognize friend from foe, form tribes and understand the emotional implications of changes in facial musculature. This skill relies on a distributed network of brain regions, but how these regions interact is poorly understood. Here we integrate anatomical and functional connectivity measurements with behavioural assays to create a global model of the face connectome. We dissect key features, such as the network topology and fibre composition. We propose a neurocognitive model with three core streams; face processing along these streams occurs in a parallel and reciprocal manner. Although long-range fibre paths are important, the face network is dominated by short-range fibres. Finally, we provide evidence that the well-known right lateralization of face processing arises from imbalanced intra- and interhemispheric connections. In summary, the face network relies on dynamic communication across highly structured fibre tracts, enabling coherent face processing that underpins behaviour and cognition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All data used in the present study were obtained from the WU-Minn HCP Consortium S900 Release. They are publicly available at https://www.humanconnectome.org.
Code availability
Most analyses were conducted using common software (FSL, SPM) or an open source toolbox that can be downloaded from GitHub (Louvain community detection algorithm, NetworkX, or AFQ toolbox). Custom codes can be accessed at https://github.com/mirrorneuronwang/HCP_face_connectome and are available from the corresponding authors on request.
References
Duchaine, B. & Yovel, G. A revised neural framework for face processing. Annu. Rev. Vis. Sci. 1, 393–416 (2015).
Ku, S. P., Tolias, A. S., Logothetis, N. K. & Goense, J. FMRI of the face-processing network in the ventral temporal lobe of awake and anesthetized macaques. Neuron 70, 352–362 (2011).
Kravitz, D. J., Saleem, K. S., Baker, C. I., Ungerleider, L. G. & Mishkin, M. The ventral visual pathway: an expanded neural framework for the processing of object quality. Trends Cogn. Sci. 17, 26–49 (2013).
Freiwald, W., Duchaine, B. & Yovel, G. Face processing systems: from neurons to real-world social perception. Annu. Rev. Neurosci. 39, 325–346 (2016).
Haxby, J. V., Hoffman, E. A. & Gobbini, M. I. The distributed human neural system for face perception. Trends Cogn. Sci. 4, 223–233 (2000).
Gobbini, M. I. & Haxby, J. V. Neural systems for recognition of familiar faces. Neuropsychologia 45, 32–41 (2007).
Pitcher, D., Walsh, V. & Duchaine, B. The role of the occipital face area in the cortical face perception network. Exp. Brain Res. 209, 481–493 (2011).
Collins, J. A. & Olson, I. R. Beyond the FFA: the role of the ventral anterior temporal lobes in face processing. Neuropsychologia 61, 65–79 (2014).
Wang, Y. et al. A dynamic neural architecture for social knowledge retrieval. Proc. Natl Acad. Sci. USA 114, E3305–E3314 (2017).
Mende-Siedlecki, P., Said, C. P. & Todorov, A. The social evaluation of faces: a meta-analysis of functional neuroimaging studies. Soc. Cogn. Affect. Neurosci 8, 285–299 (2013).
Landi, S. M. & Freiwald, W. A. Two areas for familiar face recognition in the primate brain. Science 357, 591–595 (2017).
Allison, T., Puce, a & McCarthy, G. Social perception from visual cues: role of the STS region. Trends Cogn. Sci. 4, 267–278 (2000).
Chan, A. W.-Y. & Downing, P. E. Faces and eyes in human lateral prefrontal cortex. Front. Hum. Neurosci. 5, 51 (2011).
O’ Scalaidhe, S. P., Wilson, F. A. & Goldman-Rakic, P. S. Areal segregation of face-processing neurons in prefrontal cortex. Science 278, 1135–1138 (1997).
Troiani, V., Dougherty, C. C., Michael, A. M. & Olson, I. R. Characterization of face-selective patches in orbitofrontal cortex. Front. Hum. Neurosci. 10, 279 (2016).
Barat, E., Wirth, S. & Duhamel, J.-R. Face cells in orbitofrontal cortex represent social categories. Proc. Natl Acad. Sci. USA 115, E11158–E11167 (2018).
Tsao, D. Y., Schweers, N., Moeller, S. & Freiwald, W. A. Patches of face-selective cortex in the macaque frontal lobe. Nat. Neurosci. 11, 877–879 (2008).
Tsao, D. Y., Moeller, S. & Freiwald, W. A. Comparing face patch systems in macaques and humans. Proc. Natl Acad. Sci. USA 105, 19514–19519 (2008).
O’Doherty, J. et al. Beauty in a smile: the role of medial orbitofrontal cortex in facial attractiveness. Neuropsychologia 41, 147–155 (2003).
Haxby, J. V. & Gobbini, M. I. Distributed neural systems for face perception. in Oxford Handbook of Face Perception (eds Calder, A. J., Rhodes, G., Johnson, M. H. & Haxby, J. V.) 93–110 (Oxford Univ. Press, 2012).
Grimaldi, P., Saleem, K. S. & Tsao, D. Anatomical connections of the functionally defined ‘face patches’ in the macaque monkey. Neuron 90, 1325–1342 (2016).
Wang, Y., Metoki, A., Alm, K. H. & Olson, I. R. White matter pathways and social cognition. Neurosci. Biobehav. Rev. 90, 350–370 (2018).
Gschwind, M., Pourtois, G., Schwartz, S., Van De Ville, D. & Vuilleumier, P. White-matter connectivity between face-responsive regions in the human brain. Cereb. Cortex 22, 1564–1576 (2012).
Pyles, J. A., Verstynen, T. D., Schneider, W. & Tarr, M. J. Explicating the face perception network with white matter connectivity. PLoS One 8, e61611 (2013).
Fairhall, S. L. & Ishai, A. Effective connectivity within the distributed cortical network for face perception. Cereb. Cortex 17, 2400–2406 (2007).
Davies-Thompson, J. & Andrews, T. J. Intra- and interhemispheric connectivity between face-selective regions in the human brain. J. Neurophysiol. 108, 3087–3095 (2012).
Van Essen, D. C. et al. The WU-Minn human connectome project: an overview. Neuroimage 80, 62–79 (2013).
Pestilli, F., Yeatman, J. D., Rokem, A., Kay, K. N. & Wandell, B. A. Evaluation and statistical inference for human connectomes. Nat. Methods 11, 1058–1063 (2014).
Maier-Hein, K. H. et al. The challenge of mapping the human connectome based on diffusion tractography. Nat. Commun. 8, 1349 (2017).
Thomas, C. et al. Anatomical accuracy of brain connections derived from diffusion MRI tractography is inherently limited. Proc. Natl Acad. Sci. USA 111, 16574–16579 (2014).
Gao, Y. et al. Validation of DTI tractography-based measures of primary motor area connectivity in the squirrel monkey brain. PLoS One 8, e75065 (2013).
Fillard, P. et al. Quantitative evaluation of 10 tractography algorithms on a realistic diffusion MR phantom. Neuroimage 56, 220–234 (2011).
Delettre, C. et al. Comparison between diffusion MRI tractography and histological tract-tracing of cortico-cortical structural connectivity in the ferret brain. Netw. Neurosci. 3, 1038–1050 (2019).
Avena-Koenigsberger, A., Misic, B. & Sporns, O. Communication dynamics in complex brain networks. Nat. Rev. Neurosci. 19, 17–33 (2017).
Crofts, J. J. & Higham, D. J. A weighted communicability measure applied to complex brain networks. J. R. Soc. Interface 6, 411–414 (2009).
Andreotti, J. et al. Validation of network communicability metrics for the analysis of brain structural networks. PLoS One 9, e0115503 (2014).
Yeatman, J. D., Dougherty, R. F., Myall, N. J., Wandell, B. A. & Feldman, H. M. Tract profiles of white matter properties: automating fiber-tract quantification. PLoS One 7, e49790 (2012).
Schuz, A. & Braitenberg, V. in Cortical Areas: Unity and Diversity (eds Shuez, A. & Miller, R.) 377–384 (Taylor & Francis, 2002).
Jbabdi, S., Sotiropoulos, S. N., Haber, S. N., Van Essen, D. C. & Behrens, T. E. Measuring macroscopic brain connections in vivo. Nat. Neurosci. 18, 1546–1555 (2015).
Wandell, B. A. Clarifying human white matter. Annu. Rev. Neurosci. 39, 103–128 (2016).
Mori, S. et al. Stereotaxic white matter atlas based on diffusion tensor imaging in an ICBM template. Neuroimage 40, 570–582 (2008).
Guevara, M. et al. Reproducibility of superficial white matter tracts using diffusion-weighted imaging tractography. Neuroimage 147, 703–725 (2017).
Román, C. et al. Clustering of whole-brain white matter short association bundles using HARDI data. Front. Neuroinform. 11, 73 (2017).
Smith, S. M. et al. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl Acad. Sci. USA 106, 13040–13045 (2009).
Stephan, K. E. On the role of general system theory for functional neuroimaging. J. Anat. 205, 443–470 (2004).
Friston, K. J. et al. Psychophysiological and modulatory interactions in neuroimaging. Neuroimage 6, 218–229 (1997).
Friston, K. J., Harrison, L. & Penny, W. Dynamic causal modelling. Neuroimage 19, 1273–1302 (2003).
Cole, M. W., Bassett, D. S., Power, J. D., Braver, T. S. & Petersen, S. E. Intrinsic and task-evoked network architectures of the human brain. Neuron 83, 238–251 (2014).
Gratton, C. et al. Functional brain networks are dominated by stable group and individual factors, not cognitive or daily variation. Neuron 98, 439–452 (2018).
Krienen, F. M., Yeo, B. T. & Buckner, R. L. Reconfigurable task-dependent functional coupling modes cluster around a core functional architecture. Proc. R. Soc. B 369, 20130526 (2014).
Friston, K. J. Functional and effective connectivity: a review. Brain Connect. 1, 13–36 (2011).
Smith, D. V., Gseir, M., Speer, M. E. & Delgado, M. R. Toward a cumulative science of functional integration: a meta-analysis of psychophysiological interactions. Hum. Brain Mapp. 37, 2904–2917 (2016).
Gerchen, M. F., Bernal-Casas, D. & Kirsch, P. Analyzing task-dependent brain network changes by whole-brain psychophysiological interactions: a comparison to conventional analysis. Hum. Brain Mapp. 35, 5071–5082 (2014).
Stephan, K. E. et al. Ten simple rules for dynamic causal modeling. Neuroimage 49, 3099–3109 (2010).
Honey, C. J. et al. Predicting human resting-state functional connectivity from structural connectivity. Proc. Natl Acad. Sci. USA 106, 2035–2040 (2009).
Warren, D. E. et al. Surgically disconnected temporal pole exhibits resting functional connectivity with remote brain regions. Preprint at bioRxiv https://doi.org/10.1101/127571 (2017).
Tyszka, J. M., Kennedy, D. P., Adolphs, R. & Paul, L. K. Intact bilateral resting-state networks in the absence of the corpus callosum. J. Neurosci. 31, 15154–15162 (2011).
Goni, J. et al. Resting-brain functional connectivity predicted by analytic measures of network communication. Proc. Natl Acad. Sci. USA 111, 833–838 (2014).
Grayson, D. S. et al. The rhesus monkey connectome predicts disrupted functional networks resulting from pharmacogenetic inactivation of the amygdala. Neuron 91, 453–466 (2016).
Rousselet, G. A. & Pernet, C. R. Improving standards in brain-behavior correlation analyses. Front. Hum. Neurosci 6, 119 (2012).
Arbabshirani, M. R., Plis, S., Sui, J. & Calhoun, V. D. Single subject prediction of brain disorders in neuroimaging: promises and pitfalls. Neuroimage 145, 137–165 (2017).
Fang, Y. et al. Semantic representation in the white matter pathway. PLoS Biol. 16, e2003993 (2018).
Gottfried, J. A., Doherty, J. O. & Dolan, R. J. Encoding predictive reward value in human amygdala and orbitofrontal cortex. Science 301, 1104–1107 (2003).
Grill-Spector, K., Weiner, K. S., Kay, K. & Gomez, J. The functional neuroanatomy of human face perception. Annu. Rev. Vis. Sci. 3, 167–196 (2017).
Rossion, B. Constraining the cortical face network by neuroimaging studies of acquired prosopagnosia. Neuroimage 40, 423–426 (2008).
Weiner, K. S. et al. The face-processing network is resilient to focal resection of human visual cortex. J. Neurosci. 36, 8425–8440 (2016).
Lohse, M. et al. Effective connectivity from early visual cortex to posterior occipitotemporal face areas supports face selectivity and predicts developmental prosopagnosia. J. Neurosci. 36, 3821–3828 (2016).
Chen, C. C., Henson, R. N., Stephan, K. E., Kilner, J. M. & Friston, K. J. NeuroImage forward and backward connections in the brain: a DCM study of functional asymmetries. Neuroimage 45, 453–462 (2009).
He, W. & Johnson, B. W. Developmental Cognitive neuroscience development of face recognition: dynamic causal modelling of MEG data. Dev. Cogn. Neurosci. 30, 13–22 (2018).
Bassett, D. S. & Bullmore, E. T. Small-world brain networks revisited. Neuroscientist 23, 499–516 (2017).
Betzel, R. F. & Bassett, D. S. Specificity and robustness of long-distance connections in weighted, interareal connectomes. Proc. Natl Acad. Sci. USA 115, E4880–E4889 (2018).
Bullmore, E. & Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nat. Rev. Neurosci. 10, 312–312 (2009).
Wang, Y., Olson, I. R., Wang, Y. & Olson, I. R. The original social network: white matter and social cognition. Trends Cogn. Sci. 22, 504–516 (2018).
Willems, R. M., Der Haegen, L. Van, Fisher, S. E. & Francks, C. On the other hand: including left-handers in cognitive neuroscience and neurogenetics. Nat. Rev. Neurosci. 15, 193–201 (2014).
Levine, S. C., Banich, M. T. & Koch-Weser, M. P. Face recognition: a general or specific right hemisphere capacity? Brain Cogn. 8, 303–325 (1988).
Bukowski, H., Dricot, L., Hanseeuw, B. & Rossion, B. Cerebral lateralization of face-sensitive areas in left-handers: only the FFA does not get it right. Cortex 49, 2583–2589 (2013).
Eimer, M. The face-Sensitive N170 component of the event-related brain potential. in Oxford Handbook of Face Perception (eds Calder, A. J., Rhodes, G., Johnson, M. H. & Haxby, J. V.) 329–344 (Oxford Univ. Press, 2012).
Rangarajan, V. et al. Electrical stimulation of the left and right human fusiform gyrus causes different effects in conscious face perception. J. Neurosci. 34, 12828–12836 (2014).
Ringo, J. L., Don, R. W., Demeter, S. & Simard, P. Y. Time is of the essence: a conjecture that hemispheric specialization arises from interhemispheric conduction delay. Cereb. Cortex 4, 331–343 (1994).
Karolis, V., Corbetta, M. & Thiebaut de Schotten, M. Architecture of functional lateralisation and its relationship to callosal connectivity in the human brain. Nat. Commun. 10, 1417 (2019).
Uddin, L. Q. et al. Residual functional connectivity in the split-brain revealed with resting-state functional MRI. Neuroreport 19, 703–709 (2008).
Shen, K., Hutchison, R. M., Bezgin, G., Everling, S. & McIntosh, A. R. Network structure shapes spontaneous functional connectivity dynamics. J. Neurosci. 35, 5579–5588 (2015).
Chamberland, M. et al. On the origin of individual functional connectivity variability: the role of white matter architecture. Brain Connect. 7, 491–503 (2017).
Tsang, A. et al. White matter structural connectivity is not correlated to cortical resting-state functional connectivity over the healthy adult lifespan. Front. Aging Neurosci. 9, 144 (2017).
Liang, H. & Wang, H. Structure-function network mapping and its assessment via persistent homology. PLoS Comput. Biol. 13, e1005325 (2017).
Medaglia, J. D. et al. Functional alignment with anatomical networks is associated with cognitive flexibility. Nat. Hum. Behav 2, 156–164 (2018).
Becker, C. O. et al. Spectral mapping of brain functional connectivity from diffusion imaging. Sci. Rep. 8, 1411 (2018).
Philippi, C. L., Mehta, S., Grabowski, T., Adolphs, R. & Rudrauf, D. Damage to association fiber tracts impairs recognition of the facial expression of emotion. J. Neurosci. 29, 15089–15099 (2009).
Westfall, J. & Yarkoni, T. Statistically controlling for confounding constructs is harder than you think. PLoS One 11, e0152719 (2016).
Genon, S., Reid, A., Langner, R., Amunts, K. & Eickhoff, S. B. How to characterize the function of a brain region. Trends Cogn. Sci. 22, 350–364 (2018).
Morcom, A. M. & Fletcher, P. C. Does the brain have a baseline? Why we should be resisting a rest. Neuroimage 37, 1073–1082 (2007).
Buckner, R. L., Krienen, F. M. & Yeo, B. T. T. Opportunities and limitations of intrinsic functional connectivity MRI. Nat. Neurosci. 16, 832–837 (2013).
Turchi, J. et al. The basal forebrain regulates global resting-state fMRI fluctuations. Neuron 97, 940–952 (2018).
Reveley, C. et al. Superficial white matter fiber systems impede detection of long-range cortical connections in diffusion MR tractography. Proc. Natl Acad. Sci. USA 112, E2820–E2828 (2015).
King, D. R., de Chastelaine, M., Elward, R. L., Wang, T. H. & Rugg, M. D. Recollection-related increases in functional connectivity predict individual differences in memory accuracy. J. Neurosci. 35, 1763–1772 (2015).
Cole, M. W. Task activations produce spurious but systematic inflation of task functional connectivity estimates. Neuroimage 189, 1–18 (2019).
Anzellotti, S., Kliemann, D., Jacoby, N. & Saxe, R. Directed network discovery with dynamic network modelling. Neuropsychologia 99, 1–11 (2017).
Gates, K. M. & Molenaar, P. C. M. Group search algorithm recovers effective connectivity maps for individuals in homogeneous and heterogeneous samples. Neuroimage 63, 310–319 (2012).
Gates, K. M., Molenaar, P. C. M., Hillary, F. G. & Slobounov, S. Extended unified SEM approach for modeling event-related fMRI data. Neuroimage 54, 1151–1158 (2011).
Smith, S. M. et al. Network modelling methods for FMRI. Neuroimage 54, 875–891 (2011).
Gur, R. C. et al. A cognitive neuroscience-based computerized battery for efficient measurement of individual differences: standardization and initial construct validation. J. Neurosci. Methods 187, 254–262 (2010).
McKay, N. S., Iwabuchi, S. J., Häberling, I. S., Corballis, M. C. & Kirk, I. J. Atypical white matter microstructure in left-handed individuals. Laterality 22, 257–267 (2017).
Van Essen, D. C. et al. The human connectome project: a data acquisition perspective. Neuroimage 62, 2222–2231 (2012).
Smith, S. M. et al. Functional connectomics from resting-state fMRI. Trends Cogn. Sci. 17, 666–682 (2013).
Barch, D. M. et al. Function in the human connectome: task-fMRI and individual differences in behavior. Neuroimage 80, 169–189 (2013).
Glasser, M. F. et al. The minimal preprocessing pipelines for the human connectome project. Neuroimage 80, 105–124 (2013).
Behrens, T. E. J., Berg, H. J., Jbabdi, S., Rushworth, M. F. S. & Woolrich, M. W. Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? Neuroimage 34, 144–155 (2007).
Griffanti, L. et al. ICA-based artefact removal and accelerated fMRI acquisition for improved resting state network imaging. Neuroimage 95, 232–247 (2014).
Weiner, K. S. et al. The mid-fusiform sulcus: a landmark identifying both cytoarchitectonic and functional divisions of human ventral temporal cortex. Neuroimage 84, 453–465 (2014).
Coalson, T. S., Van Essen, D. C. & Glasser, M. F. The impact of traditional neuroimaging methods on the spatial localization of cortical areas. Proc. Natl Acad. Sci. USA 115, E6356–E6365 (2018).
Robinson, E. C. et al. MSM: a new flexible framework for multimodal surface matching. Neuroimage 100, 414–426 (2014).
Fox, C. J., Iaria, G. & Barton, J. J. S. Defining the face processing network: optimization of the functional localizer in fMRI. Hum. Brain Mapp. 30, 1637–1651 (2009).
Stigliani, X. A., Weiner, X. K. S. & Grill-spector, X. K. Temporal processing capacity in high-level visual cortex is domain specific. J. Neurosci. 35, 12412–12424 (2015).
Palla, G., Derényi, I., Farkas, I. & Vicsek, T. Uncovering the overlapping community structure of complex networks in nature and society. Nature 435, 814–818 (2005).
McLaren, D. G., Ries, M. L., Xu, G. & Johnson, S. C. A generalized form of context-dependent psychophysiological interactions (gPPI): a comparison to standard approaches. Neuroimage 61, 1277–1286 (2012).
Di, X. & Biswal, B. B. Psychophysiological interactions in a visual checkerboard task: reproducibility, reliability, and the effects of deconvolution. Front. Neurosci. 11, 573 (2017).
Hillebrandt, H., Friston, K. J. & Blakemore, S.-J. Effective connectivity during animacy perception—dynamic causal modelling of human connectome project data. Sci. Rep. 4, 6240 (2015).
Stephan, K. E., Penny, W. D., Daunizeau, J., Moran, R. J. & Friston, K. J. Bayesian model selection for group studies. Neuroimage 46, 1004–1017 (2009).
Acknowledgements
We thank K. Zhang, N. Asadi, S. Zhang, H. Zhang and R. H. Hyon for their advisory help in data analysis; A. Kohlmeyer for assistance with high-performance cluster computing; 30 undergraduates from Temple University for their work in generating and inspecting all coordinates of face ROIs, especially R. Ho, I. Hanik, P. Coleman and L. J. Hoffman. The superficial white matter atlas (LNAO-SWM79) was provided by P. Guevara. This work was supported by a National Institute of Health grant to I.R.O. (RO1 MH091113). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The work also used Temple University High Performance Cluster Service (Owlsnest), which was supported by a National Science Foundation grant (no. 1625061). The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. HCP data were provided by the HCP, WU-Minn Consortium (principal investigators: D. Van Essen and K. Ugurbil; 1U54MH091657) funded by the 16 NIH Institutes and Centers that support the NIH Blueprint for Neuroscience Research; and by the McDonnell Center for Systems Neuroscience at Washington University.
Author information
Authors and Affiliations
Contributions
Y.W. conceived and designed this research. Y.W., A.M., D.V.S., J.D.M., Y.Z., S.B., H.P. and Y.L. performed research and analysed data. Y.W., A.M. and I.R.O. wrote the paper. I.R.O. was principal investigator of the laboratory.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Primary Handling Editor: Marike Schiffer.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 The Interhemispheric Fibers Connecting Bilateral Face ROIs.
Probabilistic tractography indicated that bilateral posterior ROIs (EVC, OFA, FFA, STS and PCC) are connected through the splenium of the corpus callosum whereas bilateral frontal ROIs are connected through the genu (IFG) or rostrum (OFC) of the corpus callosum. Bilateral ATLs have two separate interhemispheric connections, either via the splenium of the corpus callosum (by climbing up posteriorly along the temploral lobe) or the anterior commissure. Each amygdala is connected to the other by the anterior commissure. These findings accord well with previous work on the callosal fiber organization 118, AMG/ATL interhemispheric connections 119, and EVC interhemispheric connections which begin at the boundary of V1 and V2 120–122. Upper row: medial views; Lower row: axial views. Abbreviations: EVC: early visual cortex; OFA: occipital face area; FFA: fusiform face area; ATL: anterior temporal lobe; STS: superior temporal sulcus; IFG: inferior frontal gyrus; AMG: amygdala; OFC: orbitofrontal cortex; PCC: posterior cingulate cortex.
Extended Data Fig. 2 Additional DCM Analysis With Larger Model Space.
For simplicity, we only compared two DCM models in Fig. 4. These two models, however, have limitations, given that they were merely built from preceding PPI results. For instance, the EVC→OFA connectivity is theoretically important but was not significant in the left hemisphere (LH) of PPI results (that’s why we did not included it in the original feedforward model). In addition, one might also be interested in exploring the relative contribution of each recurrent connection to the right hemisphere (RH) face processing. To address these questions, we built larger model space with seven competing models (feedforward models in red colour and recurrent ones in blue colour). (a) The first two models were the ones we used in Fig. 4. Model 1 was feedforward (based on of PPI results in LH) and Model 2 was recurrent (based on of PPI results in RH). Since Model 1 had no direct feedforward connectivity from EVC to OFA, we next built Model 3 with additional EVC→OFA. Model 4 to 7 were recurrent models modified from Model 2. As there were four recurrent connections in Model 2, we removed one feedback connection each time to examine their respective importance to the recurrent model (that is how much the model performance would suffer in the absence of a particular recurrent connection). Model 4 removed EVC←OFA; Model 5 removed EVC←FFA; Model 6 removed EVC←STS; and Model 7 removed OFA←STS. (b) Bayesian model selection (both FFX and RFX) indicates the EVC→OFA connection is important for the feedforward model, as Model 3 performs better than Model 1 in LH. This is consistent with the Haxby model suggesting the critical role of the OFA receiving information from EVC to initiate the face processing. In addition, both feedforward models perform better than any recurrent models in LH, whereas the PPI-derived recurrent model (Model 2) performed the best in RH. These results accord well with our results in Fig. 4. Moreover, among all four recurrent connections, two feedback connections (EVC←OFA, OFA←STS) seem to be particularly important for RH recurrent processing, since removal of either one can lead to enormous drop of model performance (that is Model 4 and 7). In sum, this additional DCM analysis supports our claims in Fig. 4: the LH is dominant with feedforward face processing whereas the RH is dominant with recurrent processing.
Supplementary information
Supplementary Information
Supplementary Tables 1 and 2 and Supplementary Figs. 1–6.
Rights and permissions
About this article
Cite this article
Wang, Y., Metoki, A., Smith, D.V. et al. Multimodal mapping of the face connectome. Nat Hum Behav 4, 397–411 (2020). https://doi.org/10.1038/s41562-019-0811-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-019-0811-3
This article is cited by
-
Functional connectivity of the human face network exhibits right hemispheric lateralization from infancy to adulthood
Scientific Reports (2023)
-
EEG alpha band functional connectivity reveals distinct cortical dynamics for overt and covert emotional face processing
Scientific Reports (2023)
-
Diffusion tensor imaging helps identify shunt-responsive normal pressure hydrocephalus patients among probable iNPH cohort
Neurosurgical Review (2023)
-
Age- and gender-related dispersion of brain networks across the lifespan
GeroScience (2023)
-
Individual differences in white matter microstructure of the face processing brain network are more differentiated from global fibers with increasing ability
Scientific Reports (2022)